Personalization of Repetitive Transcranial Magnetic Stimulation for the Treatment of Chronic Subjective Tinnitus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Study Procedure
2.3. Repetitive Transcranial Magnetic Stimulation (rTMS)
2.4. Questionnaires and Outcome Measures
2.5. Statistical Analysis
3. Results
3.1. Sample Characteristics and rTMS Side Effects
3.2. rTMS Personalization
3.3. Treatment Results
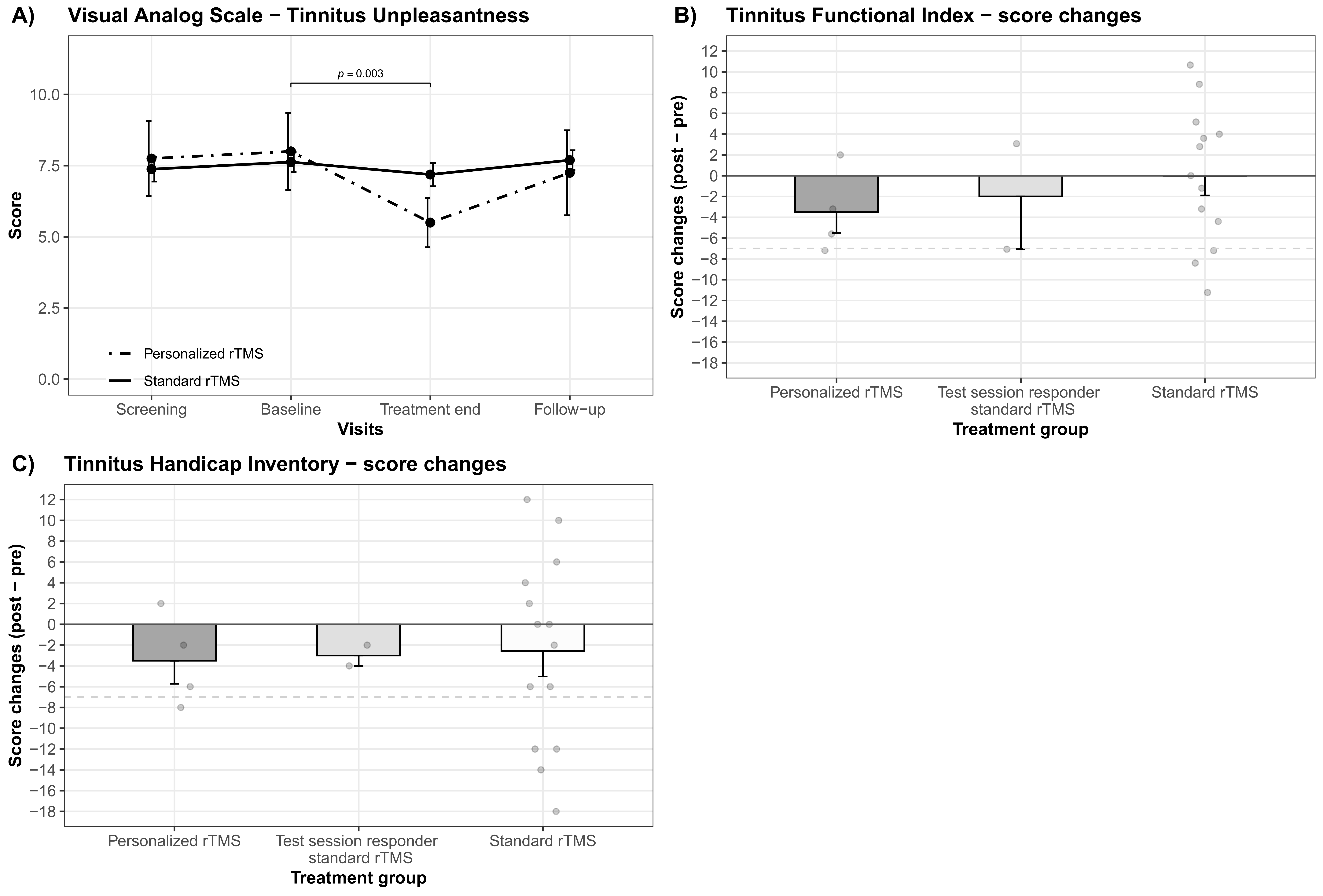
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Model | R2 (Marginal) | R2 (Conditional) | df | AIC | BIC | logLIK | LRT | p |
|---|---|---|---|---|---|---|---|---|
| TFI | ||||||||
| Intercept only: response ~1 + (1|id) | 0 | 0.78 | 579.83 | 586.74 | −286.91 | |||
| Fitted model: response ~ time + test session responder + treatment protocol + test session responder × treatment protocol + (1|id) | 0.13 | 0.82 | 5 | 574.42 | 592.85 | −279.21 | 15.41 | 0.009 |
| THI | ||||||||
| Intercept only: response ~1 + (1|id) | 0 | 0.79 | 600.27 | 607.30 | −297.13 | |||
| Fitted model: response ~ time + test session responder + treatment protocol + test session responder × treatment protocol + (1|id) | 0.14 | 0.82 | 5 | 597.08 | 615.83 | −290.54 | 13.19 | 0.022 |
| WHOQOL-BREF domain 1 | ||||||||
| Intercept only: response ~ 1 + (1|id) | 0 | 0.87 | 240.88 | 247.92 | −117.44 | |||
| Fitted model: response ~ time + test session responder + treatment protocol + test session responder × treatment protocol + (1|id) | 0.14 | 0.88 | 5 | 240.26 | 259.01 | 112.13 | 10.62 | 0.059 |
| WHOQOL-BREF domain 2 | ||||||||
| Intercept only: response ~1 + (1|id) | 0 | 0.83 | 265.71 | 272.74 | −129.85 | |||
| Fitted model: response ~ time + test session responder + treatment protocol + time × treatment protocol + test session responder × treatment protocol + (1|id) | 0.10 | 0.86 | 8 | 265.86 | 291.64 | −121.93 | 15.86 | 0.045 |
| WHOQOL-BREF domain 3 | ||||||||
| Intercept only: response ~1 + (1|id) | 0 | 0.57 | 5 | 342.61 | 349.64 | −168.30 | ||
| Fitted model: response ~ time + test session responder + treatment protocol + test session responder × treatment protocol + (1|id) | 0.20 | 0.65 | 334.56 | 353.31 | −159.28 | 18.05 | 0.003 | |
| VAS tinnitus unpleasantness | ||||||||
| Intercept only: response ~1 + (1|id) | 0 | 0.58 | 281.04 | 288.07 | −137.52 | |||
| Fitted model: response ~ time + test session responder + treatment protocol + time × treatment protocol + test session responder × treatment protocol + (1|id) | 0.08 | 0.67 | 8 | 282.08 | 307.86 | −130.04 | 14.96 | 0.059 |
| numDF | denDF | F | p | |
|---|---|---|---|---|
| TFI | ||||
| Time | 3 | 54.31 | 5.01 | 0.004 |
| THI | ||||
| Time | 3 | 57.01 | 3.85 | 0.014 |
| WHOQOL-BREF domain 1 | ||||
| Time | 3 | 57.11 | 2.78 | 0.049 |
| WHOQOL-BREF domain 2 | ||||
| Time × treatment protocol | 3 | 57.05 | 3.89 | 0.013 |
| WHOQOL-BREF domain 3 | ||||
