Patient and Therapist In-Session Cortisol as Predictor of Post-Session Patient Reported Affect
Abstract
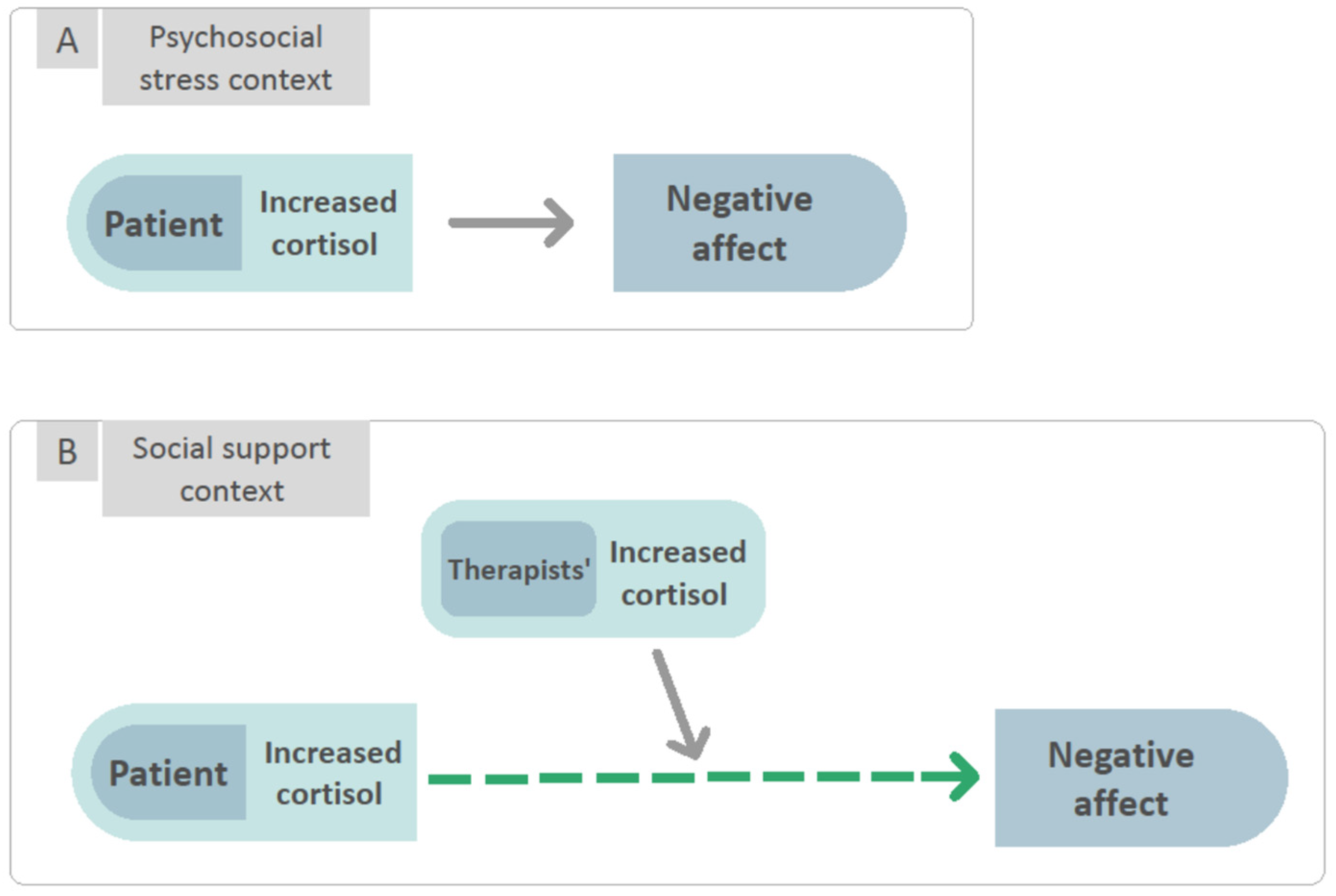
:1. Introduction
2. Materials and Methods
2.1. Participants and Treatments
2.2. Therapists
2.3. Procedure and Measures
2.3.1. Salivary Cortisol
2.3.2. Patient Positive and Negative Affect Experiences
2.4. Data Analysis Strategy
3. Results
3.1. Patients’ Cortisol as a Predictor of NA and PA
3.2. To Examine the Interaction between Patients’ and Therapists’ Cortisol in Predicting NA and PA Reported by the Patients Following Each Session
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Depression and Other Common Mental Disorders: Global Health Estimates; No. WHO/MSD/MER/2017.2; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Dotson, V.M. Variability in Depression: What Have We Been Missing? Am. J. Geriatr. Psychiatry 2017, 25, 23–24. [Google Scholar] [CrossRef]
- NICE. Depression in Adults: Recognition and Management. 2009. Available online: https://www.nice.org.uk/guidance/CG90 (accessed on 11 July 2021).
