1. Introduction
At any instance in time, our visual world provides a vast amount of visual input. Since human mental capacity is limited, only a fraction of the available information is selected for purposes such as perception, memory and action control, while the rest of the information is ignored. In the visual modality, this selectivity is called
selective visual attention [
1]. One important issue concerns the role of selective visual attention in the division of labor between unconscious (or subliminal) visual processing on the one hand and conscious vision on the other. A related important question is how this division of labor is implemented in the human brain’s visual system.
One hypothesis about the relationship between conscious and unconscious vision is that selective visuo-spatial attention (
i.e., the selection of positions or areas in the visual field) is a necessary (though not sufficient [
2]) prerequisite for at least some forms of conscious visual perception [
3,
4], but see [
5]. For example, according to feature-integration theory, selective visuo-spatial attention is necessary for the binding of different visual features (e.g., red color and round shape) into one perceived object (e.g., a tomato) [
6]. Crucially, if it is true that attention serves conscious visual perception, it follows that attention should (at least partly) operate before conscious vision—that is, visuo-spatial attention should operate on the basis of subliminal visual information, too [
3].
This has been demonstrated in cueing experiments with abrupt-onset singleton cues [
7,
8]. In cueing experiments, participants have to search for targets, and in each trial, one target is shown at one out of several positions [
9,
10]. The participants have to find this target, discriminate it, and respond to it. When presenting a single abrupt-onset briefly before the target with a Stimulus Onset Asynchrony (SOA) of up to 200–300 ms at the same position (SP) as the target, locating the target is facilitated. This facilitation is reflected in faster responses to targets in SP conditions than to targets at a different position (DP) than the cue. This facilitative cueing effect presumably reflects the capture of attention by the cue [
11]. As explained, target perception requires that attention is shifted towards target locations [
3,
6]. Since the cue captures attention towards its location, in SP conditions attention is already at the target position. In contrast, in DP conditions attention has to be directed away from the cue to the target position, which can only be performed after target onset. This capture effect (
i.e., an SP-DP performance difference) is found with single abrupt-onset cues that are not predictive for the most likely target position [
9,
10] and it turns into Inhibition Of Return (IOR)—that is, slower responses for SP than DP targets—with SOAs beyond 300 ms [
12,
13,
14]. IOR could be due to a sequence of capture by the cue, subsequent deallocation of attention away from the cue during the longer SOA, and a reluctance to reorient to the cued position if the target is finally presented there [
13,
15]. IOR might also reflect motor inhibition [
14] or result from perceptual adaptation [
12].
Whatever the exact origin of IOR, in line with subliminal attention, both the facilitative cueing effect with the short SOA and IOR with longer SOAs have also been found with subliminal abrupt-onset cues that were not seen by the participants (
i.e., could not be reported with better than chance accuracy) [
7,
8,
15,
16]. For example, presenting one disk as a single abrupt-onset cue on the left or on the right of a computer screen with a head start of 16 ms before two additional disks (at screen center and on the opposite screen side), this abrupt-onset cue can barely be seen: Due to the short onset difference, the participants were unable to decide whether a cue was presented on the left or on the right during a cue-discrimination task at the end of the experiment [
15]. These subliminal cues captured attention [
15,
17]: Target detection was facilitated with a short cue-target SOA of 16 ms in SP conditions compared to DP conditions. In addition, with a longer SOA of 1,016 ms the subliminal cue led to IOR [
15,
17].
Exactly how the brain’s visual system brings about subliminal vision in general and subliminal attention in particular, however, is currently debated. One party of researchers thinks that at least some forms of unconscious vision reflect processing along the visual system’s parvocellular projection, leading from the retina to the cortex, via the Lateral Geniculate Nucleus (LGN) of the thalamus [
18], and many forms of unconscious visual processing are probably of cortical origin [
19,
20].
