SSTR2 Mediates the Inhibitory Effect of SST/CST on Lipolysis in Chicken Adipose Tissue
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals, Primers, Peptides and Antibodies
2.2. Animals
2.3. Tissues Collection
2.4. RT-PCR and Quantitative Real-Time PCR Assays
2.5. Tissue Expression Analysis of Chicken SSTR2 Using RNA-Seq Data
2.6. Immunohistochemical Staining
2.7. Functional Characterization of Chicken SSTR2
2.8. Western Blot
2.9. Cell Counting Assay
2.10. Culture of Chicken Adipose Tissue and Glycerol Detection
2.11. Data Analysis
3. Results
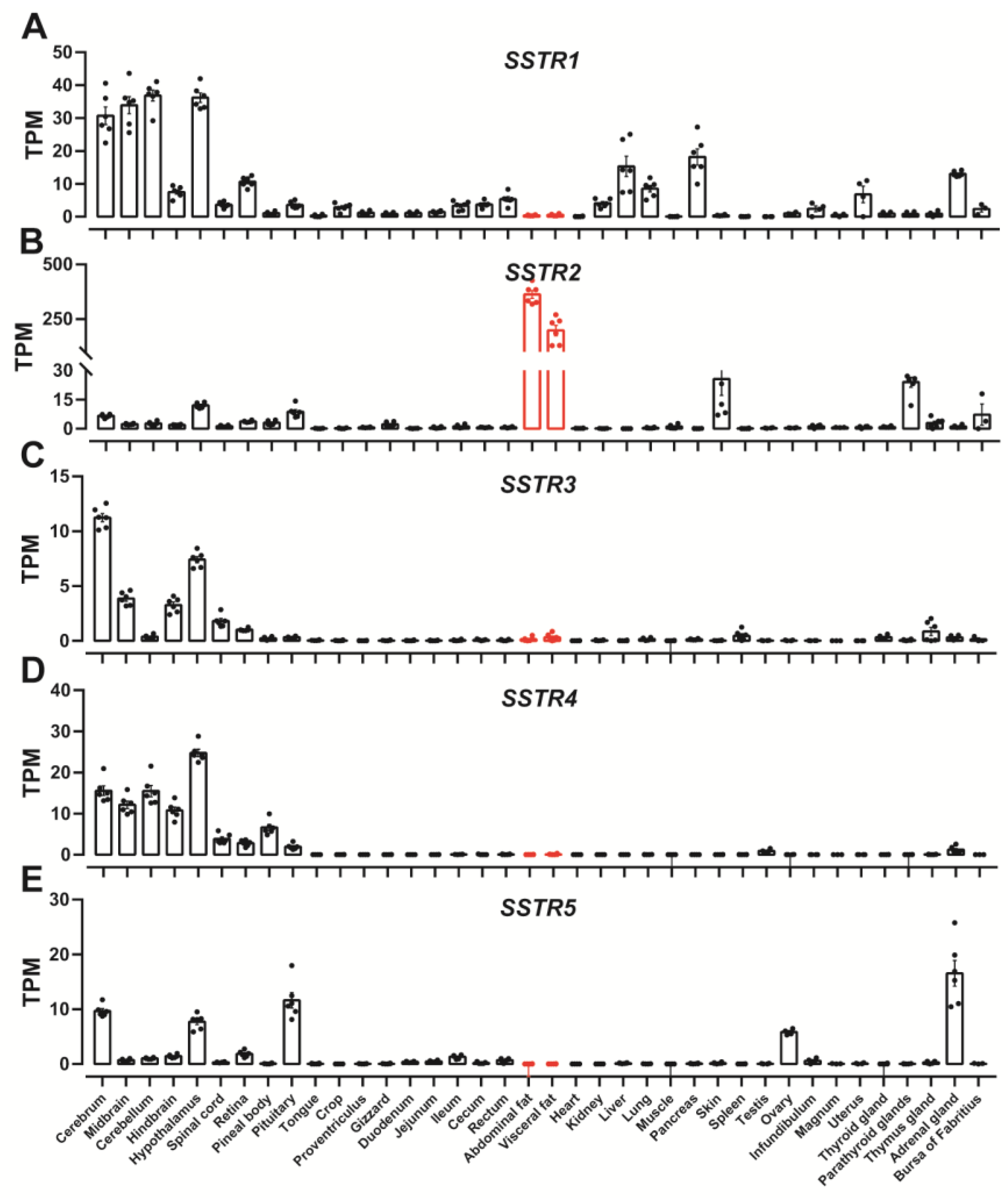
3.1. Expression of SSTR1-5 in Chickens
3.2. Validation of SSTR2 Expression in Chicken Adipose Tissue
3.3. Expression of SSTR2 in Red Jungle Fowl and J-Line Chickens
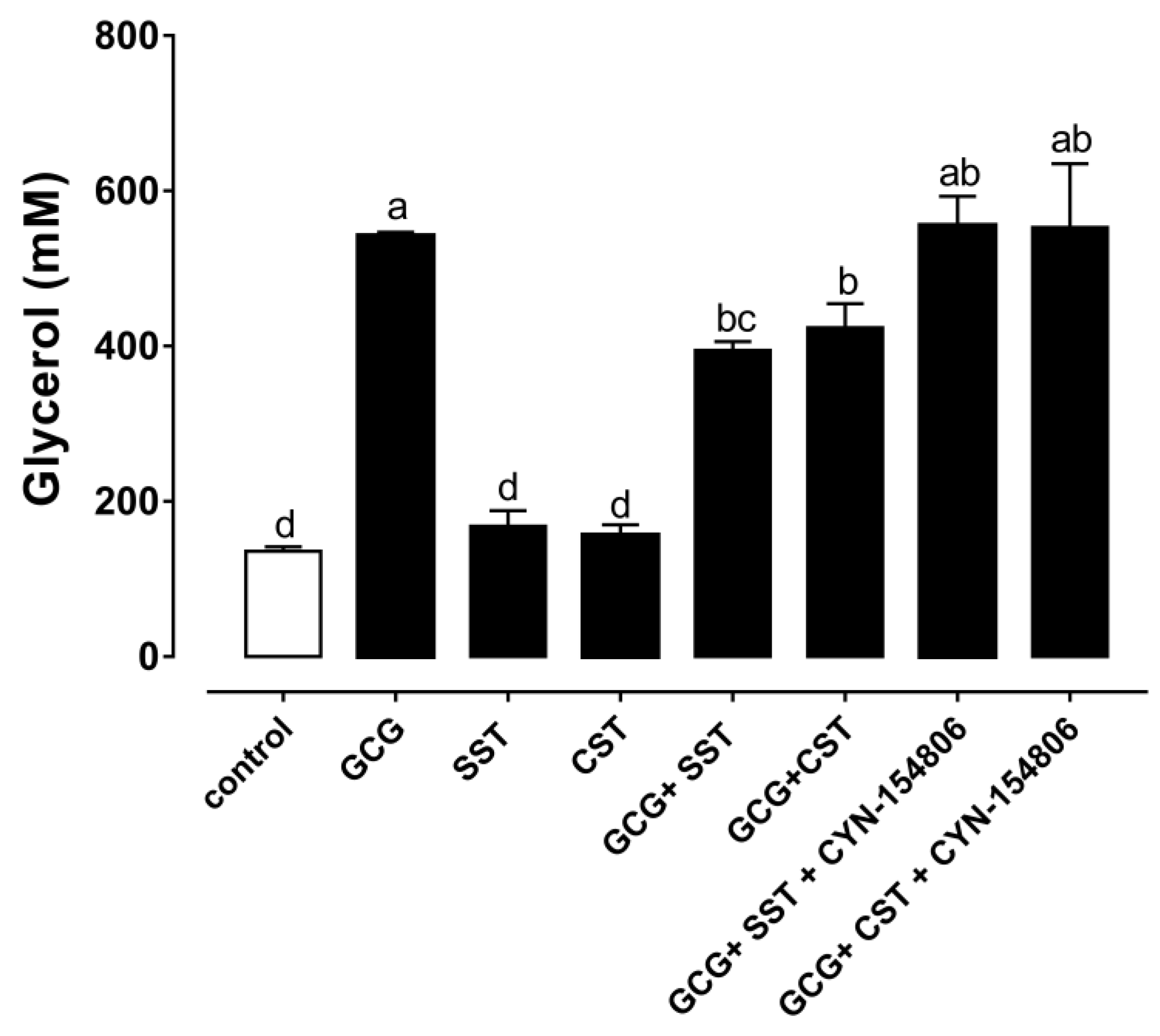
3.4. The Anti-Lipolytic Effect Mediated by SSTR2 in Chicken Adipose Tissue
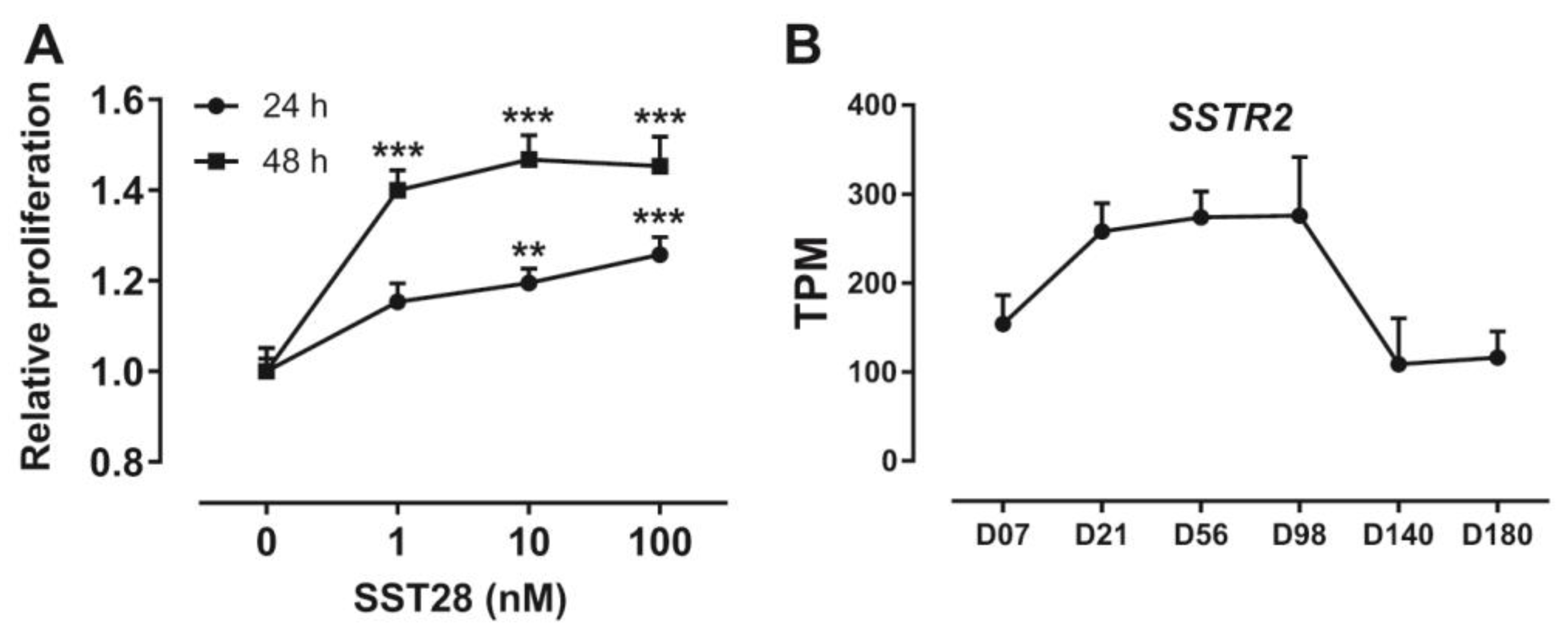
3.5. Activation of SSTR2 Stimulates the Proliferation of ICP-1 Cells
3.6. Developmental Expression Pattern of SSTR2 in Chickens
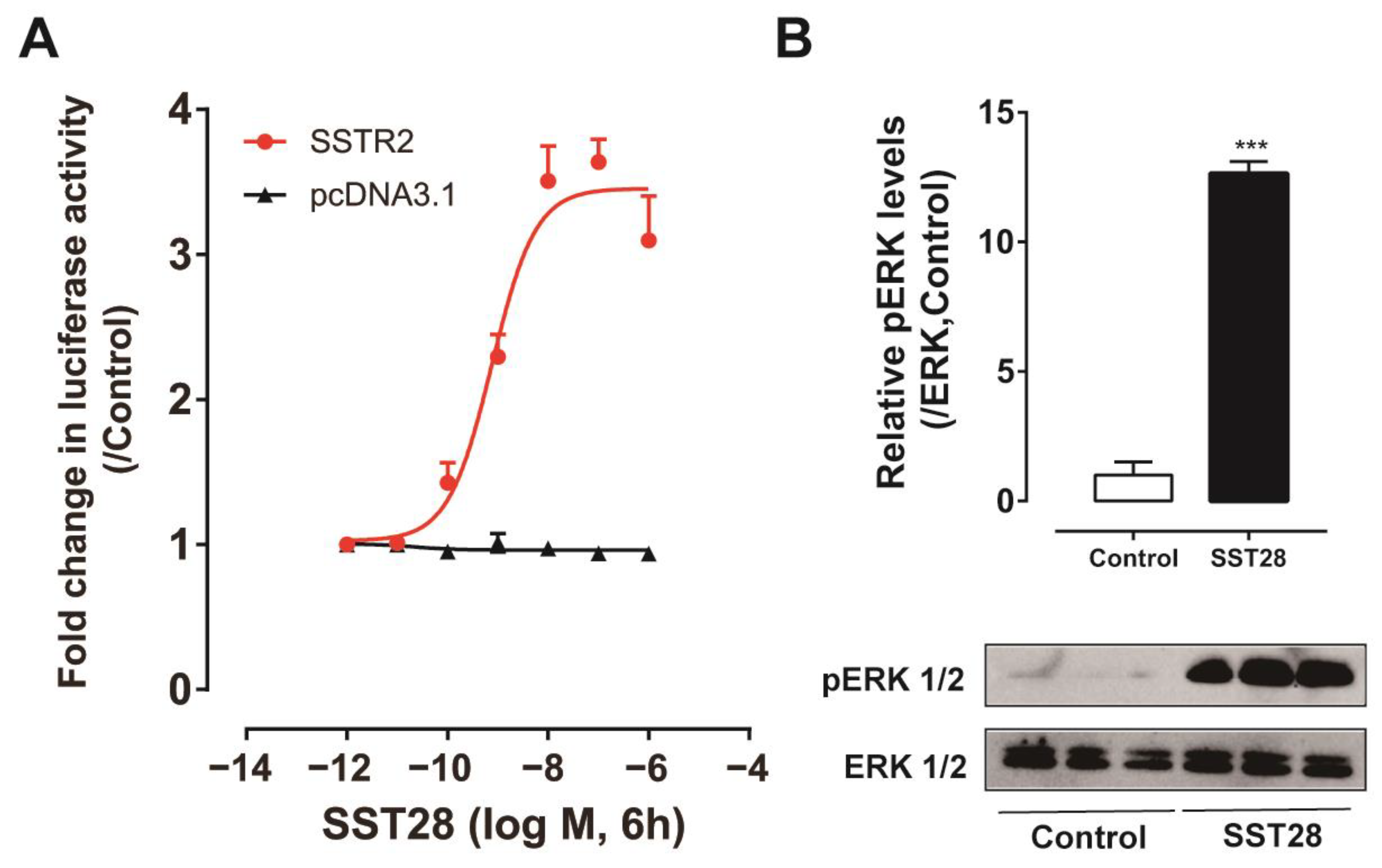
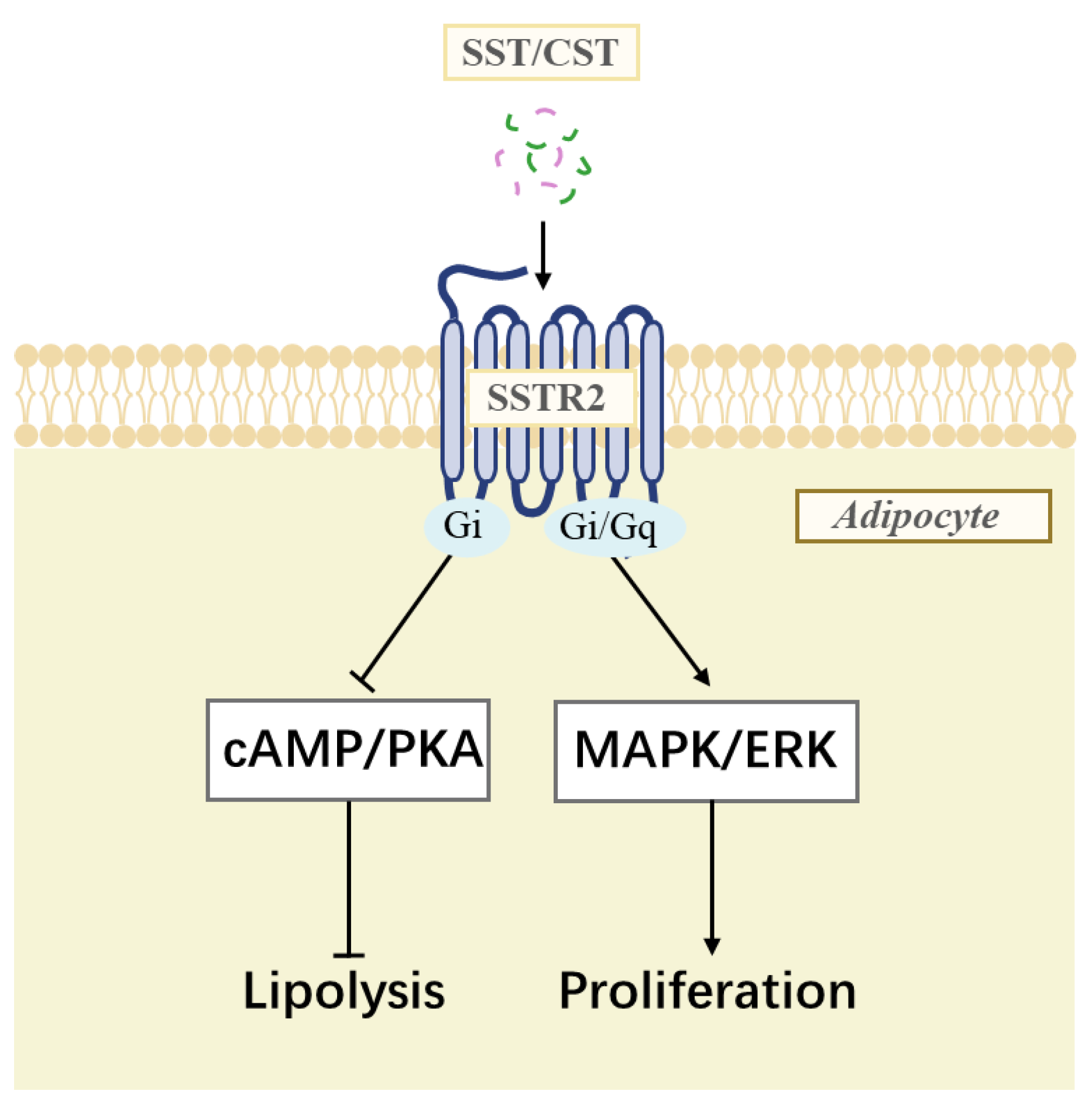
3.7. Identification of SSTR2 Signaling Pathway
4. Discussion
4.1. SSTR2 Is Expressed in Chicken Adipose Tissue
4.2. SSTR2 Mediates the Anti-Lipolytic Effect in Chicken Adipose Tissue
4.3. SSTR2 May Stimulate the Proliferation of Chicken Preadipocytes by Activating the MAPK/ERK Signaling Pathway
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brazeau, P.; Vale, W.; Burgus, R.; Ling, N.; Butcher, M.; Rivier, J.; Guillemin, R. Hypothalamic polypeptide that inhibits the secretion of immunoreactive pituitary growth hormone. Science 1973, 179, 77–79. [Google Scholar] [CrossRef]
- Pradayrol, L.; Jörnvall, H.; Mutt, V.; Ribet, A. N-terminally extended somatostatin: The primary structure of somatostatin-28. FEBS Lett. 1980, 109, 55–58. [Google Scholar] [CrossRef] [PubMed]
- Tostivint, H.; Lihrmann, I.; Bucharles, C.; Vieau, D.; Coulouarn, Y.; Boutelet, I.; Fournier, A.; Conlon, J.M.; Vaudry, H. A second somatostatin gene is expressed in the brain of the frog. Ann. N. Y. Acad. Sci. 1998, 839, 496–497. [Google Scholar] [CrossRef] [PubMed]
