Cognitive Remediation in Psychiatric Disorders: State of the Evidence, Future Perspectives, and Some Bold Ideas
Abstract
:1. The Importance of Cognition in Psychiatric Disorders
2. Can We Help?
3. How Can We Do It Right?
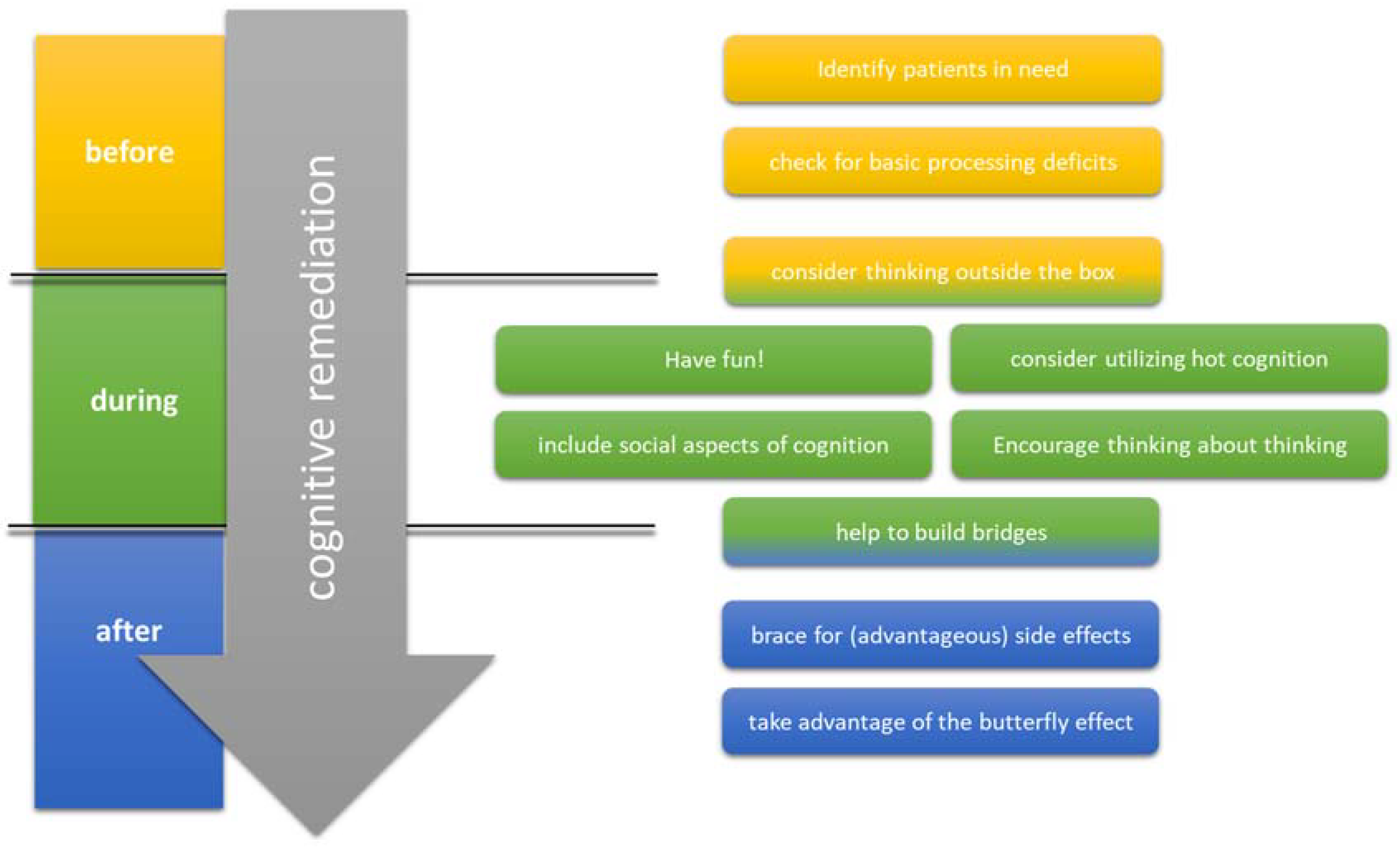
3.1. Identify Patients in Need
3.2. Check for Basic Processing Deficits
3.3. Consider Thinking Outside the Box
3.3.1. Social Cognition Training in CR Settings
3.3.2. Combination of Physical Exercise and CR
3.3.3. CR as a Tool to Improve Clinical Symptoms
3.4. Have Fun!
3.5. Consider Utilizing Hot Cognition
3.6. Include Social Aspects of Cognition
3.7. Encourage Thinking about Thinking
3.8. Help to Build Bridges
3.9. Brace for (Advantageous) Side Effects
3.10. Take Advantage of the Butterfly Effect
4. Conclusions
- Choosing a gamified approach that lets participants have as much fun as possible;
- Combining CR with previous perceptual training if basic processing deficits are present;
- Including and stimulating metacognition;
- Using group settings to stimulate social cognition;
- Letting CR reach out into everyday life as much as possible;
- Utilizing the effects of hot cognition on information processing;
- Keeping in mind that the effects of CR interventions might be subtle at the beginning but grow stronger over time;
- Extending the classic CR paradigm by including cognitive group tasks that require social interaction, combining CR with physical exercise, and considering using CR also in settings where the primary target is to improve clinical symptoms rather than ameliorate cognitive deficits.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Kim, E.J.; Bahk, Y.-C.; Oh, H.; Lee, W.-H.; Lee, J.-S.; Choi, K.-H. Current Status of Cognitive Remediation for Psychiatric Disorders: A Review. Front. Psychiatry 2018, 9, 461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keshavan, M.S.; Vinogradov, S.; Rumsey, J.; Sherrill, J.; Wagner, A. Cognitive Training in Mental Disorders: Update and Future Directions. AJP 2014, 171, 510–522. [Google Scholar] [CrossRef] [PubMed]
- McGurk, S.R.; Twamley, E.W.; Sitzer, D.I.; McHugo, G.J.; Mueser, K.T. A Meta-Analysis of Cognitive Remediation in Schizophrenia. Am. J. Psychiatry 2007, 164, 1791–1802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wykes, T.; Huddy, V.; Cellard, C.; McGurk, S.R.; Czobor, P. A Meta-Analysis of Cognitive Remediation for Schizophrenia: Methodology and Effect Sizes. Am. J. Psychiatry 2011, 168, 472–485. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, A.O. Cognitive Remediation for Schizophrenia. Focus 2020, 18, 436–439. [Google Scholar] [CrossRef]
- MacQueen, G.M.; Memedovich, K.A. Cognitive Dysfunction in Major Depression and Bipolar Disorder: Assessment and Treatment Options. Psychiatry Clin. Neurosci. 2017, 71, 18–27. [Google Scholar] [CrossRef] [Green Version]
- Motter, J.N.; Pimontel, M.A.; Rindskopf, D.; Devanand, D.P.; Doraiswamy, P.M.; Sneed, J.R. Computerized Cognitive Training and Functional Recovery in Major Depressive Disorder: A Meta-Analysis. J. Affect. Disord. 2016, 189, 184–191. [Google Scholar] [CrossRef]
- Hill, N.T.M.; Mowszowski, L.; Naismith, S.L.; Chadwick, V.L.; Valenzuela, M.; Lampit, A. Computerized Cognitive Training in Older Adults with Mild Cognitive Impairment or Dementia: A Systematic Review and Meta-Analysis. Am. J. Psychiatry 2017, 174, 329–340. [Google Scholar] [CrossRef]
- García-Casal, J.A.; Loizeau, A.; Csipke, E.; Franco-Martín, M.; Perea-Bartolomé, M.V.; Orrell, M. Computer-Based Cognitive Interventions for People Living with Dementia: A Systematic Literature Review and Meta-Analysis. Aging Ment. Health 2017, 21, 454–467. [Google Scholar] [CrossRef]
- Heinrichs, R.W.; Zakzanis, K.K. Neurocognitive Deficit in Schizophrenia: A Quantitative Review of the Evidence. Neuropsychology 1998, 12, 426–445. [Google Scholar] [CrossRef]
- Dickinson, D.; Ramsey, M.E.; Gold, J.M. Overlooking the Obvious: A Meta-Analytic Comparison of Digit Symbol Coding Tasks and Other Cognitive Measures in Schizophrenia. Arch. Gen. Psychiatry 2007, 64, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Cannon, M.; Caspi, A.; Moffitt, T.E.; Harrington, H.; Taylor, A.; Murray, R.M.; Poulton, R. Evidence for Early-Childhood, Pan-Developmental Impairment Specific to Schizophreniform Disorder: Results from a Longitudinal Birth Cohort. Arch. Gen. Psychiatry 2002, 59, 449–456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asarnow, R.F.; Nuechterlein, K.H.; Subotnik, K.L.; Fogelson, D.L.; Torquato, R.D.; Payne, D.L.; Asamen, J.; Mintz, J.; Guthrie, D. Neurocognitive Impairments in Nonpsychotic Parents of Children with Schizophrenia and Attention-Deficit/Hyperactivity Disorder: The University of California, Los Angeles Family Study. Arch. Gen. Psychiatry 2002, 59, 1053–1060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nuechterlein, K.H.; Asarnow, R.F.; Subotnik, K.L.; Fogelson, D.L.; Payne, D.L.; Kendler, K.S.; Neale, M.C.; Jacobson, K.C.; Mintz, J. The Structure of Schizotypy: Relationships between Neurocognitive and Personality Disorder Features in Relatives of Schizophrenic Patients in the UCLA Family Study. Schizophr. Res. 2002, 54, 121–130. [Google Scholar] [CrossRef] [Green Version]
- Fett, A.-K.J.; Viechtbauer, W.; Dominguez, M.-G.; Penn, D.L.; van Os, J.; Krabbendam, L. The Relationship between Neurocognition and Social Cognition with Functional Outcomes in Schizophrenia: A Meta-Analysis. Neurosci. Biobehav. Rev. 2011, 35, 573–588. [Google Scholar] [CrossRef] [Green Version]
