Repetitive Neuromuscular Magnetic Stimulation for Pediatric Headache Disorders: Muscular Effects and Factors Affecting Level of Response
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Study Design
2.3. rNMS Intervention
2.4. PPT Assessments
2.5. Headache Characteristics
2.6. Data Management
2.7. Statistical Analysis
3. Results
3.1. Subjects
3.2. Patient and Treatment Characteristics
3.3. Pressure Pain Thresholds
3.4. Headache Characteristics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Albers, L.; von Kries, R.; Heinen, F.; Straube, A. Headache in school children: Is the prevalence increasing? Curr. Pain Headache Rep. 2015, 19, 4. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Lim, S.S.; Abbafati, C.; Abbas, K.M.; Abbasi, M.; Abbasifard, M.; Abbasi-Kangevari, M.; Abbastabar, H.; Abd-Allah, F.; Abdelalim, A.; et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Youssef, P.E.; Mack, K.J. Episodic and chronic migraine in children. Dev. Med. Child Neurol. 2020, 62, 34–41. [Google Scholar] [CrossRef]
- Abu-Arafeh, I.; Razak, S.; Sivaraman, B.; Graham, C. Prevalence of headache and migraine in children and adolescents: A systematic review of population-based studies. Dev. Med. Child Neurol. 2010, 52, 1088–1097. [Google Scholar] [CrossRef] [PubMed]
- Stovner, L.; Hagen, K.; Jensen, R.; Katsarava, Z.; Lipton, R.; Scher, A.; Steiner, T.; Zwart, J.A. The global burden of headache: A documentation of headache prevalence and disability worldwide. Cephalalgia 2007, 27, 193–210. [Google Scholar] [CrossRef]
- Bonfert, M.; Straube, A.; Schroeder, A.S.; Reilich, P.; Ebinger, F.; Heinen, F. Primary headache in children and adolescents: Update on pharmacotherapy of migraine and tension-type headache. Neuropediatrics 2013, 44, 3–19. [Google Scholar] [CrossRef] [Green Version]
- Rho, Y.I.; Chung, H.J.; Lee, K.H.; Eun, B.L.; Eun, S.H.; Nam, S.O.; Kim, W.S.; Kim, Y.O.; Park, H.J.; Kim, H.S. Prevalence and clinical characteristics of primary headaches among school children in South Korea: A nationwide survey. Headache 2012, 52, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Jensen, R.H. Tension-Type Headache—The Normal and Most Prevalent Headache. Headache 2018, 58, 339–345. [Google Scholar] [CrossRef]
- Seshia, S.S. Mixed migraine and tension-type: A common cause of recurrent headache in children. Can. J. Neurol. Sci. 2004, 31, 315–318. [Google Scholar] [CrossRef] [Green Version]
- Kuczynski, A.; Crawford, S.; Bodell, L.; Dewey, D.; Barlow, K.M. Characteristics of post-traumatic headaches in children following mild traumatic brain injury and their response to treatment: A prospective cohort. Dev. Med. Child Neurol. 2013, 55, 636–641. [Google Scholar] [CrossRef]
- McEvoy, H.; Borsook, D.; Holmes, S.A. Clinical features and sex differences in pediatric post-traumatic headache: A retrospective chart review at a Boston area concussion clinic. Cephalalgia 2020, 40, 701–711. [Google Scholar] [CrossRef] [PubMed]
- Howard, L.; Schwedt, T.J. Posttraumatic headache: Recent progress. Curr. Opin. Neurol. 2020, 33, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, M.A.; Meehan, W.P., 3rd; Mannix, R. Duration and course of post-concussive symptoms. Pediatrics 2014, 133, 999–1006. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. The ICD-10 Classification of Mental and Behavioural Disorders: Diagnostic Criteria For Research; World Health Organization: Geneva, Switzerland, 1993. [Google Scholar]
- Bartsch, T.; Goadsby, P.J. The trigeminocervical complex and migraine: Current concepts and synthesis. Curr. Pain Headache Rep. 2003, 7, 371–376. [Google Scholar] [CrossRef]
