Neuromuscular Mechanisms Underlying Changes in Force Production during an Attentional Focus Task
Abstract
:1. Introduction
2. Materials and Methods
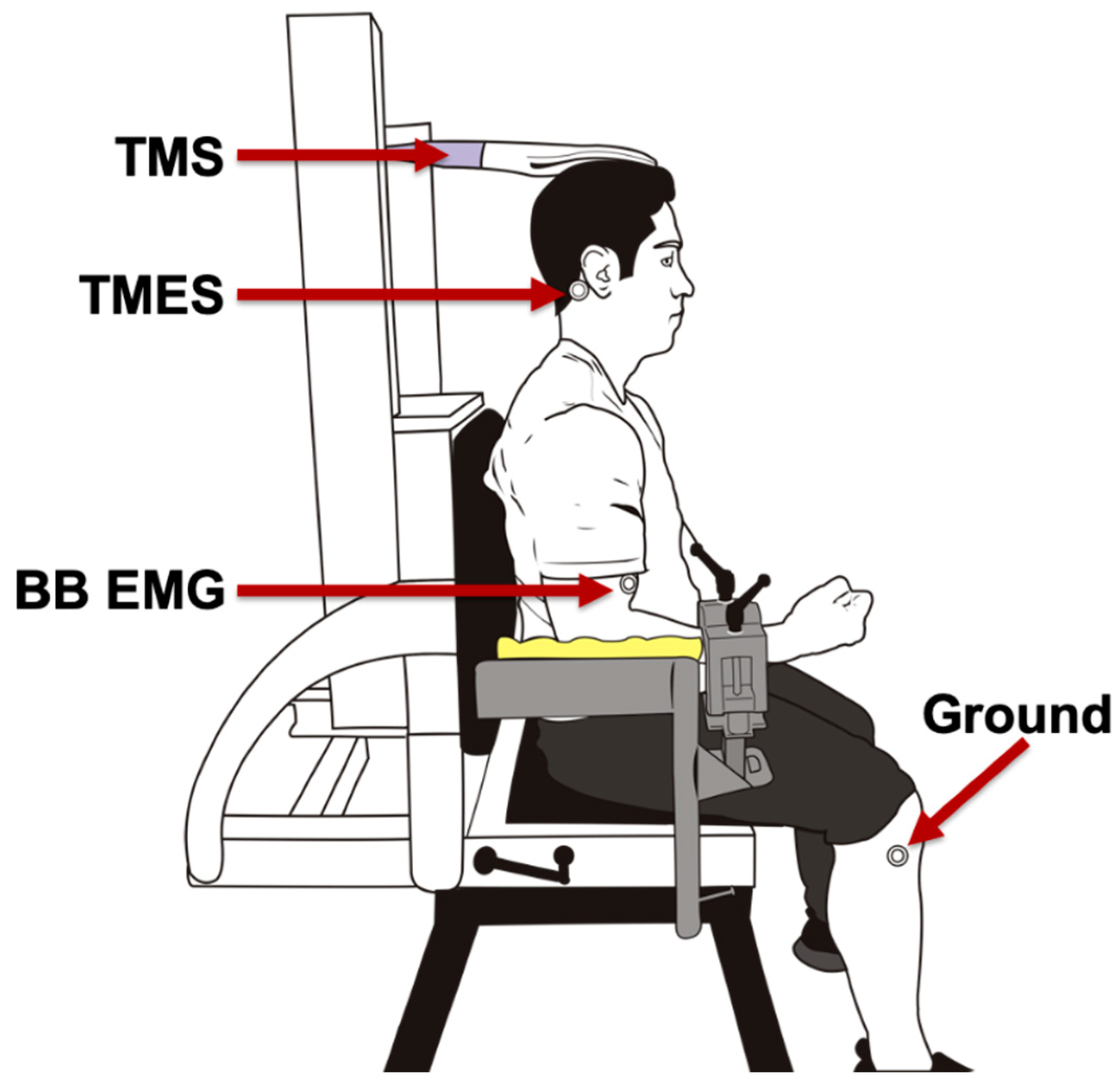
2.1. Elbow Flexor Force
2.2. Electromyography
2.3. Stimulation Conditions
2.3.1. Transcranial Magnetic Stimulation (TMS)
2.3.2. Transmastoid Electrical Stimulation (TMES)
2.3.3. Brachial Plexus Stimulation
2.4. Experimental Protocol
2.4.1. Familiarization Session
2.4.2. Stimulation Session
2.4.3. Non-Stimulation Session
2.5. Data Analysis
2.6. Statistical Analysis
3. Results
3.1. Data Distribution
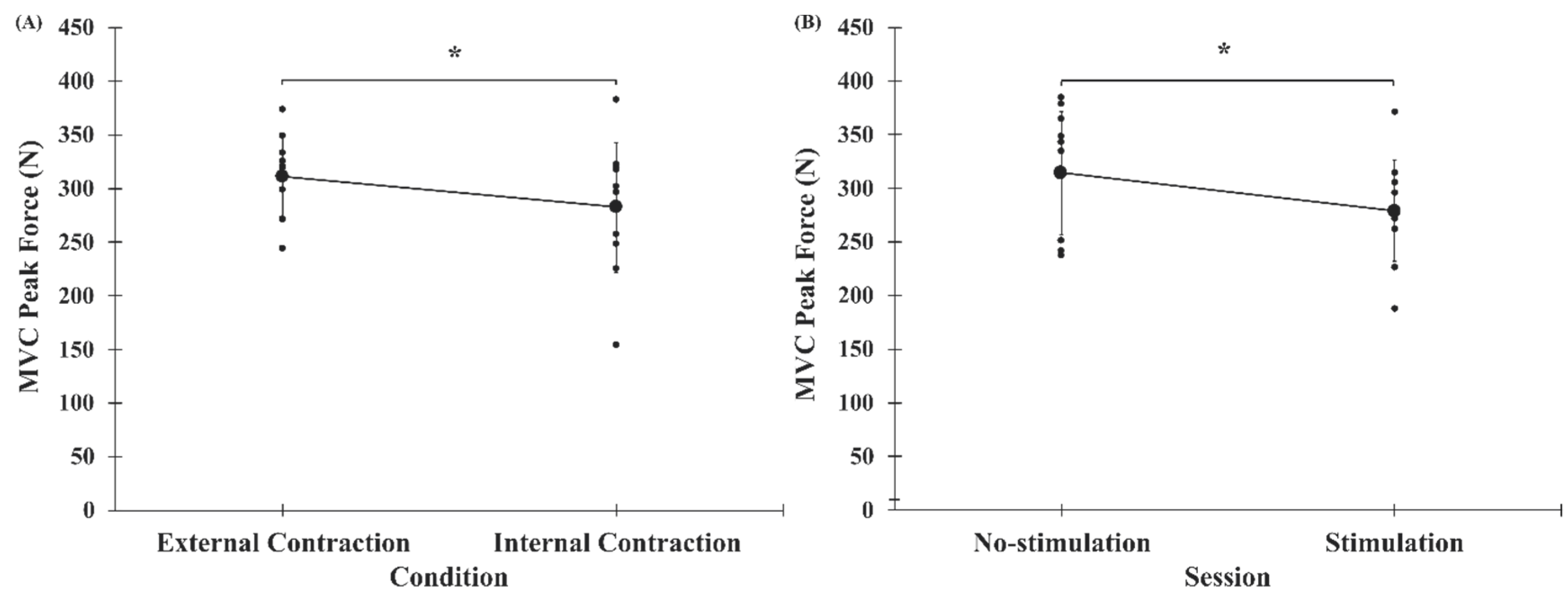
3.2. Peak Force
3.3. Electromyography (EMG)
3.3.1. Biceps Brachii
3.3.2. Triceps Brachii
3.3.3. Co-Activation
3.4. Corticospinal Excitability (CSE)
3.5. Co-Activation/MVC Peak Force
3.6. Relationship between Change in Peak Force and Co-Activation
Correlations
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wulf, G. Attentional focus and motor learning: A review of 15 years. Int. Rev. Sport Exerc. Psychol. 2013, 6, 77–104. [Google Scholar] [CrossRef]
- Lauber, B.; Keller, M. Improving motor performance: Selected aspects of augmented feedback in exercise and health. Eur. J. Sport Sci. 2014, 14, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Porter, J.; Nolan, R.; Ostrowski, E.; Wulf, G. Directing attention externally enhances agility performance: A qualitative and quantitative analysis of the efficacy of using verbal instructions to focus attention. Front. Psychol. 2010, 1, 216–223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wulf, G.; Lewthwaite, R. Optimizing performance through intrinsic motivation and attention for learning: The OPTIMAL theory of motor learning. Psychon. Bull. Rev. 2016, 23, 1382–1414. [Google Scholar] [CrossRef] [PubMed]
- Marchant, D.C.; Greig, M.; Bullough, J.; Hitchen, D. Instructions to adopt an external focus enhance muscular endurance. Res. Q. Exerc. Sport 2011, 82, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Marchant, D.C.; Greig, M.; Scott, C. Attentional focusing instructions influence force production and muscular activity during isokinetic elbow flexions. J. Strength Cond. Res. 2009, 23, 2358–2366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greig, M.; Marchant, D. Speed dependant influence of attentional focusing instructions on force production and muscular activity during isokinetic elbow flexions. Hum. Mov. Sci. 2014, 33, 135–148. [Google Scholar] [CrossRef]
