Functional Integration and Segregation in a Multilayer Network Model of Patients with Schizophrenia
Abstract
:1. Introduction
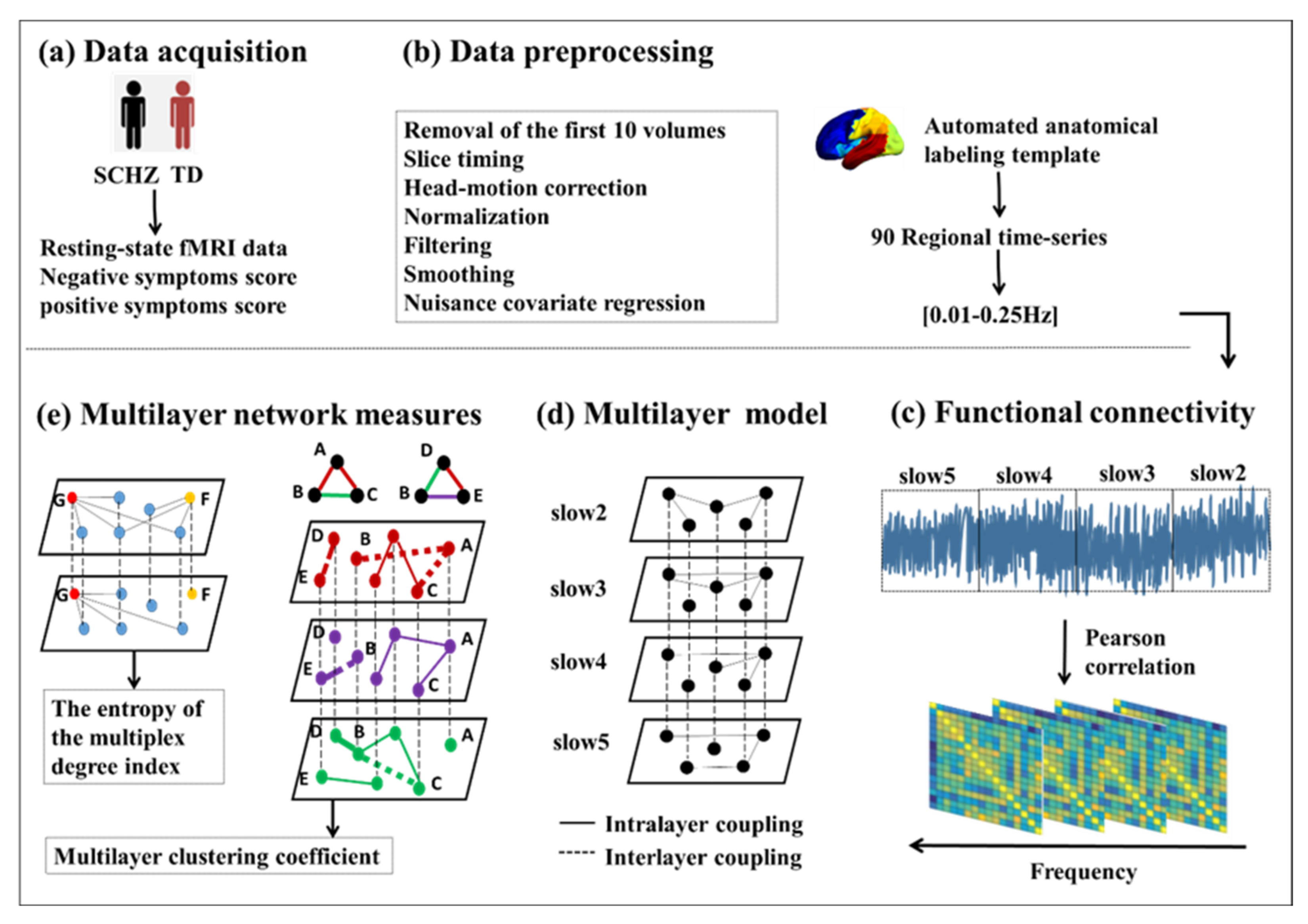
2. Materials and Methods
2.1. Participants and Data Acquisition
2.2. Data Preprocessing
2.3. Single-Layer Network Construction
2.4. Multilayer Network Construction
2.5. Network Measures
2.6. Single-Layer Network Measures of Segregation and Integration
2.7. Multilayer Network Segregation
2.8. Multilayer Network Integration
2.9. Parcellation into RSNs
2.10. Statistical Analysis
3. Results
3.1. Network Integration and Segregation
3.2. RSNs Differences
3.3. Node Vulnerability
3.4. Correlation between Network Measures and Cognitive Scores
4. Discussion
4.1. High Integration and Segregation in the Multilayer Network of Patients with Schizophrenia
4.2. Aberrant RSNs in Patients with Schizophrenia
4.3. Active Subcortical Network in Patients with Schizophrenia
4.4. Methodological Considerations and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stephan, K.E.; Friston, K.J.; Frith, C.D. Dysconnection in Schizophrenia: From Abnormal Synaptic Plasticity to Failures of Self-monitoring. Schizophr. Bull. 2009, 35, 509–527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buzsáki, G.; Logothetis, N.; Singer, W. Scaling Brain Size, Keeping Timing: Evolutionary Preservation of Brain Rhythms. Neuron 2013, 80, 751–764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xing, X.-X. Globally Aging Cortical Spontaneous Activity Revealed by Multiple Metrics and Frequency Bands Using Resting-State Functional MRI. Front. Aging Neurosci. 2021, 13, 803436. [Google Scholar] [CrossRef] [PubMed]
- Lynall, M.E.; Bassett, D.S.; Kerwin, R.; McKenna, P.J.; Kitzbichler, M.; Muller, U.; Bullmore, E. Functional connectivity and brain networks in schizophrenia. J. Neurosci. 2010, 30, 9477–9487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, R.; Chien, Y.L.; Wang, H.L.S.; Liu, C.M.; Liu, C.C.; Hwang, T.J.; Hsieh, M.H.; Hwu, H.G.; Tseng, W.Y.I. Frequency-specific alternations in the amplitude of low-frequency fluctuations in schizophrenia. Hum. Brain Mapp. 2014, 35, 627–637. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, A.; Chella, F.; Marshall, T.R.; Pizzella, V.; Romani, G.L.; Jensen, O.; Marzetti, L. Alpha and alpha-beta phase synchronization mediate the recruitment of the visuospatial attention network through the Superior Longitudinal Fasciculus. NeuroImage 2019, 188, 722–732. [Google Scholar] [CrossRef] [PubMed]
- Brookes, M.J.; Tewarie, P.K.; Hunt, B.; Robson, S.E.; Gascoyne, L.E.; Liddle, E.; Liddle, P.F.; Morris, P.G. A multi-layer network approach to MEG connectivity analysis. NeuroImage 2016, 132, 425–438. [Google Scholar] [CrossRef]
- Moran, L.V.; Hong, L.E. High vs low frequency neural oscillations in schizophrenia. Schizophr. Bull. 2011, 37, 659–663. [Google Scholar] [CrossRef]
- Kivelä, M.; Arenas, A.; Barthelemy, M.; Gleeson, J.P.; Moreno, Y.; Porter, M.A. Multilayer networks. J. Complex Netw. 2014, 2, 203–271. [Google Scholar] [CrossRef] [Green Version]