- Ashina, M.; Hansen, J.M.; Do, T.P.; Melo-Carrillo, A.; Burstein, R.; Moskowitz, M.A. Migraine and the trigeminovascular system—40 years and counting. Lancet Neurol. 2019, 18, 795–804. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, T.; Goadsby, P.J. Stimulation of the greater occipital nerve induces increased central excitability of dural afferent input. Brain 2002, 125 Pt 7, 1496–1509. [Google Scholar] [CrossRef] [Green Version]
- Busch, V.; Frese, A.; Bartsch, T. The trigemino-cervical complex. Integration of peripheral and central pain mechanisms in primary headache syndromes. Schmerz 2004, 18, 404–410. [Google Scholar] [CrossRef]
- Fernandez-De-Las-Penas, C.; Arendt-Nielsen, L. Improving understanding of trigger points and widespread pressure pain sensitivity in tension-type headache patients: Clinical implications. Expert Rev. Neurother. 2017, 17, 933–939. [Google Scholar] [CrossRef]
- Bartsch, T. Migraine and the neck: New insights from basic data. Curr. Pain Headache Rep. 2005, 9, 191–196. [Google Scholar] [CrossRef]
- Blaschek, A.; Decke, S.; Albers, L.; Schroeder, A.S.; Lehmann, S.; Straube, A.; Landgraf, M.N.; Heinen, F.; von Kries, R. Self-reported neck pain is associated with migraine but not with tension-type headache in adolescents. Cephalalgia 2014, 34, 895–903. [Google Scholar] [CrossRef]
- Landgraf, M.N.; Ertl-Wagner, B.; Koerte, I.K.; Thienel, J.; Langhagen, T.; Straube, A.; von Kries, R.; Reilich, P.; Pomschar, A.; Heinen, F. Alterations in the trapezius muscle in young patients with migraine—A pilot case series with MRI. Eur. J. Paediatr. Neurol. 2015, 19, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Landgraf, M.N.; von Kries, R.; Heinen, F.; Langhagen, T.; Straube, A.; Albers, L. Self-reported neck and shoulder pain in adolescents is associated with episodic and chronic migraine. Cephalalgia 2016, 36, 807–811. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blaschek, A.; Milde-Busch, A.; Straube, A.; Schankin, C.; Langhagen, T.; Jahn, K.; Schroder, S.A.; Reiter, K.; von Kries, R.; Heinen, F. Self-reported muscle pain in adolescents with migraine and tension-type headache. Cephalalgia 2012, 32, 241–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landgraf, M.N.; Biebl, J.T.; Langhagen, T.; Hannibal, I.; Eggert, T.; Vill, K.; Gerstl, L.; Albers, L.; von Kries, R.; Straube, A.; et al. Children with migraine: Provocation of headache via pressure to myofascial trigger points in the trapezius muscle?—A prospective controlled observational study. Eur. J. Pain 2018, 22, 385–392. [Google Scholar] [CrossRef] [PubMed]
- Simons, D.G.; Travell, J.G.; Simons, L.S. Travell & Simons’ Myofascial Pain and Dysfunction: Upper Half of Body; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 1999. [Google Scholar]
- Giamberardino, M.A.; Tafuri, E.; Savini, A.; Fabrizio, A.; Affaitati, G.; Lerza, R.; Di Ianni, L.; Lapenna, D.; Mezzetti, A. Contribution of myofascial trigger points to migraine symptoms. J. Pain 2007, 8, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, X.; Tan, S.; Huang, Q. Understanding of myofascial trigger points. Chin. Med. J. 2014, 127, 4271–4277. [Google Scholar] [PubMed]
- Ferracini, G.N.; Florencio, L.L.; Dach, F.; Grossi, D.B.; Palacios-Ceña, M.; Ordás-Bandera, C.; Chaves, T.C.; Speciali, J.G.; Fernández-de-Las-Peñas, C. Musculoskeletal disorders of the upper cervical spine in women with episodic or chronic migraine. Eur. J. Phys. Rehabil. Med. 2017, 53, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Fernández-de-Las-Peñas, C. Myofascial Head Pain. Curr. Pain Headache Rep. 2015, 19, 28. [Google Scholar] [CrossRef]