- Rotem-Lehrer, N.; Laufer, Y. Effect of focus of attention on transfer of a postural control task following an ankle sprain. J. Orthop. Sports Phys. Ther. 2007, 37, 564–569. [Google Scholar] [CrossRef] [Green Version]
- Lessa, H.T.; Chiviacowsky, S. Self-controlled practice benefits motor learning in older adults. Hum. Mov. Sci. 2015, 40, 372–380. [Google Scholar] [CrossRef]
- Halperin, I.; Chapman, D.W.; Martin, D.T.; Abbiss, C. The effects of attentional focus instructions on punching velocity and impact forces among trained combat athletes. J. Sports Sci. 2017, 35, 500–507. [Google Scholar] [CrossRef]
- Zentgraf, K.; Lorey, B.; Bischoff, M.; Zimmermann, K.; Stark, R.; Munzert, J. Neural correlates of attentional focusing during finger movements: A fMRI study. J. Mot. Behav. 2009, 41, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Van der Lubbe, R.H.; Bundt, C.; Abrahamse, E.L. Internal and external spatial attention examined with lateralized EEG power spectra. Brain Res. 2014, 1583, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Crews, D.J.; Landers, D.M. Electroencephalographic measures of attentional patterns prior to the golf putt. Med. Sci. Sports Exerc. 1993, 25, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Benedek, M.; Schickel, R.J.; Jauk, E.; Fink, A.; Neubauer, A.C. Alpha power increases in right parietal cortex reflects focused internal attention. Neuropsychologia 2014, 56, 393–400. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ray, W.J.; Cole, H.W. EEG alpha activity reflects attentional demands, and beta activity reflects emotional and cognitive processes. Science 1985, 228, 750–752. [Google Scholar] [CrossRef]
- Zachry, T.; Wulf, G.; Mercer, J.; Bezodis, N. Increased movement accuracy and reduced EMG activity as the result of adopting an external focus of attention. Brain Res. Bull. 2005, 67, 304–309. [Google Scholar] [CrossRef]
- Wulf, G.; Dufek, J.S.; Lozano, L.; Pettigrew, C. Increased jump height and reduced EMG activity with an external focus. Hum. Mov. Sci. 2010, 29, 440–448. [Google Scholar] [CrossRef]
- McNeil, C.J.; Butler, J.E.; Taylor, J.L.; Gandevia, S.C. Testing the excitability of human motoneurons. Front. Hum. Neurosci. 2013, 7, 152–161. [Google Scholar] [CrossRef] [Green Version]
- Rossini, P.M.; Rossi, S. Clinical applications of motor evoked potentials. Electroencephalogr. Clin. Neurophysiol. 1998, 106, 180–194. [Google Scholar] [CrossRef]
- Kobayashi, M.; Pascual-Leone, A. Transcranial magnetic stimulation in neurology. Lancet Neurol. 2003, 2, 145–156. [Google Scholar] [CrossRef]