- Nuechterlein, K.H.; Subotnik, K.L.; Green, M.F.; Ventura, J.; Asarnow, R.F.; Gitlin, M.J.; Yee, C.M.; Gretchen-Doorly, D.; Mintz, J. Neurocognitive Predictors of Work Outcome in Recent-Onset Schizophrenia. Schizophr. Bull. 2011, 37 (Suppl. 2), S33–S40. [Google Scholar] [CrossRef] [Green Version]
- Holthausen, E.A.E.; Wiersma, D.; Cahn, W.; Kahn, R.S.; Dingemans, P.M.; Schene, A.H.; van den Bosch, R.J. Predictive Value of Cognition for Different Domains of Outcome in Recent-Onset Schizophrenia. Psychiatry Res. 2007, 149, 71–80. [Google Scholar] [CrossRef]
- Wölwer, W.; Brinkmeyer, J.; Riesbeck, M.; Freimüller, L.; Klimke, A.; Wagner, M.; Möller, H.-J.; Klingberg, S.; Gaebel, W. German Study Group on First Episode Schizophrenia Neuropsychological Impairments Predict the Clinical Course in Schizophrenia. Eur. Arch. Psychiatry Clin. Neurosci. 2008, 258 (Suppl. 5), 28–34. [Google Scholar] [CrossRef]
- Trapp, W.; Landgrebe, M.; Hoesl, K.; Lautenbacher, S.; Gallhofer, B.; Günther, W.; Hajak, G. Cognitive Remediation Improves Cognition and Good Cognitive Performance Increases Time to Relapse—Results of a 5 Year Catamnestic Study in Schizophrenia Patients. BMC Psychiatry 2013, 13, 184. [Google Scholar] [CrossRef] [Green Version]
- Habtewold, T.D.; Rodijk, L.H.; Liemburg, E.J.; Sidorenkov, G.; Boezen, H.M.; Bruggeman, R.; Alizadeh, B.Z. A Systematic Review and Narrative Synthesis of Data-Driven Studies in Schizophrenia Symptoms and Cognitive Deficits. Transl. Psychiatry 2020, 10, 244. [Google Scholar] [CrossRef]
- Alkan, E.; Evans, S.L. Clustering of Cognitive Subtypes in Schizophrenia Patients and Their Siblings: Relationship with Regional Brain Volumes. Schizophrenia 2022, 8, 50. [Google Scholar] [CrossRef]
- Vaskinn, A.; Haatveit, B.; Melle, I.; Andreassen, O.; Ueland, T.; Sundet, K. Cognitive Heterogeneity across Schizophrenia and Bipolar Disorder: A Cluster Analysis of Intellectual Trajectories. J. Int. Neuropsychol. Soc. 2020, 26, 860–872. [Google Scholar] [CrossRef] [PubMed]
- Bortolato, B.; Miskowiak, K.W.; Köhler, C.A.; Vieta, E.; Carvalho, A.F. Cognitive Dysfunction in Bipolar Disorder and Schizophrenia: A Systematic Review of Meta-Analyses. Neuropsychiatr. Dis. Treat. 2015, 11, 3111–3125. [Google Scholar] [CrossRef] [Green Version]
- Bourne, C.; Aydemir, Ö.; Balanzá-Martínez, V.; Bora, E.; Brissos, S.; Cavanagh, J.T.O.; Clark, L.; Cubukcuoglu, Z.; Dias, V.V.; Dittmann, S.; et al. Neuropsychological Testing of Cognitive Impairment in Euthymic Bipolar Disorder: An Individual Patient Data Meta-Analysis. Acta Psychiatr. Scand. 2013, 128, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Martino, D.J.; Samamé, C.; Ibañez, A.; Strejilevich, S.A. Neurocognitive Functioning in the Premorbid Stage and in the First Episode of Bipolar Disorder: A Systematic Review. Psychiatry Res. 2015, 226, 23–30. [Google Scholar] [CrossRef]
- de la Serna, E.; Vila, M.; Sanchez-Gistau, V.; Moreno, D.; Romero, S.; Sugranyes, G.; Baeza, I.; Llorente, C.; Rodriguez-Toscano, E.; Sánchez-Gutierrez, T.; et al. Neuropsychological Characteristics of Child and Adolescent Offspring of Patients with Bipolar Disorder. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 65, 54–59. [Google Scholar] [CrossRef]
- Li, W.; Zhou, F.-C.; Zhang, L.; Ng, C.H.; Ungvari, G.S.; Li, J.; Xiang, Y.-T. Comparison of Cognitive Dysfunction between Schizophrenia and Bipolar Disorder Patients: A Meta-Analysis of Comparative Studies. J. Affect. Disord. 2020, 274, 652–661. [Google Scholar] [CrossRef]
- Daban, C.; Martinez-Aran, A.; Torrent, C.; Tabarés-Seisdedos, R.; Balanzá-Martínez, V.; Salazar-Fraile, J.; Selva-Vera, G.; Vieta, E. Specificity of Cognitive Deficits in Bipolar Disorder versus Schizophrenia. A Systematic Review. Psychother. Psychosom. 2006, 75, 72–84. [Google Scholar] [CrossRef]
- Wingo, A.P.; Harvey, P.D.; Baldessarini, R.J. Neurocognitive Impairment in Bipolar Disorder Patients: Functional Implications. Bipolar Disord. 2009, 11, 113–125. [Google Scholar] [CrossRef]
- Tse, S.; Chan, S.; Ng, K.L.; Yatham, L.N. Meta-Analysis of Predictors of Favorable Employment Outcomes among Individuals with Bipolar Disorder. Bipolar Disord. 2014, 16, 217–229. [Google Scholar] [CrossRef]
- Träger, C.; Decker, L.; Wæhrens, E.E.; Knorr, U.; Miskowiak, K.; Vinberg, M. Influences of Patient Informed Cognitive Complaints on Activities of Daily Living in Patients with Bipolar Disorder. An Exploratory Cross-Sectional Study. Psychiatry Res. 2017, 249, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Bora, E.; Harrison, B.J.; Yücel, M.; Pantelis, C. Cognitive Impairment in Euthymic Major Depressive Disorder: A Meta-Analysis. Psychol. Med. 2013, 43, 2017–2026. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.S.C.; Hermens, D.F.; Porter, M.A.; Redoblado-Hodge, M.A. A Meta-Analysis of Cognitive Deficits in First-Episode Major Depressive Disorder. J. Affect. Disord. 2012, 140, 113–124. [Google Scholar] [CrossRef]
- Snyder, H.R. Major Depressive Disorder Is Associated with Broad Impairments on Neuropsychological Measures of Executive Function: A Meta-Analysis and Review. Psychol. Bull. 2013, 139, 81–132. [Google Scholar] [CrossRef] [Green Version]
- Grahek, I.; Shenhav, A.; Musslick, S.; Krebs, R.M.; Koster, E.H.W. Motivation and Cognitive Control in Depression. Neurosci. Biobehav. Rev. 2019, 102, 371–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elgamal, S.; Denburg, S.; Marriott, M.; MacQueen, G. Clinical Factors That Predict Cognitive Function in Patients with Major Depression. Can. J. Psychiatry 2010, 55, 653–661. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jaeger, J.; Berns, S.; Uzelac, S.; Davis-Conway, S. Neurocognitive Deficits and Disability in Major Depressive Disorder. Psychiatry Res. 2006, 145, 39–48. [Google Scholar] [CrossRef]