- Fernandez-de-las-Penas, C.; Madeleine, P.; Caminero, A.B.; Cuadrado, M.L.; Arendt-Nielsen, L.; Pareja, J.A. Generalized neck-shoulder hyperalgesia in chronic tension-type headache and unilateral migraine assessed by pressure pain sensitivity topographical maps of the trapezius muscle. Cephalalgia 2010, 30, 77–86. [Google Scholar] [CrossRef]
- Fernandez-de-las-Penas, C.; Cuadrado, M.L.; Arendt-Nielsen, L.; Simons, D.G.; Pareja, J.A. Myofascial trigger points and sensitization: An updated pain model for tension-type headache. Cephalalgia 2007, 27, 383–393. [Google Scholar] [CrossRef]
- Luedtke, K.; Starke, W.; May, A. Musculoskeletal dysfunction in migraine patients. Cephalalgia 2018, 38, 865–875. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-de-las-Penas, C.; Alonso-Blanco, C.; Cuadrado, M.L.; Gerwin, R.D.; Pareja, J.A. Trigger points in the suboccipital muscles and forward head posture in tension-type headache. Headache 2006, 46, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Goadsby, P.J.; Holland, P.R.; Martins-Oliveira, M.; Hoffmann, J.; Schankin, C.; Akerman, S. Pathophysiology of Migraine: A Disorder of Sensory Processing. Physiol. Rev. 2017, 97, 553–622. [Google Scholar] [CrossRef] [PubMed]
- Buitenhuis, J.; de Jong, P.J.; Jaspers, J.P.; Groothoff, J.W. Work disability after whiplash: A prospective cohort study. Spine (Phila Pa 1976) 2009, 34, 262–267. [Google Scholar] [CrossRef] [Green Version]
- Pearce, J.M. Headaches in the whiplash syndrome. Spinal Cord 2001, 39, 228–233. [Google Scholar] [CrossRef]
- Ashina, H.; Porreca, F.; Anderson, T.; Amin, F.M.; Ashina, M.; Schytz, H.W.; Dodick, D.W. Post-traumatic headache: Epidemiology and pathophysiological insights. Nat. Rev. Neurol. 2019, 15, 607–617. [Google Scholar] [CrossRef]
- Bonfert, M.V.; Borner, C.; Gerstl, L.; Hannibal, I.; Mathonia, N.; Huss, K.; Rahmsdorf, B.; Kainz, C.; Klose, B.; Koenig, H.; et al. Migraine in childhood and adolescence-neurostimulation as a future innovative approach in terms of a multimodal treatment regimen. Bundesgesundheitsblatt-Gesundh. Gesundh. 2020, 63, 872–880. [Google Scholar] [CrossRef]
- Borner, C.; Urban, G.; Beaulieu, L.D.; Sollmann, N.; Krieg, S.M.; Straube, A.; Renner, T.; Schandelmaier, P.; Lang, M.; Lechner, M.; et al. The bottom-up approach: Non-invasive peripheral neurostimulation methods to treat migraine: A scoping review from the child neurologist’s perspective. Eur. J. Paediatr. Neurol. 2021, 32, 16–28. [Google Scholar] [CrossRef]
- Staisch, J.; Börner, C.; Lang, M.; Hauser, A.; Hannibal, I.; Huß, K.; Klose, B.; Lechner, M.F.; Sollmann, N.; Heinen, F.; et al. Repetitive neuromuscular magnetic stimulation in children with headache. Eur. J. Paediatr. Neurol. 2022, 39, 40–48. [Google Scholar] [CrossRef]
- Smania, N.; Corato, E.; Fiaschi, A.; Pietropoli, P.; Aglioti, S.M.; Tinazzi, M. Repetitive magnetic stimulation: A novel therapeutic approach for myofascial pain syndrome. J. Neurol. 2005, 252, 307–314. [Google Scholar] [CrossRef]
- Pujol, J.; Pascual-Leone, A.; Dolz, C.; Delgado, E.; Dolz, J.L.; Aldomà, J. The effect of repetitive magnetic stimulation on localized musculoskeletal pain. Neuroreport 1998, 9, 1745–1748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartsch, T.; Goadsby, P.J. Central mechanisms of peripheral nerve stimulation in headache disorders. Prog. Neurol. Surg. 2011, 24, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Charles, A. The pathophysiology of migraine: Implications for clinical management. Lancet Neurol. 2018, 17, 174–182. [Google Scholar] [CrossRef]
- ICHD-3 German-Translation-2018. 2018. Available online: https://ichd-3.org/ (accessed on 11 July 2022).