| Time | 3 | 57.35 | 5.20 | 0.003 |
| VAS tinnitus unpleasantness | ||||
| Time | 3 | 57.01 | 5.29 | 0.003 |
| Time × treatment protocol | 3 | 57.01 | 3.00 | 0.038 |
References
- Eichhammer, P.; Langguth, B.; Marienhagen, J.; Kleinjung, T.; Hajak, G. Neuronavigated repetitive transcranial magnetic stimulation in patients with tinnitus: A short case series. Biol. Psychiatry 2003, 54, 862–865. [Google Scholar] [CrossRef]
- Langguth, B.; Eichhammer, P.; Wiegand, R.; Marienhegen, J.; Maenner, P.; Jacob, P.; Hajak, G. Neuronavigated rTMS in a patient with chronic tinnitus. Effects of 4 weeks treatment. Neuroreport 2003, 14, 977–980. [Google Scholar] [CrossRef] [PubMed]
- Landgrebe, M.; Hajak, G.; Wolf, S.; Padberg, F.; Klupp, P.; Fallgatter, A.J.; Polak, T.; Höppner, J.; Haker, R.; Cordes, J.; et al. 1-Hz rTMS in the treatment of tinnitus: A sham-controlled, randomized multicenter trial. Brain Stimul. 2017, 10, 1112–1120. [Google Scholar] [CrossRef] [PubMed]
- Folmer, R.L.; Theodoroff, S.M.; Casiana, L.; Shi, Y.; Griest, S.; Vachhani, J. Repetitive Transcranial Magnetic Stimulation Treatment for Chronic Tinnitus: A Randomized Clinical Trial. JAMA Otolaryngol. Head Neck Surg. 2015, 141, 716–722. [Google Scholar] [CrossRef] [Green Version]
- Schoisswohl, S.; Agrawal, K.; Simoes, J.; Neff, P.; Schlee, W.; Langguth, B.; Schecklmann, M. RTMS parameters in tinnitus trials: A systematic review. Sci. Rep. 2019, 9, 12190. [Google Scholar] [CrossRef]
- Vanneste, S.; De Ridder, D. Differences between a single session and repeated sessions of 1 Hz TMS by double-cone coil prefrontal stimulation for the improvement of tinnitus. Brain Stimul. 2013, 6, 155–159. [Google Scholar] [CrossRef]
- Meeus, O.; Blaivie, C.; Ost, J.; De Ridder, D.; Van de Heyning, P. Influence of Tonic and Burst Transcranial Magnetic Stimulation Characteristics on Acute Inhibition of Subjective Tinnitus. Otol. Neurotol. 2009, 30, 697–703. [Google Scholar] [CrossRef]
- Lorenz, I.; Müller, N.; Schlee, W.; Langguth, B.; Weisz, N. Short-Term Effects of Single Repetitive TMS Sessions on Auditory Evoked Activity in Patients With Chronic Tinnitus. J. Neurophysiol. 2010, 104, 1497–1505. [Google Scholar] [CrossRef] [Green Version]
- Schoisswohl, S.; Langguth, B.; Schecklmann, M. Short-Term Tinnitus Suppression With Electric-Field Guided rTMS for Individualizing rTMS Treatment: A Technical Feasibility Report. Front. Neurol. 2020, 11, 86. [Google Scholar] [CrossRef]
- Yin, L.; Chen, X.; Lu, X.; An, Y.; Zhang, T.; Yan, J. An updated meta-analysis: Repetitive transcranial magnetic stimulation for treating tinnitus. J. Int. Med. Res. 2021, 49, 300060521999549. [Google Scholar] [CrossRef]