- Xu, F.; Zhang, J.; Jin, M.; Huang, S.; Fang, T. Chimera states and synchronization behavior in multilayer memristive neural networks. Nonlinear Dyn. 2018, 94, 775–783. [Google Scholar] [CrossRef]
- Micheloyannis, S.; Pachou, E.; Stam, C.J.; Breakspear, M.; Bitsios, P.; Vourkas, M.; Erimaki, S.; Zervakis, M. Small-world networks and disturbed functional connectivity in schizophrenia. Schizophr. Res. 2006, 87, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.; Hilgetag, C.C. Nonoptimal component placement, but short processing paths, due to long-distance projections in neural systems. PLoS Comput. Biol. 2006, 2, e95. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.X.; Guo, F.; Zhu, Y.Q.; Wang, H.N.; Liu, W.M.; Li, C.; Wang, X.R.; Cui, L.B.; Xi, Y.B.; Yin, H. Effect of second-generation antipsychotics on brain network topology in first-episode schizophrenia: A longitudinal rs-fMRI study. Schizophr. Res. 2019, 208, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Mehta, U.M.; Ibrahim, F.A.; Sharma, M.S.; Venkatasubramanian, G.; Thirthalli, J.; Bharath, R.D.; Bolo, N.R.; Gangadhar, B.N.; Keshavan, M.S. Resting-state functional connectivity predictors of treatment response in schizophrenia—A systematic review and meta-analysis. Schizophr. Res. 2021, 237, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Doucet, G.; Moser, D.; Luber, M.J.; Leibu, E.; Frangou, S. Baseline brain structural and functional predictors of clinical outcome in the early course of schizophrenia. Mol. Psychiatry 2020, 25, 863–872. [Google Scholar] [CrossRef]
- Cui, L.-B.; Cai, M.; Wang, X.-R.; Zhu, Y.-Q.; Wang, L.-X.; Xi, Y.-B.; Wang, H.-N.; Zhu, X.; Yin, H. Prediction of early response to overall treatment for schizophrenia: A functional magnetic resonance imaging study. Brain Behav. 2019, 9, e01211. [Google Scholar] [CrossRef] [Green Version]
- Anticevic, A.; Hu, X.; Xiao-Jing, W.; Hu, J.; Li, F.; Bi, F.; Cole, M.; Savic, A.; Yang, G.J.; Repovs, G.; et al. Early-Course Unmedicated Schizophrenia Patients Exhibit Elevated Prefrontal Connectivity Associated with Longitudinal Change. J. Neurosci. 2015, 35, 267–286. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Li, X.; Zhu, Y.; Lin, B.; Bo, Q.; Li, F.; Wang, C.-Y. Discriminative Analysis of Symptom Severity and Ultra-High Risk of Schizophrenia Using Intrinsic Functional Connectivity. Int. J. Neural Syst. 2020, 30, 2050047. [Google Scholar] [CrossRef]
- Maximo, J.; Nelson, E.A.; Armstrong, W.P.; Kraguljac, N.V.; Lahti, A.C. Duration of Untreated Psychosis Correlates With Brain Connectivity and Morphology in Medication-Naïve Patients With First-Episode Psychosis. Biol. Psychiatry: Cogn. Neurosci. Neuroimaging 2020, 5, 231–238. [Google Scholar] [CrossRef]
- Andreasen, N.C. Scale for the Assessment of Positive Symptoms (SAPS); University of Iowa: Iowa City, IA, USA, 1984. [Google Scholar]
