Abstract
Jatropha L. species, in particular, J. curcas and J. gossypiifolia, are well known medicinal plants used for treating various diseases. In the present study, leaf and stem bark extracts of J. curcas and J. gossypiifolia obtained by maceration or homogenizer assisted extraction, were investigated for their phytochemical contents and biological potential as antioxidants, enzyme inhibitors and neuromodulators. In this regard, the gene expression of tumor necrosis factor α (TNFα) and brain-derived neurotrophic factor (BDNF) was investigated in hypothalamic HypoE22 cells. Finally, a bioinformatics analysis was carried out with the aim to unravel the putative mechanisms consistent with both metabolomic fingerprints and pharmacological effects. The leaf extracts of J. curcas showed higher total phenolic content (TPC) and total flavonoid content (TFC) than the stem bark extracts (range: 5.79–48.95 mg GAE/g and 1.64–13.99 mg RE/g, respectively), while J. gossypiifolia possessed TPC and TFC in the range of 42.62–62.83 mg GAE/g and 6.97–17.63 mg RE/g, respectively. HPLC-MS/MS analysis revealed that the leaf extracts of both species obtained by homogenizer assisted extraction are richer in phytochemical compounds compared to the stem bark extracts obtained by the same extraction method. In vitro antioxidant potentials were also demonstrated in different assays (DPPH: 6.89–193.93 mg TE/g, ABTS: 20.20–255.39 mg TE/g, CUPRAC: 21.07–333.30 mg TE/g, FRAP: 14.02–168.93 mg TE/g, metal chelating activity: 3.21–17.51 mg EDTAE/g and phosphomolybdenum assay: 1.76–3.55 mmol TE/g). In particular, the leaf extract of J. curcas and the stem bark extract of J. gossypiifolia, both obtained by homogenizer assisted extraction, showed the most potent antioxidant capacity in terms of free radical scavenging and reducing activity, which could be related to their higher TPC and TFC. Furthermore, anti-neurodegenerative (acetylcholinesterase inhibition: 1.12–2.36 mg GALAE/g; butyrylcholinetserase inhibition: 0.50–3.68 mg GALAE/g), anti-hyperpigmentation (tyrosinase inhibition: 38.14–57.59 mg KAE/g) and antidiabetic (amylase inhibition: 0.28–0.62 mmol ACAE/g; glucosidase inhibition: 0.65–0.81 mmol ACAE/g) properties were displayed differentially by the different extracts. Additionally, the extracts were effective in reducing the gene expression of both TNFα and BDNF, which could be partially mediated by phenolic compounds such as naringenin, apigenin and quercetin. Indeed, the scientific data obtained from the present study complement the several other reports highlighting the pharmacological potentials of these two species, thus supporting their uses as therapeutically active plants.
1. Introduction
The genus Jatropha L., which belongs to the tribe Joannesieae in the Euphorbiaceae family, contains approximately 170 known species. The name Jatropha is derived from the Greek word ‘‘jatros’’ (doctor) and ‘‘trophe”(food), which implies its medicinal uses [1]. Jatropha species are widely used in traditional folklore medicine to cure various ailments in Africa, Asia and Latin America and are also used as ornamental plants and energy crops [2].
Jatropha species have been used as medicinal plants by native people in many tropical and subtropical countries. For instance, Jatropha species are famous for the purgative effect of the seed oil. This purgative effect has been directed to cure digestive system symptoms like diarrhoea, dysentery, vomiting, retching and stomachache. Additionally, some parts of Jatropha plants are employed to heal skin-related ailments. The seed oil, leaf, latex, stem bark or root of Jatropha plants are pounded and applied on infected skin such as eczema, itches, mouth blisters, carbuncles, wounds and swellings. They are also believed to cure venereal diseases and urinary discharge. Moreover, the roots of some Jatropha species have long been applied on people suffering from leprosy and gonorrhea [3].
Several reviews have been conducted on the different species of the genus Jatropha covering various aspects such as their ethnobotany, medicinal properties, phytochemistry, and toxicity among others [3,4,5]. Phytochemical studies of the genus Jatropha have increased in recent years due to the high potential of these species as natural sources of bioactive compounds. Investigations of the chemical constituents of Jatropha plants resulted in the isolation of a number of alkaloids, cyclic peptides, terpenes (monoterpene, sesquiterpenes, diterpenes and triterpenes), flavonoids, lignans, coumarins, coumarino-lignoids, a non-cyanogenic glucoside, phloroglucinols, ester ferulates, phenolics, deoxypreussomerins and fatty acids [3]. Moreover, extracts and isolated compounds from various species of this genus have been found to possess properties of cytotoxicity, antimicrobial, anti-inflammatory, antioxidant, insecticidal, larvicidal, cholinesterase inhibition, and toxicity activities [6].
In particular, among the various Jatropha species, J. gossypiifolia has been documented to exhibit promising biological effects. For instance, its stem latex has been reported to possess coagulating features by reducing clotting and bleeding times in experiments, thereby providing a scientific basis for its use as a haemostatic agent [7]. Furthermore, jatrophone, an active compound isolated from J. gossypiifolia, has been reported to show a better anticancer effect against hepatocellular carcinoma (Hep G2 1886) compared to standard anticancer drugs like sorafenib and arsenic trioxyde [8].
Another important species of the genus J. curcas has also been appraised for its broad spectrum of pharmacological activities. As example, extracts of this plant were found to display antiviral activity on human immunodeficiency virus [9], while others reported remarkable anti-inflammatory and antibacterial, cosmetic and wound healing properties [10,11,12].
Therefore, taking into consideration the striking scientific data gathered so far, the present study was conducted to investigate the pharmacological properties further, in terms of the antioxidant, antidiabetic, anti-neurodegenerative and anti-hyperpigmentation, of methanolic extracts of different parts (leaf and stem bark) of J. curcas L. and J. gossypiifolia L., two important species of the genus Jatropha using different extraction methods (maceration and homogenizer assisted extraction). The protective and neuromodulatory effects of the extracts were evaluated in hypothalamic HypoE22 cells. In this regard, the gene expression of tumor necrosis factor α (TNFα) and brain-derived neurotrophic factor (BDNF) was measured. This study also attempted to analyze the total phenolic and flavonoid contents using spectrophometric analysis, as well as detect and characterize the phytochemical profiles of the extracts using HPLC-MS/MS. Finally, a bioinformatics analysis was carried out with the aim to unravel the putative mechanisms consistent with both metabolomic fingerprints and pharmacological effects.