- Tostivint, H.; Joly, L.; Lihrmann, I.; Ekker, M.; Vaudry, H. Chromosomal localization of three somatostatin genes in zebrafish.: Evidence that the [Pro]-somatostatin-14 isoform and cortistatin are encoded by orthologous genes. J. Mol. Endocrinol. 2004, 33, R1–R8. [Google Scholar] [CrossRef] [PubMed]
- Nishi, R.; Stubbusch, J.; Hulce, J.J.; Hruska, M.; Pappas, A.; Bravo, M.C.; Huber, L.P.; Bakondi, B.; Soltys, J.; Rohrer, H. The Cortistatin Gene PSS2 Rather Than the Somatostatin Gene PSS1 Is Strongly Expressed in Developing Avian Autonomic Neurons. J. Comp. Neurol. 2010, 518, 839–850. [Google Scholar] [CrossRef]
- Spier, A.D.; de Lecea, L. Cortistatin: A member of the somatostatin neuropeptide family with distinct physiological functions. Brain Res. Rev. 2000, 33, 228–241. [Google Scholar] [CrossRef]
- Barnett, P. Somatostatin and somatostatin receptor physiology. Endocrine 2003, 20, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Gahete, M.D.; Durán-Prado, M.; Luque, R.M.; Martínez-Fuentes, A.J.; Vázquez-Martínez, R.; Malagón, M.M.; Castaño, J.P. Are somatostatin and cortistatin two siblings in regulating endocrine secretions? In vitro work ahead. Mol. Cell. Endocrinol. 2008, 286, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Broglio, F.; Papotti, M.; Muccioli, G.; Ghigo, E. Brain-gut communication: Cortistatin, somatostatin and ghrelin. Trends Endocrin. Met. 2007, 18, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Patel, Y.C. Somatostatin and its receptor family. Front. Neuroendocr. 1999, 20, 157–198. [Google Scholar] [CrossRef] [PubMed]
- Fukusumi, S.; Kitada, C.; Takekawa, S.; Sakamoto, J.; Miyamoto, M.; Hinuma, S.; Kitano, K.; Fujino, M. Identification and characterization of a novel human cortistatin-like peptide. Biochem. Biophys. Res. Commun. 1997, 232, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Panetta, R.; Greenwood, M.T.; Warszynska, A.; Demchyshyn, L.L.; Day, R.; Niznik, H.B.; Srikant, C.B.; Patel, Y.C. Molecular-Cloning, Functional-Characterization, and Chromosomal Localization of a Human Somatostatin Receptor (Somatostatin Receptor Type-5) with Preferential Affinity for Somatostatin-28. Mol. Pharmacol. 1994, 45, 417–427. [Google Scholar] [PubMed]