- Lefebvre-Demers, M.; Doyon, N.; Fecteau, S. Non-invasive neuromodulation for tinnitus: A meta-analysis and modeling studies. Brain Stimul. 2021, 14, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-J.; Zeng, B.-S.; Wu, C.-N.; Stubbs, B.; Carvalho, A.F.; Brunoni, A.R.; Su, K.-P.; Tu, Y.-K.; Wu, Y.-C.; Chen, T.-Y.; et al. Association of Central Noninvasive Brain Stimulation Interventions With Efficacy and Safety in Tinnitus Management: A Meta-analysis. JAMA Otolaryngol. Head Neck Surg. 2020, 146, 801–809. [Google Scholar] [CrossRef]
- Langguth, B. Non-Invasive Neuromodulation for Tinnitus. J. Audiol. Otol. 2020, 24, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Cima, R.F.F.; Mazurek, B.; Haider, H.; Kikidis, D.; Lapira, A.; Noreña, A.; Hoare, D.J. A multidisciplinary European guideline for tinnitus: Diagnostics, assessment, and treatment. HNO 2019, 67, 10–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lefaucheur, J.-P.; Aleman, A.; Baeken, C.; Benninger, D.H.; Brunelin, J.; Di Lazzaro, V.; Filipović, S.R.; Grefkes, C.; Hasan, A.; Hummel, F.C.; et al. Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018). Clin. Neurophysiol. 2020, 131, 474–528. [Google Scholar] [CrossRef]
- Khedr, E.M.; Abo-Elfetoh, N.; Rothwell, J.C.; El-Atar, A.; Sayed, E.; Khalifa, H. Contralateral versus ipsilateral rTMS of temporoparietal cortex for the treatment of chronic unilateral tinnitus: Comparative study. Eur. J. Neurol. 2010, 17, 976–983. [Google Scholar] [CrossRef]
- Khedr, E.M.; Rothwell, J.C.; Ahmed, M.A.; El-Atar, A. Effect of daily repetitive transcranial magnetic stimulation for treatment of tinnitus: Comparison of different stimulus frequencies. J. Neurol. Neurosurg. Psychiatry 2008, 79, 212–215. [Google Scholar] [CrossRef]
- Schecklmann, M.; Giani, A.; Tupak, S.; Langguth, B.; Raab, V.; Polak, T.; Varallyay, C.; Grossmann, W.; Herrmann, M.J.; Fallgatter, A.J. Neuronavigated left temporal continuous theta burst stimulation in chronic tinnitus. Restor. Neurol. Neurosci. 2016, 34, 165–175. [Google Scholar] [CrossRef]
- De Ridder, D.; Song, J.-J.; Vanneste, S. Frontal cortex TMS for tinnitus. Brain Stimul. 2013, 6, 355–362. [Google Scholar] [CrossRef]
- Kreuzer, P.M.; Landgrebe, M.; Schecklmann, M.; Poeppl, T.B.; Vielsmeier, V.; Hajak, G.; Kleinjung, T.; Langguth, B. Can Temporal Repetitive Transcranial Magnetic Stimulation be Enhanced by Targeting Affective Components of Tinnitus with Frontal rTMS? A Randomized Controlled Pilot Trial. Front. Syst. Neurosci. 2011, 5, 88. [Google Scholar] [CrossRef] [Green Version]
- Lehner, A.; Schecklmann, M.; Greenlee, M.W.; Rupprecht, R.; Langguth, B. Triple-site rTMS for the treatment of chronic tinnitus: A randomized controlled trial. Sci. Rep. 2016, 6, 22302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noh, T.-S.; Kyong, J.S.; Chang, M.Y.; Park, M.-K.; Lee, J.-H.; Oh, S.-H.; Kim, J.S.; Chung, C.K.; Suh, M.-W. Comparison of Treatment Outcomes Following Either Prefrontal Cortical-only or Dual-site Repetitive Transcranial Magnetic Stimulation in Chronic Tinnitus Patients: A Double-blind Randomized Study. Otol. Neurotol. 2017, 38, 296–303. [Google Scholar] [PubMed]