2. Materials and Methods
2.1. Plant Materials
The Jatropha species (J. curcas and J. gossipiifolia) were collected in the village of Lolodo (district of Yamoussoukro) of Côte d’Ivoire in the year 2019 and authenticated by the botanist Ouattara Katinan Etienne (Université Félix Houphouet Boigny, Abidjan, Ivory Coast). Voucher specimens were deposited in Science Faculty, Selcuk University. The stem bark and leaf samples were randomly collected from ten plants in the same population. The stem bark samples were stripped vertically while using a knife to limit it to the cambium layer. The plant materials were dried under shade for 10 days.
2.2. Extraction
The plant materials were ground and then 10 g were extracted with methanol by using maceration (MAC) and homogenizer-assisted extraction (HAE) techniques. In MAC, the plant materials (5 g) were macerated with 100 mL of methanol at room temperature (about 25 ± 2 °C) for 24 h. Regarding HAE, the plant materials (5 g) were extracted with methanol (100 mL) by using one ultra-turrax (6000× g) for 5 min at room temperature (about 25 ± 2 °C). All extracts were filtered by using Whatman No.1 filter papers and then the extracts were evaporated to dryness and stored at 4 °C until analysis.
2.3. Total Phenolic and Flavonoid Content
Spectrophotometric methods were used to determine total phenolic and flavonoid content as conducted previously. Standard equivalents (gallic acid equivalent (GAE) for phenolic and rutin equivalent (RE) for flavonoid) were used to assess the bioactive content in the plant extracts [13,14].
2.4. HPLC Analysis
Chromatographic separation was accomplished with a Dionex Ultimate 3000RS HPLC instrument, equipped with a Thermo Accucore C18 (100 mm × 2.1 mm i. d., 2.6 μm) analytical column for separation of compounds. Water (A) and methanol (B) containing 0.1% formic acid were employed as mobile phases, respectively. The total run time was 70 min; the elution profile and all exact analytical conditions have been published [15].
2.5. Determination of Antioxidant and Enzyme Inhibitory Effects
Antioxidant protocols included reducing power (cupric reducing antioxidant capacity (CUPRAC) and ferric reducing power (FRAP)), metal chelating, phosphomolybdenum (PBD) and free radical scavenging (2,2-diphenyl-1-picrylhydrazyl (DPPH) and 3-ethylbenzothiazoline-6-sulphonic acid (ABTS)) activities. Experimental details were as described previously by [16]. Inhibitory effects of the extracts were tested against different enzymes (tyrosinase, α-amylase, α-glucosidase and cholinesterases). Trolox and ethylenediaminetetraacetic acid (EDTA) for antioxidant, galantamine for cholinesterases, kojic acid for tyrosinase, and acarbose for α-amylase and α-glucosidase were used to express antioxidant and enzyme inhibitory results.
In the antioxidant and enzyme inhibitory assays, one-way ANOVA with Tukey comparison test were performed to display significance level among the extracts at a confidence level of 95%. Xlstat 2016 was used for statistical analyses.
2.6. Artemia salina Lethality Bioassay
Artemia salina cysts were hatched in oxygenated artificial sea water (1 g cysts/L). After 24 h, brine shrimp larvae were gently transferred with a pipette into 6-well plates containing 2 mL of herbal extracts at different concentrations (0.1–20 mg/mL) in artificial sea water. Ten larvae per well were incubated at 25–28 °C for 24 h. After 24 h, the number of living napulii were counted under light microscope and compared to the control untreated group. Results were expressed as percentage of mortality calculated as: ((T − S)/T) ∗ 100. T is the total number of incubated larvae and S is the number of survival napulii. Living nauplii were considered those exhibiting light activating movements during 10 s of observation. For each experimental condition, two replicates per plate were performed and experimental triplicates were performed in separate plates.
2.7. Cell Cultures and Viability Test
HypoE22 cells were purchased from Cedarlane Cellution Biosystem and cultured in DMEM (Euroclone) supplemented with 10% (v/v) heat-inactivated fetal bovine serum and 1.2% (v/v) penicillin G/streptomycin in 75 cm2 tissue culture flasks (n = 5 individual culture flasks for each condition). The cultured cells were maintained in a humidified incubator with 5% CO2 at 37 °C. For cell differentiation, cell suspension at a density of 1 × 106 cells/mL was treated with various concentrations (10, 50, and 100 ng/mL) of phorbol myristate acetate (PMA, Fluka) for 24 h or 48 h (induction phase). Thereafter, the PMA-treated cells were washed twice with pH 7.4 phosphate buffer solution (PBS) to remove PMA and non-adherent cells, whereas the adherent cells were further maintained for 48 h (recovery phase). Morphology of cells was examined under an inverted phase-contrast microscope. To assess the basal cytotoxicity of herbal extract, a viability test was performed on 96 microwell plates, using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) test. Cells were incubated with extracts (ranging concentration 1–100 µg/mL) for 24 h. After the treatment period, 10 μL of MTT (5 mg/mL) were added to each well and incubated for 3 h. The formazan dye formed was extracted with dimethyl sulfoxide and absorbance recorded. Effects on cell viability were evaluated in comparison to the untreated control group.
2.8. RNA Extraction, Reverse Transcription, and Real-Time Reverse Transcription Polymerase Chain Reaction (Real-Time RT PCR)
Total RNA was extracted from the cells using TRI Reagent (Sigma-Aldrich, St. Louis, MO, USA), according to the manufacturer’s protocol. Contaminating DNA was removed using two units of RNase-free DNase 1 (DNA-free kit, Ambion, Austin, TX, USA). The RNA solution was quantified at 260 nm by spectrophotometer reading (BioPhotometer, Eppendorf, Hamburg, Germany) and its purity was assessed by the ratio at 260 and 280 nm readings. The quality of the extracted RNA samples was also determined by electrophoresis through agarose gels and staining with ethidium bromide under UV light. Ine microgram of total RNA was reverse transcribed using a High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA). Reactions were incubated in a 2720 Thermal Cycler (Applied Biosystems, Foster City, CA, USA) initially at 25 °C for 10 min, then at 37 °C for 120 min, and finally at 85 °C for 5 s. Gene expression was determined by quantitative real-time PCR using TaqMan probe-based chemistry (Applied Biosystems, Foster City, CA, USA). PCR primers and TaqMan probes were obtained from Applied Biosystems (Assays-on-Demand Gene Expression Products, Rn02531967_s1 for BDNF gene; Rn01525859_g1 for TNF-α). β-actin (Applied Biosystems, Foster City, CA, USA, Part No. 4352340E) was used as the housekeeping gene. The real-time PCR was carried out in triplicate for each cDNA sample in relation to each of the investigated genes. Data were elaborated with the Sequence Detection System (SDS) software version 2.3 (Applied Biosystems, Foster City, CA, USA).