- Withall, A.; Harris, L.M.; Cumming, S.R. The Relationship between Cognitive Function and Clinical and Functional Outcomes in Major Depressive Disorder. Psychol. Med. 2009, 39, 393–402. [Google Scholar] [CrossRef]
- Morimoto, S.S.; Gunning, F.M.; Kanellopoulos, D.; Murphy, C.F.; Klimstra, S.A.; Kelly, R.E.; Alexopoulos, G.S. Semantic Organizational Strategy Predicts Verbal Memory and Remission Rate of Geriatric Depression. Int. J. Geriatr. Psychiatry 2012, 27, 506–512. [Google Scholar] [CrossRef]
- Kampf-Sherf, O.; Zlotogorski, Z.; Gilboa, A.; Speedie, L.; Lereya, J.; Rosca, P.; Shavit, Y. Neuropsychological Functioning in Major Depression and Responsiveness to Selective Serotonin Reuptake Inhibitors Antidepressants. J. Affect. Disord. 2004, 82, 453–459. [Google Scholar] [CrossRef]
- McLennan, S.N.; Mathias, J.L. The Depression-Executive Dysfunction (DED) Syndrome and Response to Antidepressants: A Meta-Analytic Review. Int. J. Geriatr. Psychiatry 2010, 25, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, A.; Eubig, P.A.; Schantz, S.L. Attention Deficit/Hyperactivity Disorder: A Focused Overview for Children’s Environmental Health Researchers. Environ. Health Perspect. 2010, 118, 1646–1653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaidya, C.J.; Stollstorff, M. Cognitive Neuroscience of Attention Deficit Hyperactivity Disorder: Current Status and Working Hypotheses. Dev. Disabil. Res. Rev. 2008, 14, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Ramey, T.; Regier, P.S. Cognitive Impairment in Substance Use Disorders. CNS Spectr. 2018, 24, 102–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nardo, T.; Batchelor, J.; Berry, J.; Francis, H.; Jafar, D.; Borchard, T. Cognitive Remediation as an Adjunct Treatment for Substance Use Disorders: A Systematic Review. Neuropsychol. Rev. 2021, 32, 161–191. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Serrano, M.J.; Pérez-García, M.; Verdejo-García, A. What Are the Specific vs. Generalized Effects of Drugs of Abuse on Neuropsychological Performance? Neurosci. Biobehav. Rev. 2011, 35, 377–406. [Google Scholar] [CrossRef] [PubMed]
- Kwako, L.E.; Momenan, R.; Litten, R.Z.; Koob, G.F.; Goldman, D. Addictions Neuroclinical Assessment: A Neuroscience-Based Framework for Addictive Disorders. Biol. Psychiatry 2016, 80, 179–189. [Google Scholar] [CrossRef] [Green Version]
- Horner, M.D.; Hamner, M.B. Neurocognitive Functioning in Posttraumatic Stress Disorder. Neuropsychol. Rev. 2002, 12, 15–30. [Google Scholar] [CrossRef]
- Twamley, E.W.; Allard, C.B.; Thorp, S.R.; Norman, S.B.; Hami Cissell, S.; Hughes Berardi, K.; Grimes, E.M.; Stein, M.B. Cognitive Impairment and Functioning in PTSD Related to Intimate Partner Violence. J. Int. Neuropsychol. Soc. 2009, 15, 879–887. [Google Scholar] [CrossRef]
- Penadés, R.; Catalán, R.; Andrés, S.; Salamero, M.; Gastó, C. Executive Function and Nonverbal Memory in Obsessive-Compulsive Disorder. Psychiatry Res. 2005, 133, 81–90. [Google Scholar] [CrossRef]
- Tchanturia, K.; Harrison, A.; Davies, H.; Roberts, M.; Oldershaw, A.; Nakazato, M.; Stahl, D.; Morris, R.; Schmidt, U.; Treasure, J. Cognitive Flexibility and Clinical Severity in Eating Disorders. PLoS ONE 2011, 6, e20462. [Google Scholar] [CrossRef] [PubMed]
- Quinones, M.M.; Gallegos, A.M.; Lin, F.V.; Heffner, K. Dysregulation of Inflammation, Neurobiology, and Cognitive Function in PTSD: An Integrative Review. Cogn. Affect. Behav. Neurosci. 2020, 20, 455–480. [Google Scholar] [CrossRef] [PubMed]
- Benzina, N.; Mallet, L.; Burguière, E.; N’Diaye, K.; Pelissolo, A. Cognitive Dysfunction in Obsessive-Compulsive Disorder. Curr. Psychiatry Rep. 2016, 18, 80. [Google Scholar] [CrossRef] [Green Version]
- Millan, M.J.; Agid, Y.; Brüne, M.; Bullmore, E.T.; Carter, C.S.; Clayton, N.S.; Connor, R.; Davis, S.; Deakin, B.; DeRubeis, R.J.; et al. Cognitive Dysfunction in Psychiatric Disorders: Characteristics, Causes and the Quest for Improved Therapy. Nat. Rev. Drug Discov. 2012, 11, 141–168. [Google Scholar] [CrossRef]
- Bowie, C.R.; Bell, M.D.; Fiszdon, J.M.; Johannesen, J.K.; Lindenmayer, J.-P.; McGurk, S.R.; Medalia, A.A.; Penadés, R.; Saperstein, A.M.; Twamley, E.W.; et al. Cognitive Remediation for Schizophrenia: An Expert Working Group White Paper on Core Techniques. Schizophr. Res. 2020, 215, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Grynszpan, O.; Perbal, S.; Pelissolo, A.; Fossati, P.; Jouvent, R.; Dubal, S.; Perez-Diaz, F. Efficacy and Specificity of Computer-Assisted Cognitive Remediation in Schizophrenia: A Meta-Analytical Study. Psychol. Med. 2011, 41, 163–173. [Google Scholar] [CrossRef]
- Wykes, T.; Spaulding, W.D. Thinking about the Future Cognitive Remediation Therapy—What Works and Could We Do Better? Schizophr. Bull. 2011, 37 (Suppl. 2), S80–S90. [Google Scholar] [CrossRef] [Green Version]
- Revell, E.R.; Neill, J.C.; Harte, M.; Khan, Z.; Drake, R.J. A Systematic Review and Meta-Analysis of Cognitive Remediation in Early Schizophrenia. Schizophr. Res. 2015, 168, 213–222. [Google Scholar] [CrossRef]
- Legemaat, A.M.; Semkovska, M.; Brouwer, M.; Geurtsen, G.J.; Burger, H.; Denys, D.; Bockting, C.L. Effectiveness of Cognitive Remediation in Depression: A Meta-Analysis. Psychol. Med. 2021; Online anhead of print. [Google Scholar] [CrossRef]
- Hoorelbeke, K.; Koster, E.H.W. Internet-Delivered Cognitive Control Training as a Preventive Intervention for Remitted Depressed Patients: Evidence from a Double-Blind Randomized Controlled Trial Study. J. Consult. Clin. Psychol. 2017, 85, 135–146. [Google Scholar] [CrossRef] [Green Version]
