Transferrin Receptor 2 in Canine Testicular Tumors: An Emerging Key Role in Seminomas
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissue Samples
2.2. Histology
2.3. Immunohistochemistry
2.4. Western Blot
3. Results
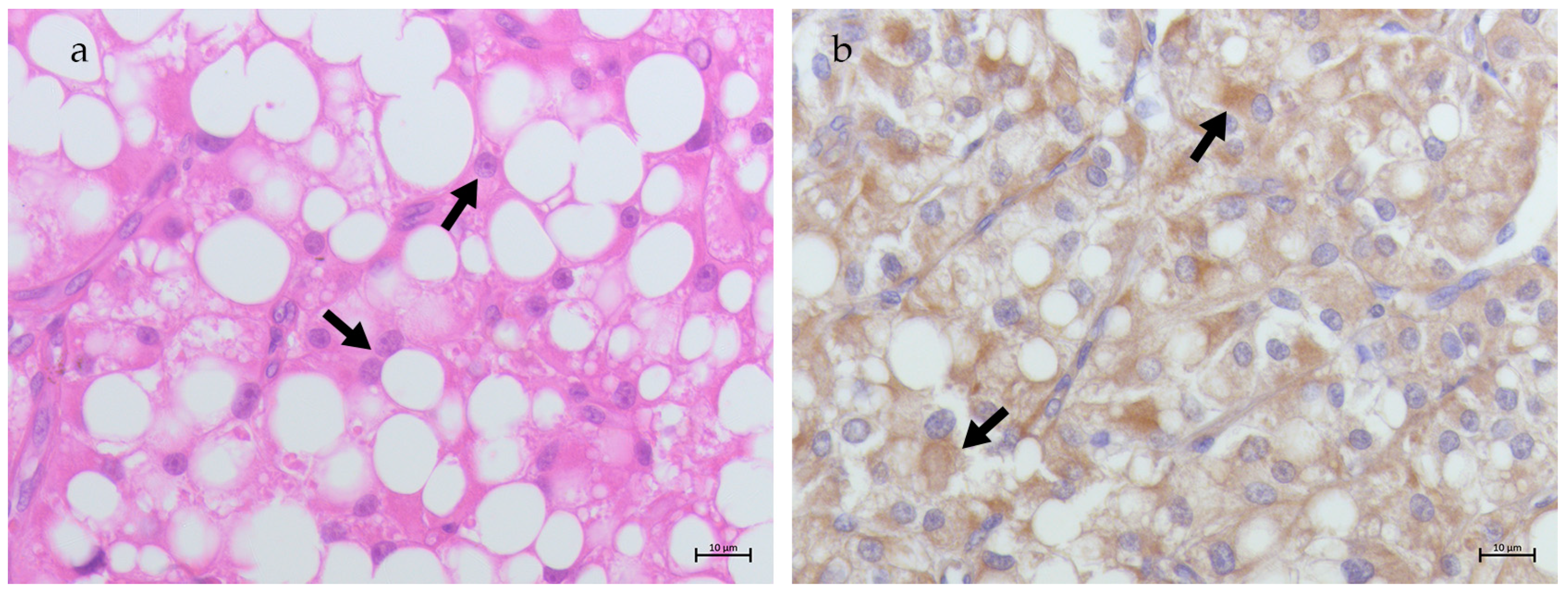
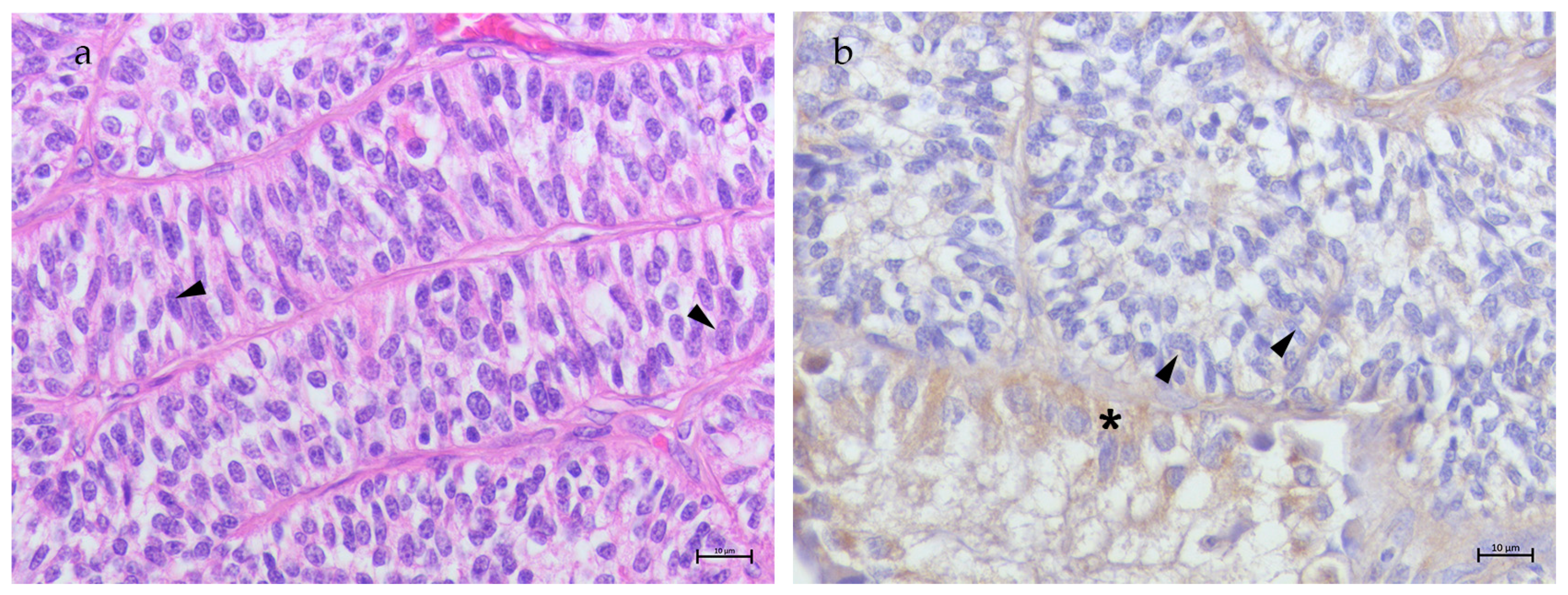
3.1. Non-Neoplastic Testis
3.2. Neoplastic Testis
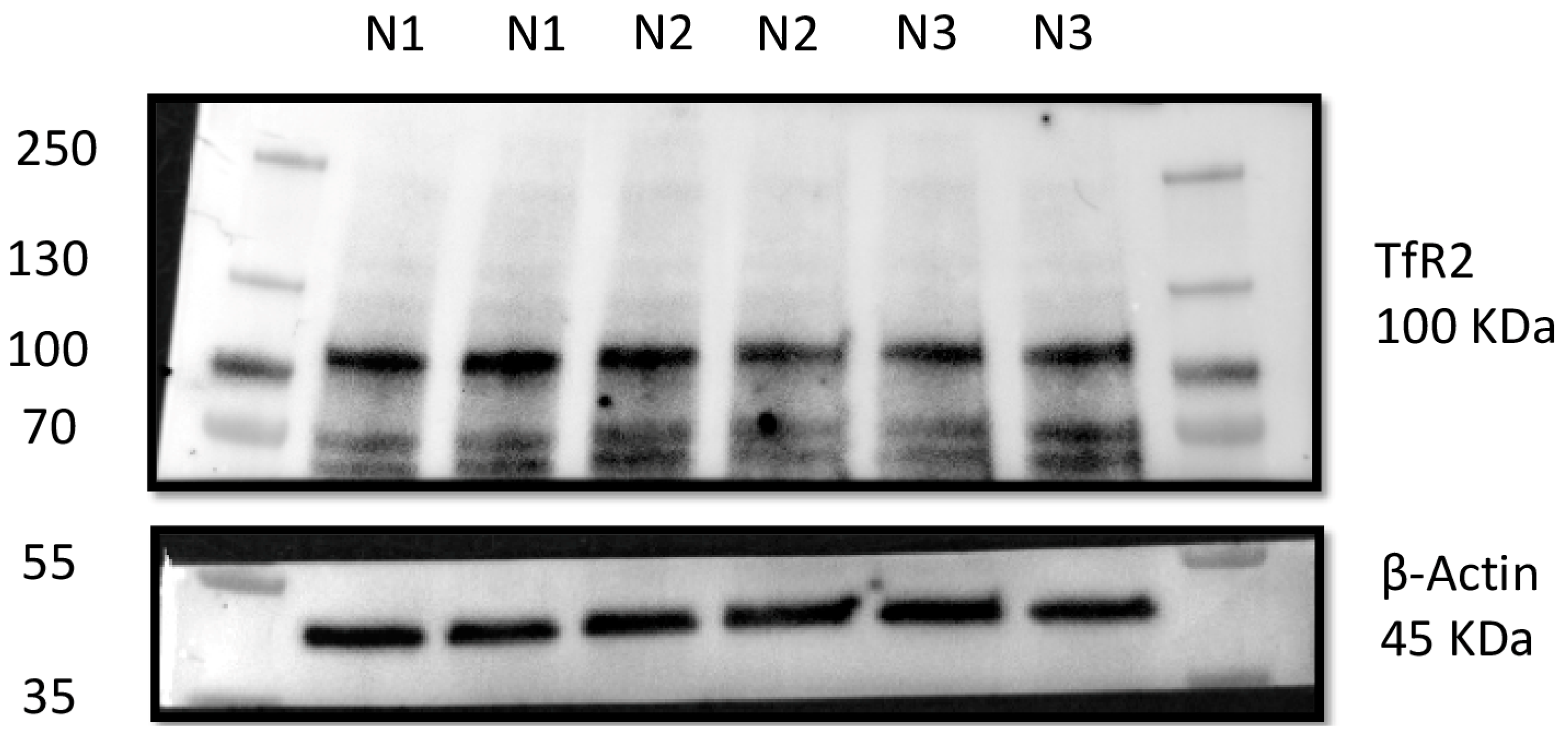
3.3. Western Blot Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Baioni, E.; Scanziani, E.; Vincenti, M.C.; Leschiera, M.; Bozzetta, E.; Pezzolato, M.; Desiato, R.; Bertolini, S.; Maurella, C.; Ru, G. Estimating Canine Cancer Incidence: Findings from a Population-Based Tumour Registry in Northwestern Italy. BMC Vet. Res. 2017, 13, 203. [Google Scholar] [CrossRef] [PubMed]
- Canadas, A.; Romão, P.; Gärtner, F. Multiple Cutaneous Metastasis of a Malignant Leydig Cell Tumour in a Dog. J. Comp. Pathol. 2016, 155, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Grüntzig, K.; Graf, R.; Hässig, M.; Welle, M.; Meier, D.; Lott, G.; Erni, D.; Schenker, N.S.; Guscetti, F.; Boo, G.; et al. The Swiss Canine Cancer Registry: A Retrospective Study on the Occurrence of Tumours in Dogs in Switzerland from 1955 to 2008. J. Comp. Pathol. 2015, 152, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Kudo, T.; Kamiie, J.; Aihara, N.; Doi, M.; Sumi, A.; Omachi, T.; Shirota, K. Malignant Leydig Cell Tumor in Dogs: Two Cases and a Review of the Literature. J. Vet. Diagn. Investig. 2019, 31, 557–561. [Google Scholar] [CrossRef]
- Kennedy, P.C. Histological Classification of Tumors of the Genital System of Domestic Animals. In World Health Organization International Histological Classification of Tumors of Domestic Animals, 2nd ed.; Second Series; Armed Forces Institute of Pathology: Washington, DC, USA, 1998; ISBN 978-1-881041-55-9. [Google Scholar]
- Agnew, D.W.; MacLachlan, N.J. Tumors of the Genital Systems. In Tumors in Domestic Animals; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2016; pp. 689–722. ISBN 978-1-119-18120-0. [Google Scholar]