2.9. Bioinformatics
The chemical structures were prepared with ChemSketch software and the related canonical SMILES were then processed by the STITCH platform, for predicting putative pharmacological targets. The identification of predicted targets was confirmed through the use of UniProt database. Protein–protein interactions were predicted through STRINGH bioinformatics platform. Docking calculations were conducted through the Autodock Vina of PyRx 0.8 software. Crystal structures of target proteins were derived from the Protein Data Bank (PDB) with PDB IDs as follows: 5FDR (Induced myeloid leukemia cell differentiation protein (MCL1)); 1QKU (Estrogen receptor 1 (ESR1)). Discovery studio 2020 visualizer was employed to investigate the protein–ligand non-bonding interactions.
2.10. Statistical Analysis
GraphPad Prism for Windows v5.01 (GraphPad Software, San Diego, CA, USA) was used to analyze the experimental results. The means ± SD were determined for each experimental group and analyzed using one-way analysis of variance (ANOVA), followed by a Newman–Keuls comparison multiple test.
3. Results and Discussion
In the present study, two extraction methods, namely maceration and homogenizer assisted extraction were used to see if there was an effect on the yield of bioactive compounds and biological properties of the extracts. The maceration technique was selected to preserve thermolabile compounds in the tested plant materials. Regarding homogenizer assisted extraction, this technique was used as one of green extraction techniques with shorter extraction time. Thus, the traditional (maceration) and green extraction (homogenizer assisted extracts) methods were compared.
Spectrophotometry is one of the relatively simple techniques for quantification of plant total phenolics and total flavonoids [17]. In the present study, spectrophotometric determination of extracts of J. curcas were found to possess significantly higher total phenolic contents (TPC) in the leaf extracts than stem bark extracts (range: 5.79–48.95 mg GAE/g). Conversely, the highest TPC was yielded in the stem bark extract of J. gossypiifolia obtained by homogenizer assisted extraction (62.83 ± 2.05 mg GAE/g) compared with the other extracts of the plant (42.62–49.05 mg GAE/g) (Table 1).

Table 1.
Total bioactive contents (TPC and TFC) and total antioxidant capacity (phosphomolybdenum assay) of the tested extracts.
A similar trend was noted for the extracts with regard to their contents of total flavonoids. For instance, the leaf extracts of J. curcas showed significantly higher total flavonoid contents (TFC) than the stem bark extracts (range: 1.64–13.99 mg RE/g). On the other hand, for J. gossypiifolia, the highest and lowest TFC were yielded by the stem bark extract and leaf extract, respectively, both obtained by homogenizer assisted extraction (17.63 ±0.34 mg RE/g and 6.97 ± 0.32 mg RE/g, respectively). The leaf and stem bark extracts of J. gossypiifolia obtained by maceration showed TFC 11.04 ± 0.59 mg RE/g, and 12.71 ± 0.10 mg RE/g, respectively (Table 1).
In particular, J. gossypiifolia was found to yield the highest TPC and TFC when homogenizer assisted extraction was used. Indeed, other studies have also shown homogenizer assisted extraction to present high potential for extracting phenolics and antioxidant compounds [18]. Interestingly, several studies have also demonstrated that extraction techniques play a crucial role in the yield of phenolic content from plant extracts [19,20].
Other researchers also determined the TPC and TFC from different parts of J. curcas and J. gossypiifolia. For instance, investigation of the methanolic extracts of J. gossypiifolia revealed the leaves to have higher total phenolic content (65.66 mg GAE/g) compared to the stem portion (33.332 mg GAE/g) [21]. Additionally, the total phenolic content of crude extract J. curcas fruit was found to possess TPC 7.04 ± 0.10 mg GAE/g of extract and 0.22–18.61 mg GAE/g of extract for its fraction [22]. The polyphenolic contents of the ethanol, methanol and aqueous extracts of the stem bark of J. curcas were also assessed by Igbinosa, et al. [23], whereby the total phenol and total flavonoid were obtained in amounts of 10.92–28.87 mg tannic acid/g extract and 6.28–11.18 mg quercetin/g extract, respectively.
HPLC-MS/MS analysis was also performed on extracts obtained by homogenizer assisted extraction. A total of 68 compounds were revealed to be present in the leaf extract of J. curcas, whereas only 44 compounds were detected in the stem bark extract. However, many compounds were found in both extracts, such as loliolide, orientin, soorientin, vitexin, isovitexin, isoquercitrin, quercetin, jasmonic acid, luteolin, sebacic acid, apigenin, 12-oxo phytodienoic acid, hydroxyoctadecatrienoic acid, hydroxyoctadecadienoic acid, hydroxyhexadecenoic acid, α-linolenic acid, linoleic acid, palmitic acid, oleic acid and stearic acid (Table 2 and Table 3). Detailed chemical composition is also available as Supplementary Materials.

Table 2.
Chemical composition of J. curcas leaves (HAE).

Table 3.
Chemical composition of J. curcas stem bark (HAE).
On the other hand, 78 compounds were identified in J. gossypifolia leaf extract obtained by homogenizer assisted extraction, while 64 compounds were detected in the stem bark extract of J. gossypifolia obtained by the same method. Many compounds were also found to be present in both extracts of J. gossypiifolia, such as quinic acid, catechin, epiatechin, scopoletin, ferulic acid, loliolide, vicenin-1, orientin, vicenin-3, vitexin, isoorientin, dihydrokaempferol, isovitexin, luteolin-7-O-glucoside, isoquercitrin, quercetin, dodecanedioic acid, undecanedioic acid, isorhamnetin, apigenin, sebacic acid, naringenin, jasmonic acid, luteolin, kaempferol, hydroxyoctadecatrienoic acid, hydroxyoctadecadienoic acid, α-linolenic acid, linoleic acid, palmitic acid, oleic acid, stearic acid, 12-oxo phytodienoic acid, stearidonic acid, and 12-oxo phytodienoic acid (Table 4 and Table 5).

Table 4.
Chemical composition of J. gossypifolia leaves (HAE).

Table 5.
Antioxidant properties of the tested extracts.
Indeed, for both studied Jatropha species, HPLC-MS/MS analysis showed the leaf extracts to be richer in phytochemical compounds compared to the stem bark extracts. However, the chemical profiles of both Jatropha species indicate that some compounds were uniformly distributed throughout the plant, that is the leaves and the stem bark. It has been suggested that some compounds are more concentrated in the roots and seeds and others in the green tissues of the aerial part such as stems and leaves. This is because each organ has a specialization that it must fulfill according to its physiological function. Interestingly however, in a previous study, the contents of each phenolic compound from the leaves and stems of two other Jatropha species, J. cinerea and J. cordata were found to significantly differ between species and plant organs [28]. Similarly, aqueous leaf extracts of J. gossypiifolia and J. mollissima prepared by decoction showed quantitatively different chemical profiles by HPLC-DAD [29].