However, another party of researchers recently proposed that subliminal attention at least partly reflects contrast-elicited attentional capture mediated by midbrain structures, namely neurons in the Superior Colliculi (SC) [
15,
17]. Functionally, the SC has a prominent role in the elicitation of saccadic eye movements and it contains cells sensitive to visual contrast and motion [
21,
22,
23]. Anatomically, the SC is part of the magnocellular projection leading from the retina to the human cortex (via the nucleus pulvinaris of the thalamus), and it receives its input mainly from luminance-sensitive retinal ganglion cells [
24,
25]. As a consequence of these characteristics, the SC is very sensitive to the visual
contrast changes elicited by luminance transients and abrupt visual onsets.
In line with a collicular origin of subliminal attention, the unconscious cueing effect occurs when contrast increases (e.g., produced by sudden onsets) and independently of contrast polarities (
i.e., for both, lighter and darker items) [
26]: When presenting subliminal light or dark rings (with the same Weber contrast but different signs) as abrupt-onset cues, similar facilitative cueing effects for both contrast polarities but no IOR were found [
27]. Also, in general agreement with the hypothesis that the SC could be responsible for subliminal capture, the SC seems to (partly) account for “blindsight”—that is, the spared subliminal visual capabilities concerning input from visual field areas of scotoma after damage to primary visual cortex [
28,
29], but see [
30]—and the SC has an active modulating role in visual attention [
31].
Probably also in line with the SC hypothesis of subliminal attention are the differences between subliminal cueing by single abrupt-onset cues on the one hand [
7,
15,
16] and by subliminal color cues on the other [
32,
33]. As explained, subliminal abrupt-onset cueing is found with abrupt-onset targets [
7,
15,
16], and regardless of cue-target “color” (or luminance polarity) [
27]. This is different with subliminal color cues during color search—that is, searches for a color-defined target: Subliminal cues with a color similar to the target capture attention but subliminal cues with a color different from the searched-for target fail to capture attention [
32,
33]. For example, if the target is red and presented together with either of three differently colored distractors (e.g., a green, a blue, and a gray distractor), it is difficult (if not impossible) for the participants to search for contrasts to find the target. The reason for this difficulty and the resulting cueing effect differences is probably simple. In comparison to a single abrupt-onset target that creates a single new contrast change, during color search each of the color stimuli (target and distractors) creates an (individually varying) different contrast change. This is true even if all stimuli are objectively equated for their luminance because individuals differ in their sensitivity for different colors [
34,
35] (see
Figure 1).
Figure 1.
Schematic picture of a color-target display with two additional color distractors (top row). Below that, fictitious color-contrast dependent response functions for three different participants are plotted for all three different hues (rows 2–4). Colors with objectively the same luminance are able to produce different subjectively perceived contrasts in different observers due to varying sensitivity to particular hues. The figure demonstrates how three fictitious participants differ in their sensitivity and have their respective maximal contrast-dependent response in the range of red (row 2), blue (row 3), or green (row 4) colors. This figure illustrates the heterogeneity of the resulting contrast differences between participants, as well as the fact that the contrast differences are relatively small. The small size of the color contrast differences can be inferred from a comparison of the respective sizes of differences of: (1) background luminance responses (at 0) and the asymptotic maximum value of each color-elicited response; and (2) the background and black (this is the starting value of each color-elicited contrast-dependent response function).
Figure 1 also depicts the way in which colors of targets and distractors were realized in the current Experiments 4 to 6: as changes from black to red, green, or blue.
Figure 1.
Schematic picture of a color-target display with two additional color distractors (top row). Below that, fictitious color-contrast dependent response functions for three different participants are plotted for all three different hues (rows 2–4). Colors with objectively the same luminance are able to produce different subjectively perceived contrasts in different observers due to varying sensitivity to particular hues. The figure demonstrates how three fictitious participants differ in their sensitivity and have their respective maximal contrast-dependent response in the range of red (row 2), blue (row 3), or green (row 4) colors. This figure illustrates the heterogeneity of the resulting contrast differences between participants, as well as the fact that the contrast differences are relatively small. The small size of the color contrast differences can be inferred from a comparison of the respective sizes of differences of: (1) background luminance responses (at 0) and the asymptotic maximum value of each color-elicited response; and (2) the background and black (this is the starting value of each color-elicited contrast-dependent response function).