- Patel, Y.C.; Greenwood, M.T.; Warszynska, A.; Panetta, R.; Srikant, C.B. All five cloned human somatostatin receptors (hSSTR1-5) are functionally coupled to adenylyl cyclase. Biochem. Biophys. Res. Commun. 1994, 198, 605–612. [Google Scholar] [CrossRef] [PubMed]
- Masyuk, T.V.; Masyuk, A.I.; Torres, V.E.; Harris, P.C.; Larusso, N.F. Octreotide inhibits hepatic cystogenesis in a rodent model of polycystic liver disease by reducing cholangiocyte adenosine 3′,5′-cyclic monophosphate. Gastroenterology 2007, 132, 1104–1116. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shlomo, A.; Melmed, S. Pituitary somatostatin receptor signaling. Trends Endocrin. Met. 2010, 21, 123–133. [Google Scholar] [CrossRef]
- Kumar, U.; Sasi, R.; Suresh, S.; Patel, A.; Thangaraju, M.; Metrakos, P.; Patel, S.C.; Patel, Y.C. Subtype-selective expression of the five somatostatin receptors (hSSTR1-5) in human pancreatic islet cells—A quantitative double-label immunohistochemical analysis. Diabetes 1999, 48, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Shimon, I.; Taylor, J.E.; Dong, J.Z.; Bitonte, R.A.; Kim, S.; Morgan, B.; Coy, D.H.; Culler, M.D.; Melmed, S. Somatostatin receptor subtype specificity in human fetal pituitary cultures-Differential role of SSTR2 and SSTR5 for growth hormone, thyroid-stimulating hormone, and prolactin regulation. J. Clin. Investig. 1997, 99, 789–798. [Google Scholar] [CrossRef]
- Strowski, M.Z.; Parmar, R.M.; Blake, A.D.; Schaeffer, J.M. Somatostatin inhibits insulin and glucagon secretion via two receptor subtypes:: An study of pancreatic islets from somatostatin receptor 2 knockout mice. Endocrinology 2000, 141, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Gong, A.Y.; Tietz, P.S.; Muff, M.A.; Splinter, P.L.; Huebert, R.C.; Strowski, M.Z.; Chen, X.M.; LaRusso, N.F. Somatostatin stimulates ductal bile absorption and inhibits ductal bile secretion in mice via SSTR2 on cholangiocytes. Am. J. Physiol.-Cell Physiol. 2003, 284, C1205–C1214. [Google Scholar] [CrossRef] [PubMed]
- Meng, F.; Huang, G.; Gao, S.; Li, J.; Yan, Z.; Wang, Y. Identification of the receptors for somatostatin (SST) and cortistatin (CST) in chickens and investigation of the roles of cSST28, cSST14, and cCST14 in inhibiting cGHRH1–27NH2-induced growth hormone secretion in cultured chicken pituitary cells. Mol. Cell. Endocrinol. 2014, 384, 83–95. [Google Scholar] [CrossRef] [PubMed]
