Screening of Crucial Cytosolicproteins Interconnecting the Endoplasmic Reticulum and Mitochondria in Parkinson’s Disease and the Impact of Anti-Parkinson Drugs in the Preservation of Organelle Connectivity
Abstract
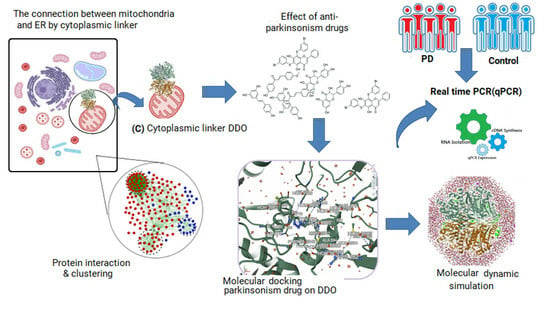
:1. Introduction
2. Materials and Methods
2.1. Data Collection and Protein Interactome Construction
2.2. Cluster Analysis and Pathway Enrichment
2.3. Molecular Modelling of Crucial Cytosolic Proteins
2.4. Ligand Preparation and Molecular Docking
2.5. Molecular Dynamic Simulation
2.6. Patient Recruitment and Clinical Assessment
2.7. Gene Expression Profiling
2.8. Statistical Analysis
3. Results
3.1. Data Collection and Meta-Analysis of Cluster Proteins
3.2. Interactome and Pathway Enrichment Analysis
3.3. Molecular Docking and Molecular Dynamic Simulation
3.4. Gene Expression of DDO Gene in PD
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tanner, C.M.; Goldman, S.M. Epidemiology of Parkinson’s disease. Neurol. Clin. 1996, 14, 317–335. [Google Scholar] [CrossRef] [PubMed]
- Kouli, A.; Torsney, K.M.; Kuan, W.L. Parkinson’s Disease: Etiology, Neuropathology, and Pathogenesis; Exon Publications: Brisbane, Australia, 2018; pp. 3–26. [Google Scholar]
- Dawson, T.M.; Dawson, V.L. Rare genetic mutations shed light on the pathogenesis of Parkinson disease. J. Clin. Investig. 2003, 111, 145–151. [Google Scholar] [CrossRef]
- Gcwensa, N.Z.; Russell, D.L.; Cowell, R.M.; Volpicelli-Daley, L.A. Molecular mechanisms underlying synaptic and axon degeneration in Parkinson’s disease. Front. Cell Neurosci. 2021, 15, 626128. [Google Scholar] [CrossRef] [PubMed]
- Ryan, K.C.; Ashkavand, Z.; Norman, K.R. The Role of Mitochondrial Calcium Homeostasis in Alzheimer’s and Related Diseases. Int. J. Mol. Sci. 2020, 21, 9153. [Google Scholar] [CrossRef] [PubMed]
- Lebiedzinska, M.; Szabadkai, G.; Jones, A.W.; Duszynski, J.; Wieckowski, M.R. Interactions between the endoplasmic reticulum, mitochondria, plasma membrane and other subcellular organelles. Int. J. Biochem. Cell Biol. 2009, 41, 1805–1816. [Google Scholar] [CrossRef] [PubMed]
- Szymański, J.; Janikiewicz, J.; Michalska, B.; Patalas-Krawczyk, P.; Perrone, M.; Ziółkowski, W.; Duszyński, J.; Pinton, P.; Dobrzyń, A.; Więckowski, M.R. Interaction of Mitochondria with the Endoplasmic Reticulum and Plasma Membrane in Calcium Homeostasis, Lipid Trafficking and Mitochondrial Structure. Int. J. Mol. Sci. 2017, 18, 1576. [Google Scholar] [CrossRef]
- Kodavati, M.; Wang, H.; Hegde, M.L. Altered Mitochondrial Dynamics in Motor Neuron Disease: An Emerging Perspective. Cells 2020, 9, 1065. [Google Scholar] [CrossRef] [PubMed]
- Vrijsen, S.; Vrancx, C.; Del Vecchio, M.; Swinnen, J.V.; Agostinis, P.; Winderickx, J.; Vangheluwe, P.; Annaert, W. Inter-organellar Communication in Parkinson’s and Alzheimer’s Disease: Looking Beyond Endoplasmic Reticulum-Mitochondria Contact Sites. Front. Neurosci. 2022, 16, 900338. [Google Scholar] [CrossRef]