- De Ridder, D.; Adhia, D.; Langguth, B. Tinnitus and Brain Stimulation. Curr. Top. Behav. Neurosci. 2021, 51, 249–293. [Google Scholar] [PubMed]
- Guerra, A.; López-Alonso, V.; Cheeran, B.; Suppa, A. Variability in non-invasive brain stimulation studies: Reasons and results. Neurosci. Lett. 2017, 719, 133330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polanía, R.; Nitsche, M.A.; Ruff, C.C. Studying and modifying brain function with non-invasive brain stimulation. Nat. Neurosci. 2018, 21, 174–187. [Google Scholar] [CrossRef]
- Hesse, G. Evidence and evidence gaps in tinnitus therapy. GMS Curr. Top. Otorhinolaryngol. Head Neck Surg. 2016, 15, Doc04. [Google Scholar]
- Kleinjung, T.; Langguth, B. Avenue for Future Tinnitus Treatments. Otolaryngol. Clin. N. Am. 2020, 53, 667–683. [Google Scholar] [CrossRef]
- Cederroth, C.R.; Gallus, S.; Hall, D.A.; Kleinjung, T.; Langguth, B.; Maruotti, A.; Meyer, M.; Norena, A.; Probst, T.; Pryss, R.; et al. Editorial: Towards an Understanding of Tinnitus Heterogeneity. Front. Aging Neurosci. 2019, 11, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tyler, R.S.; Oleson, J.; Noble, W.; Coelho, C.; Ji, H. Clinical trials for tinnitus: Study populations, designs, measurement variables, and data analysis. Prog. Brain Res. 2007, 166, 499–509. [Google Scholar]
- Schoisswohl, S.; Langguth, B.; Hebel, T.; Abdelnaim, M.A.; Volberg, G.; Schecklmann, M. Heading for Personalized rTMS in Tinnitus: Reliability of Individualized Stimulation Protocols in Behavioral and Electrophysiological Responses. J. Pers. Med. 2021, 11, 536. [Google Scholar] [CrossRef]
- Kreuzer, P.M.; Poeppl, T.B.; Rupprecht, R.; Vielsmeier, V.; Lehner, A.; Langguth, B.; Schecklmann, M. Individualized Repetitive Transcranial Magnetic Stimulation Treatment in Chronic Tinnitus? Front. Neurol. 2017, 8, 126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Genitsaridi, E.; Partyka, M.; Gallus, S.; Lopez-Escamez, J.A.; Schecklmann, M.; Mielczarek, M.; Trpchevska, N.; Santacruz, J.L.; Schoisswohl, S.; Riha, C.; et al. Standardised profiling for tinnitus research: The European School for Interdisciplinary Tinnitus Research Screening Questionnaire (ESIT-SQ). Hear. Res. 2019, 377, 353–359. [Google Scholar] [CrossRef]
- Awiszus, F. Chapter 2 TMS and threshold hunting. In Supplements to Clinical Neurophysiology; Transcranial Magnetic Stimulation and Transcranial Direct Current Stimulation; Paulus, W., Tergau, F., Nitsche, M.A., Rothwell, J.G., Ziemann, U., Hallett, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; Volume 56, pp. 13–23. [Google Scholar]
- Chen, R.; Classen, J.; Gerloff, C.; Celnik, P.; Wassermann, E.M.; Hallett, M.; Cohen, L.G. Depression of motor cortex excitability by low-frequency transcranial magnetic stimulation. Neurology 1997, 48, 1398–1403. [Google Scholar] [CrossRef] [PubMed]