- Hoorelbeke, K.; Van den Bergh, N.; De Raedt, R.; Wichers, M.; Koster, E.H.W. Preventing Recurrence of Depression: Long-Term Effects of a Randomized Controlled Trial on Cognitive Control Training for Remitted Depressed Patients. Clin. Psychol. Sci. 2021, 9, 615–633. [Google Scholar] [CrossRef]
- Deckersbach, T.; Nierenberg, A.A.; Kessler, R.; Lund, H.G.; Ametrano, R.M.; Sachs, G.; Rauch, S.L.; Dougherty, D. RESEARCH: Cognitive Rehabilitation for Bipolar Disorder: An Open Trial for Employed Patients with Residual Depressive Symptoms. CNS Neurosci. Ther. 2010, 16, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Torrent, C.; Bonnin, C.d.M.; Martínez-Arán, A.; Valle, J.; Amann, B.L.; González-Pinto, A.; Crespo, J.M.; Ibáñez, Á.; Garcia-Portilla, M.P.; Tabarés-Seisdedos, R.; et al. Efficacy of Functional Remediation in Bipolar Disorder: A Multicenter Randomized Controlled Study. Am. J. Psychiatry 2013, 170, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Demant, K.M.; Vinberg, M.; Kessing, L.V.; Miskowiak, K.W. Effects of Short-Term Cognitive Remediation on Cognitive Dysfunction in Partially or Fully Remitted Individuals with Bipolar Disorder: Results of a Randomised Controlled Trial. PLoS ONE 2015, 10, e0127955. [Google Scholar] [CrossRef] [Green Version]
- Ott, C.V.; Vinberg, M.; Kessing, L.V.; Bowie, C.R.; Forman, J.L.; Miskowiak, K.W. Effect of Action-Based Cognitive Remediation on Cognitive Impairment in Patients with Remitted Bipolar Disorder: A Randomized Controlled Trial. Bipolar Disord. 2020, 23, 487–499. [Google Scholar] [CrossRef]
- Lewandowski, K.E.; Sperry, S.H.; Cohen, B.M.; Norris, L.A.; Fitzmaurice, G.M.; Ongur, D.; Keshavan, M.S. Treatment to Enhance Cognition in Bipolar Disorder (TREC-BD): Efficacy of a Randomized Controlled Trial of Cognitive Remediation Versus Active Control. J. Clin. Psychiatry 2017, 78, e1242–e1249. [Google Scholar] [CrossRef] [Green Version]
- Cortese, S.; Ferrin, M.; Brandeis, D.; Buitelaar, J.; Daley, D.; Dittmann, R.W.; Holtmann, M.; Santosh, P.; Stevenson, J.; Stringaris, A.; et al. Cognitive Training for Attention-Deficit/Hyperactivity Disorder: Meta-Analysis of Clinical and Neuropsychological Outcomes from Randomized Controlled Trials. J. Am. Acad. Child Adolesc. Psychiatry 2015, 54, 164–174. [Google Scholar] [CrossRef] [Green Version]
- Lambez, B.; Harwood-Gross, A.; Golumbic, E.Z.; Rassovsky, Y. Non-Pharmacological Interventions for Cognitive Difficulties in ADHD: A Systematic Review and Meta-Analysis. J. Psychiatr. Res. 2020, 120, 40–55. [Google Scholar] [CrossRef]
- Dovis, S.; der Oord, S.V.; Wiers, R.W.; Prins, P.J.M. Improving Executive Functioning in Children with ADHD: Training Multiple Executive Functions within the Context of a Computer Game. A Randomized Double-Blind Placebo Controlled Trial. PLoS ONE 2015, 10, e0121651. [Google Scholar] [CrossRef]
- Veloso, A.; Vicente, S.G.; Filipe, M.G. Effectiveness of Cognitive Training for School-Aged Children and Adolescents with Attention Deficit/Hyperactivity Disorder: A Systematic Review. Front. Psychol. 2020, 10, 2983. [Google Scholar] [CrossRef] [Green Version]
- Rapport, M.D.; Orban, S.A.; Kofler, M.J.; Friedman, L.M. Do Programs Designed to Train Working Memory, Other Executive Functions, and Attention Benefit Children with ADHD? A Meta-Analytic Review of Cognitive, Academic, and Behavioral Outcomes. Clin. Psychol. Rev. 2013, 33, 1237–1252. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.R.; Katz, B.; Buschkuehl, M.; Jaeggi, S.M.; Shah, P. Exploring N-Back Cognitive Training for Children with ADHD. J. Atten. Disord. 2020, 24, 704–719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyer, K.N.; Santillana, R.; Miller, B.; Clapp, W.; Way, M.; Bridgman-Goines, K.; Sheridan, M.A. Computer-Based Inhibitory Control Training in Children with Attention-Deficit/Hyperactivity Disorder (ADHD): Evidence for Behavioral and Neural Impact. PLoS ONE 2020, 15, e0241352. [Google Scholar] [CrossRef]
- Al-Saad, M.S.H.; Al-Jabri, B.; Almarzouki, A.F. A Review of Working Memory Training in the Management of Attention Deficit Hyperactivity Disorder. Front. Behav. Neurosci. 2021, 15, 686873. [Google Scholar] [CrossRef] [PubMed]
- Bühren, K.; Mainz, V.; Herpertz-Dahlmann, B.; Schäfer, K.; Kahraman-Lanzerath, B.; Lente, C.; Konrad, K. Cognitive Flexibility in Juvenile Anorexia Nervosa Patients before and after Weight Recovery. J. Neural Transm. 2012, 119, 1047–1057. [Google Scholar] [CrossRef]
- Danner, U.N.; Sanders, N.; Smeets, P.A.M.; van Meer, F.; Adan, R.A.H.; Hoek, H.W.; van Elburg, A.A. Neuropsychological Weaknesses in Anorexia Nervosa: Set-Shifting, Central Coherence, and Decision Making in Currently Ill and Recovered Women. Int. J. Eat. Disord. 2012, 45, 685–694. [Google Scholar] [CrossRef]
- Hagan, K.E.; Christensen, K.A.; Forbush, K.T. A Preliminary Systematic Review and Meta-Analysis of Randomized-Controlled Trials of Cognitive Remediation Therapy for Anorexia Nervosa. Eat. Behav. 2020, 37, 101391. [Google Scholar] [CrossRef]
- Tchanturia, K.; Giombini, L.; Leppanen, J.; Kinnaird, E. Evidence for Cognitive Remediation Therapy in Young People with Anorexia Nervosa: Systematic Review and Meta-Analysis of the Literature. Eur. Eat. Disord. Rev. 2017, 25, 227–236. [Google Scholar] [CrossRef] [Green Version]
- Brockmeyer, T.; Schmidt, H.; Leiteritz-Rausch, A.; Zimmermann, J.; Wünsch-Leiteritz, W.; Leiteritz, A.; Friederich, H.-C. Cognitive Remediation Therapy in Anorexia Nervosa-A Randomized Clinical Trial. J. Consult. Clin. Psychol. 2021, 89, 805–815. [Google Scholar] [CrossRef]