- Grieco, V.; Riccardi, E.; Greppi, G.F.; Teruzzi, F.; Iermanò, V.; Finazzi, M. Canine Testicular Tumours: A Study on 232 Dogs. J. Comp. Pathol. 2008, 138, 86–89. [Google Scholar] [CrossRef]
- Liao, A.T.; Chu, P.-Y.; Yeh, L.-S.; Lin, C.-T.; Liu, C.-H. A 12-Year Retrospective Study of Canine Testicular Tumors. J. Vet. Med. Sci. 2009, 71, 919–923. [Google Scholar] [CrossRef]
- Manuali, E.; Forte, C.; Porcellato, I.; Brachelente, C.; Sforna, M.; Pavone, S.; Ranciati, S.; Morgante, R.; Crescio, I.M.; Ru, G.; et al. A Five-Year Cohort Study on Testicular Tumors from a Population-Based Canine Cancer Registry in Central Italy (Umbria). Prev. Vet. Med. 2020, 185, 105201. [Google Scholar] [CrossRef]
- Hohšteter, M.; Artuković, B.; Severin, K.; Kurilj, A.G.; Beck, A.; Šoštarić-Zuckermann, I.-C.; Grabarević, Ž. Canine Testicular Tumors: Two Types of Seminomas Can Be Differentiated by Immunohistochemistry. BMC Vet. Res. 2014, 10, 169. [Google Scholar] [CrossRef]
- Lawrence, J.; Saba, C. Tumors of the Male Reproductive System. In Withrow & MacEwen’s Small Animal Clinical Oncology; Elsevier: Amsterdam, The Netherlands, 2013; pp. 557–571. [Google Scholar]
- Ru, G.; Terracini, B.; Glickman, L.T. Host Related Risk Factors for Canine Osteosarcoma. Vet. J. 1998, 156, 31–39. [Google Scholar] [CrossRef]
- Villamil, J.A.; Henry, C.J.; Hahn, A.W.; Bryan, J.N.; Tyler, J.W.; Caldwell, C.W. Hormonal and Sex Impact on the Epidemiology of Canine Lymphoma. J. Cancer Epidemiol. 2009, 2009, 591753. [Google Scholar] [CrossRef]
- Ware, W.A.; Hopper, D.L. Cardiac Tumors in Dogs: 1982–1995. J. Vet. Intern. Med. 1999, 13, 95–103. [Google Scholar] [CrossRef] [PubMed]