- Sohrabi, T.; Mirzaei-Behbahani, B.; Zadali, R.; Pirhaghi, M.; Morozova-Roche, L.A.; Meratan, A.A. Common Mechanisms Underlying α-Synuclein-Induced Mitochondrial Dysfunction in Parkinson’s Disease. J. Mol. Biol. 2023, 435, 167992. [Google Scholar] [CrossRef]
- Sultana, M.A.; Hia, R.A.; Akinsiku, O.; Hegde, V. Peripheral Mitochondrial Dysfunction: A Potential Contributor to the Development of Metabolic Disorders and Alzheimer’s Disease. Biology 2023, 12, 1019. [Google Scholar] [CrossRef]
- Rodríguez-Arribas, M.; Yakhine-Diop, S.M.S.; Pedro, J.M.B.-S.; Gómez-Suaga, P.; Gómez-Sánchez, R.; Martínez-Chacón, G.; Fuentes, J.M.; González-Polo, R.A.; Niso-Santano, M. Mitochondria-Associated Membranes (MAMs): Overview and Its Role in Parkinson’s Disease. Mol. Neurobiol. 2014, 54, 6287–6303. [Google Scholar] [CrossRef] [PubMed]
- Kozjak-Pavlovic, V. The MICOS complex of human mitochondria. Cell Tissue Res. 2017, 367, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Eramo, M.J.; Lisnyak, V.; Formosa, L.E.; Ryan, M.T. The ‘mitochondrial contact site and cristae organising system’ (MICOS) in health and human disease. J. Biochem. 2020, 167, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Suaga, P.; Bravo-San Pedro, J.M.; González-Polo, R.A.; Fuentes, J.M.; Niso-Santano, M. ER-mitochondria signaling in Parkinson’s disease. Cell Death Dis. 2018, 9, 337. [Google Scholar] [CrossRef]
- Krols, M.; van Isterdael, G.; Asselbergh, B.; Kremer, A.; Lippens, S.; Timmerman, V.; Janssens, S. Mitochondria-associated membranes as hubs for neurodegeneration. Acta Neuropathol. 2016, 131, 505–523. [Google Scholar] [CrossRef]
- Harischandra, D.S.; Ghaisas, S.; Zenitsky, G.; Jin, H.; Kanthasamy, A.; Anantharam, V.; Kanthasamy, A.G. Manganese-induced neurotoxicity: New insights into the triad of protein misfolding, mitochondrial impairment, and neuroinflammation. Front. Neurosci. 2019, 13, 654. [Google Scholar] [CrossRef]
- Ramalingam, M.; Huh, Y.-J.; Lee, Y.-I. The Impairments of α-Synuclein and Mechanistic Target of Rapamycin in Rotenone-Induced SH-SY5Y Cells and Mice Model of Parkinson’s Disease. Front. Neurosci. 2019, 13, 1028. [Google Scholar] [CrossRef]
- Valdinocci, D.; Simões, R.F.; Kovarova, J.; Cunha-Oliveira, T.; Neuzil, J.; Pountney, D.L. Intracellular and intercellular mitochondrial dynamics in Parkinson’s disease. Front. Neurosci. 2019, 13, 930. [Google Scholar] [CrossRef]
- Paillusson, S.; Stoica, R.; Gomez-Suaga, P.; Lau, D.H.; Mueller, S.; Miller, T.; Miller, C.C. There’s something wrong with my MAM; the ER–mitochondria axis and neurodegenerative diseases. Trends Neurosci. 2016, 39, 146–157. [Google Scholar] [CrossRef]
- Lee, K.-S.; Huh, S.; Lee, S.; Wu, Z.; Kim, A.-K.; Kang, H.-Y.; Lu, B. Altered ER–mitochondria contact impacts mitochondria calcium homeostasis and contributes to neurodegeneration in vivo in disease models. Proc. Natl. Acad. Sci. USA 2018, 115, E8844–E8853. [Google Scholar] [CrossRef]