- van Passel, B.; Danner, U.N.; Dingemans, A.E.; Aarts, E.; Sternheim, L.C.; Becker, E.S.; van Elburg, A.A.; van Furth, E.F.; Hendriks, G.-J.; Cath, D.C. Cognitive Remediation Therapy Does Not Enhance Treatment Effect in Obsessive-Compulsive Disorder and Anorexia Nervosa: A Randomized Controlled Trial. Psychother. Psychosom. 2020, 89, 228–241. [Google Scholar] [CrossRef]
- Gates, N.J.; Vernooij, R.W.; Di Nisio, M.; Karim, S.; March, E.; Martínez, G.; Rutjes, A.W. Computerised Cognitive Training for Preventing Dementia in People with Mild Cognitive Impairment. Cochrane Database Syst. Rev. 2019, 3, CD012279. [Google Scholar] [CrossRef] [PubMed]
- Marshall, G.A.; Rentz, D.M.; Frey, M.T.; Locascio, J.J.; Johnson, K.A.; Sperling, R.A. Alzheimer’s Disease Neuroimaging Initiative Executive Function and Instrumental Activities of Daily Living in Mild Cognitive Impairment and Alzheimer’s Disease. Alzheimers Dement. 2011, 7, 300–308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, F.; Heffner, K.L.; Ren, P.; Tivarus, M.E.; Brasch, J.; Chen, D.-G.; Mapstone, M.; Porsteinsson, A.P.; Tadin, D. Cognitive and Neural Effects of Vision-Based Speed-of-Processing Training in Older Adults with Amnestic Mild Cognitive Impairment: A Pilot Study. J. Am. Geriatr. Soc. 2016, 64, 1293–1298. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sitzer, D.I.; Twamley, E.W.; Jeste, D.V. Cognitive Training in Alzheimer’s Disease: A Meta-Analysis of the Literature. Acta Psychiatr. Scand. 2006, 114, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Aguirre, E.; Woods, R.T.; Spector, A.; Orrell, M. Cognitive Stimulation for Dementia: A Systematic Review of the Evidence of Effectiveness from Randomised Controlled Trials. Ageing Res. Rev. 2013, 12, 253–262. [Google Scholar] [CrossRef]
- Reeder, C.; Huddy, V.; Cella, M.; Taylor, R.; Greenwood, K.; Landau, S.; Wykes, T. A New Generation Computerised Metacognitive Cognitive Remediation Programme for Schizophrenia (CIRCuiTS): A Randomised Controlled Trial. Psychol. Med. 2017, 47, 2720–2730. [Google Scholar] [CrossRef] [Green Version]
- Minzenberg, M.J.; Laird, A.R.; Thelen, S.; Carter, C.S.; Glahn, D.C. Meta-Analysis of 41 Functional Neuroimaging Studies of Executive Function in Schizophrenia. Arch. Gen. Psychiatry 2009, 66, 811–822. [Google Scholar] [CrossRef] [Green Version]
- Trapp, W.; Dotterweich, S.; Hintner, L.; Wollny, H.; Lautenbacher, S.; Hajak, G. Speed and Capacity of Working Memory and Executive Function in Schizophrenia Compared to Unipolar Depression. Schizophr. Res. Cogn. 2017, 10, 1–6. [Google Scholar] [CrossRef]
- Best, M.W.; Milanovic, M.; Iftene, F.; Bowie, C.R. A Randomized Controlled Trial of Executive Functioning Training Compared with Perceptual Training for Schizophrenia Spectrum Disorders: Effects on Neurophysiology, Neurocognition, and Functioning. Am. J. Psychiatry 2019, 176, 297–306. [Google Scholar] [CrossRef]
- Medalia, A.; Saperstein, A.M.; Qian, M.; Javitt, D.C. Impact of Baseline Early Auditory Processing on Response to Cognitive Remediation for Schizophrenia. Schizophr. Res. 2019, 208, 397–405. [Google Scholar] [CrossRef]
- Vinogradov, S. Has the Time Come for Cognitive Remediation in Schizophrenia…Again? AJP 2019, 176, 262–264. [Google Scholar] [CrossRef] [PubMed]
- Sandoval, L.R.; González, B.L.; Stone, W.S.; Guimond, S.; Rivas, C.T.; Sheynberg, D.; Kuo, S.S.; Eack, S.; Keshavan, M.S. Effects of Peer Social Interaction on Performance during Computerized Cognitive Remediation Therapy in Patients with Early Course Schizophrenia: A Pilot Study. Schizophr. Res. 2019, 203, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Trapp, W.; Engel, S.; Hajak, G.; Lautenbacher, S.; Gallhofer, B. Cognitive Remediation for Depressed Inpatients: Results of a Pilot Randomized Controlled Trial. Aust. N. Z. J. Psychiatry 2016, 50, 46–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trapp, W.; Hasmann, A.; Gallhofer, B.; Schwerdtner, J.; Guenther, W.; Dobmeier, M. Cognitive Improvement of Schizophrenia Patients: Enhancing Cognition While Enjoying Computer-Aided Cognitive Training. Clin. Schizophr. Relat. Psychoses 2007, 2, 307–316. [Google Scholar] [CrossRef]
- Trapp, W.; Kilic, O.; Röder, S.; Lautenbacher, S.; Hajak, G. The Bamberg Tablet-Based Training Battery (BTTB) Pilot Data Regarding Attractiveness, Validity and Usability of a Cognitive Stimulation App for Dementia Patients (Preprint). JMIR, 2019; preprint. [Google Scholar]
- Falkai, P.; Malchow, B.; Schmitt, A. Aerobic Exercise and Its Effects on Cognition in Schizophrenia. Curr. Opin Psychiatry 2017, 30, 171–175. [Google Scholar] [CrossRef]
- Firth, J.; Stubbs, B.; Rosenbaum, S.; Vancampfort, D.; Malchow, B.; Schuch, F.; Elliott, R.; Nuechterlein, K.H.; Yung, A.R. Aerobic Exercise Improves Cognitive Functioning in People with Schizophrenia: A Systematic Review and Meta-Analysis. Schizophr. Bull. 2017, 43, 546–556. [Google Scholar] [CrossRef] [Green Version]
- Greer, T.L.; Furman, J.L.; Trivedi, M.H. Evaluation of the Benefits of Exercise on Cognition in Major Depressive Disorder. Gen. Hosp. Psychiatry 2017, 49, 19–25. [Google Scholar] [CrossRef]
- Ströhle, A.; Schmidt, D.K.; Schultz, F.; Fricke, N.; Staden, T.; Hellweg, R.; Priller, J.; Rapp, M.A.; Rieckmann, N. Drug and Exercise Treatment of Alzheimer Disease and Mild Cognitive Impairment: A Systematic Review and Meta-Analysis of Effects on Cognition in Randomized Controlled Trials. Am. J. Geriatr. Psychiatry 2015, 23, 1234–1249. [Google Scholar] [CrossRef]