- Elsevier. Buck’s 2020 ICD-10-CM Hospital Edition E-Book; Elsevier Health Sciences: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Renner, T.; Sollmann, N.; Trepte-Freisleder, F.; Albers, L.; Mathonia, N.M.; Bonfert, M.V.; Konig, H.; Klose, B.; Krieg, S.M.; Heinen, F.; et al. Repetitive Peripheral Magnetic Stimulation (rPMS) in Subjects With Migraine-Setup Presentation and Effects on Skeletal Musculature. Front. Neurol. 2019, 10, 738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lacourt, T.E.; Houtveen, J.H.; van Doomen, L.J.P. Experimental pressure-pain Assessments: Test-retest reliability, convergence and dimensionality. Scand. J. Pain 2012, 3, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Barón, J.; Ruiz, M.; Palacios-Ceña, M.; Madeleine, P.; Guerrero, Á.L.; Arendt-Nielsen, L.; Fernández-de-Las-Peñas, C. Differences in Topographical Pressure Pain Sensitivity Maps of the Scalp Between Patients With Migraine and Healthy Controls. Headache 2017, 57, 226–235. [Google Scholar] [CrossRef]
- Florencio, L.L.; Giantomassi, M.C.; Carvalho, G.F.; Gonçalves, M.C.; Dach, F.; Fernández-de-Las-Peñas, C.; Bevilaqua-Grossi, D. Generalized Pressure Pain Hypersensitivity in the Cervical Muscles in Women with Migraine. Pain Med. 2015, 16, 1629–1634. [Google Scholar] [CrossRef] [Green Version]
- Palacios-Cena, M.; Florencio, L.L.; Ferracini, G.N.; Baron, J.; Guerrero, A.L.; Ordas-Bandera, C.; Arendt-Nielsen, L.; Fernandez-de-Las-Penas, C. Women with Chronic and Episodic Migraine Exhibit Similar Widespread Pressure Pain Sensitivity. Pain Med. 2016, 17, 2127–2133. [Google Scholar] [CrossRef] [Green Version]
- Castien, R.F.; van der Wouden, J.C.; De Hertogh, W. Pressure pain thresholds over the cranio-cervical region in headache: A systematic review and meta-analysis. J. Headache Pain 2018, 19, 9. [Google Scholar] [CrossRef] [Green Version]
- Renner, T.; Sollmann, N.; Heinen, F.; Albers, L.; Trepte-Freisleder, F.; Klose, B.; Konig, H.; Krieg, S.M.; Bonfert, M.V.; Landgraf, M.N. Alleviation of migraine symptoms by application of repetitive peripheral magnetic stimulation to myofascial trigger points of neck and shoulder muscles—A randomized trial. Sci. Rep. 2020, 10, 5954. [Google Scholar] [CrossRef]