- Barth, J.; Munder, T.; Gerger, H.; Nüesch, E.; Trelle, S.; Znoj, H.; Jüni, P.; Cuijpers, P. Comparative Efficacy of Seven Psychotherapeutic Interventions for Patients with Depression: A Network Meta-Analysis. Focus 2016, 14, 229–243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leichsenring, F.; Schauenburg, H. Empirically supported methods of short-term psychodynamic therapy in depression–Towards an evidence-based unified protocol. J. Affect. Disord. 2014, 169, 128–143. [Google Scholar] [CrossRef]
- Burum, B.A.; Goldfried, M.R. The centrality of emotion to psychological change. Clin. Psychol. Sci. Pr. 2007, 14, 407–413. [Google Scholar] [CrossRef]
- Pascual-Leone, A.; Yeryomenko, N. The client “experiencing” scale as a predictor of treatment outcomes: A meta-analysis on psychotherapy process. Psychother. Res. 2017, 27, 653–665. [Google Scholar] [CrossRef] [PubMed]
- Husen, K.; Rafaeli, E.; Rubel, J.; Bar-Kalifa, E.; Lutz, W. Daily affect dynamics predict early response in CBT: Feasibility and predictive validity of EMA for outpatient psychotherapy. J. Affect. Disord. 2016, 206, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Boumparis, N.; Karyotaki, E.; Kleiboer, A.; Hofmann, S.G.; Cuijpers, P. The effect of psychotherapeutic interventions on positive and negative affect in depression: A systematic review and meta-analysis. J. Affect. Disord. 2016, 202, 153–162. [Google Scholar] [CrossRef]
- Maust, D.; Cristancho, M.; Gray, L.; Rushing, S.; Tjoa, C.; Thase, M.E. Psychiatric rating scales. Handb. Clin. Neurol. 2012, 106, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Stetler, C.; Miller, G.E. Depression and Hypothalamic-Pituitary-Adrenal Activation: A Quantitative Summary of Four Decades of Research. Psychosom. Med. 2011, 73, 114–126. [Google Scholar] [CrossRef]
- Fischer, S.; Macare, C.; Cleare, A.J. Hypothalamic-pituitary-adrenal (HPA) axis functioning as predictor of antidepressant response–Meta-analysis. Neurosci. Biobehav. Rev. 2017, 83, 200–211. [Google Scholar] [CrossRef] [Green Version]
- Fischer, S.; Strawbridge, R.; Vives, A.R.H.; Cleare, A.J. Cortisol as a predictor of psychological therapy response in depressive disorders: Systematic review and meta-analysis. Br. J. Psychiatry 2017, 210, 105–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laufer, S.; Engel, S.; Knaevelsrud, C.; Schumacher, S. Cortisol and alpha-amylase assessment in psychotherapeutic intervention studies: A systematic review. Neurosci. Biobehav. Rev. 2018, 95, 235–262. [Google Scholar] [CrossRef] [PubMed]
- Fischer, S.; Zilcha-Mano, S. Why Does Psychotherapy Work and for Whom? Hormonal Answers. Under Review.