- Kumru, H.; Albu, S.; Rothwell, J.; Leon, D.; Flores, C.; Opisso, E.; Tormos, J.M.; Valls-Sole, J. Modulation of motor cortex excitability by paired peripheral and transcranial magnetic stimulation. Clin. Neurophysiol. 2017, 128, 2043–2047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langguth, B.; Goodey, R.; Azevedo, A.; Bjorne, A.; Cacace, A.; Crocetti, A.; Del Bo, L.; De Ridder, D.; Diges, I.; Elbert, T.; et al. Consensus for tinnitus patient assessment and treatment outcome measurement: Tinnitus Research Initiative meeting, Regensburg, July 2006. Prog. Brain Res. 2007, 166, 525–536. [Google Scholar]
- Meikle, M.B.; Henry, J.A.; Griest, S.E.; Stewart, B.J.; Abrams, H.B.; McArdle, R.; Myers, P.J.; Newman, C.W.; Sandridge, S.; Turk, D.C.; et al. The Tinnitus Functional Index: Development of a New Clinical Measure for Chronic, Intrusive Tinnitus. Ear Hear. 2012, 33, 153–176. [Google Scholar] [CrossRef]
- Kleinjung, T.; Fischer, B.; Langguth, B.; Sand, P.G.; Hajak, G.; Dvorakova, J.; Eichhammer, P. Validierung einer deutschsprachigen Version des “Tinnitus Handicap Inventory”. Psychiatr. Prax. 2007, 34, S140–S142. [Google Scholar] [CrossRef]
- Newman, C.W.; Jacobson, G.P.; Spitzer, J.B. Development of the Tinnitus Handicap Inventory. Arch. Otolaryngol. Head Neck Surg. 1996, 122, 143–148. [Google Scholar] [CrossRef]
- Hiller, W.; Goebel, G. Rapid assessment of tinnitus-related psychological distress using the Mini-TQ. Int. J. Audiol. 2004, 43, 600–604. [Google Scholar] [CrossRef]
- Bech, P.; Rasmussen, N.A.; Olsen, L.R.; Noerholm, V.; Abildgaard, W. The sensitivity and specificity of the Major Depression Inventory, using the Present State Examination as the index of diagnostic validity. J. Affect. Disord. 2001, 66, 159–164. [Google Scholar] [CrossRef]
- Harper, A.; Power, M.; WHOQOL Group, X. Development of the World Health Organization WHOQOL-Bref quality of life assessment. Psychol. Med. 1998, 28, 551–558. [Google Scholar]
- Adamchic, I.; Tass, P.A.; Langguth, B.; Hauptmann, C.; Koller, M.; Schecklmann, M.; Zeman, F.; Landgrebe, M. Linking the Tinnitus Questionnaire and the subjective Clinical Global Impression: Which differences are clinically important? Health Qual. Life Outcomes 2012, 10, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrison, X.A.; Donaldson, L.; Correa-Cano, M.E.; Evans, J.; Fisher, D.N.; Goodwin, C.E.D.; Robinson, B.S.; Hodgson, D.J.; Inger, R. A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ 2018, 6, e4794. [Google Scholar] [CrossRef] [Green Version]