- Lee, J.; Song, C.-H. Effect of Reactive Oxygen Species on the Endoplasmic Reticulum and Mitochondria during Intracellular Pathogen Infection of Mammalian Cells. Antioxidants 2021, 10, 872. [Google Scholar] [CrossRef]
- Su, L.; Chen, S.; Zheng, C.; Wei, H.; Song, X. Meta-analysis of gene expression and identification of biological regulatory mechanisms in Alzheimer’s disease. Front. Neurosci. 2019, 13, 633. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.I.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Ge, S.X.; Jung, D.; Yao, R. ShinyGO: A graphical gene-set enrichment tool for animals and plants. Bioinformatics 2020, 36, 2628–2629. [Google Scholar] [CrossRef]
- Shen, J.; Zhang, W.; Fang, H.; Perkins, R.; Tong, W.; Hong, H. Homology modeling, molecular docking, and molecular dynamics simulations elucidated α-fetoprotein binding modes. BMC Bioinform. 2013, 14 (Suppl. S14), S6. [Google Scholar] [CrossRef] [PubMed]
- Brogi, S.; Rossi, S.; Ibba, R.; Butini, S.; Calderone, V.; Campiani, G.; Gemma, S. In Silico Analysis of Peptide-Based Derivatives Containing Bifunctional Warheads Engaging Prime and Non-Prime Subsites to Covalent Binding SARS-CoV-2 Main Protease (Mpro). Computation 2022, 10, 69. [Google Scholar] [CrossRef]
- Robertson, M.J.; Qian, Y.; Robinson, M.C.; Tirado-Rives, J.; Jorgensen, W.L. Development and testing of the OPLS-AA/M force field for RNA. J. Chem. Theory Comput. 2019, 15, 2734–2742. [Google Scholar] [CrossRef] [PubMed]
- Gajjar, N.D.; Dhameliya, T.M.; Shah, G.B. In search of RdRp and Mpro inhibitors against SARS CoV-2: Molecular docking, molecular dynamic simulations and ADMET analysis. J. Mol. Struct. 2021, 1239, 130488. [Google Scholar] [CrossRef] [PubMed]
- Gouda, M.A. Common Pitfalls in Reporting the Use of SPSS Software. Med. Princ. Pract. 2015, 24, 300. [Google Scholar] [CrossRef] [PubMed]
- Ota, N.; Shi, T.; Sweedler, J.V. d-Aspartate acts as a signaling molecule in nervous and neuroendocrine systems. Amino Acids 2012, 43, 1873–1886. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Homma, H.; Lee, J.-A.; Fukushima, T.; Santa, T.; Tashiro, K.; Iwatsubo, T.; Imai, K. Emergence of d-aspartic acid in the differentiating neurons of the rat central nervous system. Brain Res. 1998, 808, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Wolosker, H.; D’aniello, A.; Snyder, S. d-Aspartate disposition in neuronal and endocrine tissues: Ontogeny, biosynthesis and release. Neuroscience 2000, 100, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Van Veldhoven, P.P.; Brees, C.; Mannaerts, G.P. d-Aspartate oxidase, a peroxisomal enzyme in liver of rat and man. Biochim. Biophys. Acta (BBA)-Gen. Subj. 1991, 1073, 203–208. [Google Scholar] [CrossRef]