- Sollmann, N.; Trepte-Freisleder, F.; Albers, L.; Jung, N.H.; Mall, V.; Meyer, B.; Heinen, F.; Krieg, S.M.; Landgraf, M.N. Magnetic stimulation of the upper trapezius muscles in patients with migraine—A pilot study. Eur. J. Paediatr. Neurol. 2016, 20, 888–897. [Google Scholar] [CrossRef] [PubMed]
- Magis, D.; Sava, S.; d’Elia, T.S.; Baschi, R.; Schoenen, J. Safety and patients’ satisfaction of transcutaneous Supraorbital NeuroStimulation (tSNS) with the Cefaly device in headache treatment: A survey of 2,313 headache sufferers in the general population. J. Headache Pain 2013, 14, 95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schoenen, J.; Vandersmissen, B.; Jeangette, S.; Herroelen, L.; Vandenheede, M.; Gérard, P.; Magis, D. Migraine prevention with a supraorbital transcutaneous stimulator: A randomized controlled trial. Neurology 2013, 80, 697–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamed, N.S. Supraorbital electrical stimulation in management of chronic type tension headache: A randomized controlled study. Physiother. Theory Pract. 2018, 34, 101–110. [Google Scholar] [CrossRef]
- Liu, Y.; Dong, Z.; Wang, R.; Ao, R.; Han, X.; Tang, W.; Yu, S. Migraine Prevention Using Different Frequencies of Transcutaneous Occipital Nerve Stimulation: A Randomized Controlled Trial. J. Pain 2017, 18, 1006–1015. [Google Scholar] [CrossRef]
- Bono, F.; Salvino, D.; Mazza, M.R.; Curcio, M.; Trimboli, M.; Vescio, B.; Quattrone, A. The influence of ictal cutaneous allodynia on the response to occipital transcutaneous electrical stimulation in chronic migraine and chronic tension-type headache: A randomized, sham-controlled study. Cephalalgia 2015, 35, 389–398. [Google Scholar] [CrossRef]
- Goadsby, P.J.; Grosberg, B.M.; Mauskop, A.; Cady, R.; Simmons, K.A. Effect of noninvasive vagus nerve stimulation on acute migraine: An open-label pilot study. Cephalalgia 2014, 34, 986–993. [Google Scholar] [CrossRef]
- Grazzi, L.; Egeo, G.; Calhoun, A.H.; McClure, C.K.; Liebler, E.; Barbanti, P. Non-invasive Vagus Nerve Stimulation (nVNS) as mini-prophylaxis for menstrual/menstrually related migraine: An open-label study. J. Headache Pain 2016, 17, 91. [Google Scholar] [CrossRef] [Green Version]