- Kandel, E.R.; Schwartz, J.H.; Jessell, T.M.; Biochemistry, D.o.; Jessell, M.B.T.; Siegelbaum, S.; Hudspeth, A. Principles of Neural Science, 5th ed.; McGraw Hill Professional: New York, NY, USA, 2012; Volume 4, p. 1760. [Google Scholar]
- Specterman, M.; Bhuiya, A.; Kuppuswamy, A.; Strutton, P.; Catley, M.; Davey, N. The effect of an energy drink containing glucose and caffeine on human corticospinal excitability. Physiol. Behav. 2005, 83, 723–728. [Google Scholar] [CrossRef] [PubMed]
- Fadiga, L.; Buccino, G.; Craighero, L.; Fogassi, L.; Gallese, V.; Pavesi, G. Corticospinal excitability is specifically modulated by motor imagery: A magnetic stimulation study. Neuropsychologia 1998, 37, 147–158. [Google Scholar] [CrossRef]
- Carroll, T.J.; Riek, S.; Carson, R.G. The sites of neural adaptation induced by resistance training in humans. J. Physiol. 2002, 544, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Falvo, M.J.; Sirevaag, E.J.; Rohrbaugh, J.W.; Earhart, G.M. Resistance training induces supraspinal adaptations: Evidence from movement-related cortical potentials. Eur. J. Appl. Physiol. 2010, 109, 923–933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pearcey, G.E.; Power, K.E.; Button, D.C. Differences in supraspinal and spinal excitability during various force outputs of the biceps brachii in chronic-and non-resistance trained individuals. PLoS ONE 2014, 9, e98468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Philpott, D.T.; Pearcey, G.E.; Forman, D.; Power, K.E.; Button, D.C. Chronic resistance training enhances the spinal excitability of the biceps brachii in the non-dominant arm at moderate contraction intensities. Neurosci. Lett. 2015, 585, 12–16. [Google Scholar] [CrossRef] [Green Version]
- Rossi, S.; Hallett, M.; Rossini, P.M.; Pascual-Leone, A.; Safety of TMS Consensus Group. Safety, ethical considerations, and application guidelines for the use of transcranial magnetic stimulation in clinical practice and research. Clin. Neurophysiol. 2009, 120, 2008–2039. [Google Scholar] [CrossRef] [Green Version]
- Lahouti, B.; Lockyer, E.J.; Wiseman, S.; Power, K.E.; Button, D.C. Short-interval intracortical inhibition of the biceps brachii in chronic-resistance versus non-resistance-trained individuals. Exp. Brain Res. 2019, 237, 3023–3032. [Google Scholar] [CrossRef]
- Lockyer, E.J.; Hosel, K.; Nippard, A.P.; Button, D.C.; Power, K.E. Corticospinal-Evoked Responses from the Biceps Brachii during Arm Cycling across Multiple Power Outputs. Brain Sci. 2019, 9, 205. [Google Scholar] [CrossRef] [Green Version]