- Cotman, C.W.; Berchtold, N.C. Exercise: A Behavioral Intervention to Enhance Brain Health and Plasticity. Trends Neurosci. 2002, 25, 295–301. [Google Scholar] [CrossRef]
- Eriksson, P.S.; Perfilieva, E.; Björk-Eriksson, T.; Alborn, A.-M.; Nordborg, C.; Peterson, D.A.; Gage, F.H. Neurogenesis in the Adult Human Hippocampus. Nat. Med. 1998, 4, 1313–1317. [Google Scholar] [CrossRef] [PubMed]
- van Praag, H. Exercise and the Brain: Something to Chew On. Trends Neurosci. 2009, 32, 283–290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ainslie, P.N.; Cotter, J.D.; George, K.P.; Lucas, S.; Murrell, C.; Shave, R.; Thomas, K.N.; Williams, M.J.A.; Atkinson, G. Elevation in Cerebral Blood Flow Velocity with Aerobic Fitness throughout Healthy Human Ageing. J. Physiol. 2008, 586, 4005–4010. [Google Scholar] [CrossRef] [PubMed]
- Oertel-Knöchel, V.; Mehler, P.; Thiel, C.; Steinbrecher, K.; Malchow, B.; Tesky, V.; Ademmer, K.; Prvulovic, D.; Banzer, W.; Zopf, Y.; et al. Effects of Aerobic Exercise on Cognitive Performance and Individual Psychopathology in Depressive and Schizophrenia Patients. Eur. Arch. Psychiatry Clin. Neurosci. 2014, 264, 589–604. [Google Scholar] [CrossRef]
- Nuechterlein, K.H.; Ventura, J.; McEwen, S.C.; Gretchen-Doorly, D.; Vinogradov, S.; Subotnik, K.L. Enhancing Cognitive Training Through Aerobic Exercise After a First Schizophrenia Episode: Theoretical Conception and Pilot Study. Schizophr. Bull. 2016, 42 (Suppl. 1), S44–S52. [Google Scholar] [CrossRef]
- Malchow, B.; Keller, K.; Hasan, A.; Dörfler, S.; Schneider-Axmann, T.; Hillmer-Vogel, U.; Honer, W.G.; Schulze, T.G.; Niklas, A.; Wobrock, T.; et al. Effects of Endurance Training Combined with Cognitive Remediation on Everyday Functioning, Symptoms, and Cognition in Multiepisode Schizophrenia Patients. Schizophr. Bull. 2015, 41, 847–858. [Google Scholar] [CrossRef] [Green Version]
- Torre, M.M.; Temprado, J.-J. Effects of Exergames on Brain and Cognition in Older Adults: A Review Based on a New Categorization of Combined Training Intervention. Front. Aging Neurosci. 2022, 14, 859715. [Google Scholar] [CrossRef]
- Swinnen, N.; Vandenbulcke, M.; de Bruin, E.D.; Akkerman, R.; Stubbs, B.; Firth, J.; Vancampfort, D. The Efficacy of Exergaming in People with Major Neurocognitive Disorder Residing in Long-Term Care Facilities: A Pilot Randomized Controlled Trial. Alzheimers Res. Ther. 2021, 13, 70. [Google Scholar] [CrossRef]
- Carl, E.; Stein, A.T.; Levihn-Coon, A.; Pogue, J.R.; Rothbaum, B.; Emmelkamp, P.; Asmundson, G.J.G.; Carlbring, P.; Powers, M.B. Virtual Reality Exposure Therapy for Anxiety and Related Disorders: A Meta-Analysis of Randomized Controlled Trials. J. Anxiety Disord. 2019, 61, 27–36. [Google Scholar] [CrossRef]
- Keller, N.E.; Hennings, A.C.; Dunsmoor, J.E. Behavioral and Neural Processes in Counterconditioning: Past and Future Directions. Behav. Res. Ther. 2020, 125, 103532. [Google Scholar] [CrossRef]
- Nakagami, E.; Hoe, M.; Brekke, J.S. The Prospective Relationships Among Intrinsic Motivation, Neurocognition, and Psychosocial Functioning in Schizophrenia. Schizophr. Bull. 2010, 36, 935–948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silverstein, S.M. Bridging the Gap Between Extrinsic and Intrinsic Motivation in the Cognitive Remediation of Schizophrenia. Schizophr. Bull. 2010, 36, 949–956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miley, K.; Fisher, M.; Nahum, M.; Howard, E.; Rowlands, A.; Brandrett, B.; Woolley, J.; Hooker, C.I.; Biagianti, B.; Ramsay, I.; et al. Six Month Durability of Targeted Cognitive Training Supplemented with Social Cognition Exercises in Schizophrenia. Schizophr. Res. Cogn. 2020, 20, 100171. [Google Scholar] [CrossRef] [PubMed]
- Lumsden, J.; Edwards, E.A.; Lawrence, N.S.; Coyle, D.; Munafò, M.R. Gamification of Cognitive Assessment and Cognitive Training: A Systematic Review of Applications and Efficacy. JMIR Serious Games 2016, 4, e5888. [Google Scholar] [CrossRef] [Green Version]
- Young, D.A.; Freyslinger, M.G. Scaffolded Instruction and the Remediation of Wisconsin Card Sorting Test Deficits in Chronic Schizophrenia. Schizophr. Res. 1995, 16, 199–207. [Google Scholar] [CrossRef]
- Baddeley, A. Working Memory. Science 1992, 255, 556–559. [Google Scholar] [CrossRef]
- Baddeley, A. The Episodic Buffer: A New Component of Working Memory? Trends Cogn. Sci. 2000, 4, 417–423. [Google Scholar] [CrossRef]
- Beck, A.T. The Evolution of the Cognitive Model of Depression and Its Neurobiological Correlates. Am. J. Psychiatry 2008, 165, 969–977. [Google Scholar] [CrossRef]
- Beck, A.T.; Brown, G.; Steer, R.A.; Eidelson, J.I.; Riskind, J.H. Differentiating Anxiety and Depression: A Test of the Cognitive Content-Specificity Hypothesis. J. Abnorm. Psychol. 1987, 96, 179–183. [Google Scholar] [CrossRef]
- Bower, G.H. Mood and Memory. Am. Psychol. 1981, 36, 129–148. [Google Scholar] [CrossRef]
- Roiser, J.P.; Sahakian, B.J. Hot and Cold Cognition in Depression. CNS Spectr. 2013, 18, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Moritz, S.; Gläscher, J.; Brassen, S. Investigation of Mood-Congruent False and True Memory Recognition in Depression. Depress. Anxiety 2005, 21, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Matt, G.E.; Vázquez, C.; Campbell, W.K. Mood-Congruent Recall of Affectively Toned Stimuli: A Meta-Analytic Review. Clin. Psychol. Rev. 1992, 12, 227–255. [Google Scholar] [CrossRef]
- Wells, T.T.; Beevers, C.G. Biased Attention and Dysphoria: Manipulating Selective Attention Reduces Subsequent Depressive Symptoms. Cogn. Emot. 2010, 24, 719–728. [Google Scholar] [CrossRef]