- Naren, P.; Cholkar, A.; Kamble, S.; Khan, S.S.; Srivastava, S.; Madan, J.; Mehra, N.; Tiwari, V.; Singh, S.B.; Khatri, D.K. Pathological and Therapeutic Advances in Parkinson’s Disease: Mitochondria in the Interplay. J. Alzheimer’s Dis. 2023, 94, S399–S428. [Google Scholar] [CrossRef] [PubMed]
- Errico, F.; Napolitano, F.; Nisticò, R.; Usiello, A. New insights on the role of free D-aspartate in the mammalian brain. Amino Acids 2012, 43, 1861–1871. [Google Scholar] [CrossRef] [PubMed]
- Errico, F.; Nisticò, R.; Napolitano, F.; Mazzola, C.; Astone, D.; Pisapia, T.; Giustizieri, M.; D’Aniello, A.; Mercuri, N.B.; Usiello, A. Increased D-aspartate brain content rescues hippocampal age-related synaptic plasticity deterioration of mice. Neurobiol. Aging 2011, 32, 2229–2243. [Google Scholar] [CrossRef]
- Errico, F.; Rossi, S.; Napolitano, F.; Catuogno, V.; Topo, E.; Fisone, G.; D’Aniello, A.; Centonze, D.; Usiello, A. D-Aspartate Prevents Corticostriatal Long-Term Depression and Attenuates Schizophrenia-Like Symptoms Induced by Amphetamine and MK-801. J. Neurosci. 2008, 28, 10404–10414. [Google Scholar] [CrossRef]
- Errico, F.; Bonito-Oliva, A.; Bagetta, V.; Vitucci, D.; Romano, R.; Zianni, E.; Napolitano, F.; Marinucci, S.; Di Luca, M.; Calabresi, P.; et al. Higher free d-aspartate and N-methyl-d-aspartate levels prevent striatal depotentiation and anticipate l-DOPA-induced dyskinesia. Exp. Neurol. 2011, 232, 240–250. [Google Scholar] [CrossRef]
- Cristino, L.; Luongo, L.; Squillace, M.; Paolone, G.; Mango, D.; Piccinin, S.; Zianni, E.; Imperatore, R.; Iannotta, M.; Longo, F.; et al. d-Aspartate oxidase influences glutamatergic system homeostasis in mammalian brain. Neurobiol. Aging 2015, 36, 1890–1902. [Google Scholar] [CrossRef]
- Punzo, D.; Errico, F.; Cristino, L.; Sacchi, S.; Keller, S.; Belardo, C.; Luongo, L.; Nuzzo, T.; Imperatore, R.; Florio, E.; et al. Age-Related changes in D-aspartate oxidase promoter methylation control extracellular D-aspartate levels and prevent precocious cell death during brain aging. J. Neurosci. 2016, 36, 3064–3078. [Google Scholar] [CrossRef]
- Olivares, D.; Deshpande, V.K.; Shi, Y.; Lahiri, D.K.; Greig, N.H.; Rogers, J.T.; Huang, X. N-methyl D-aspartate (NMDA) receptor antagonists and memantine treatment for Alzheimer’s disease, vascular dementia and Parkinson’s disease. Curr. Alzheimer Res. 2012, 9, 746–758. [Google Scholar] [CrossRef]
- Meredith, G.E.; Rademacher, D.J. MPTP mouse models of Parkinson’s disease: An update. J. Park. Dis. 2011, 1, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Santiago, B.; Brunet, A.; Sobrino, B.; Serra-Juhé, C.; Flores, R.; Armengol, L.; Vilella, E.; Gabau, E.; Guitart, M.; Guillamat, R.; et al. Association of common copy number variants at the glutathione S-transferase genes and rare novel genomic changes with schizophrenia. Mol. Psychiatry 2009, 15, 1023–1033. [Google Scholar] [CrossRef]