Figure 1 also depicts the way in which colors of targets and distractors were realized in the current Experiments 4 to 6: as changes from black to red, green, or blue.
![Brainsci 02 00033 g001]()
As a consequence of at least two simultaneous contrast changes during color search, the differences between the individual contrasts of the color-defined target on the one hand and the individual contrasts of the misleading color distractors on the other are mitigated (in comparison to a situation with a single abrupt-onset target). Even worse, the target could be of a lower contrast than at least some of the color distractors. Say, for example, that the target is blue. In this situation, if a participant is more sensitive to green than to blue colors (see
Figure 1, 4th row), it would be difficult for this person to prioritize search for the target by searching for the target’s contrast. A low-contrast target among higher contrast-distractors could not be prioritized on the basis of the collicular contrast response because the collicular response is proportional to the strength of the contrast [
26,
36]. In this situation, the balance of attention-guiding channels in the brain’s network of biased competition [
37,
38] would therefore be tipped away from contrast as an attention-guiding principle and towards color. This is simply because the smaller contrast differences between relevant color target and irrelevant color disractors would be discriminated too late to dominate over the stronger and easier to discriminate color differences. In line with this, prior studies have shown that participants actually disregard small luminance differences during a color discrimination task [
39]. In addition, because at least the superficial layers of SC are color-blind [
40], the balance would thus also be tipped away from the SC and the magnocellular projection towards color-sensitive regions of the human visual system in the parvocellular projection (e.g., V2 [
41]). In the following we refer to this possibility as the “SC hypothesis”, meaning here that contrast-driven subliminal capture based on the SC could also be shunted once color differences among relevant targets and irrelevant distractors are strong and contrast or luminance differences are weak.
Overview of the Experiments
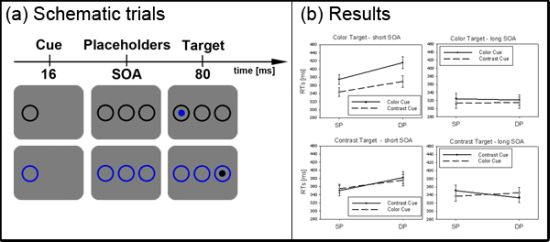
In the current study, we set out to test the SC hypothesis. First, we had to establish that under standard subliminal color cueing conditions with abrupt onset targets, it is possible to find a subliminal cueing effect. So far, it is unclear whether subliminal color cues capture attention when participants can search for a contrast-defined abrupt onset target. This possibility is tested in the present Experiment 1. For each trial, we presented a single subliminal color cue or contrast (black) cue (see also
Figure 2). Importantly, the luminance of the color cue was objectively equated to the luminance of the gray background. Participants had to search for an abrupt-onset dark (
i.e., black) target or for a single color target. The black target can thus be located by its luminance or contrast change. Likewise, if it is true that the color target elicits an (individually varying) contrast, the color target could also be located by its contrast change. In this situation, we expected subliminal capture by the contrast cue and by the color cue. Due to the participants’ individual differences in their color sensitivities [
34,
35,
42], the color cues should have led to a single luminance change. Also, because subliminal capture should not depend on contrast polarity [
27], it should not have mattered whether the individual color sensitivities led to a positive or to a negative contrast polarity between color cue and background. Therefore, any contrast change elicited by the subliminal color cue was expected to capture attention when the relevant black target could be located by its contrast change (or its abrupt onset). (Of course, the black contrast cues which were darker than the background were expected to attract attention on the basis of their elicitation of a contrast change anyway. They were included as a safeguard to verify the sensitivity of our methods in the case that color cues failed to attract attention.)