- Andreasen, N.C. The Scale for the Assessment of Negative Symptoms (SANS): Conceptual and Theoretical Foundations. Br. J. Psychiatry Suppl. 1989, 155, 49–58. [Google Scholar] [CrossRef]
- Yan, C.; Zang, Y. DPARSF: A MATLAB toolbox for “pipeline” data analysis of resting-state fMRI. Front. Syst. Neurosci. 2010, 4, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eickhoff, S.B.; Stephan, K.E.; Mohlberg, H.; Grefkes, C.; Fink, G.R.; Amunts, K.; Zilles, K. A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. NeuroImage 2005, 25, 1325–1335. [Google Scholar] [CrossRef] [PubMed]
- Zuo, X.-N.; Di Martino, A.; Kelly, C.; Shehzad, Z.E.; Gee, D.; Klein, D.F.; Castellanos, F.; Biswal, B.B.; Milham, M.P. The oscillating brain: Complex and reliable. NeuroImage 2010, 49, 1432–1445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tzoutio-Mazoyera, N.; Landeau, B.; Papathanassiou, D.; Crivello, F.; Etard, O.; Delcroix, N.; Tzourio-Mazoyer, B.; Joliot, M. Automated Anatomical Labeling of Activations in SPM Using a Macroscopic Anatomical Parcellation of the MNI MRI Single-Subject Brain. NeuroImage 2002, 15, 273–289. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.; Xiang, J.; Cui, X.; Wang, X.; Li, D.; Cheng, C.; Wang, B. Abnormal Dynamic Community Structure of Patients with Attention-Deficit/Hyperactivity Disorder in the Resting State. J. Atten. Disord. 2020, 26, 34–47. [Google Scholar] [CrossRef]
- Wang, X.; Cui, X.; Ding, C.; Li, D.; Cheng, C.; Wang, B.; Xiang, J. Deficit of Cross-Frequency Integration in Mild Cognitive Impairment and Alzheimer’s Disease: A Multilayer Network Approach. J. Magn. Reson. Imaging 2021, 53, 1387–1398. [Google Scholar] [CrossRef]
- Wang, J.; Wang, X.; Xia, M.; Liao, X.; Evans, A.; He, Y. GRETNA: A graph theoretical network analysis toolbox for imaging connectomics. Front. Hum. Neurosci. 2015, 9, 386. [Google Scholar]
- Donges, J.F.; Schultz, H.C.H.; Marwan, N.; Zou, Y.; Kurths, J. Investigating the topology of interacting networks. Eur. Phys. J. B 2011, 84, 635–651. [Google Scholar] [CrossRef] [Green Version]
- Podobnik, B.; Horvatic, D.; Dickison, M.; Stanley, H.E. Preferential attachment in the interaction between dynamically generated interdependent networks. Eur. Lett. 2012, 100, 50004. [Google Scholar] [CrossRef]
- Parshani, R.; Rozenblat, C.; Ietri, D.; Ducruet, C.; Havlin, S. Inter-similarity between coupled networks. Eur. Lett. 2010, 92, 68002. [Google Scholar] [CrossRef] [Green Version]
- Barrett, L.; Henzi, S.P.; Lusseau, D. Taking sociality seriously: The structure of multi-dimensional social networks as a source of information for individuals. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 2108–2118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Battiston, F.; Nicosia, V.; Latora, V. Structural measures for multiplex networks. Phys. Rev. E 2014, 89, 032804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Wang, J.; Wang, L.; Chen, Z.J.; Yan, C.-G.; Yang, H.; Tang, H.; Zhu, C.; Gong, Q.; Zang, Y.; et al. Uncovering Intrinsic Modular Organization of Spontaneous Brain Activity in Humans. PLoS ONE 2009, 4, e5226. [Google Scholar] [CrossRef] [Green Version]