- Li, Y.J.; Oliveira, S.A.; Xu, P.; Martin, E.R.; Stenger, J.E.; Scherzer, C.R.; Hauser, M.A.; Scott, W.K.; Small, G.W.; Nance, M.A.; et al. Glutathione S-transferase omega-1 modifies age-at-onset of Alzheimer disease and Parkinson disease. Hum. Mol. Genet. 2003, 12, 3259–3267. [Google Scholar] [CrossRef]
- Dringen, R. Metabolism and functions of glutathione in brain. Prog. Neurobiol. 2000, 62, 649–671. [Google Scholar] [CrossRef] [PubMed]
- Isaacson, S.H.; Betté, S.; Pahwa, R. Istradefylline for OFF Episodes in Parkinson’s Disease: A US Perspective of Common Clinical Scenarios. Degener. Neurol. Neuromuscul. Dis. 2022, 12, 97–109. [Google Scholar] [CrossRef]
- Naderi, S.; Faghih, H.; Aqamolaei, A.; Mortazavi, S.H.; Mortezaei, A.; Sahebolzamani, E.; Rezaei, F.; Akhondzadeh, S. Amantadine as adjuvant therapy in the treatment of moderate to severe obsessive–compulsive disorder: A double-blind randomized trial with placebo control. Psychiatry Clin. Neurosci. 2019, 73, 169–174. [Google Scholar] [CrossRef]
- Dragašević-Mišković, N.; Petrović, I.; Stanković, I.; Kostić, V.S. Chemical management of levodopa-induced dyskinesia in Parkinson’s disease patients. Expert Opin. Pharmacother. 2019, 20, 219–230. [Google Scholar] [CrossRef]
- Machado-Alba, J.E.; Calvo-Torres, L.F.; Gaviria-Mendoza, A.; Castrillón-Spitia, J.D. Prescribing patterns of antiparkinson drugs in a group of Colombian patients, 2015. Biomedica 2018, 38, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Schreiner, W.; Karch, R.; Knapp, B.; Ilieva, N. Relaxation estimation of RMSD in molecular dynamics immune simulations. Comput. Math. Methods Med. 2012, 2012, 173521. [Google Scholar] [CrossRef]
- Sokkar, P.; Harms, M.; Sturzel, C.; Gilg, A.; Kizilsavas, G.; Raasholm, M.; Preising, N.; Wagner, M.; Kirchhoff, F.; Standker, L.; et al. Computational modeling and experimental validation of the EPI-X4/CXCR4 complex allows rational design of small peptide antagonists. Commun. Biol. 2021, 4, 1113. [Google Scholar] [CrossRef] [PubMed]
- Daison, F.A.; Kumar, N.; Balakrishnan, S.; Venugopal, K.; Elango, S.; Sokkar, P. Molecular Dynamics Studies on the Bacterial Membrane Pore Formation by Small Molecule Antimicrobial Agents. J. Chem. Inf. Model. 2021, 62, 40–48. [Google Scholar] [CrossRef] [PubMed]






| Parameter | Istradefylline PD (n = 25) | Amantadine PD (n = 25) | Healthy Control (HC) (n = 25) | p-Value |
|---|---|---|---|---|
| Gender (Male/Female) | 13/12 | 8/17 | 17/8 | 0.527 |
| Age (mean ± SD) | 56.51 ± 1.40 | 55.45 ± 1.81 | 51.18 ± 1.33 | 0.007 |
| Age at onset (mean ± SD) | 48.46 ± 10.23 | 46.72 ± 11.68 | ||
| Disease Duration Year (mean ± SD) | 6.92 ± 3.42 | 6.89 ± 3.14 | ||
| UPDRS total (mean ± SD) | 42.45 ± 19.02 | 41.35 ± 18.02 | ||
| UPDRS I | 6.57 ± 4.15 | 6.47 ± 3.05 | ||
| UPDRS II | 11.09 ± 8.36 | 11.29 ± 7.58 | ||
| UPDRS III | 23.57 ± 14.34 | 23.71 ± 13.14 | ||
| UPDRS IV | 1.85 ± 5.48 | 1.91 ± 5.18 | ||
| Hoehn and Yahr Scale | 2.12 ± 0.60 | 2.32 ± 0.50 |