- Lucas, S. Posttraumatic Headache: Clinical Characterization and Management. Curr. Pain Headache Rep. 2015, 19, 48. [Google Scholar] [CrossRef]
- Schneider, C.; Zangrandi, A.; Sollmann, N.; Bonfert, M.V.; Beaulieu, L.D. Checklist on the Quality of the Repetitive Peripheral Magnetic Stimulation (rPMS) Methods in Research: An International Delphi Study. Front. Neurol. 2022, 13, 852848. [Google Scholar] [CrossRef]
- Oskoui, M.; Pringsheim, T.; Holler-Managan, Y.; Potrebic, S.; Billinghurst, L.; Gloss, D.; Hershey, A.D.; Licking, N.; Sowell, M.; Victorio, M.C.; et al. Practice guideline update summary: Acute treatment of migraine in children and adolescents: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology and the American Headache Society. Neurology 2019, 93, 487–499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaptchuk, T.J.; Goldman, P.; Stone, D.A.; Stason, W.B. Do medical devices have enhanced placebo effects? J. Clin. Epidemiol. 2000, 53, 786–792. [Google Scholar] [CrossRef]
- Raucci, U.; Boni, A.; Evangelisti, M.; Della Vecchia, N.; Velardi, M.; Ursitti, F.; Terrin, G.; Di Nardo, G.; Reale, A.; Villani, A.; et al. Lifestyle Modifications to Help Prevent Headache at a Developmental Age. Front. Neurol. 2020, 11, 618375. [Google Scholar] [CrossRef] [PubMed]
- Papetti, L.; Loro, P.A.D.; Tarantino, S.; Grazzi, L.; Guidetti, V.; Parisi, P.; Raieli, V.; Sciruicchio, V.; Termine, C.; Toldo, I.; et al. I stay at home with headache. A survey to investigate how the lockdown for COVID-19 impacted on headache in Italian children. Cephalalgia 2020, 40, 1459–1473. [Google Scholar] [CrossRef] [PubMed]


| Group | n (Patients) | n (Interventions) |
|---|---|---|
| Total sample | 20 | 25 |
| PPT analysis (total sample) | 17 | 22 |
| Headache diagnosis analysis | ||
| Primary headaches | 13 | 14 |
| PTH | 7 | 11 |
| Neck pain analysis | ||
| With neck pain | 13 | 15 |
| Without neck pain | 7 | 10 |
| Treatment time frame analysis | ||
| <2x/week | 8 | 9 |
| 2x/week | 7 | 10 |
| >2x/week | 5 | 6 |
| PPT | Test Values | Mean (SD) | Post-Hoc Test | ||||
|---|---|---|---|---|---|---|---|
| F | p | η2 | Pre | Post | FU | p | |
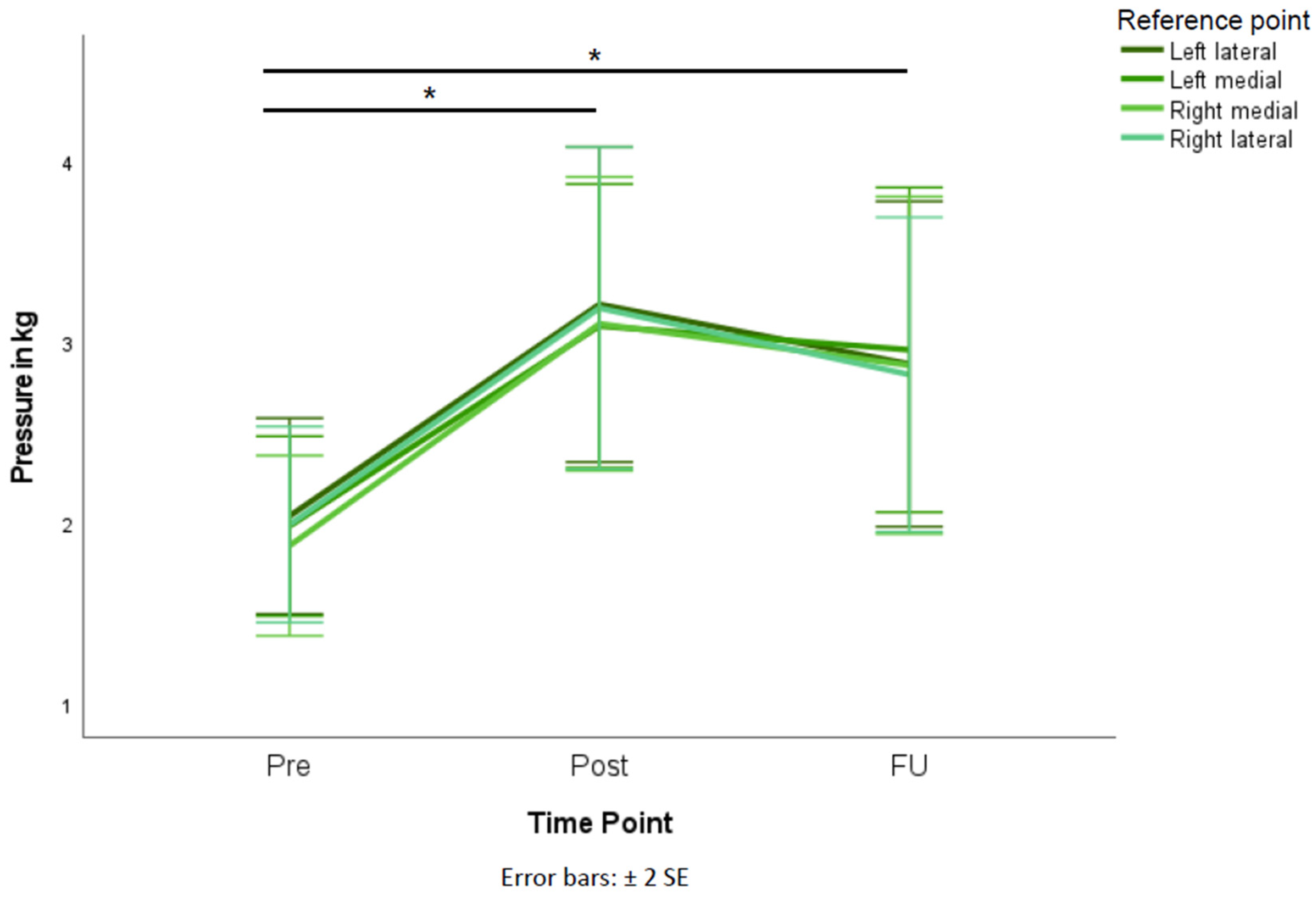