Second, the situation should dramatically change when the target-elicited luminance- or contrast-changes are smaller and therefore cannot be used as easily to locate the target. Since the attentional effect depending on the SC is assumed to be gradually dependent on contrast strength [
36], cueing effects should be stronger for high- than low-contrast cues. This prediction was tested in Experiment 2.
Third, another prediction by the SC hypothesis concerns the naso-temporal hemifield asymmetry of the retinal projection to the SC. Because the retino-tectal projection is much stronger for the temporal compared to the nasal aspect of the projection, on the basis of the SC hypothesis, with contrast cues and contrast-defined targets a stronger cueing effect after temporally than nasally presented cues was expected [
43]. This hypothesis concerning the naso-temporal asymmetry of the subliminal cueing effect was tested in Experiment 3.
Figure 2.
Experiment 1. (a) Depicted are schematic examples of trials. The top row shows a contrast cue and a blue target (same position (SP) condition). The bottom row depicts a color cue and a black target (different position (DP) condition). (b) Depicted are mean reaction times (RTs) and standard errors of the mean (error bars) of all participants, plotted separately for the short (left side) and long Stimulus Onset Asynchrony (SOA) (right side) and for color targets (top row) and contrast targets (bottom row). Results are shown for SP (left side of each panel) and DP conditions (right side of each panel). Results are plotted separately in each panel for cues of the same contrast or color (solid line) and different contrast or color cues (dashed line).
Figure 2.
Experiment 1. (a) Depicted are schematic examples of trials. The top row shows a contrast cue and a blue target (same position (SP) condition). The bottom row depicts a color cue and a black target (different position (DP) condition). (b) Depicted are mean reaction times (RTs) and standard errors of the mean (error bars) of all participants, plotted separately for the short (left side) and long Stimulus Onset Asynchrony (SOA) (right side) and for color targets (top row) and contrast targets (bottom row). Results are shown for SP (left side of each panel) and DP conditions (right side of each panel). Results are plotted separately in each panel for cues of the same contrast or color (solid line) and different contrast or color cues (dashed line).
Experiment 4 tested whether abrupt-onset cues have the potential to capture attention during color search. According to the SC hypothesis, this should not be the case. The test was achieved by two measures, (A) and (B) (see also
Figure 1). (A) We used three black disks, only one of which turned into the color-defined target (e.g., a red stimulus). Importantly, at target onset all disks simultaneously changed their colors. As a consequence, the color target was presented together with two color distractors of different colors. For example, if the target turned red, one distractor became blue, and the other green. In this way, the color-elicited contrast changes of all disks, including the target, were relatively small (in comparison to the contrast between the black disks and the background). (B) The relevant color target was unveiled by a color change at the same time as the two color changes in the additional placeholders. In this situation, for at least those participants that were less sensitive for the target colors than for either of the distractor colors, it would have been difficult to prioritize the localization of the target by searching for color-elicited contrasts. In Experiment 4, the subliminal SC contrast-capture effect was therefore expected to be shunted, and no subliminal cueing effect was expected even though a single subliminal black abrupt-onset cue of high cue-background contrast was used. We also ran a control experiment to test whether top-down singleton search provided a better account of our results (Experiment 5). This will be explained in more detail below.
Finally, Experiment 6 was yet another control. We tested whether top-down contingent capture by color could account for the absence of the subliminal cueing effects under color-search conditions [
44]. To that end, we used color-change cues instead of abrupt-onset cues, when the participants searched for a color-change target. If top-down contingent capture by color provided a better account of the lacking cueing effects of subliminal onset cues under color-search conditions, a cueing effect was to be expected in control Experiment 6 at least for the cues that changed into the same color as the target. This prediction holds true for clearly visible stimuli [
44]. However, if the SC hypothesis of subliminal capture by abrupt-onset cues holds true, no cueing effect was to be expected in Experiment 6, too, because the targets (and cues) were defined as color changes and the SC is only sensitive to contrasts of abrupt-onset cues but color-blind.