Antioxidant properties of the tested extracts were investigated by different methods and the results are summarized in Table 5. In the present study, all extracts were found to possess free radical scavenging ability in both DPPH and ABTS assays. In the case of J. curcas extracts, the scavenging capacity in the DPPH assay ranged from 6.89 to124.70 mg TE/g, whereas in the ABTS assay, it ranged from 20.20 to 149.12 mg TE/g. For J. gossypiifolia extracts, the scavenging potential ranges were 48.14–193.93 mg TE/g and 86.88–160.00 mg TE/g in DPPH and ABTS assays, respectively. Interestingly, the leaf extracts of J. curcas were observed to exhibit significantly higher scavenging activity than the stem bark extracts, with the leaf extract obtained by the HAE method showing the highest activity. On the other hand, the stem bark-HAE extract of J. gossypiifolia was found to be the most prominent radical scavenger (Table 5).
In the present work, the extracts of J. curcas showed reducing activity of 21.07–256.21 mg TE/g and 14.02–97.03 mg TE/g in CUPRAC and FRAP assays, respectively. Remarkably, the same trend as in the radical scavenging assays (DPPH and ABTS) could be observed in the reducing assays (CUPRAC and FRAP). The leaf extracts of J. curcas showed better reducing activity compared to the stem bark extracts in both CUPRAC and FRAP assays. As for J. gossypiifolia extracts, reducing activities of 243.59–333.30 mg TE/g and 101.32–168.93 mg TE/g were obtained in CUPRAC and FRAP assays, respectively, with the highest activity displayed by stem bark-HAE extract (Table 6).

Table 6.
Enzyme inhibitory effects of the tested extracts.
Moreover, the extracts of both species were found to act as metal chelators (J. curcas: 3.21–10.98 mg EDTAE/g; J. gossypiifolia: 13.67–18.98 mg EDTAE/g). However, it was revealed that the leaf extracts of both J. curcas and J. gossypiifolia showed higher metal chelating activity compared to the stem bark extracts (Table 6). Interestingly, this could be due to the higher number of phytochemicals detected in the leaf extracts obtained by the homogenizer assisted extraction compared to the stem bark extracts.
In the phosphomolybdenum assay, the highest total antioxidant capacity was shown by stem bark extracts of J. curcas (3.55 mM TE/g and 3.34 mM TE/g in extracts obtained by maceration and homogenizer assisted extraction, respectively), in contrast to the leaf extracts of J. curcas (2.27 and 2.57 mM TE/g). The total antioxidant capacity of the J. gossypifolia extracts ranged from 1.76 to 2.44 mM TE/g, with the lowest and highest activity demonstrated by stem bark/maceration and leaf/homogenizer assisted extraction extracts respectively (Table 6).
Numerous previous studies have also confirmed the antioxidant potential of J. curcas and J. gossypiifolia using various experimental models. For instance, using DPPH assay, Rofida [30] determined the antioxidant activity of ethanolic leaf, fruit, stem bark and root extracts of J. curcas (IC50: 26.44–420.98 µg/mL) and J. gossypiifolia (IC50: 10.79–98.63 µg/mL), obtained by maceration. Furthermore, the results showed that J. curcas stem bark extract possessed higher antioxidant activity, whereas in J. gossypiifolia, the leaves and stem bark extracts displayed better antioxidant activity [30]. In addition, based on phosphomolybdate assay and DPPH radical scavenging activity, the ethyl acetate extract of J. gossipiifolia was found to have high antioxidant activity when compared to other extracts studied by Saishri, et al. [31]. Even though the extract yield of ethyl acetate extract (4.6%) was lower when compared to the yield of ethanol extract (9.6%) and water extract (18%), the high antioxidant power exhibited by the ethyl acetate extract was suggested to be due to the presence of bioactive constituents.
Moreover, in the study of Saosoong, Litthanapongsatorn and Ruangviriyachai [22], the antioxidant activity of the crude extract of J. curcas fruit was found to be 270.98 ± 0.59 μmol Fe/g of extract using the phenanthroline method, while the extract gave an IC50 of 14.09 ± 0.05 mg/mL with the DPPH method. In particular, the methanolic fraction showed the highest antioxidant activity with an IC50 of 0.04 ± 0.02 mg/mL with the DPPH method and an antioxidant activity of 207.53 ± 2.58 μmol Fe/g of extract with the phenanthroline method. A good correlation among antioxidant activity in both methods and total phenolic content was also observed.
In fact, significant strong correlations have been previously established between TPC and antioxidant potentials of plant extracts, signifying that the polyphenolic compounds present in the plant extracts contributed to their antioxidant activity and reducing capability [32]. These findings were in agreement with the results of the present study, showing extracts with higher TPC exhibiting higher antioxidant activity.
Cholinesterase inhibitors function by inhibiting cholinesterase from hydrolyzing acetylcholine into its components of acetate and choline. This allows for an increase in the availability and duration of action of acetylcholine in neuromuscular junctions. Most commonly, their use is in treating neurogenerative diseases such as Alzheimer disease, Parkinson disease, and Lewy body dementia. Indeed, plants have been widely assessed as potent sources of natural cholinesterase inhibitors [33,34]. In the present study, while the leaf extract of J. curcas obtained by maceration did not show any AChE inhibition, leaf extracts obtained by homogenizer assisted extraction and stem bark extracts of J. curcas showed AChE inhibitory potential ranging from 2.04 to 2.36 mg GALAE/g. Comparatively, all extracts of J. gossypifolia were found to be active as AChE inhibitors (1.12–2.06 mg GALAE/g). Additionally, BChE inhibition was exhibited by all extracts of J. curcas, with the stem bark extracts showing higher potential than leaf extracts (1.59–3.68 mg GALAE/g). However, with the exception of the leaf/maceration extract of J. gossypifolia, which showed no activity against BChE, all other extracts of J. gossypifolia were found to inhibit BChE with an inhibition range of 0.50–0.72 mg GALAE/g (Table 6).
Eighteen species belonging to Convolvulaceae, Crassulaceae, Euphorbiaceae, Leguminosae, Malvaceae, Moraceae, Nyctaginaceae and Rutaceae families were tested for their anti-AChE in the study of Feitosa, et al. [35], whereby among the most active plants, J. curcas (IC50 = 0.25 mg/mL) and J. gossypiifolia (IC50 = 0.05 mg/mL) were also found to possess promising anti-AChE activity compared to galantamine (IC50 = 0.37 × 10−3 mg/mL). The authors suggested that there could be compounds with a similar activity to galanthamine present in the plant extracts. Saleem, et al. [36] also reported the cholinesterase inhibitory potentials of J. gossypiifolia. For instance, the root dichloromethane fraction (% inhibition: 65.43 ± 0.11%), root methanol fraction (62.79 ± 0.34%) and leaf dichloromethane fraction (57.71 ± 0.15%) of J. gossypiifolia showed significant AChE inhibitory activity relative to other tested fractions when compared with the standard, eserine (91.29 ± 1.17%). Furthermore, BChE enzyme inhibitory results showed that the root dichloromethane fraction (80.46 ± 0.44%) and leaf ethyl acetate extract (77.34 ± 0.34%) displayed significant BChE enzyme inhibitory activity relative to other tested fractions when compared with the standard, eserine (82.82 ± 1.09%).