| Left lateral | 9.77 | <0.001 * | 0.318 | 2.00 (1.37) | 3.28 (2.21) | 2.87 (2.11) | |
| Pre-post | 0.001 * | ||||||
| Pre-FU | 0.034 * | ||||||
| Post-FU | 0.415 | ||||||
| Left medial | 11.38 | <0.001 * | 0.351 | 1.96 (1.27) | 3.17 (1.99) | 2.95 (2.11) | |
| Pre-post | 0.002 * | ||||||
| Pre-FU | 0.007 * | ||||||
| Post-FU | 0.988 | ||||||
| Right medial | 11.98 | <0.001 * | 0.363 | 1.83 (1.26) | 3.17 (2.06) | 2.95 (2.11) | |
| Pre-post | 0.001 * | ||||||
| Pre-FU | 0.004 * | ||||||
| Post-FU | 0.788 | ||||||
| Right lateral | 9.49 | <0.001 * | 0.311 | 1.94 (1.37) | 3.24 (2.25) | 2.81 (2.04) | |
| Pre-post | 0.002 * | ||||||
| Pre-FU | 0.020 * | ||||||
| Post-FU | 0.510 | ||||||
| PPT | Test Values | Mean (SE) | ||||
|---|---|---|---|---|---|---|
| F | p | η2 | Pre | Post | FU | |
| Left lateral | ||||||
| Headache diagnosis | 5.82 | 0.026 * | 0.225 | |||
| Time | 10.50 | <0.001 * | 0.344 | |||
| Time * headache diagnosis | 2.57 | 0.089 | 0.114 | |||
| Primary headache | 1.49 (0.39) | 2.13 (0.58) | 2.07 (0.60) | |||
| PTH | 2.50 (0.39) | 4.43 (0.58) | 3.68 (0.60) | |||
| Left medial | ||||||
| Headache diagnosis | 6.56 | 0.019 * | 0.247 | |||
| Time | 11.62 | <0.001 * | 0.368 | |||
| Time * headache diagnosis | 1.45 | 0.247 | 0.068 | |||
| Primary headache | 1.38 (0.35) | 2.13 (0.52) | 2.13 (0.60) | |||
| PTH | 2.54 (0.35) | 4.20 (0.52) | 3.78 (0.60) | |||
| Right medial | ||||||
| Headache diagnosis | 6.47 | 0.019 * | 0.244 | |||
| Time | 12.66 | <0.001 * | 0.388 | |||
| Time * headache diagnosis | 2.19 | 0.125 | 0.099 | |||
| Primary headache | 1.33 (0.36) | 2.11 (0.54) | 1.93 (0.61) | |||
| PTH | 2.34 (0.36) | 4.22 (0.54) | 3.80 (0.61) | |||
| Right lateral | ||||||
| Headache diagnosis | 6.34 | 0.020 * | 0.241 | |||
| Time | 9.94 | <0.001 * | 0.332 | |||
| Time * headache diagnosis | 2.00 | 0.148 | 0.091 | |||
| Primary headache | 1.41 (0.39) | 2.12 (0.60) | 1.97 (0.57) | |||
| PTH | 2.46 (0.39) | 4.37 (0.60) | 3.66 (0.57) | |||
| Responder Rate | Headache Diagnosis Comparison n (%) | Neck Pain Comparison n (%) | Treatment Time Frame Comparison n (%) | ||||
|---|---|---|---|---|---|---|---|
| Primary Headache | PTH | Neck Pain | No Neck Pain | <2x/Week | 2x/Week | >2x/Week | |