- White, C.R.; Hohenhaus, A.E.; Kelsey, J.; Procter-Gray, E. Cutaneous MCTs: Associations with Spay/Neuter Status, Breed, Body Size, and Phylogenetic Cluster. J. Am. Anim. Hosp. Assoc. 2011, 47, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.S.; Carvalho, S.; Cabral, J.; Reis, C.A.; Gärtner, F. Canine Tumors: A Spontaneous Animal Model of Human Carcinogenesis. Transl. Res. 2012, 159, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Ciaputa, R.; Nowak, M.; Madej, J.A.; Janus, I.; Górzyńska, E.; Kandefer-Gola, M. Expression of Selected Markers in Immunohistochemical Diagnosis of Canine and Human Testicular Tumours. Bull. Vet. Inst. Pulawy 2015, 59, 99–106. [Google Scholar] [CrossRef]
- Anderson, G.J.; Frazer, D.M. Current Understanding of Iron Homeostasis. Am. J. Clin. Nutr. 2017, 106, 1559S–1566S. [Google Scholar] [CrossRef]
- Chen, C.; Paw, B.H. Cellular and Mitochondrial Iron Homeostasis in Vertebrates. Biochim. Biophys. Acta BBA-Mol. Cell Res. 2012, 1823, 1459–1467. [Google Scholar] [CrossRef]
- Ru, Q.; Li, Y.; Chen, L.; Wu, Y.; Min, J.; Wang, F. Iron Homeostasis and Ferroptosis in Human Diseases: Mechanisms and Therapeutic Prospects. Signal Transduct. Target. Ther. 2024, 9, 271. [Google Scholar] [CrossRef]
- Papanikolaou, G.; Pantopoulos, K. Iron Metabolism and Toxicity. Toxicol. Appl. Pharmacol. 2005, 202, 199–211. [Google Scholar] [CrossRef]
- Watt, R.K.; Hilton, R.J.; Graff, D.M. Oxido-Reduction Is Not the Only Mechanism Allowing ions to Traverse the Ferritin Protein Shell. Biochim. Biophys. Acta 2010, 1800, 745–759. [Google Scholar] [CrossRef]
- Tsao, C.-W.; Liao, Y.-R.; Chang, T.-C.; Liew, Y.-F.; Liu, C.-Y. Effects of Iron Supplementation on Testicular Function and Spermatogenesis of Iron-Deficient Rats. Nutrients 2022, 14, 2063. [Google Scholar] [CrossRef]
- Yokonishi, T.; McKey, J.; Ide, S.; Capel, B. Sertoli Cell Ablation and Replacement of the Spermatogonial Niche in Mouse. Nat. Commun. 2020, 11, 40. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Sun, Z.; Ji, G.; Hu, H. Emerging Roles of Ferroptosis in Male Reproductive Diseases. Cell Death Discov. 2023, 9, 358. [Google Scholar] [CrossRef] [PubMed]