- Baert, S.; De Raedt, R.; Schacht, R.; Koster, E.H.W. Attentional Bias Training in Depression: Therapeutic Effects Depend on Depression Severity. J. Behav. Ther. Exp. Psychiatry 2010, 41, 265–274. [Google Scholar] [CrossRef]
- De Raedt, R.; Koster, E.H.W. Understanding Vulnerability for Depression from a Cognitive Neuroscience Perspective: A Reappraisal of Attentional Factors and a New Conceptual Framework. Cogn. Affect. Behav. Neurosci. 2010, 10, 50–70. [Google Scholar] [CrossRef] [Green Version]
- De Raedt, R.; Koster, E.H.W.; Joormann, J. Attentional Control in Depression: A Translational Affective Neuroscience Approach. Cogn. Affect. Behav. Neurosci. 2010, 10, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Li, X.; Han, B.; Liu, X. Effects of Cognitive Bias Modification on Social Anxiety: A Meta-Analysis. PLoS ONE 2017, 12, e0175107. [Google Scholar] [CrossRef]
- Hang, Y.; Xu, L.; Wang, C.; Zhang, G.; Zhang, N. Can Attention Bias Modification Augment the Effect of CBT for Anxiety Disorders? A Systematic Review and Meta-Analysis. Psychiatry Res. 2021, 299, 113892. [Google Scholar] [CrossRef]
- Basanovic, J.; Grafton, B.; Ford, A.; Hirani, V.; Glance, D.; MacLeod, C.; Almeida, O.P. Cognitive Bias Modification to Prevent Depression (COPE): Results of a Randomised Controlled Trial. Psychol. Med. 2020, 50, 2514–2525. [Google Scholar] [CrossRef] [Green Version]
- Cristea, I.A.; Kok, R.N.; Cuijpers, P. Efficacy of Cognitive Bias Modification Interventions in Anxiety and Depression: Meta-Analysis. Br. J. Psychiatry 2015, 206, 7–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woolridge, S.M.; Harrison, G.W.; Best, M.W.; Bowie, C.R. Attention Bias Modification in Depression: A Randomized Trial Using a Novel, Reward-Based, Eye-Tracking Approach. J. Behav. Ther. Exp. Psychiatry 2021, 71, 101621. [Google Scholar] [CrossRef] [PubMed]
- McDermott, L.M.; Ebmeier, K.P. A Meta-Analysis of Depression Severity and Cognitive Function. J. Affect. Disord. 2009, 119, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Nuechterlein, K.H.; Ventura, J.; Subotnik, K.L.; Gretchen-Doorly, D.; Turner, L.R.; Casaus, L.R.; Luo, J.; Boucher, M.L.; Hayata, J.N.; Bell, M.D.; et al. A Randomized Controlled Trial of Cognitive Remediation and Long-Acting Injectable Risperidone after a First Episode of Schizophrenia: Improving Cognition and Work/School Functioning. Psychol. Med. 2020, 1–10. [Google Scholar] [CrossRef]
- Fiske, S.T.; Taylor, S.E. Social Cognition, 2nd ed.; Mcgraw-Hill Book Company: New York, NY, USA; London, UK, 1991; Volume xviii, p. 717. ISBN 978-0-07-021191-9. [Google Scholar]
- Green, M.F.; Horan, W.P.; Lee, J. Social Cognition in Schizophrenia. Nat. Rev. Neurosci. 2015, 16, 620–631. [Google Scholar] [CrossRef]
- de Jong, J.J.; de Gelder, B.; Hodiamont, P.P.P.G. Sensory Processing, Neurocognition, and Social Cognition in Schizophrenia: Towards a Cohesive Cognitive Model. Schizophr. Res. 2013, 146, 209–216. [Google Scholar] [CrossRef]
- Weightman, M.J.; Air, T.M.; Baune, B.T. A Review of the Role of Social Cognition in Major Depressive Disorder. Front. Psychiatry 2014, 5, 179. [Google Scholar] [CrossRef] [Green Version]
- Javed, A.; Charles, A. The Importance of Social Cognition in Improving Functional Outcomes in Schizophrenia. Front. Psychiatry 2018, 9, 157. [Google Scholar] [CrossRef]
- Rose, A.; Vinogradov, S.; Fisher, M.; Green, M.F.; Ventura, J.; Hooker, C.; Merzenich, M.; Nahum, M. Randomized Controlled Trial of Computer-Based Treatment of Social Cognition in Schizophrenia: The TRuSST Trial Protocol. BMC Psychiatry 2015, 15, 142. [Google Scholar] [CrossRef] [Green Version]
- Nahum, M.; Fisher, M.; Loewy, R.; Poelke, G.; Ventura, J.; Nuechterlein, K.H.; Hooker, C.I.; Green, M.F.; Merzenich, M.; Vinogradov, S. A Novel, Online Social Cognitive Training Program for Young Adults with Schizophrenia: A Pilot Study. Schizophr. Res. Cogn. 2014, 1, e11–e19. [Google Scholar] [CrossRef] [Green Version]
- Biagianti, B.; Schlosser, D.; Nahum, M.; Woolley, J.; Vinogradov, S. Creating Live Interactions to Mitigate Barriers (CLIMB): A Mobile Intervention to Improve Social Functioning in People with Chronic Psychotic Disorders. JMIR Ment. Health 2016, 3, e52. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.; Bassett, E.; Iftene, F.; Bowie, C.R. Functional Outcomes in Schizophrenia: Understanding the Competence-Performance Discrepancy. J. Psychiatr. Res. 2012, 46, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Nemoto, T.; Uchino, T.; Aikawa, S.; Saito, J.; Matsumoto, H.; Funatogawa, T.; Yamaguchi, T.; Katagiri, N.; Tsujino, N.; Mizuno, M. Social Anxiety and Negative Symptoms as the Characteristics of Patients with Schizophrenia Who Show Competence-Performance Discrepancy in Social Functioning. Psychiatry Clin. Neurosci. 2019, 73, 394–399. [Google Scholar] [CrossRef] [PubMed]
- MEDALIA, A.; FREILICH, B. The Neuropsychological Educational Approach to Cognitive Remediation (NEAR) Model: Practice Principles and Outcome Studies. Am. J. Psychiatr. Rehabil. 2008, 11, 123–143. [Google Scholar] [CrossRef]
- Danion, J.M.; Meulemans, T.; Kauffmann-Muller, F.; Vermaat, H. Intact Implicit Learning in Schizophrenia. Am. J. Psychiatry 2001, 158, 944–948. [Google Scholar] [CrossRef]
- Harrison, B.E.; Son, G.-R.; Kim, J.; Whall, A.L. Preserved Implicit Memory in Dementia: A Potential Model for Care. Am. J. Alzheimers Dis. Other Dement. 2007, 22, 286–293. [Google Scholar] [CrossRef]
- Vauth, R.; Corrigan, P.W.; Clauss, M.; Dietl, M.; Dreher-Rudolph, M.; Stieglitz, R.-D.; Vater, R. Cognitive Strategies versus Self-Management Skills as Adjunct to Vocational Rehabilitation. Schizophr. Bull. 2005, 31, 55–66. [Google Scholar] [CrossRef]