| 75% responder | 2 (14.3) | 5 (45.6) | 6 (40) | 1 (10) | 2 (22.2) | 4 (40) | 1 (16.7) |
| 50% responder | 5 (35.7) | 5 (45.6) | 8 (53.3) | 2 (20) | 2 (22.2) | 6 (60) | 2 (33.3) |
| 25% responder | 6 (42.9) | 5 (45.6) | 9 (60) * | 2 (20) * | 3 (33.3) | 6 (60) | 2 (33.3) |
| No responder | 8 (57) | 6 (54.6) | 6 (40) * | 8 (80) * | 6 (66.7) | 4 (40) | 4 (66.7) |
| Group | Correlation r | p |
|---|---|---|
| Headache diagnosis groups | ||
| Primary headache a | −0.05 | 0.857 |
| PTH b | 0.23 | 0.492 |
| Neck pain groups | ||
| Neck pain a | −0.04 | 0.881 |
| No neck pain a | 0.34 | 0.334 |
| Treatment time frame groups | ||
| <2x/week a | 0.53 | 0.139 |
| 2x/week a | −0.03 | 0.943 |
| >2x/week a | 0.03 | 0.955 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Börner, C.; Staisch, J.; Lang, M.; Hauser, A.; Hannibal, I.; Huß, K.; Klose, B.; Lechner, M.F.; Sollmann, N.; Heinen, F.; et al. Repetitive Neuromuscular Magnetic Stimulation for Pediatric Headache Disorders: Muscular Effects and Factors Affecting Level of Response. Brain Sci. 2022, 12, 932. https://doi.org/10.3390/brainsci12070932
Börner C, Staisch J, Lang M, Hauser A, Hannibal I, Huß K, Klose B, Lechner MF, Sollmann N, Heinen F, et al. Repetitive Neuromuscular Magnetic Stimulation for Pediatric Headache Disorders: Muscular Effects and Factors Affecting Level of Response. Brain Sciences. 2022; 12(7):932. https://doi.org/10.3390/brainsci12070932
Chicago/Turabian StyleBörner, Corinna, Jacob Staisch, Magdalena Lang, Ari Hauser, Iris Hannibal, Kristina Huß, Birgit Klose, Matthias F. Lechner, Nico Sollmann, Florian Heinen, and et al. 2022. "Repetitive Neuromuscular Magnetic Stimulation for Pediatric Headache Disorders: Muscular Effects and Factors Affecting Level of Response" Brain Sciences 12, no. 7: 932. https://doi.org/10.3390/brainsci12070932
APA StyleBörner, C., Staisch, J., Lang, M., Hauser, A., Hannibal, I., Huß, K., Klose, B., Lechner, M. F., Sollmann, N., Heinen, F., Landgraf, M. N., & Bonfert, M. V. (2022). Repetitive Neuromuscular Magnetic Stimulation for Pediatric Headache Disorders: Muscular Effects and Factors Affecting Level of Response. Brain Sciences, 12(7), 932. https://doi.org/10.3390/brainsci12070932