| Gene | Primer Sequence (5′->3′) | Ampliconic Size | |
|---|---|---|---|
| DDO | Forward Sequence | GGAGCTGAAATCTCTCACCTGG | 193 |
| Reverse Sequence | CTCATGGACACAGCACGGAT | ||
| GAPDH | Forward Sequence | TGTCATCAACGGAAAGGC | 183 |
| Reverse Sequence | GCATCAGCAGAAGGAGCA | ||
| Sl. No | Drug ID | Drug Name | DOCKING SCORE (kcal/mol) |
|---|---|---|---|
| 1 | DB11757 | Istradefylline | −9.073 |
| 2 | DB06654 | Safinamide | −8.862 |
| 3 | DB00843 | Donepezil | −8.861 |
| 4 | DB00246 | Ziprasidone | −8.381 |
| 5 | DB06477 | Sumanirole | −8.256 |
| 6 | DB00494 | Entacapone | −8.153 |
| 7 | DB00490 | Buspirone | −8.074 |
| 8 | DB06454 | Sarizotan | −7.918 |
| 9 | DB01202 | Lavetriacetam | −7.879 |
| 10 | DB00323 | Tolcapone | −7.703 |
| 11 | DB00486 | Nabilone | −7.455 |
| 12 | DB00413 | Pramipexole | −7.246 |
| 13 | DB12551 | Idazoxan | −7.082 |
| 14 | DB06585 | Fipamezol | −7.025 |
| 15 | DB01367 | Rasagiline | −6.788 |
| 16 | DB00745 | Modafinil | −6.766 |
| 17 | DB00363 | Clozapine | −6.711 |
| 18 | DB05271 | Rotigotine | −6.679 |
| 19 | DB01235 | Levodopa | −6.601 |
| 20 | DB00571 | Propranol | −6.551 |
| 21 | DB06156 | Tesofensine | −6.517 |
| 22 | DB05814 | Gpi-1485 | −6.468 |
| 23 | DB01224 | Quetiapine | −6.333 |
| 24 | DB00472 | Fluoxetine | −6.31 |
| 25 | DB00268 | Ropinirole | −6.046 |
| 26 | DB00190 | Carbidopa | −6.025 |
| 27 | DB00674 | Galantamine | −6.012 |
| 28 | DB01183 | Naloxone | −5.886 |
| 29 | DB04982 | Talampanel | −5.768 |
| 30 | DB00714 | Apomorphine | −5.743 |
| 31 | DB01043 | Memantine | −5.605 |
| 32 | DB00989 | Rivastigmine | −5.486 |
| 33 | DB00334 | Olanzapine | −5.295 |
| 34 | DB01037 | Selegiline | −4.823 |
| 35 | DB00915 | Amantadine | −4.543 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anirudhan, A.; Mahema, S.; Ahmad, S.F.; Emran, T.B.; Ahmed, S.S.S.J.; Paramasivam, P. Screening of Crucial Cytosolicproteins Interconnecting the Endoplasmic Reticulum and Mitochondria in Parkinson’s Disease and the Impact of Anti-Parkinson Drugs in the Preservation of Organelle Connectivity. Brain Sci. 2023, 13, 1551. https://doi.org/10.3390/brainsci13111551
Anirudhan A, Mahema S, Ahmad SF, Emran TB, Ahmed SSSJ, Paramasivam P. Screening of Crucial Cytosolicproteins Interconnecting the Endoplasmic Reticulum and Mitochondria in Parkinson’s Disease and the Impact of Anti-Parkinson Drugs in the Preservation of Organelle Connectivity. Brain Sciences. 2023; 13(11):1551. https://doi.org/10.3390/brainsci13111551
Chicago/Turabian StyleAnirudhan, Athira, S. Mahema, Sheikh F. Ahmad, Talha Bin Emran, Shiek S. S. J. Ahmed, and Prabu Paramasivam. 2023. "Screening of Crucial Cytosolicproteins Interconnecting the Endoplasmic Reticulum and Mitochondria in Parkinson’s Disease and the Impact of Anti-Parkinson Drugs in the Preservation of Organelle Connectivity" Brain Sciences 13, no. 11: 1551. https://doi.org/10.3390/brainsci13111551