- Stefanelli, L.; Lockyer, E.J.; Collins, B.W.; Snow, N.J.; Crocker, J.; Kent, C.; Power, K.E.; Button, D.C. Delayed-Onset Muscle Soreness and Topical Analgesic Alter Corticospinal Excitability of the Biceps Brachii. Med. Sci. Sports Exerc. 2019, 51, 2344–2356. [Google Scholar] [CrossRef]
- Taylor, J.L.; Todd, G.; Gandevia, S.C. Evidence for a supraspinal contribution to human muscle fatigue. Clin. Exp. Pharmacol. Physiol. 2006, 33, 400–405. [Google Scholar] [CrossRef]
- Cadigan, E.W.; Collins, B.W.; Philpott, D.T.; Kippenhuck, G.; Brenton, M.; Button, D.C. Maximal voluntary activation of the elbow flexors is under predicted by transcranial magnetic stimulation compared to motor point stimulation prior to and following muscle fatigue. Front. Physiol. 2017, 8, 707–718. [Google Scholar] [CrossRef] [PubMed]
- Levenez, M.; Garland, S.J.; Klass, M.; Duchateau, J. Cortical and spinal modulation of antagonist coactivation during a submaximal fatiguing contraction in humans. J. Neurophysiol. 2008, 99, 554–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calder, K.M.; Hall, L.-A.; Lester, S.M.; Inglis, J.G.; Gabriel, D.A. Reliability of the biceps brachii M-wave. J. Neuroeng. Rehabil. 2005, 2, 33–41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gandevia, S.; Petersen, N.; Butler, J.; Taylor, J. Impaired response of human motoneurones to corticospinal stimulation after voluntary exercise. J. Physiol. 1999, 521, 749–759. [Google Scholar] [CrossRef]
- Gamst, G.; Meyers, L.S.; Guarino, A.J. Analysis of Variance Designs: A Conceptual and Computational Approach with SPSS and SAS; Cambridge University Press: New York, NY, USA, 2008; p. 594. [Google Scholar]
- Halaki, M.; Ginn, K.A. Normalization of EMG Signals: To Normalize or Not to Normalize and What to Normalize to? In Computational Intelligence in Electromyography Analysis: A Perspective on Current Applications and Future Challenges; InTech: Rijeka, Crotia, 2012; pp. 175–194. [Google Scholar]
- Mukaka, M.M. A guide to appropriate use of correlation coefficient in medical research. Malawi Med. J. 2012, 24, 69–71. [Google Scholar] [PubMed]
- Halperin, I.; Williams, K.J.; Martin, D.T.; Chapman, D.W. The effects of attentional focusing instructions on force production during the isometric midthigh pull. J. Strength Cond. Res. 2016, 30, 919–923. [Google Scholar] [CrossRef]
- Ashbridge, E.; Walsh, V.; Cowey, A. Temporal aspects of visual search studied by transcranial magnetic stimulation. Neuropsychologia 1997, 35, 1121–1131. [Google Scholar] [CrossRef]