- Nakagawa, S.; Johnson, P.C.D.; Schielzeth, H. The coefficient of determination R2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J. R. Soc. Interface 2017, 14, 20170213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeman, F.; Koller, M.; Figueiredo, R.; Aazevedo, A.; Rates, M.; Coelho, C.; Kleinjung, T.; De Ridder, D.; Langguth, B.; Landgrebe, M. Tinnitus handicap inventory for evaluating treatment effects: Which changes are clinically relevant? Otolaryngol. Head Neck Surg. 2011, 145, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Goebel, G.; Hiller, W. The tinnitus questionnaire. A standard instrument for grading the degree of tinnitus. Results of a multicenter study with the tinnitus questionnaire. HNO 1994, 42, 166–172. [Google Scholar]
- Adams, M.E.; Huang, T.C.; Nagarajan, S.; Cheung, S.W. Tinnitus Neuroimaging. Otolaryngol. Clin. N. Am. 2020, 53, 583–603. [Google Scholar] [CrossRef]
- Elgoyhen, A.B.; Langguth, B.; De Ridder, D.; Vanneste, S. Tinnitus: Perspectives from human neuroimaging. Nat. Rev. Neurosci. 2015, 16, 632–642. [Google Scholar] [CrossRef]
- Schlee, W.; Mueller, N.; Hartmann, T.; Keil, J.; Lorenz, I.; Weisz, N. Mapping cortical hubs in tinnitus. BMC Biol. 2009, 7, 80. [Google Scholar] [CrossRef] [Green Version]
- Vanneste, S.; De Ridder, D. The auditory and non-auditory brain areas involved in tinnitus. An emergent property of multiple parallel overlapping subnetworks. Front. Syst. Neurosci. 2012, 6, 31. [Google Scholar] [CrossRef] [Green Version]
- Ciminelli, P.; Machado, S.; Palmeira, M.; Coutinho, E.S.F.; Sender, D.; Nardi, A.E. Dorsomedial Prefrontal Cortex Repetitive Transcranial Magnetic Stimulation for Tinnitus: Promising Results of a Blinded, Randomized, Sham-Controlled Study. Ear Hear. 2020, 42, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Langguth, B.; Landgrebe, M.; Frank, E.; Schecklmann, M.; Sand, P.G.; Vielsmeier, V.; Hajak, G.; Kleinjung, T. Efficacy of different protocols of transcranial magnetic stimulation for the treatment of tinnitus: Pooled analysis of two randomized controlled studies. World J. Biol. Psychiatry 2014, 15, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Lehner, A.; Schecklmann, M.; Poeppl, T.B.; Kreuzer, P.M.; Vielsmeier, V.; Rupprecht, R.; Landgrebe, M.; Langguth, B. Multisite rTMS for the treatment of chronic tinnitus: Stimulation of the cortical tinnitus network--a pilot study. Brain Topogr. 2013, 26, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Noh, T.-S.; Kyong, J.-S.; Park, M.K.; Lee, J.H.; Oh, S.H.; Suh, M.-W. Dual-site rTMS is More Effective than Single-site rTMS in Tinnitus Patients: A Blinded Randomized Controlled Trial. Brain Topogr. 2020, 33, 767–775. [Google Scholar] [CrossRef]
- Boecking, B.; Brueggemann, P.; Kleinjung, T.; Mazurek, B. All for One and One for All?—Examining Convergent Validity and Responsiveness of the German Versions of the Tinnitus Questionnaire (TQ), Tinnitus Handicap Inventory (THI), and Tinnitus Functional Index (TFI). Front. Psychol. 2021, 12, 630. [Google Scholar] [CrossRef]
- Fackrell, K.; Hall, D.A.; Barry, J.G.; Hoare, D.J. Psychometric properties of the Tinnitus Functional Index (TFI): Assessment in a UK research volunteer population. Hear. Res. 2016, 335, 220–235. [Google Scholar] [CrossRef]