- Geris, K.L.; De Groef, B.; Rohrer, S.P.; Geelissen, S.; Kühn, E.R.; Darras, V.M. Identification of somatostatin receptors controlling growth hormone and thyrotropin secretion in the chicken using receptor subtype-specific agonists. J. Endocrinol. 2003, 177, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Bossis, I.; Porter, T.E. Identification of the somatostatin receptor subtypes involved in regulation of growth hormone secretion in chickens. Mol. Cell. Endocrinol. 2001, 182, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Strosser, M.T.; Discalaguenot, D.; Koch, B.; Mialhe, P. Inhibitory Effect and Mode of Action of Somatostatin on Lipolysis in Chicken Adipocytes. Biochim. Biophys. Acta 1983, 763, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Oscar, T.P. Glucagon-Stimulated Lipolysis of Primary Cultured Broiler Adipocytes. Poultry Sci. 1991, 70, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Freeman, B.M.; Manning, A.C.C. The prandial state and the glycaemic and lipolytic responses of Callus domesticus to catecholamines and glucagon. Comp. Biochem. Physiol. Part A Physiol. 1974, 47, 1145–1152. [Google Scholar] [CrossRef] [PubMed]
- Langslow, D.R.; Hales, C.N. Lipolysis in Chicken Adipose Tissue in Vitro. J. Endocrinol. 1969, 43, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Goodridge, A.G. Lipolysis in vitro in adipose tissue from embryonic and growing chicks. Am. J. Physiol.-Leg. Content 1968, 214, 902–907. [Google Scholar] [CrossRef] [PubMed]
- Strosser, M.T.; Foltzer, C.; Cohen, L.; Mialhe, P. Evidence for an Indirect Effect of Somatostatin on Glucagon-Secretion Via Inhibition of Free Fatty-Acids Release in the Duck. Horm. Metab. Res. 1983, 15, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.K.; Luo, Q.; Zhang, J.N.; Mo, C.H.; Wang, Y.J.; Li, J. Endothelins (EDN1, EDN2, EDN3) and their receptors (EDNRA, EDNRB, EDNRB2) in chickens: Functional analysis and tissue distribution. Gen. Comp. Endocr. 2019, 283, 113231. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, X.; Lv, C.; Wan, Y.; Zhang, X.; Li, J.; Wang, Y. A gene expression atlas of Lohmann white chickens. bioRxiv 2022. [Google Scholar] [CrossRef]