- Smith, D.T.; Jackson, S.R.; Rorden, C. Repetitive transcranial magnetic stimulation over frontal eye fields disrupts visually cued auditory attention. Brain Stimul. 2009, 2, 81–87. [Google Scholar] [CrossRef] [Green Version]
- Bolognini, N.; Ro, T. Transcranial magnetic stimulation: Disrupting neural activity to alter and assess brain function. J. Neurosci. 2010, 30, 9647–9650. [Google Scholar] [CrossRef] [Green Version]
- Conte, A.; Gilio, F.; Iezzi, E.; Frasca, V.; Inghilleri, M.; Berardelli, A. Attention influences the excitability of cortical motor areas in healthy humans. Exp. Brain Res. 2007, 182, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Button, D.C.; Behm, D.G. The effect of stimulus anticipation on the interpolated twitch technique. J. Sports Sci. Med. 2008, 7, 520–524. [Google Scholar]
- Lohse, K.R.; Sherwood, D.E.; Healy, A.F. Neuromuscular effects of shifting the focus of attention in a simple force production task. J. Mot. Behav. 2011, 43, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Hasegawa, Y.; Kasai, T.; Tsuji, T.; Yahagi, S. Further insight into the task-dependent excitability of motor evoked potentials in first dorsal interosseous muscle in humans. Exp. Brain Res. 2001, 140, 387–396. [Google Scholar] [CrossRef] [PubMed]



| No-Stimulation | Stimulation | |||
|---|---|---|---|---|
| External Cue (Range) | Internal Cue (Range) | External Cue (Range) | Internal Cue (Range) | |
| MVC Peak Force (N) | 333.5 ± 43.7, (242.6–375.5) | 294.8 ± 76.7, (184.2–398.6) | 287.9 ± 38.7, (245.3–376.0) | 270.1 ± 62.4, (124.3–367.1) |
| Biceps Brachii rmsEMG | 0.73 ± 0.51, (0.27–1.88) | 0.60 ± 0.38, (0.16–1.47) | 0.59 ± 0.33, (0.26–1.41) | 0.53 ± 0.25, (0.21–1.04) |
| Triceps Brachii rmsEMG | 0.12 ± 0.03, (0.07–0.18) | 0.15 ± 0.09, (0.05–0.34) | 0.11 ± 0.04, (0.05–0.17) | 0.12 ± 0.04, (0.05–0.17) |
| Co-activation (% Triceps/Biceps rmsEMG) | 22.2 ± 13.8, (8.6–49.6) | 31.5 ± 19.5, (12.1–77.2) | 24.3 ± 15.7, (11.3–63.0) | 26.8 ± 17.5, (12.7–74.4) |
| % Co-activation per Newton Force | 0.06 ± 0.04, (0.03–0.14) | 0.12 ± 0.08, (0.03–0.24) | 0.09 ± 0.06, (0.03–0.23) | 0.11 ± 0.08, (0.03–0.29) |
| 1 (Range) | 2 (Range) | 3 (Range) | 4 (Range) | 5 (Range) | 6 (Range) | ||
|---|---|---|---|---|---|---|---|
| MVC Peak Force (N) | 313.8 ± 45.8, (229.9–373.8) d,e | 304.7 ± 42.1, (224.4–372.2) d,e | 308.1 ± 36.2, (258.9–377.7) d,e | 276.0 ± 34.1, (227.5–336.1) a,b,c | 284.0 ± 30.4, (23.4–320.9) a,b,c | 288.6 ± 34.8, (219.4–335.6) | |
| Biceps Brachii rmsEMG | S | 0.72 ± 0.46, (0.25–1.83) | 0.70 ± 0.46, (0.22–1.76) e | 0.69 ± 0.45, (0.21–1.71) d | 0.64 ± 0.43, (0.21–1.59) c | 0.64 ± 0.45, (0.23–1.67) b | 0.61 ± 0.40, (0.17–1.50) |