- Dickerson, S.S.; Kemeny, M.E. Acute Stressors and Cortisol Responses: A Theoretical Integration and Synthesis of Laboratory Research. Psychol. Bull. 2004, 130, 355–391. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrewijn, A.; Vidal-Ribas, P.; Clore-Gronenborn, K.; Jackson, S.M.; Pisano, S.; Pine, D.S.; Stringaris, A. Associations between brain activity and endogenous and exogenous cortisol—A systematic review. Psychoneuroendocrinology 2020, 120, 104775. [Google Scholar] [CrossRef] [PubMed]
- Joseph, N.T.; Jiang, Y.; Zilioli, S. Momentary emotions and salivary cortisol: A systematic review and meta-analysis of ecological momentary assessment studies. Neurosci. Biobehav. Rev. 2021, 125, 365–379. [Google Scholar] [CrossRef]
- Booij, S.H.; Bos, E.H.; de Jonge, P.; Oldehinkel, A.J. The temporal dynamics of cortisol and affective states in depressed and non-depressed individuals. Psychoneuroendocrinology 2016, 69, 16–25. [Google Scholar] [CrossRef]
- Zeiders, K.H.; Hoyt, L.; Adam, E.K. Associations between self-reported discrimination and diurnal cortisol rhythms among young adults: The moderating role of racial–ethnic minority status. Psychoneuroendocrinology 2014, 50, 280–288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, S. Social Relationships and Health. Am. Psychol. 2004, 59, 676–684. [Google Scholar] [CrossRef]
- Engert, V.; Linz, R.; Grant, J.A. Embodied stress: The physiological resonance of psychosocial stress. Psychoneuroendocrinology 2019, 105, 138–146. [Google Scholar] [CrossRef]
- Gonzalez-Liencres, C.; Breidenstein, A.; Wolf, O.T.; Brüne, M. Sex-dependent effects of stress on brain correlates to empathy for pain. Int. J. Psychophysiol. 2016, 105, 47–56. [Google Scholar] [CrossRef]
- Wingenfeld, K.; Duesenberg, M.; Fleischer, J.; Roepke, S.; Dziobek, I.; Otte, C.; Wolf, O.T. Psychosocial stress differentially affects emotional empathy in women with borderline personality disorder and healthy controls. Acta Psychiatr. Scand. 2018, 137, 206–215. [Google Scholar] [CrossRef]
- Wolf, O.T.; Schulte, J.M.; Drimalla, H.; Hamacher-Dang, T.C.; Knoch, D.; Dziobek, I. Enhanced emotional empathy after psychosocial stress in young healthy men. Stress 2015, 18, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Tomova, L.; von Dawans, B.; Heinrichs, M.; Silani, G.; Lamm, C. Is stress affecting our ability to tune into others? Evidence for gender differences in the effects of stress on self-other distinction. Psychoneuroendocrinology 2014, 43, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Buruck, G.; Wendsche, J.; Melzer, M.; Strobel, A.; Dãrfel, D. Acute psychosocial stress and emotion regulation skills modulate empathic reactions to pain in others. Front. Psychol. 2014, 5, 517. [Google Scholar] [CrossRef] [Green Version]
- Doorn, K.A.-V.; Barber, J.P. Systematic Review of In-Session Affect Experience in Cognitive Behavioral Therapy for Depression. Cogn. Ther. Res. 2017, 41, 807–828. [Google Scholar] [CrossRef]
- Zilcha-Mano, S.; Goldstein, P.; Dolev- Amit, T.; Ben David Sela, T.; Barber, J. A randomized controlled trial for identifying the most suitable treatment for depression based on patients’ attachment orientation. J. Consult. Clin. Psych. in press.