- Merkin, J.; Russell, C.; Chen, P.; Burge, C.B. Evolutionary Dynamics of Gene and Isoform Regulation in Mammalian Tissues. Science 2012, 338, 1593–1599. [Google Scholar] [CrossRef] [PubMed]
- Xing, S.Y.; Liu, R.R.; Zhao, G.P.; Liu, L.; Groenen, M.A.M.; Madsen, O.; Zheng, M.Q.; Yang, X.T.; Crooijmans, R.P.M.A.; Wen, J. RNA-Seq Analysis Reveals Hub Genes Involved in Chicken Intramuscular Fat and Abdominal Fat Deposition during Development. Front. Genet. 2020, 11, 1009. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Bu, G.X.; Wan, Y.P.; Zhang, J.N.; Mo, C.H.; Li, J.; Wang, Y.J. Evidence for Neuropeptide W Acting as a Physiological Corticotropin-releasing Inhibitory Factor in Male Chickens. Endocrinology 2022, 163, bqac073. [Google Scholar] [CrossRef] [PubMed]
- Wan, Y.P.; Zhang, Z.; Lin, D.L.; Wang, X.L.; Huang, T.J.; Su, J.C.; Zhang, J.N.; Li, J.; Wang, Y.J. Characterization of CRH-Binding Protein (CRHBP) in Chickens: Molecular Cloning, Tissue Distribution and Investigation of Its Role as a Negative Feedback Regulator within the Hypothalamus-Pituitary-Adrenal Axis. Genes 2022, 13, 1680. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhang, T.M.; Wu, C.Y.; Wang, S.S.; Wang, Y.X.; Li, H.; Wang, N. Immortalization of chicken preadipocytes by retroviral transduction of chicken TERT and TR. PLoS ONE 2017, 12, e0177348. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Everaert, N.; Decuypere, E.; Buyse, J. Adipose tissue and lipid metabolism. In Sturkie’s Avian Physiology; Elsevier: Amsterdam, The Netherlands, 2022; pp. 647–660. [Google Scholar]
- Al-Nasser, A.; Al-Khalaifa, H.; Al-Saffar, A.; Khalil, F.; Albahouh, M.; Ragheb, G.; Al-Haddad, A.; Mashaly, M. Overview of chicken taxonomy and domestication. World Poultry Sci. J. 2007, 63, 285–300. [Google Scholar] [CrossRef]
- Kanginakudru, S.; Metta, M.; Jakati, R.D.; Nagaraju, J. Genetic evidence from Indian red jungle fowl corroborates multiple domestication of modern day chicken. BMC Evol. Biol. 2008, 8, 174. [Google Scholar] [CrossRef]
- O’Carroll, A.M. Localization of messenger ribonucleic acids for somatostatin receptor subtypes (sstr1-5) in the rat adrenal gland. J. Histochem. Cytochem. 2003, 51, 55–60. [Google Scholar] [CrossRef]