3. General Discussion
In the current study, we tested several crucial predictions of the hypothesis that the SC mediates unconscious attention effects of abrupt onsets [
15,
17]. In Experiment 1, we tested and confirmed that color cues and color targets created subliminal cueing effects. This was the case although the cue and target colors were not the same. For example, a subliminal blue cue captured attention, even if a black target was used. Likewise a subliminal black cue created a cueing effect where a blue target was searched-for. This result is in line with the SC hypothesis because the luminance of the color cues and color targets were only equated with the objective background luminance. As a consequence, different color sensitivities of the participants should have led to color-elicited contrast responses. These contrast responses to the color cues were evidently sufficient for the cueing effects because whether or not the contrasts of cues and targets were or were not the same was irrelevant for the cueing effect. The present subliminal cueing effect is therefore in line with the SC’s polarity-independent attentional effects of visible [
26] and unconscious cues [
27].
A second crucial prediction of the SC hypothesis was tested in Experiment 2. In this experiment, we investigated whether the attentional effect of the subliminal abrupt-onset cues was proportional to the contrast strength of the cues [
36]. In line with this hypothesis stronger cueing effects were found for high-contrast cues compared to low-contrast cues (Experiment 2).
A third critical prediction of the SC hypothesis was tested in Experiment 3. Prior studies showed that the SC response is larger for temporally compared to nasally presented onset stimuli [
53]. This is also true of subliminal cues [
43]. In line with prior findings [
51] a naso-temporal asymmetry of the subliminal cueing effect was found in Experiment 3.
We also reasoned that if the SC’s contrast-dependent response is responsible for the subliminal cueing effect, the contrast-elicited subliminal cueing effects should only be found when contrasts could be used to easily locate the target. After all, the participants have to find the target. Finding the target with the help of contrast should be more difficult if more than one stimulus elicits a contrast change at the time of the target. In this situation, different visual features all compete for the guidance of attention towards the relevant stimuli, and the feature that provides the easiest target-distractor discrimination and the strongest target-distractor difference should succeed in a competition for attentional guidance [
37,
38]. If it happens that the succeeding attention-guiding feature is processed outside of the SC, as would be the case with color [
25], attentional guidance would be tipped away from the SC and towards the more sensitive feature channels of the visual system [
39]. With color, these channels would lie in the parvocellular projection [
25,
41]. In this situation the SC should be shunted.
In line with this prediction, we found no subliminal cueing effects when a color target was presented together with color distractors, so that the color-elicited contrast differences between target and distractors were kept relatively small. The predicted lack of subliminal cueing was observed under these conditions when a color feature had to be used to find the target (Experiment 4), when a color difference could have been used to locate the target (Experiment 5), and even when the cue and the target shared the searched-for color.
Besides, we observed several unexpected results. First of all, cue discrimination was too high to pass a criterion of objective subliminality. This is in contrast to prior studies [
15]. Yet, our participants were subjectively unaware of the cues in all experiments, and unawareness was also confirmed by a significant offset at the point of zero visibility of the regression predicting the cueing effect on the basis of discrimination [
46].
A final unexpected observation was the similar strength of the cueing effects of colored and of black cues (Experiment 1). On the basis of the stronger objective contrast of the black cues than the color cues, an SC mediated cueing effect should have been stronger with the black cues than the color cues [
26,
36]; see also the present Experiments 2. Maybe this lack of a contrast-dependence of the cueing effect reflected strong subjective color-elicited contrast responses. We cannot reject this possibility because we did not quantify individual color-elicited contrast perception. Nevertheless, we also found attentional effects in line with this assumption of cueing effects proportional to the strength of the cue’s contrast (Experiment 2).