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Publishing: Washington, DC, USA, 2013; Volume 21. [Google Scholar]
- Hamilton, M. Development of a Rating Scale for Primary Depressive Illness. Br. J. Soc. Clin. Psychol. 1967, 6, 278–296. [Google Scholar] [CrossRef]
- Sheehan, D.V.; Lecrubier, Y.; Sheehan, K.H.; Amorim, P.; Janavs, J.; Weiller, E.; Hergueta, T.; Baker, R.; Dunbar, G.C. The Mini-International Neuropsychiatric Interview (M.I.N.I.): The development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 1998, 59, 22–33, quiz 34–57. [Google Scholar]
- Zilcha-Mano, S.; Dolev, T.; Leibovich, L.; Barber, J. Identifying the most suitable treatment for depression based on patients’ attachment: Study protocol for a randomized controlled trial of supportive-expressive vs. supportive treatments. BMC Psychiatry 2018, 18, 362. [Google Scholar] [CrossRef] [Green Version]
- Campbell, J.; Ehlert, U. Acute psychosocial stress: Does the emotional stress response correspond with physiological responses? Psychoneuroendocrinology 2012, 37, 1111–1134. [Google Scholar] [CrossRef]
- Hanneman, S.K.; Cox, C.D.; Green, K.E.; Kang, D.-H. Estimating Intra- and Inter-Assay Variability in Salivary Cortisol. Biol. Res. Nurs. 2011, 13, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.; Clark, L.A.; Tellegen, A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Pers. Soc. Psychol. 1988, 54, 1063–1070. [Google Scholar] [CrossRef]
- Raudenbush, S.W.; Bryk, A.S. Hierarchical Linear Models: Applications and Data Analysis Methods; Sage: London, UK, 2002; Volume 1. [Google Scholar]
- Baldwin, S.A.; Imel, Z.E.; Braithwaite, S.R.; Atkins, D.C. Analyzing multiple outcomes in clinical research using multivariate multilevel models. J. Consult. Clin. Psychol. 2014, 82, 920–930. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peluso, P.R.; Freund, R.R. Therapist and client emotional expression and psychotherapy outcomes: A meta-analysis. Psychotherapy 2018, 55, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Carryer, J.R.; Greenberg, L.S. Optimal levels of emotional arousal in experiential therapy of depression. J. Consult. Clin. Psychol. 2010, 78, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Zilcha-Mano, S.; Porat, Y.; Dolev, T.; Shamay-Tsoory, S. Oxytocin as a Neurobiological Marker of Ruptures in the Working Alliance. Psychother. Psychosom. 2018, 87, 126–127. [Google Scholar] [CrossRef] [PubMed]
- Zilcha-Mano, S.; Shamay-Tsoory, S.; Dolev-Amit, T.; Zagoory-Sharon, O.; Feldman, R. Oxytocin as a biomarker of the formation of therapeutic alliance in psychotherapy and counseling psychology. J. Couns. Psychol. 2020, 67, 523–535. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, S.; Gaudlitz, K.; Plag, J.; Miller, R.; Kirschbaum, C.; Fehm, L.; Fydrich, T.; Ströhle, A. Who is stressed? A pilot study of salivary cortisol and alpha-amylase concentrations in agoraphobic patients and their novice therapists undergoing in vivo exposure. Psychoneuroendocrinology 2014, 49, 280–289. [Google Scholar] [CrossRef]