Tyrosinase (EC 1.14.18.0) is a copper-containing mixed-function oxidase that is ubiquitously expressed in animals, plants, and microorganisms. Furthermore, tyrosinase is a key rate-limiting enzyme that can catalyze enzyme browning and melanin synthesis. In humans, the overexpression of tyrosinase leads to the overproduction of melanin in the skin, which can trigger hyperpigmentation effects such as melasma, freckles, age spots, and melanoma [37]. In the present study, all extracts of J. curcas and J. gossypiifolia displayed anti-tyrosinase potential (J. curcas: 38.14–56.30 mg KAE/g; J. gossypiifolia: 50.43–57.59 mg KAE/g). However, while the leaf extracts of J. curcas exhibited the most potent activity against tyrosinase, the highest anti-tyrosinase effect was shown by the stem bark extracts of J. gossypiifolia (Table 6).
Interestingly, the higher anti-tyrosinase effect shown by the J. curcas leaf extracts and J. gossipiifolia extracts in the present study, were found to be correlated with the high antioxidant potentials of those extracts. In fact, an extremely interesting and delicate relationship exists between antioxidant defense systems and melanogenesis. This relationship is associated with ROS scavenging. The synergistic effect in this relationship increases the effectiveness of antioxidants in scavenging free radicals, while tyrosinase inhibitors work, thus reducing melanin production [38]. Additionally, in a previous study, the fraction of water extracts of new and fallen Sapium sebiferum (L.) Roxb. leaves were found to possess great antioxidant and tyrosinase inhibition activities, even better than those of the positive control (BHT and arbutin). Moreover, the tyrosinase inhibition effect was significantly and positively correlated with its copper chelating activity, which was suggested to be the mechanism of tyrosinase inhibition [39].