- Xia, M.; Wang, J.; He, Y. BrainNet Viewer: A network visualization tool for human brain connectomics. PLoS ONE 2013, 8, e68910. [Google Scholar] [CrossRef] [Green Version]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Braun, U.; Schäfer, A.; Bassett, D.S.; Rausch, F.; Schweiger, J.I.; Bilek, E.; Erk, S.; Romanczuk-Seiferth, N.; Grimm, O.; Geiger, L.S.; et al. Dynamic brain network reconfiguration as a potential schizophrenia genetic risk mechanism modulated by NMDA receptor function. Proc. Natl. Acad. Sci. USA 2016, 113, 12568–12573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gifford, G.; Crossley, N.; Kempton, M.J.; Morgan, S.; Dazzan, P.; Young, J.; McGuire, P. Resting State fMRI Based Multilayer Network Configuration in Patients with Schizophrenia. NeuroImage Clin. 2020, 25, 102169. [Google Scholar] [CrossRef] [PubMed]
- Pettersson-Yeo, W.; Allen, P.; Benetti, S.; McGuire, P.; Mechelli, A. Dysconnectivity in schizophrenia: Where are we now? Neurosci. Biobehav. Rev. 2011, 35, 1110–1124. [Google Scholar] [CrossRef]
- Heuvel, M.; Fornito, A. Brain Networks in Schizophrenia. Neuropsychol. Rev. 2014, 24, 32–48. [Google Scholar] [CrossRef]
- Hummer, T.A.; Yung, M.G.; Goñi, J.; Conroy, S.K.; Francis, M.M.; Mehdiyoun, N.F.; Breier, A. Functional network connectivity in early-stage schizophrenia. Schizophr. Res. 2020, 218, 107–115. [Google Scholar] [CrossRef]
- Lerman-Sinkoff, D.B.; Barch, D.M. Network community structure alterations in adult schizophrenia: Identification and localization of alterations. NeuroImage Clin. 2015, 10, 96–106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salgado-Pineda, P.; Fakra, E.; Delaveau, P.; McKenna, P.; Pomarol-Clotet, E.; Blin, O. Correlated structural and functional brain abnormalities in the default mode network in schizophrenia patients. Schizophr. Res. 2011, 125, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Duan, M.; Xie, Q.; Lai, Y.; Dong, L.; Cao, W.; Yao, D.; Luo, C. Functional disconnection between the visual cortex and the sensorimotor cortex suggests a potential mechanism for self-disorder in schizophrenia. Schizophr. Res. 2015, 166, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Woodward, N.D.; Rogers, B.; Heckers, S.J.S.R. Functional resting-state networks are differentially affected in schizophrenia. Schizophr. Res. 2011, 130, 86–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bassett, D.S.; Bullmore, E.; Verchinski, B.A.; Mattay, V.S.; Weinberger, D.R.; Meyer-Lindenberg, A. Hierarchical Organization of Human Cortical Networks in Health and Schizophrenia. J. Neurosci. 2008, 28, 9239–9248. [Google Scholar] [CrossRef] [Green Version]
- Kanahara, N.; Sekine, Y.; Haraguchi, T.; Uchida, Y.; Hashimoto, K.; Shimizu, E.; Iyo, M. Orbitofrontal cortex abnormality and deficit schizophrenia. Schizophr. Res. 2013, 143, 246–252. [Google Scholar] [CrossRef]