- Leichtmann-Bardoogo, Y.; Cohen, L.A.; Weiss, A.; Marohn, B.; Schubert, S.; Meinhardt, A.; Meyron-Holtz, E.G. Compartmentalization and Regulation of Iron Metabolism Proteins Protect Male Germ Cells from Iron Overload. Am. J. Physiol.-Endocrinol. Metab. 2012, 302, E1519–E1530. [Google Scholar] [CrossRef] [PubMed]
- De Domenico, I.; McVey Ward, D.; Kaplan, J. Regulation of Iron Acquisition and Storage: Consequences for Iron-Linked Disorders. Nat. Rev. Mol. Cell Biol. 2008, 9, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Hsu, V.W.; Bai, M.; Li, J. Getting Active: Protein Sorting in Endocytic Recycling. Nat. Rev. Mol. Cell Biol. 2012, 13, 323–328. [Google Scholar] [CrossRef]
- Knutson, M.D. Steap Proteins: Implications for Iron and Copper Metabolism. Nutr. Rev. 2007, 65, 335–340. [Google Scholar] [CrossRef]
- Harrison, P.M.; Arosio, P. The Ferritins: Molecular Properties, Iron Storage Function and Cellular Regulation. Biochim. Biophys. Acta 1996, 1275, 161–203. [Google Scholar] [CrossRef]
- Lelièvre, P.; Sancey, L.; Coll, J.-L.; Deniaud, A.; Busser, B. Iron Dysregulation in Human Cancer: Altered Metabolism, Biomarkers for Diagnosis, Prognosis, Monitoring and Rationale for Therapy. Cancers 2020, 12, 3524. [Google Scholar] [CrossRef]
- Leandri, R.; Power, K.; Buonocore, S.; De Vico, G. Preliminary Evidence of the Possible Roles of the Ferritinophagy-Iron Uptake Axis in Canine Testicular Cancer. Animals 2024, 14, 2619. [Google Scholar] [CrossRef]
- De Vico, G.; Martano, M.; Maiolino, P.; Carella, F.; Leonardi, L. Expression of Transferrin Receptor-1 (TFR-1) in Canine Osteosarcomas. Vet. Med. Sci. 2020, 6, 272–276. [Google Scholar] [CrossRef]
- Evaluation of Serum Ferritin as a Tumor Marker for Canine Histiocytic Sarcoma—Friedrichs—2010—Journal of Veterinary Internal Medicine—Wiley Online Library. Available online: https://onlinelibrary.wiley.com/doi/abs/10.1111/j.1939-1676.2010.0543.x (accessed on 26 December 2024).