| NS | 0.63 ± 0.28, (0.31–1.10) | 0.57 ± 0.31, (0.18–1.27) | 0.58 ± 0.33, (0.20–1.38) | 0.53 ± 0.33, (0.18–1.35) | 0.52 ± 0.26, (0.21–1.14) | 0.53 ± 0.27, (0.20–1.15) | |
| Triceps Brachii rmsEMG (mV·s) | S | 0.14 ± 0.05, (0.07–0.23) | 0.15 ± 0.06, (0.07–0.26) | 0.14 ± 0.06, (0.06–0.25) | 0.14 ± 0.08, (0.06–0.35) | 0.13 ± 0.06 (0.06–0.25) | 0.12 ± 0.05, (0.06–0.20) |
| NS | 0.12 ± 0.04, (0.05–0.16) | 0.11 ± 0.04, (0.05–0.17) | 0.12 ± 0.04, (0.06–0.17) | 0.11 ± 0.04, (0.05–0.18) | 0.12 ± 0.05, (0.05–0.19) | 0.11 ± 0.04, (0.05–0.16) | |
| Co-activation (%Triceps/Biceps rmsEMG) (mV·s) | 23.9 ± 9.8, (12.8–40.5) | 27.0 ± 13.4, (13.1–53.9) | 25.5 ± 12.1, (12.3–48.7) | 27.6 ± 12.6, (12.1–55.8) | 26.8 ± 12.9, (12.5–54.5) | 26.3 ± 14.2, (11.8–55.9) | |
| %Co-activation per Newton Force | 0.08 ± 0.03, (0.03–0.13) | 0.09 ± 0.04, (0.04–0.16) | 0.08 ± 0.04, (0.03–0.15) | 0.10 ± 0.04, (0.04–0.18) | 0.09 ± 0.04, (0.04–0.17) | 0.09 ± 0.05, (0.04–0.18) | |
| External Cue (Range) | Internal Cue (Range) | |
|---|---|---|
| Mmax Amplitude (mV) | 8.62 ± 4.97, (3.0–20.1) | 8.48 ± 5.0, (1.63–17.47) |
| MEP Amplitude (Ratio of Mmax) | 0.83 ± 0.36, (0.48–1.65) | 1.01 ± 0.78, (0.48–2.95) |
| CMEP Amplitude (Ratio of Mmax) | 0.64 ± 0.38, (0.30–1.76) | 0.79 ± 0.71, (0.23–2.20) |
| MEP/CMEP Ratio | 1.64 ± 0.99, (0.92–3.64) | 1.97 ± 1.77, (0.82–6.44) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wiseman, S.; Alizadeh, S.; Halperin, I.; Lahouti, B.; Snow, N.J.; Power, K.E.; Button, D.C. Neuromuscular Mechanisms Underlying Changes in Force Production during an Attentional Focus Task. Brain Sci. 2020, 10, 33. https://doi.org/10.3390/brainsci10010033
Wiseman S, Alizadeh S, Halperin I, Lahouti B, Snow NJ, Power KE, Button DC. Neuromuscular Mechanisms Underlying Changes in Force Production during an Attentional Focus Task. Brain Sciences. 2020; 10(1):33. https://doi.org/10.3390/brainsci10010033
Chicago/Turabian StyleWiseman, Shawn, Shahab Alizadeh, Israel Halperin, Behzad Lahouti, Nicholas J. Snow, Kevin E. Power, and Duane C. Button. 2020. "Neuromuscular Mechanisms Underlying Changes in Force Production during an Attentional Focus Task" Brain Sciences 10, no. 1: 33. https://doi.org/10.3390/brainsci10010033
APA StyleWiseman, S., Alizadeh, S., Halperin, I., Lahouti, B., Snow, N. J., Power, K. E., & Button, D. C. (2020). Neuromuscular Mechanisms Underlying Changes in Force Production during an Attentional Focus Task. Brain Sciences, 10(1), 33. https://doi.org/10.3390/brainsci10010033