- Heuvel, M.V.D.; Mandl, R.C.W.; Stam, C.J.; Kahn, R.S.; Pol, H.H. Aberrant Frontal and Temporal Complex Network Structure in Schizophrenia: A Graph Theoretical Analysis. J. Neurosci. 2010, 30, 15915–15926. [Google Scholar] [CrossRef]
- Meyer-Lindenberg, A.; Poline, J.-B.; Kohn, P.D.; Holt, J.L.; Egan, M.F.; Weinberger, D.R.; Berman, K.F. Evidence for Abnormal Cortical Functional Connectivity During Working Memory in Schizophrenia. Am. J. Psychiatry 2001, 158, 1809–1817. [Google Scholar] [CrossRef]
- Garrity, A.; Pearlson, G.; McKiernan, K.; Lloyd, D.; Kiehl, K.; Calhoun, V. Aberrant “default mode” functional connectivity in schizophrenia. Am. J. Psychiatry 2007, 164, 450–457. [Google Scholar] [CrossRef]
- Buchsbaum, M.S.J.S.B. The frontal lobes, basal ganglia, and temporal lobes as sites for schizophrenia. Schizophr. Bull. 1990, 16, 379–389. [Google Scholar] [CrossRef] [Green Version]
- Van Erp, T.G.; Hibar, D.P.; Rasmussen, J.M.; Glahn, D.C.; Pearlson, G.D.; Andreassen, O.A.; Agartz, I.; Westlye, L.T.; Haukvik, U.K.; Dale, A.M.; et al. Subcortical brain volume abnormalities in 2028 individuals with schizophrenia and 2540 healthy controls via the ENIGMA consortium. Mol. Psychiatry 2016, 21, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Okada, N.; Fukunaga, M.; Yamashita, F.; Koshiyama, D.; Yamamori, H.; Ohi, K.; Yasuda, Y.; Fujimoto, M.; Watanabe, Y.; Yahata, N.; et al. Abnormal asymmetries in subcortical brain volume in schizophrenia. Mol. Psychiatry 2016, 21, 1460–1466. [Google Scholar] [CrossRef] [PubMed] [Green Version]









| Group | TD | SCHZ | p-Value |
|---|---|---|---|
| Number of subjects | 69 | 50 | -- |
| Age (mean ± SD) | 31.83 ± 8.73 | 33.84 ± 6.51 | 0.156 1 |
| Sex (M/F) | 42/27 | 38/12 | 0.083 2 |
| SAPS (mean ± SD) | -- | 30.92 ± 21.04 | -- |
| SANS (mean ± SD) | -- | 35.9 ± 19.21 | -- |
| ROI | Name | Abbreviation | Network | P (FDR) | SCHZ (SD) | TD (SD) |
|---|---|---|---|---|---|---|
| 3 | Frontal_Sup_L | SFGdor.L | Default mode | 0.020 | 1.338 (0.058) | 1.305 (0.061) |
| 21 | Olfactory_L | OLF.L | Subcortical | 0.000 | 1.098 (0.338) | 0.132 (0.344) |
| 22 | Olfactory_R | OLF.R | Subcortical | 0.000 | 1.091 (0.398) | 0.038 (0.182) |
| 25 | Frontal_Mid_Orb_L | ORBsupmed.L | Default mode | 0.014 | 1.245 (0.122) | 1.092 (0.388) |
| 28 | Rectus_R | REC.R | Default mode | 0.009 | 1.180 (0.228) | 0.999 (0.387) |
| 35 | Cingulum_Post_L | PCG.L | Default mode | 0.009 | 1.205 (0.213) | 1.021 (0.393) |
| 36 | Cingulum_Post_R | PCG.R | Default mode | 0.000 | 1.208 (0.126) | 0.289 (0.483) |
| 39 | ParaHippocampal_L | PHG.L | Subcortical | 0.023 | 1.289 (0.098) | 1.190 (0.229) |
| 41 | Amygdala_L | AMYG.L | Subcortical | 0.000 | 1.242 (0.129) | 0.000 (0.000) |
| 42 | Amygdala_R | AMYG.R | Subcortical | 0.000 | 1.249 (0.116) | 0.026 (0.156) |
| 45 | Cuneus_L | CUN.L | Visual | 0.005 | 1.311 (0.073) | 1.268 (0.068) |
| 46 | Cuneus_R | CUN.R | Visual | 0.023 | 1.332 (0.059) | 1.300 (0.061) |