- Kazmierski, K.J.; Ogilvie, G.K.; Fettman, M.J.; Lana, S.E.; Walton, J.A.; Hansen, R.A.; Richardson, K.L.; Hamar, D.W.; Bedwell, C.L.; Andrews, G.; et al. Serum Zinc, Chromium, and Iron Concentrations in Dogs with Lymphoma and Osteosarcoma. J. Vet. Intern. Med. 2001, 15, 585–588. [Google Scholar] [CrossRef] [PubMed]
- Marques, O.; Canadas, A.; Faria, F.; Oliveira, E.; Amorim, I.; Seixas, F.; Gama, A.; Lobo-da-Cunha, A.; da Silva, B.M.; Porto, G.; et al. Expression of Iron-Related Proteins in Feline and Canine Mammary Gland Reveals Unexpected Accumulation of Iron. Biotech. Histochem. 2017, 92, 584–594. [Google Scholar] [CrossRef] [PubMed]
- Polak, K.Z.; Schaffer, P.; Donaghy, D.; Zenk, M.C.; Olver, C.S. Iron, Hepcidin, and Microcytosis in Canine Hepatocellular Carcinoma. Vet. Clin. Pathol. 2022, 51, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Rosenblum, S.L. Inflammation, Dysregulated Iron Metabolism, and Cardiovascular Disease. Front. Aging 2023, 4, 1124178. [Google Scholar] [CrossRef]
- Steinbicker, A.U.; Muckenthaler, M.U. Out of Balance-Systemic Iron Homeostasis in Iron-Related Disorders. Nutrients 2013, 5, 3034–3061. [Google Scholar] [CrossRef]
- Tian, Y.; Tian, Y.; Yuan, Z.; Zeng, Y.; Wang, S.; Fan, X.; Yang, D.; Yang, M. Iron Metabolism in Aging and Age-Related Diseases. Int. J. Mol. Sci. 2022, 23, 3612. [Google Scholar] [CrossRef]
- Kawabata, H.; Yang, R.; Hirama, T.; Vuong, P.T.; Kawano, S.; Gombart, A.F.; Koeffler, H.P. Molecular Cloning of Transferrin Receptor 2. A New Member of the Transferrin Receptor-Like Family. J. Biol. Chem. 1999, 274, 20826–20832. [Google Scholar] [CrossRef]
- Kawabata, H.; Germain, R.S.; Vuong, P.T.; Nakamaki, T.; Said, J.W.; Koeffler, H.P. Transferrin Receptor 2-Alpha Supports Cell Growth Both in Iron-Chelated Cultured Cells and In Vivo. J. Biol. Chem. 2000, 275, 16618–16625. [Google Scholar] [CrossRef]
- Herbison, C.E.; Thorstensen, K.; Chua, A.C.G.; Graham, R.M.; Leedman, P.; Olynyk, J.K.; Trinder, D. The Role of Transferrin Receptor 1 and 2 in Transferrin-Bound Iron Uptake in Human Hepatoma Cells. Am. J. Physiol. Cell Physiol. 2009, 297, C1567–C1575. [Google Scholar] [CrossRef]
- Fleming, R.E.; Migas, M.C.; Holden, C.C.; Waheed, A.; Britton, R.S.; Tomatsu, S.; Bacon, B.R.; Sly, W.S. Transferrin Receptor 2: Continued Expression in Mouse Liver in The Face of Iron Overload and in Hereditary Hemochromatosis. Proc. Natl. Acad. Sci. USA 2000, 97, 2214–2219. [Google Scholar] [CrossRef]
- Kawabata, H.; Nakamaki, T.; Ikonomi, P.; Smith, R.D.; Germain, R.S.; Koeffler, H.P. Expression of Transferrin Receptor 2 in Normal and Neoplastic Hematopoietic Cells. Blood 2001, 98, 2714–2719. [Google Scholar] [CrossRef]
- Calzolari, A.; Oliviero, I.; Deaglio, S.; Mariani, G.; Biffoni, M.; Sposi, N.M.; Malavasi, F.; Peschle, C.; Testa, U. Transferrin Receptor 2 Is Frequently Expressed in Human Cancer Cell Lines. Blood Cells Mol. Dis. 2007, 39, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Forejtnikovà, H.; Vieillevoye, M.; Zermati, Y.; Lambert, M.; Pellegrino, R.M.; Guihard, S.; Gaudry, M.; Camaschella, C.; Lacombe, C.; Roetto, A.; et al. Transferrin Receptor 2 Is a Component of The Erythropoietin Receptor Complex and Is Required for Efficient Erythropoiesis. Blood 2010, 116, 5357–5367. [Google Scholar] [CrossRef] [PubMed]
- Pagani, A.; Vieillevoye, M.; Nai, A.; Rausa, M.; Ladli, M.; Lacombe, C.; Mayeux, P.; Verdier, F.; Camaschella, C.; Silvestri, L. Regulation of Cell Surface Transferrin Receptor-2 By Iron-Dependent Cleavage and Release of a Soluble Form. Haematologica 2015, 100, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Goswami, T.; Rolfs, A.; Hediger, M.A. Iron Transport: Emerging Roles in Health and Disease. Biochem. Cell Biol. 2002, 80, 679–689. [Google Scholar] [CrossRef]