- Ballian, N.; Brunicardi, F.C.; Wang, X.P. Somatostatin and its receptors in the development of the endocrine pancreas. Pancreas 2006, 33, 1–12. [Google Scholar] [CrossRef]
- Perez, J.; Rigo, M.; Kaupmann, K.; Bruns, C.; Yasuda, K.; Bell, G.I.; Lübbert, H.; Hoyer, D. Localization of somatostatin (SRIF) SSTR-1, SSTR-2 and SSTR-3 receptor mRNA in rat brain by in situ hybridization. Naunyn-Schmiedeberg’s Arch. Pharmacol. 1994, 349, 145–160. [Google Scholar] [CrossRef] [PubMed]
- Day, R.; Dong, W.; Panetta, R.; Kraicer, J.; Greenwood, M.T.; Patel, Y.C. Expression of mRNA for somatostatin receptor (sstr) types 2 and 5 in individual rat pituitary cells. A double labeling in situ hybridization analysis. Endocrinology 1995, 136, 5232–5235. [Google Scholar] [CrossRef] [PubMed]
- Reed, D.K.; Korytko, A.I.; Hipkin, R.W.; Wehrenberg, W.B.; Schonbrunn, A.; Cuttler, L. Pituitary somatostatin receptor (sst)1-5 expression during rat development: Age-dependent expression of sst2. Endocrinology 1999, 140, 4739–4744. [Google Scholar] [CrossRef] [PubMed]
- Reynaert, H.; Rombouts, K.; Vandermonde, A.; Urbain, D.; Kumar, U.; Bioulac-Sage, P.; Pinzani, M.; Rosenbaum, J.; Geerts, A. Expression of somatostatin receptors in normal and cirrhotic human liver and in hepatocellular carcinoma. Gut 2004, 53, 1180–1189. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Fujishima, F.; Unno, M.; Sasano, H.; Suzuki, T. Somatostatin receptor 2 in 10 different types of human non-neoplastic gastrointestinal neuroendocrine cells. Pathol. Res. Pract. 2023, 244, 154418. [Google Scholar] [CrossRef] [PubMed]
- Balster, D.A.; O’Dorisio, M.S.; Summers, M.A.; Turman, M.A. Segmental expression of somatostatin receptor subtypes sst and sst in tubules and glomeruli of human kidney. Am. J. Physiol.-Renal 2001, 280, F457–F465. [Google Scholar] [CrossRef]
- Thoss, V.S.; Perez, J.; Probst, A.; Hoyer, D. Expression of five somatostatin receptor mRNAs in the human brain and pituitary. Naunyn-Schmiedeberg’s Arch. Pharmacol. 1996, 354, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Koppo, K.; Siklová-Vitková, M.; Klimcáková, E.; Polák, J.; Marques, M.A.; Berlan, M.; Van de Voorde, J.; Bulow, J.; Langin, D.; de Glisezinski, I.; et al. Catecholamine and insulin control of lipolysis in subcutaneous adipose tissue during long-term diet-induced weight loss in obese women. Am. J. Physiol.-Endocrinol. Metab. 2012, 302, E226–E232. [Google Scholar] [CrossRef] [PubMed]