- Silverstein, S.M.; Hatashita-Wong, M.; Solak, B.A.; Uhlhaas, P.; Landa, Y.; Wilkniss, S.M.; Goicochea, C.; Carpiniello, K.; Schenkel, L.S.; Savitz, A.; et al. Effectiveness of a Two-Phase Cognitive Rehabilitation Intervention for Severely Impaired Schizophrenia Patients. Psychol. Med. 2005, 35, 829–837. [Google Scholar] [CrossRef]
- Simons, J.S.; Spiers, H.J. Prefrontal and Medial Temporal Lobe Interactions in Long-Term Memory. Nat. Rev. Neurosci. 2003, 4, 637–648. [Google Scholar] [CrossRef]
- Dunning, D.L.; Holmes, J. Does Working Memory Training Promote the Use of Strategies on Untrained Working Memory Tasks? Mem. Cogn. 2014, 42, 854–862. [Google Scholar] [CrossRef] [Green Version]
- Davies, G.; Fowler, D.; Greenwood, K. Metacognition as a Mediating Variable Between Neurocognition and Functional Outcome in First Episode Psychosis. Schizophr. Bull. 2017, 43, 824–832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flavell, J.H. Metacognition and Cognitive Monitoring: A New Area of Cognitive–Developmental Inquiry. Am. Psychol. 1979, 34, 906–911. [Google Scholar] [CrossRef]
- Cella, M.; Reeder, C.; Wykes, T. Lessons Learnt? The Importance of Metacognition and Its Implications for Cognitive Remediation in Schizophrenia. Front. Psychol. 2015, 6, 1259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cella, M.; Edwards, C.; Swan, S.; Elliot, K.; Reeder, C.; Wykes, T. Exploring the Effects of Cognitive Remediation on Metacognition in People with Schizophrenia. J. Exp. Psychopathol. 2019, 10, 2043808719826846. [Google Scholar] [CrossRef] [Green Version]
- Bowie, C.R.; Grossman, M.; Gupta, M.; Holshausen, K.; Best, M.W. Action-Based Cognitive Remediation for Individuals with Serious Mental Illnesses: Effects of Real-World Simulations and Goal Setting on Functional and Vocational Outcomes. Psychiatr. Rehabil. J. 2017, 40, 53–60. [Google Scholar] [CrossRef]
- Unity Real-Time Development Platform|3D, 2D VR & AR Engine. Available online: https://unity.com/ (accessed on 6 December 2021).
- Unreal Engine. The Most Powerful Real-Time 3D Creation Tool. Available online: https://www.unrealengine.com/en-US/ (accessed on 6 December 2021).
- Trapp, W. “Ladies and Gentlemen, Please Start Your Engines!”: Why You Should Gamify Your Cognitive Stimulation Interventions for Dementia Patients. Int. Psychogeriatr. 2022, 34, 101–103. [Google Scholar] [CrossRef]
- Ahmed, A.O.; Hunter, K.M.; Goodrum, N.M.; Batten, N.-J.; Birgenheir, D.; Hardison, E.; Dixon, T.; Buckley, P.F. A Randomized Study of Cognitive Remediation for Forensic and Mental Health Patients with Schizophrenia. J. Psychiatr. Res. 2015, 68, 8–18. [Google Scholar] [CrossRef]
- O’Reilly, K.; Donohoe, G.; O’Sullivan, D.; Coyle, C.; Corvin, A.; O’Flynn, P.; O’Donnell, M.; Galligan, T.; O’Connell, P.; Kennedy, H.G. A Randomized Controlled Trial of Cognitive Remediation for a National Cohort of Forensic Patients with Schizophrenia or Schizoaffective Disorder. BMC Psychiatry 2019, 19, 27. [Google Scholar] [CrossRef] [Green Version]
- Cella, M.; Preti, A.; Edwards, C.; Dow, T.; Wykes, T. Cognitive Remediation for Negative Symptoms of Schizophrenia: A Network Meta-Analysis. Clin. Psychol. Rev. 2017, 52, 43–51. [Google Scholar] [CrossRef] [Green Version]
- Cella, M.; Bishara, A.J.; Medin, E.; Swan, S.; Reeder, C.; Wykes, T. Identifying Cognitive Remediation Change through Computational Modelling—Effects on Reinforcement Learning in Schizophrenia. Schizophr. Bull. 2014, 40, 1422–1432. [Google Scholar] [CrossRef]
- Medalia, A.; Saperstein, A. The Role of Motivation for Treatment Success. Schizophr. Bull. 2011, 37 (Suppl. 2), S122–S128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorenz, E.N. The Essence of Chaos; UCL Press: London, UK, 1993; ISBN 978-1-85728-187-3. [Google Scholar]
- Green, M.F. Cognitive Remediation in Schizophrenia: Is It Time Yet? Am. J. Psychiatry 1993, 150, 178–187. [Google Scholar] [CrossRef] [PubMed]
- WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int (accessed on 13 September 2021).
- Søraas, A.; Bø, R.; Kalleberg, K.T.; Støer, N.C.; Ellingjord-Dale, M.; Landrø, N.I. Self-Reported Memory Problems 8 Months After COVID-19 Infection. JAMA Netw. Open 2021, 4, e2118717. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Leon, S.; Wegman-Ostrosky, T.; Perelman, C.; Sepulveda, R.; Rebolledo, P.A.; Cuapio, A.; Villapol, S. More than 50 Long-Term Effects of COVID-19: A Systematic Review and Meta-Analysis. Sci. Rep. 2021, 11, 16144. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Trapp, W.; Heid, A.; Röder, S.; Wimmer, F.; Hajak, G. Cognitive Remediation in Psychiatric Disorders: State of the Evidence, Future Perspectives, and Some Bold Ideas. Brain Sci. 2022, 12, 683. https://doi.org/10.3390/brainsci12060683
Trapp W, Heid A, Röder S, Wimmer F, Hajak G. Cognitive Remediation in Psychiatric Disorders: State of the Evidence, Future Perspectives, and Some Bold Ideas. Brain Sciences. 2022; 12(6):683. https://doi.org/10.3390/brainsci12060683
Chicago/Turabian StyleTrapp, Wolfgang, Andreas Heid, Susanne Röder, Franziska Wimmer, and Göran Hajak. 2022. "Cognitive Remediation in Psychiatric Disorders: State of the Evidence, Future Perspectives, and Some Bold Ideas" Brain Sciences 12, no. 6: 683. https://doi.org/10.3390/brainsci12060683
APA StyleTrapp, W., Heid, A., Röder, S., Wimmer, F., & Hajak, G. (2022). Cognitive Remediation in Psychiatric Disorders: State of the Evidence, Future Perspectives, and Some Bold Ideas. Brain Sciences, 12(6), 683. https://doi.org/10.3390/brainsci12060683