- Zhang, S.; Xin, W.; Anderson, G.J.; Li, R.; Gao, L.; Chen, S.; Zhao, J.; Liu, S. Double-Edge Sword Roles of Iron in Driving Energy Production Versus Instigating Ferroptosis. Cell Death Dis. 2022, 13, 40. [Google Scholar] [CrossRef]
- Puig, S.; Ramos-Alonso, L.; Romero, A.M.; Martínez-Pastor, M.T. The Elemental Role of Iron in DNA Synthesis and Repair. Metallomics 2017, 9, 1483–1500. [Google Scholar] [CrossRef]
- Kurniawan, A.L.; Lee, Y.-C.; Shih, C.-K.; Hsieh, R.-H.; Chen, S.-H.; Chang, J.-S. Alteration in Iron Efflux Affects Male Sex Hormone Testosterone Biosynthesis in a Diet-Induced Obese Rat Model. Food Funct. 2019, 10, 4113–4123. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, F. Iron Homeostasis and Tumorigenesis: Molecular Mechanisms and Therapeutic Opportunities. Protein Cell 2015, 6, 88–100. [Google Scholar] [CrossRef]
- Daniels, T.R.; Bernabeu, E.; Rodríguez, J.A.; Patel, S.; Kozman, M.; Chiappetta, D.A.; Holler, E.; Ljubimova, J.Y.; Helguera, G.; Penichet, M.L. The Transferrin Receptor and the Targeted Delivery of Therapeutic Agents Against Cancer. Biochim. Biophys. Acta 2012, 1820, 291–317. [Google Scholar] [CrossRef]
- Nagai, K.; Nakahata, S.; Shimosaki, S.; Tamura, T.; Kondo, Y.; Baba, T.; Taki, T.; Taniwaki, M.; Kurosawa, G.; Sudo, Y.; et al. Development of a Complete Human Anti-Human Transferrin Receptor C Antibody as a Novel Marker of Oral Dysplasia and Oral Cancer. Cancer Med. 2014, 3, 1085–1099. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Navarrete, J.M.; Novelle, M.G.; Catalán, V.; Ortega, F.; Moreno, M.; Gomez-Ambrosi, J.; Xifra, G.; Serrano, M.; Guerra, E.; Ricart, W.; et al. Insulin Resistance Modulates Iron-Related Proteins in Adipose Tissue. Diabetes Care 2014, 37, 1092–1100. [Google Scholar] [CrossRef] [PubMed]
- Senyilmaz, D.; Virtue, S.; Xu, X.; Tan, C.Y.; Griffin, J.L.; Miller, A.K.; Vidal-Puig, A.; Teleman, A.A. Regulation of Mitochondrial Morphology and Function by Stearoylation of TFR1. Nature 2015, 525, 124–128. [Google Scholar] [CrossRef]
- Roberts, K.P.; Griswold, M.D. Characterization of Rat Transferrin Receptor cDNA: The Regulation of Transferrin Receptor mRNA in Testes and in Sertoli Cells in Culture. Mol. Endocrinol. 1990, 4, 531–542. [Google Scholar] [CrossRef] [PubMed]
- Sylvester, S.R.; Griswold, M.D. Localization of Transferrin and Transferrin Receptors in Rat Testes. Biol. Reprod. 1984, 31, 195–203. [Google Scholar] [CrossRef]
- Vannelli, B.G.; Orlando, C.; Barni, T.; Natali, A.; Serio, M.; Balboni, G.C. Immunostaining of Transferrin and Transferrin Receptor in Human Seminiferous Tubules. Fertil. Steril. 1986, 45, 536–541. [Google Scholar] [CrossRef]
- Atanasiu, V.; Manolescu, B.; Stoian, I. Hepcidin—Central Regulator of Iron Metabolism. Eur. J. Haematol. 2007, 78, 1–10. [Google Scholar] [CrossRef]
- Kemna, E.H.J.M.; Tjalsma, H.; Willems, H.L.; Swinkels, D.W. Hepcidin: From Discovery to Differential Diagnosis. Haematologica 2008, 93, 90–97. [Google Scholar] [CrossRef]
- Knutson, M.D.; Oukka, M.; Koss, L.M.; Aydemir, F.; Wessling-Resnick, M. Iron Release from Macrophages after Erythrophagocytosis Is Up-Regulated by Ferroportin 1 Overexpression and down-Regulated by Hepcidin. Proc. Natl. Acad. Sci. USA 2005, 102, 1324–1328. [Google Scholar] [CrossRef]
- Ganz, T.; Nemeth, E. Iron Imports. IV. Hepcidin and Regulation of Body Iron Metabolism. Am. J. Physiol. Gastrointest. Liver Physiol. 2006, 290, G199–G203. [Google Scholar] [CrossRef]