- Schumacher, S.; Miller, R.; Fehm, L.; Kirschbaum, C.; Fydrich, T.; Ströhle, A. Therapists’ and patients’ stress responses during graduated versus flooding in vivo exposure in the treatment of specific phobia: A preliminary observational study. Psychiatry Res. 2015, 230, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Kirschbaum, C.; Hellhammer, D.H. Salivary Cortisol in Psychobiological Research: An Overview. Neuropsychobiology 1989, 22, 150–169. [Google Scholar] [CrossRef]
- Stalder, T.; Kirschbaum, C. Analysis of cortisol in hair—State of the art and future directions. Brain Behav. Immun. 2012, 26, 1019–1029. [Google Scholar] [CrossRef] [PubMed]
- Fischer, S.; Schumacher, S.; Skoluda, N.; Strahler, J. Fingernail cortisol—State of research and future directions. Front. Neuroendocr. 2020, 58, 100855. [Google Scholar] [CrossRef] [PubMed]


| Variable | 1 | 2 | 3 | 4 | M (SD) | Skew | Kurtosis |
|---|---|---|---|---|---|---|---|
| Negative affect | - | 1.54 (0.58) | 1.46 | 1.95 | |||
| Positive affect | −0.29 ** | - | 2.91 (0.93) | 0.15 | −0.46 | ||
| Patients’ cortisol | 0.10 | 0.09 | - | −2.46 (4.95) | −0.82 | 3.26 | |
| Therapists’ cortisol | −0.11 | −0.04 | 0.17 * | - | −0.80 (2.16) | −0.45 | 6.21 |
| Model 1 Outcome: NA | Model 2 Outcome: PA | |||||
|---|---|---|---|---|---|---|
| Fixed Effects | b (SE) | p | Effect Size | b (SE) | p | Effect Size |
| Intercept | 2.12 (0.15) | <0.001 | 2.65 (0.15) | <0.0001 | ||
| Patients’ cortisol | 0.01 (0.01) | 0.10 | 0.09 | 0.00 (0.01) | 0.60 | 0.02 |
| Log time | −0.26 (0.06) | <0.001 | −0.24 | 0.26 (0.09) | 0.004 | 0.14 |
| Model 1 Outcome: NA | Model 2 Outcome: PA | |||||
|---|---|---|---|---|---|---|
| Fixed Effects | b (SE) | p | Effect Size | b (SE) | p | Effect Size |
| Intercept | 1.76 (0.09) | <0.001 | 2.65 (0.15) | <0.0001 | ||
| Patients’ cortisol | 0.01 (0.01) | 0.14 | 0.09 | 0.00 (0.01) | 0.28 | 0.00 |
| Therapists’ cortisol | −0.02 (0.02) | 0.25 | −0.07 | 0.04 (0.03) | 0.18 | 0.07 |
| Patients’*therapists’ cortisol | −0.01 (0.00) | 0.02 | −0.13 | 0.02 (0.01) | 0.026 | 0.14 |
| Log time | 0.03 (0.01) | <0.001 | −0.27 | 0.03 (0.01) | 0.001 | 0.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Levi, E.; Fischer, S.; Fisher, H.; Admon, R.; Zilcha-Mano, S. Patient and Therapist In-Session Cortisol as Predictor of Post-Session Patient Reported Affect. Brain Sci. 2021, 11, 1483. https://doi.org/10.3390/brainsci11111483
Levi E, Fischer S, Fisher H, Admon R, Zilcha-Mano S. Patient and Therapist In-Session Cortisol as Predictor of Post-Session Patient Reported Affect. Brain Sciences. 2021; 11(11):1483. https://doi.org/10.3390/brainsci11111483
Chicago/Turabian StyleLevi, Eyal, Susanne Fischer, Hadar Fisher, Roee Admon, and Sigal Zilcha-Mano. 2021. "Patient and Therapist In-Session Cortisol as Predictor of Post-Session Patient Reported Affect" Brain Sciences 11, no. 11: 1483. https://doi.org/10.3390/brainsci11111483
APA StyleLevi, E., Fischer, S., Fisher, H., Admon, R., & Zilcha-Mano, S. (2021). Patient and Therapist In-Session Cortisol as Predictor of Post-Session Patient Reported Affect. Brain Sciences, 11(11), 1483. https://doi.org/10.3390/brainsci11111483