- Arner, P. Catecholamine-induced lipolysis in obesity. Int. J. Obesity 1999, 23, 10–13. [Google Scholar] [CrossRef] [PubMed]
- Campbell, R.M.; Scanes, C.G. Growth hormone inhibition of glucagon-and cAMP-induced lipolysis by chicken adipose tissue in vitro. Proc. Soc. Exp. Biol. Med. 1987, 184, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Feniuk, W.; Jarvie, E.; Luo, J.; Humphrey, P.P.A. Selective somatostatin sst2 receptor blockade with the novel cyclic octapeptide, CYN-154806. Neuropharmacology 2000, 39, 1443–1450. [Google Scholar] [CrossRef] [PubMed]
- Córdoba-Chacón, J.; Gahete, M.D.; Pozo-Salas, A.I.; Martínez-Fuentes, A.J.; de Lecea, L.; Gracia-Navarro, F.; Kineman, R.D.; Castaño, J.P.; Luque, R.M. Cortistatin is not a somatostatin analogue but stimulates prolactin release and inhibits GH and ACTH in a gender-dependent fashion: Potential role of ghrelin. Endocrinology 2011, 152, 4800–4812. [Google Scholar] [CrossRef] [PubMed]
- Di Scala-Guenot, D.; Strosser, M.T.; Mialhe, P. Characterization of somatostatin in peripheral and portal plasma in the duck: In-vivo metabolism of somatostatin-28 and-14. J. Endocrinol. 1984, 100, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Cartwright, A.L. Adipose Cellularity in Gallus-Domesticus—Investigations to Control Body-Composition in Growing Chickens. J. Nutr. 1991, 121, 1486–1497. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Gong, F.Y.; Duan, L.; Lv, X.R.; Wu, H.J.; Tang, Y.; Zhu, H.J.; Chen, X.G. Somatostatin receptor ligands suppressed proliferation and lipogenesis in 3T3-L1 preadipocytes. Basic Clin. Pharmacol. 2022, 131, 174–188. [Google Scholar] [CrossRef] [PubMed]
- March, B.E.; Macmillan, C.; Chu, S. Characteristics of Adipose-Tissue Growth in Broiler-Type Chickens to 22 Weeks of Age and the Effects of Dietary-Protein and Lipid. Poultry Sci. 1984, 63, 2207–2216. [Google Scholar] [CrossRef] [PubMed]