- Nemeth, E.; Tuttle, M.S.; Powelson, J.; Vaughn, M.B.; Donovan, A.; Ward, D.M.; Ganz, T.; Kaplan, J. Hepcidin Regulates Cellular Iron Efflux by Binding to Ferroportin and Inducing its Internalization. Science 2004, 306, 2090–2093. [Google Scholar] [CrossRef] [PubMed]
- Silvestri, L.; Nai, A.; Pagani, A.; Camaschella, C. The Extrahepatic Role of TFR2 in Iron Homeostasis. Front. Pharmacol. 2014, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Goswami, T.; Andrews, N.C. Hereditary Hemochromatosis Protein, HFE, Interaction with Transferrin Receptor 2 Suggests a Molecular Mechanism for Mammalian Iron Sensing. J. Biol. Chem. 2006, 281, 28494–28498. [Google Scholar] [CrossRef] [PubMed]
- Rishi, G.; Wallace, D.F.; Subramaniam, V.N. Hepcidin: Regulation of the Master Iron Regulator. Biosci. Rep. 2015, 35, e00192. [Google Scholar] [CrossRef]
- Wang, X.; Shi, Q.; Gong, P.; Zhou, C.; Cao, Y. An Integrated Systematic Analysis and the Clinical Significance of Hepcidin in Common Malignancies of the Male Genitourinary System. Front. Genet. 2022, 13, 771344. [Google Scholar] [CrossRef]
- Ciaputa, R.; Nowak, M.; Kiełbowicz, M.; Antończyk, A.; Błasiak, K.; Madej, J.A. Seminoma, Sertolioma, and Leydigoma in Dogs: Clinical and Morphological Correlations. Bull. Vet. Inst. Pulawy 2012, 56, 361–367. [Google Scholar] [CrossRef]
- Papaioannou, N.; Psalla, D.; Zavlaris, M.; Loukopoulos, P.; Tziris, N.; Vlemmas, I. Immunohistochemical Expression of dogTERT in Canine Testicular Tumours in Relation to PCNA, ki67 and p53 Expression. Vet. Res. Commun. 2009, 33, 905–919. [Google Scholar] [CrossRef]
- Wauben-Penris, P.J.; Veldscholte, J.; van der Ende, A.; van der Donk, H.A. The Release of Iron by Sertoli Cells in Culture. Biol. Reprod. 1988, 38, 1105–1113. [Google Scholar] [CrossRef]
- Sylvester, S.R.; Griswold, M.D. The Testicular Iron Shuttle: A “Nurse” Function of the Sertoli Cells. J. Androl. 1994, 15, 381–385. [Google Scholar] [CrossRef]
- Henley, J.D.; Young, R.H.; Ulbright, T.M. Malignant Sertoli Cell Tumors of the Testis: A Study of 13 Examples of a Neoplasm Frequently Misinterpreted as Seminoma. Am. J. Surg. Pathol. 2002, 26, 541–550. [Google Scholar] [CrossRef]
- Dhaliwal, R.S.; Kitchell, B.E.; Knight, B.L.; Schmidt, B.R. Treatment of Aggressive Testicular Tumors in Four Dogs. J. Am. Anim. Hosp. Assoc. 1999, 35, 311–318. [Google Scholar] [CrossRef] [PubMed]
- McDonald, R.K.; Walker, M.; Legendre, A.M.; van Ee, R.T.; Gompf, R.E. Radiotherapy of Metastatic Seminoma in the Dog. Case Reports. J. Vet. Intern. Med. 1988, 2, 103–107. [Google Scholar] [CrossRef] [PubMed]
- Bryan, J.N.; Keeler, M.R.; Henry, C.J.; Bryan, M.E.; Hahn, A.W.; Caldwell, C.W. A Population Study of Neutering Status as a Risk Factor for Canine Prostate Cancer. Prostate 2007, 67, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Hart, B.L.; Hart, L.A.; Thigpen, A.P.; Willits, N.H. Long-Term Health Effects of Neutering Dogs: Comparison of Labrador Retrievers with Golden Retrievers. PLoS ONE 2014, 9, e102241. [Google Scholar] [CrossRef]
- Torres de la Riva, G.; Hart, B.L.; Farver, T.B.; Oberbauer, A.M.; Messam, L.L.M.; Willits, N.; Hart, L.A. Neutering Dogs: Effects on Joint Disorders and Cancers in Golden Retrievers. PLoS ONE 2013, 8, e55937. [Google Scholar] [CrossRef]
- Fleming, R.E.; Ahmann, J.R.; Migas, M.C.; Waheed, A.; Koeffler, H.P.; Kawabata, H.; Britton, R.S.; Bacon, B.R.; Sly, W.S. Targeted Mutagenesis of the Murine Transferrin Receptor-2 Gene Produces Hemochromatosis. Proc. Natl. Acad. Sci. USA 2002, 99, 10653–10658. [Google Scholar] [CrossRef]