- Kreuzer, P.M.; Poeppl, T.B.; Rupprecht, R.; Vielsmeier, V.; Lehner, A.; Langguth, B.; Schecklmann, M. Daily high-frequency transcranial random noise stimulation of bilateral temporal cortex in chronic tinnitus—A pilot study. Sci. Rep. 2019, 9, 12274. [Google Scholar] [CrossRef] [Green Version]
- Kreuzer, P.M.; Poeppl, T.B.; Bulla, J.; Schlee, W.; Lehner, A.; Langguth, B.; Schecklmann, M. A proof-of-concept study on the combination of repetitive transcranial magnetic stimulation and relaxation techniques in chronic tinnitus. J. Neural Transm. 2016, 123, 1147–1157. [Google Scholar] [CrossRef]
- De Ridder, D.; Vanneste, S.; Langguth, B.; Llinas, R. Thalamocortical Dysrhythmia: A Theoretical Update in Tinnitus. Front. Neurol. 2015, 6, 124. [Google Scholar] [CrossRef]
- Weisz, N.; Dohrmann, K.; Elbert, T. The relevance of spontaneous activity for the coding of the tinnitus sensation. Prog. Brain Res. 2007, 166, 61–70. [Google Scholar] [PubMed] [Green Version]
- Carter, G.; Govindan, R.; Brown, G.; Heimann, C.; Hayes, H.; Thostenson, J.; Dornhoffer, J.; Brozoski, T.; Kimbrell, T.; Hayar, A.; et al. Change in EEG Activity is Associated with a Decrease in Tinnitus Awareness after rTMS. Front. Neurol. Neurosci. Res. 2021, 2, 100010. [Google Scholar] [PubMed]
- Simoes, J.; Neff, P.; Schoisswohl, S.; Bulla, J.; Schecklmann, M.; Harrison, S.; Vesala, M.; Langguth, B.; Schlee, W. Toward Personalized Tinnitus Treatment: An Exploratory Study Based on Internet Crowdsensing. Front. Public Health 2019, 7, 157. [Google Scholar] [CrossRef]
- Lehner, A.; Schecklmann, M.; Landgrebe, M.; Kreuzer, P.M.; Poeppl, T.B.; Frank, E.; Vielsmeier, V.; Kleinjung, T.; Rupprecht, R.; Langguth, B. Predictors for rTMS response in chronic tinnitus. Front. Syst. Neurosci. 2012, 6, 11. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| N (female) | 20 (5) | |||
| Handedness (left/right/both) (3 missings) | 0/13/4 | |||
| Tinnitus laterality (left/right/both/inside head) (3 missing) | 1/1/13/2 | |||
| M ± SD | Md | Min | Max | |
| Age (years) | 57.05 ± 6.77 | 57.50 | 43.00 | 69.00 |
| Tinnitus duration (months) (2 missing) | 126.00 ± 105.83 | 102.00 | 14.00 | 420.00 |
| Hearing loss left (dB) (7 missing) | 23.60 ± 10.10 | 22.22 | 7.22 | 41.67 |
| Hearing loss right (dB) (7 missing) | 28.39 ± 14.74 | 23.89 | 7.78 | 61.86 |
| RMT (%) | 34.10 ± 4.70 | 33.50 | 27.00 | 44.00 |
| TFI score (0–100) (2 missing) | 48.08 ± 17.91 | 48.55 | 23.20 | 78.80 |
| THI score (0–100) | 46.80 ± 16.43 | 42.00 | 24.00 | 84.00 |
| Mini-TQ score (0–24) (3 missing) | 12.94 ± 4.28 | 13.00 | 7.00 | 20.00 |
| MDI score (0–50) (3 missing) | 14.82 ± 9.93 | 14.00 | 1.00 | 40.00 |
| VAS tinnitus loudness (0–10) | 7.15 ± 1.69 | 7.50 | 3.00 | 10.00 |
| VAS tinnitus discomfort (0–10) | 7.50 ± 1.61 | 8.00 | 4.00 | 10.00 |