- Hood, R.L. The Cellular Basis for Growth of the Abdominal Fat Pad in Broiler-Type Chickens. Poultry Sci. 1982, 61, 117–121. [Google Scholar] [CrossRef]
- Carmen, G.Y.; Víctor, S.M. Signalling mechanisms regulating lipolysis. Cell Signal 2006, 18, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Liu, W.Z.; Liu, T.; Feng, X.; Yang, N.; Zhou, H.F. Signaling pathway of MAPK/ERK in cell proliferation, differentiation, migration, senescence and apoptosis. J. Recept. Sig. Transd. 2015, 35, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wang, J.J.; Li, J.M.; Park, K.; Qian, X.X.; Ma, J.X.; Zhang, S.X. Pigment epithelium-derived factor suppresses adipogenesis via inhibition of the MAPK/ERK pathway in 3T3-L1 preadipocytes. Am. J. Physiol.-Endocrinol. Metab. 2009, 297, E1378–E1387. [Google Scholar] [CrossRef] [PubMed]
- Bost, F.; Aouadi, M.; Caron, L.; Binétruy, B. The role of MAPKs in adipocyte differentiation and obesity. Biochimie 2005, 87, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Martini, C.N.; Gabrielli, M.; Bonifacino, G.; Codesido, M.M.; Vila, M.D. Lead enhancement of 3T3-L1 fibroblasts differentiation to adipocytes involves ERK, C/EBPβ and PPARγ activation. Mol. Cell Biochem. 2018, 437, 37–44. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Zhang, J.; Huang, T.; Wang, X.; Su, J.; He, J.; Shi, N.; Wang, Y.; Li, J. SSTR2 Mediates the Inhibitory Effect of SST/CST on Lipolysis in Chicken Adipose Tissue. Animals 2024, 14, 1034. https://doi.org/10.3390/ani14071034
Zhang X, Zhang J, Huang T, Wang X, Su J, He J, Shi N, Wang Y, Li J. SSTR2 Mediates the Inhibitory Effect of SST/CST on Lipolysis in Chicken Adipose Tissue. Animals. 2024; 14(7):1034. https://doi.org/10.3390/ani14071034
Chicago/Turabian StyleZhang, Xiao, Jiannan Zhang, Tianjiao Huang, Xinglong Wang, Jiancheng Su, Jiliang He, Ningkun Shi, Yajun Wang, and Juan Li. 2024. "SSTR2 Mediates the Inhibitory Effect of SST/CST on Lipolysis in Chicken Adipose Tissue" Animals 14, no. 7: 1034. https://doi.org/10.3390/ani14071034
APA StyleZhang, X., Zhang, J., Huang, T., Wang, X., Su, J., He, J., Shi, N., Wang, Y., & Li, J. (2024). SSTR2 Mediates the Inhibitory Effect of SST/CST on Lipolysis in Chicken Adipose Tissue. Animals, 14(7), 1034. https://doi.org/10.3390/ani14071034