- Boer, H.; Proost, J.H.; Nuver, J.; Bunskoek, S.; Gietema, J.Q.; Geubels, B.M.; Altena, R.; Zwart, N.; Oosting, S.F.; Vonk, J.M.; et al. Long-Term Exposure to Circulating Platinum Is Associated with Late Effects of Treatment in Testicular Cancer Survivors. Ann. Oncol. 2015, 26, 2305–2310. [Google Scholar] [CrossRef]
- Kondagunta, G.V.; Sheinfeld, J.; Mazumdar, M.; Mariani, T.V.; Bajorin, D.; Bacik, J.; Bosl, G.J.; Motzer, R.J. Relapse-Free and Overall Survival in Patients with Pathologic Stage II Nonseminomatous Germ Cell Cancer Treated with Etoposide and Cisplatin Adjuvant Chemotherapy. J. Clin. Oncol. 2004, 22, 464–467. [Google Scholar] [CrossRef]
- Knapp, D.W.; Richardson, R.C.; Bonney, P.L.; Hahn, K. Cisplatin Therapy in 41 Dogs with Malignant Tumors. J. Vet. Intern. Med. 1988, 2, 41–46. [Google Scholar] [CrossRef]
- Liang, Z.; Zhao, W.; Li, X.; Wang, L.; Meng, L.; Yu, R. Cisplatin Synergizes with PRLX93936 to Induce Ferroptosis in Non-Small Cell Lung Cancer Cells. Biochem. Biophys. Res. Commun. 2021, 569, 79–85. [Google Scholar] [CrossRef]
- Guo, J.; Xu, B.; Han, Q.; Zhou, H.; Xia, Y.; Gong, C.; Dai, X.; Li, Z.; Wu, G. Ferroptosis: A Novel Anti-Tumor Action for Cisplatin. Cancer Res. Treat. 2018, 50, 445–460. [Google Scholar] [CrossRef]
- Torti, S.V.; Torti, F.M. Iron and Cancer: More Ore to Be Mined. Nat. Rev. Cancer 2013, 13, 342–355. [Google Scholar] [CrossRef]


| Samples | Breed | Age (ys) | Histological Diagnosis |
|---|---|---|---|
| S1 | Poodle | 5 | SCT |
| S2 | English Setter | 9 | SCT |
| S3 | English Setter | 9 | SCT |
| S4 | Mixed Breed | 6 | SCT |
| S5 | German Sheperd | 7 | SCT |
| S6 | Mixed Breed | 12 | ITSEM |
| S7 | Mixed Breed | 10 | ITSEM |
| S8 | Pit bull | 12 | ITSEM |
| S9 | Mixed Breed | 11 | ITSEM |
| S10 | Mixed Breed | 11 | ITSEM |
| S11 | Mixed Breed | 8 | ITSEM |
| S12 | Beagle | 11 | DSEM |
| S13 | German Sheperd | 8 | DSEM |
| S14 | German Sheperd | 10 | DSEM |
| S15 | Mixed Breed | 6 | DSEM |
| S16 | Mixed Breed | 16 | DSEM |
| S17 | West Highland Terrier | 7 | DSEM |
| S18 | Poodle | 5 | DSEM |
| S19 | English Setter | 9 | DSEM |
| S20 | English Setter | 9 | DSEM |
| S21 | Mixed Breed | 6 | DSEM |
| S22 | German Sheperd | 7 | DSEM |
| S23 | Mixed Breed | 8 | DSEM |
| S24 | Mixed Breed | 9 | LCT |
| S25 | Mixed Breed | 8 | LCT |
| S26 | German Sheperd | 10 | LCT |
| N1 | Poodle | 8 | N.n. testis |
| N2 | German Sheperd | 9 | N.n. testis |
| N3 | English Setter | 7 | N.n. testis |
| Antibody | Manufacturer/Clone | Host Species | Dilution |
|---|---|---|---|
| TfR2 | Antibodies */Polyclonal | Rabbit | 1:100 |
| Antibody | Manufacturer/Clone | Host Species | Dilution |
|---|---|---|---|
| TfR2 | Antibodies */Polyclonal | Rabbit | 1:1000 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leandri, R.; Buonocore, S.; Power, K. Transferrin Receptor 2 in Canine Testicular Tumors: An Emerging Key Role in Seminomas. Animals 2025, 15, 264. https://doi.org/10.3390/ani15020264
Leandri R, Buonocore S, Power K. Transferrin Receptor 2 in Canine Testicular Tumors: An Emerging Key Role in Seminomas. Animals. 2025; 15(2):264. https://doi.org/10.3390/ani15020264
Chicago/Turabian StyleLeandri, Rebecca, Sara Buonocore, and Karen Power. 2025. "Transferrin Receptor 2 in Canine Testicular Tumors: An Emerging Key Role in Seminomas" Animals 15, no. 2: 264. https://doi.org/10.3390/ani15020264
APA StyleLeandri, R., Buonocore, S., & Power, K. (2025). Transferrin Receptor 2 in Canine Testicular Tumors: An Emerging Key Role in Seminomas. Animals, 15(2), 264. https://doi.org/10.3390/ani15020264