| 49 | Occipital_Sup_L | SOG.L | Visual | 0.000 | 1.311 (0.071) | 1.188 (0.221) |
| 53 | Occipital_Inf_L | IOG.L | Visual | 0.000 | 1.322 (0.079) | 1.265 (0.072) |
| 59 | Parietal_Sup_L | SPG.L | Sensorimotor | 0.000 | 1.334 (0.060) | 1.260 (0.095) |
| 74 | Putamen_R | PUT.R | Subcortical | 0.043 | 1.293 (0.075) | 1.250 (0.113) |
| 75 | Pallidum_L | PAL.L | Subcortical | 0.000 | 1.227 (0.211) | 0.045 (0.216) |
| 76 | Pallidum_R | PAL.R | Subcortical | 0.000 | 1.245 (0.119) | 0.015 (0.124) |
| 77 | Thalamus_L | THA.L | Subcortical | 0.038 | 1.258 (0.118) | 1.179 (0.191) |
| 78 | Thalamus_R | THA.R | Subcortical | 0.000 | 1.269 (0.111) | 1.163 (0.154) |
| 79 | Heschl_L | HES.L | Sensorimotor | 0.000 | 1.282 (0.080) | 0.196 (0.409) |
| 80 | Heschl_R | HES.R | Sensorimotor | 0.000 | 1.302 (0.074) | 0.285 (0.482) |
| 87 | Temporal_Pole_Mid_L | TPOmid.L | Subcortical | 0.000 | 1.149 (0.278) | 0.829 (0.489) |
| ROI | Name | Abbreviation | Network | P (FDR) | SCHZ (SD) | TD (SD) |
|---|---|---|---|---|---|---|
| 21 | Olfactory_L | OLF.L | Subcortical | 0.000 | 0.207 (0.079) | 0.139(0.059) |
| 37 | Hippocampus_L | HIP.L | Subcortical | 0.000 | 0.247 (0.071) | 0.203 (0.058) |
| 38 | Hippocampus_R | HIP.R | Subcortical | 0.000 | 0.254 (0.070) | 0.212 (0.054) |
| 39 | Parahippocampal_L | PHG.L | Subcortical | 0.000 | 0.270 (0.065) | 0.221 (0.049) |
| 40 | Parahippocampal_R | PHG.R | Subcortical | 0.004 | 0.274 (0.065) | 0.236 (0.054) |
| 41 | Amygdala_L | AMYG.L | Subcortical | 0.000 | 0.230 (0.076) | 0.172 (0.057) |
| 42 | Amygdala_R | AMYG.R | Subcortical | 0.000 | 0.240 (0.064) | 0.169 (0.060) |
| 73 | Putamen_L | PUT.L | Subcortical | 0.033 | 0.249 (0.073) | 0.219 (0.062) |
| 75 | Pallidum_L | PAL.L | Subcortical | 0.000 | 0.223 (0.073) | 0.168 (0.059) |
| 76 | Pallidum_R | PAL.R | Subcortical | 0.002 | 0.213 (0.073) | 0.170 (0.058) |
| 77 | Thalamus_L | THA.L | Subcortical | 0.000 | 0.254 (0.068) | 0.199 (0.067) |
| 78 | Thalamus_R | THA.R | Subcortical | 0.002 | 0.245 (0.073) | 0.201 (0.063) |
| 87 | Temporal_Pole_Mid_L | TPOmid.L | Subcortical | 0.000 | 0.216 (0.064) | 0.160 (0.061) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, J.; Wang, X.; Cui, X.; Wang, B.; Xue, J.; Niu, Y.; Wang, Q.; Osmani, A.; Xiang, J. Functional Integration and Segregation in a Multilayer Network Model of Patients with Schizophrenia. Brain Sci. 2022, 12, 368. https://doi.org/10.3390/brainsci12030368
Wei J, Wang X, Cui X, Wang B, Xue J, Niu Y, Wang Q, Osmani A, Xiang J. Functional Integration and Segregation in a Multilayer Network Model of Patients with Schizophrenia. Brain Sciences. 2022; 12(3):368. https://doi.org/10.3390/brainsci12030368
Chicago/Turabian StyleWei, Jing, Xiaoyue Wang, Xiaohong Cui, Bin Wang, Jiayue Xue, Yan Niu, Qianshan Wang, Arezo Osmani, and Jie Xiang. 2022. "Functional Integration and Segregation in a Multilayer Network Model of Patients with Schizophrenia" Brain Sciences 12, no. 3: 368. https://doi.org/10.3390/brainsci12030368