| VAS tinnitus annoyance (0–10) | 6.60 ± 2.30 | 7.00 | 2.00 | 10.00 |
| VAS tinnitus ignorability (0–10) | 7.60 ± 2.14 | 8.00 | 3.00 | 10.00 |
| VAS tinnitus unpleasantness 0–10) | 7.45 ± 1.88 | 8.00 | 3.00 | 10.00 |
| WHOQOL-BREF domain 1 (Physical health) (4–20) | 12.35 ± 2.01 | 13.00 | 8.00 | 15.00 |
| WHOQOL-BREF domain 2 (Psychological health) (4–20) | 13.80 ± 2.19 | 14.00 | 10.00 | 18.00 |
| WHOQOL-BREF domain 3 (Social relationships) (4–20) | 14.55 ± 2.80 | 15.50 | 9.00 | 20.00 |
| WHOQOL-BREF domain 4 (Environment) (4–20) | 16.45 ± 1.99 | 16.50 | 13.00 | 19.00 |
| Contrast | M ± SD | Estimate | T (df, se) | p | d |
|---|---|---|---|---|---|
| TFI | |||||
| Treatment end—follow-up | 49.69 ± 16.80—56.68 ± 17.62 | −7.65 | −3.05 (57.40, 2.51) | 0.018 | 0.406 |
| Follow-up—screening | 56.69 ± 17.62—48.08 ± 17.91 | 9.14 | 3.50 (57.70, 2.61) | 0.005 | 0.484 |
| THI | |||||
| Treatment end—follow-up | 46.10 ± 16.31—53.76 ± 18.68 | −7.69 | −3.04 (60.40, 2.53) | 0.018 | 0.437 |
| Follow-up—screening | 53.76 ± 18.68—46.80 ± 16.43 | 6.99 | 2.76 (60.40, 2.53) | 0.037 | 0.396 |
| WHOQOL-BREF domain 3 | |||||
| Baseline—treatment end | 14.40 ± 2.82—12.85 ± 1.79 | 1.55 | 3.14 (60.20, 0.49) | 0.014 | 0.656 |
| Treatment end—screening | 12.85 ± 1.79—14.55 ± 2.80 | −1.70 | −3.45 (60.20, 0.49) | 0.005 | 0.702 |
| VAS—Tinnitus unpleasantness | |||||
| Baseline—treatment end | 7.70 ± 1.66—6.85 ± 1.76 | 1.47 | 3.50 (63.70, 0.42) | 0.004 | 0.497 |
| Treatment end—screening | 6.85 ± 1.76—7.45 ± 1.88 | −1.22 | −2.91 (63.70, 0.42) | 0.025 | 0.329 |
| Personalized rTMS | |||||
| Baseline—treatment end | 8.00 ± 2.71—5.50 ± 1.73 | 2.50 | 3.33 (63.70, 0.75) | 0.003 | 1.100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schoisswohl, S.; Langguth, B.; Hebel, T.; Vielsmeier, V.; Abdelnaim, M.A.; Schecklmann, M. Personalization of Repetitive Transcranial Magnetic Stimulation for the Treatment of Chronic Subjective Tinnitus. Brain Sci. 2022, 12, 203. https://doi.org/10.3390/brainsci12020203
Schoisswohl S, Langguth B, Hebel T, Vielsmeier V, Abdelnaim MA, Schecklmann M. Personalization of Repetitive Transcranial Magnetic Stimulation for the Treatment of Chronic Subjective Tinnitus. Brain Sciences. 2022; 12(2):203. https://doi.org/10.3390/brainsci12020203
Chicago/Turabian StyleSchoisswohl, Stefan, Berthold Langguth, Tobias Hebel, Veronika Vielsmeier, Mohamed A. Abdelnaim, and Martin Schecklmann. 2022. "Personalization of Repetitive Transcranial Magnetic Stimulation for the Treatment of Chronic Subjective Tinnitus" Brain Sciences 12, no. 2: 203. https://doi.org/10.3390/brainsci12020203
APA StyleSchoisswohl, S., Langguth, B., Hebel, T., Vielsmeier, V., Abdelnaim, M. A., & Schecklmann, M. (2022). Personalization of Repetitive Transcranial Magnetic Stimulation for the Treatment of Chronic Subjective Tinnitus. Brain Sciences, 12(2), 203. https://doi.org/10.3390/brainsci12020203