There are numerous conventional drugs available for diabetes mellitus, which vary in their mechanism of action. One of the pharmacological approaches is by using carbohydrate enzyme inhibitor drugs such as acarbose, voglibose and miglitol. These drugs inhibit both α-amylase and α-glucosidase, which are enzymes responsible for the breakdown of carbohydrates. However, these current antidiabetic drugs suffer from a number of undesirable side effects, leading researchers to seek traditional medicinal plants as alternatives for diabetic treatment [32]. In the current work, all of the extracts of J. curcas acted as dual inhibitors of amylase (0.28–0.62 mmol ACAE/g) and glucosidase (0.63–0.81 mmol ACAE/g). While the leaf extracts of J. curcas showed greater inhibition against amylase than the stem bark extracts; however, the stem bark extracts of J. curcas were found to display a better glucosidase inhibitory effect compared to the leaf extracts. On the other hand, with the exception of the stem bark-HAE extract of J. gossypifolia, which selectively inhibited amylase (0.49 ± 0.01 mmol ACAE/g), all of the other extracts of J. gossipifolia showed dual inhibition against the carbohydrate hydrolyzing enzymes (0.43–0.81 mmol ACAE/g) (Table 6).
Different extracts and fractions of the root, leaf and stem bark of J. gossypiifola were also screened for their α-glucosidase inhibitory property. n-Butanol and ethyl acetate fractions showed maximum enzyme inhibition for α-glucosidase with 67.93 ± 0.66 and 67.67 ± 0.71% and an IC50 of 218.47 ± 0.23 and 213.45 ± 0.12 μg/mL, respectively, while acarbose, used as a positive control, exhibited enzyme inhibition activity of 92.14 ± 0.38% with an IC50 of 38.24 ± 0.1 μg/mL [36].
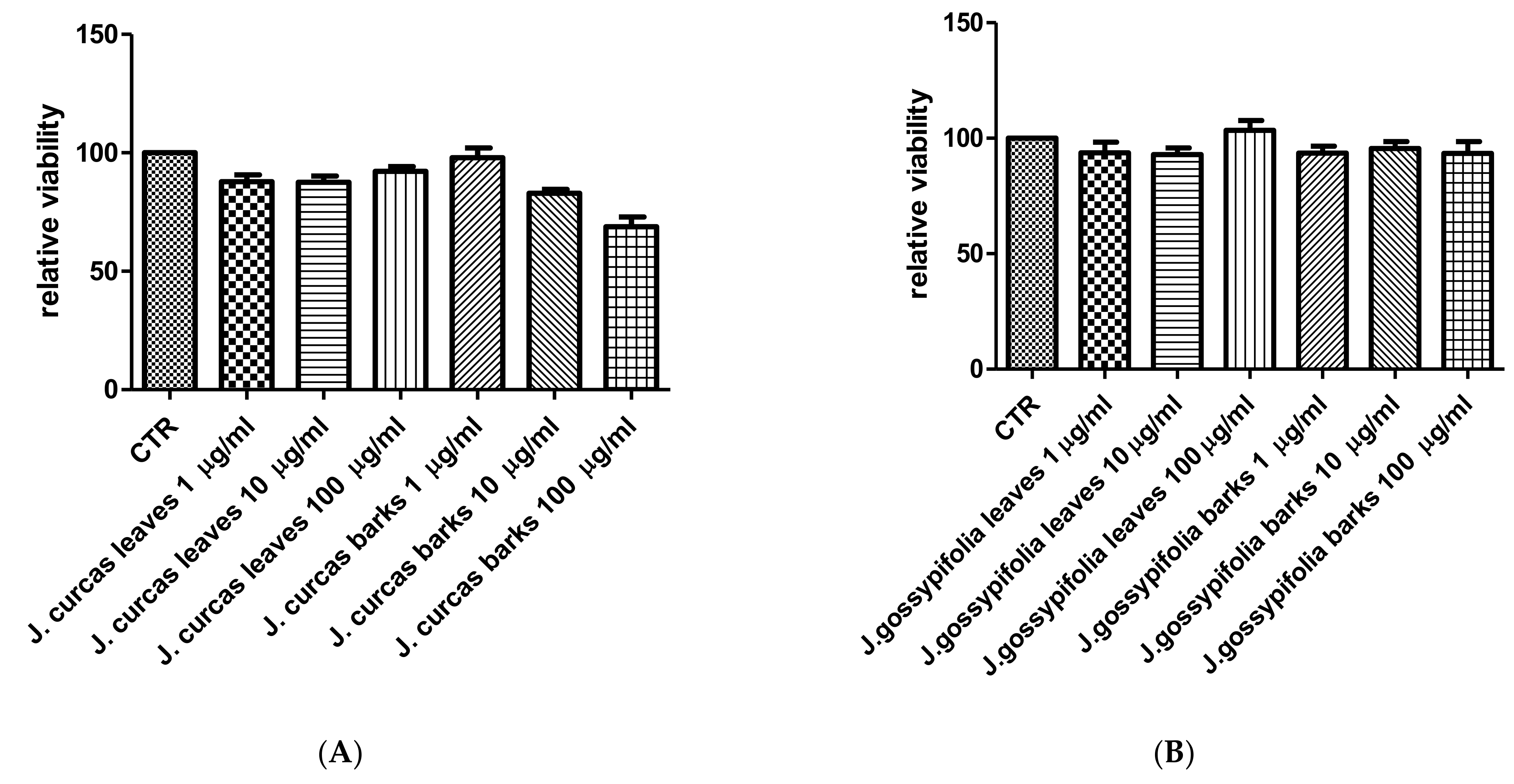
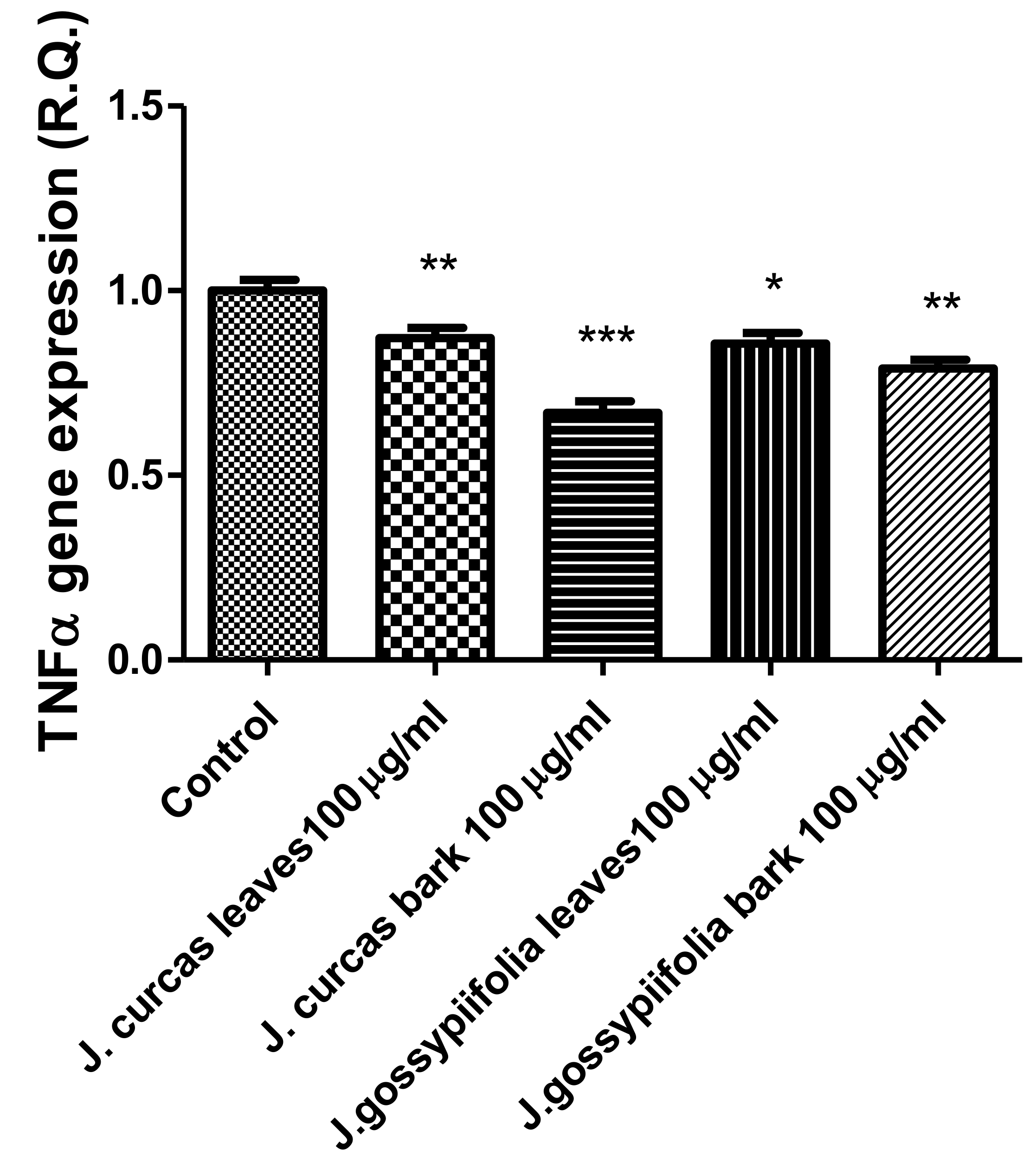
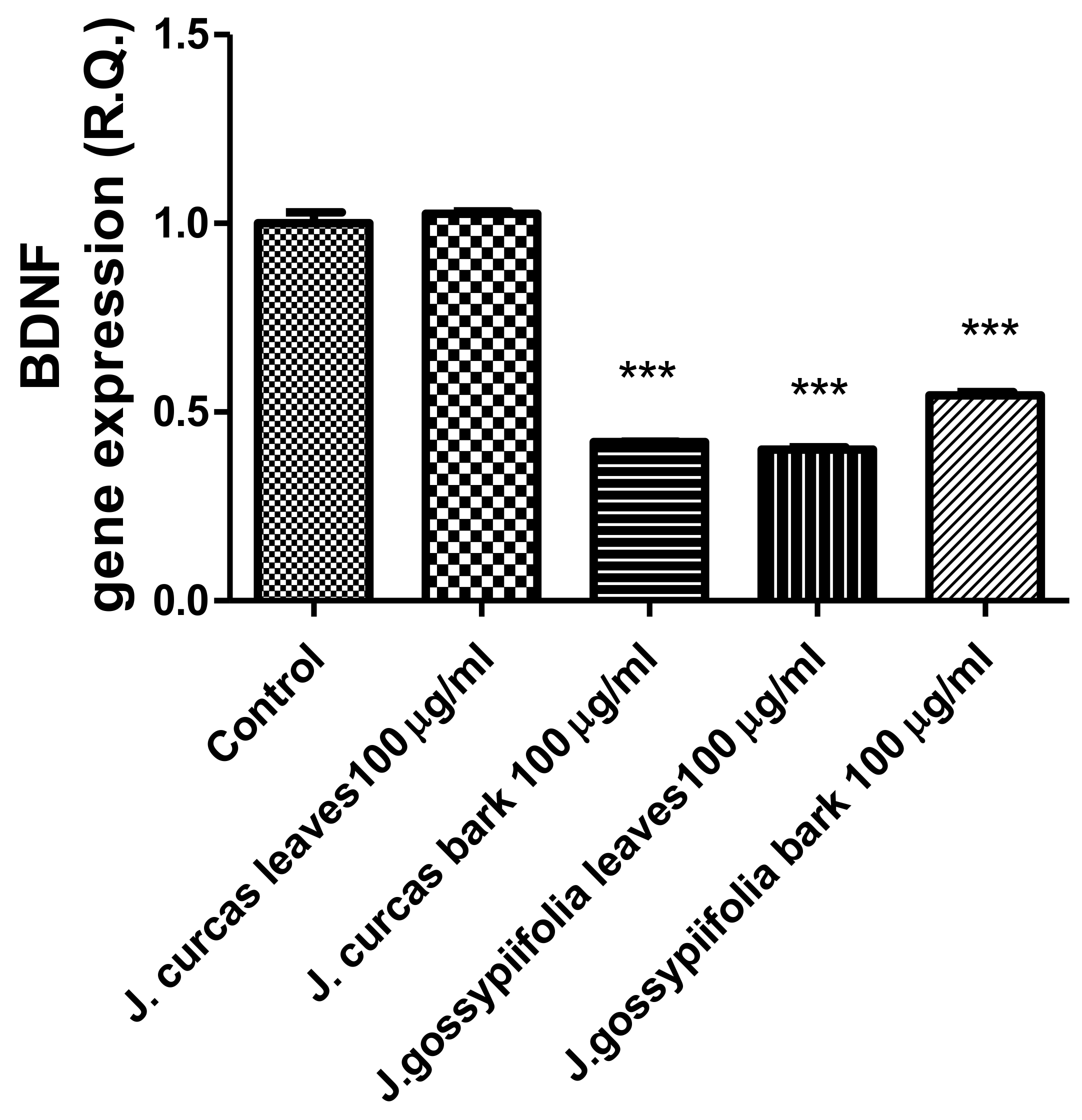
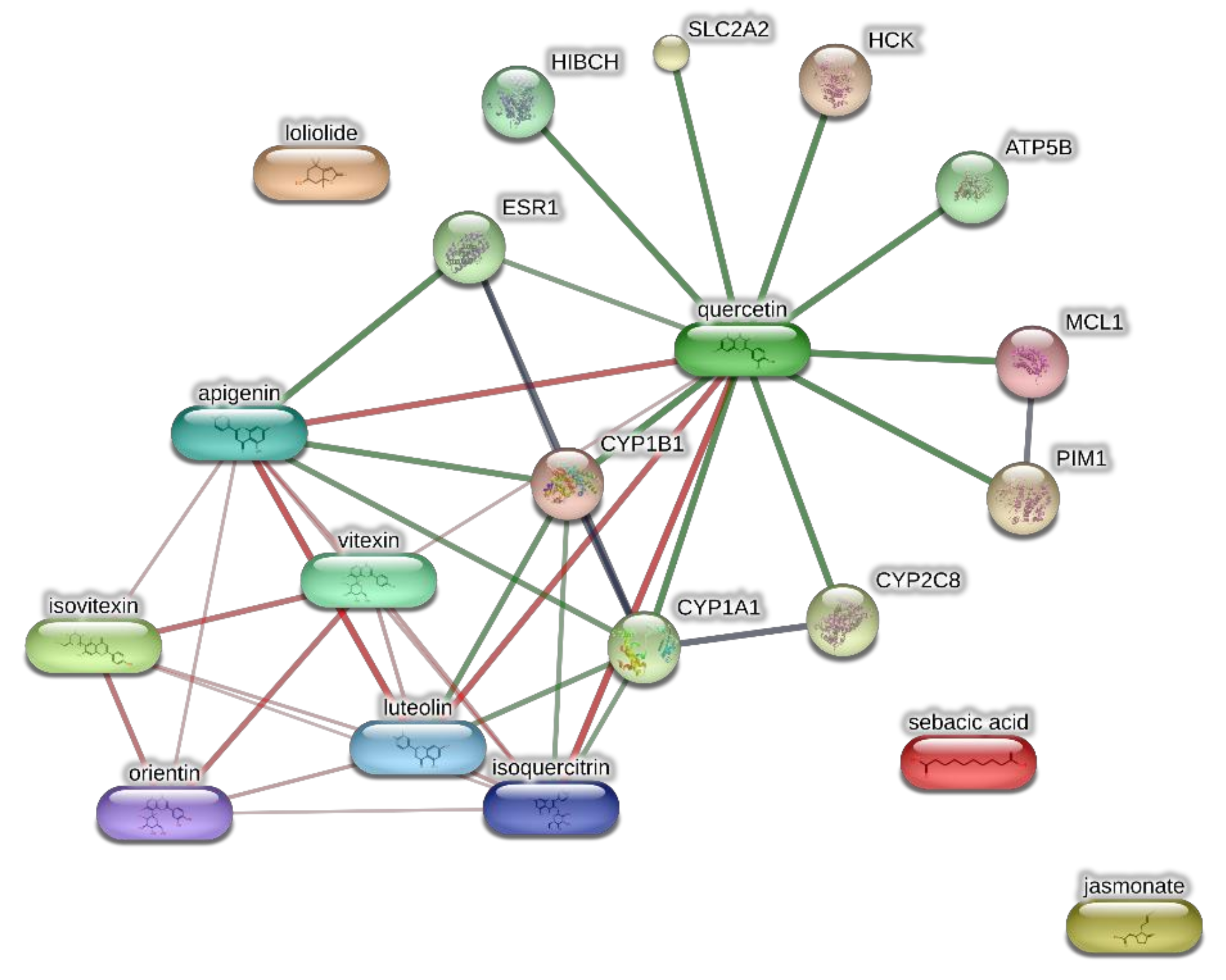
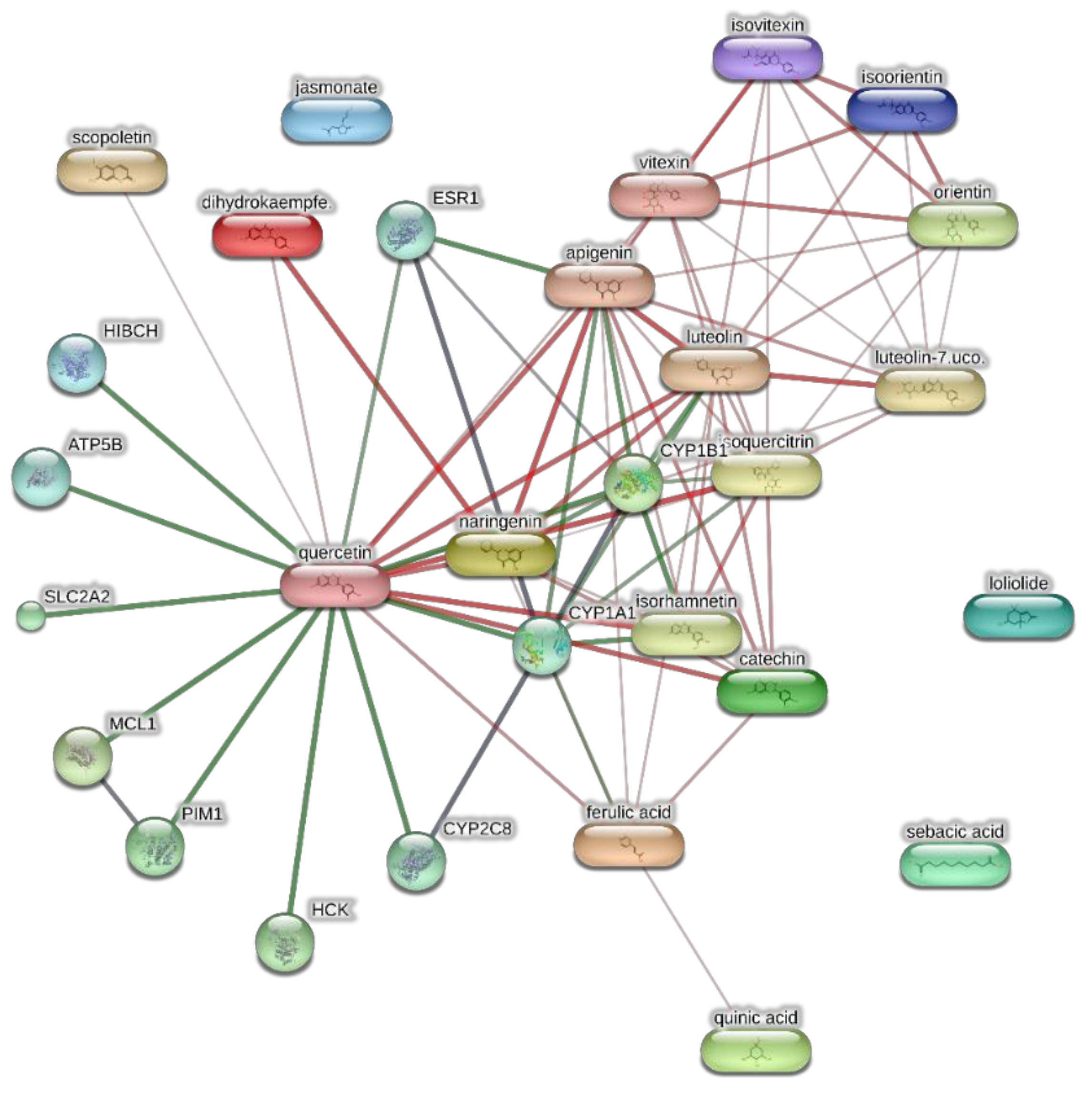
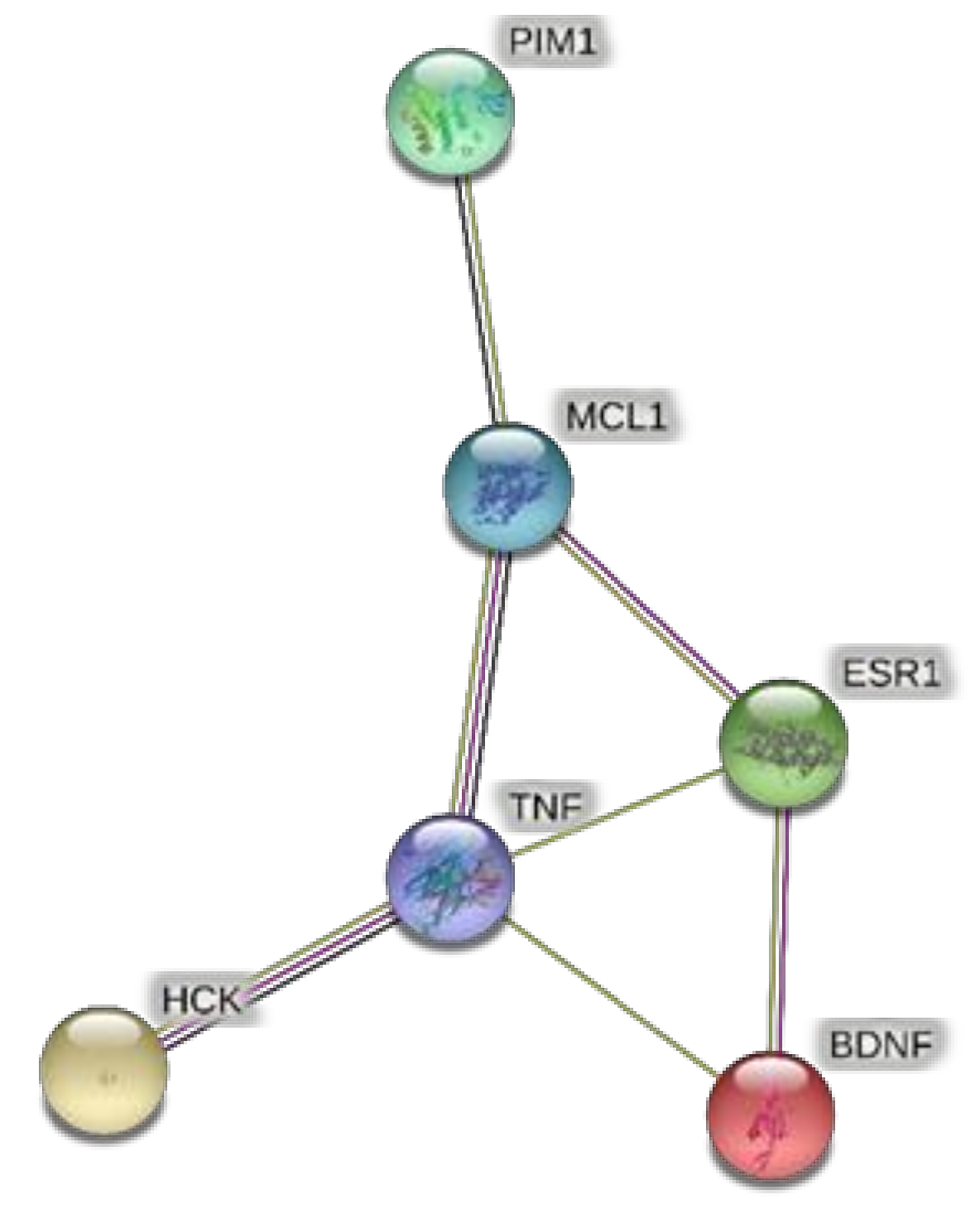
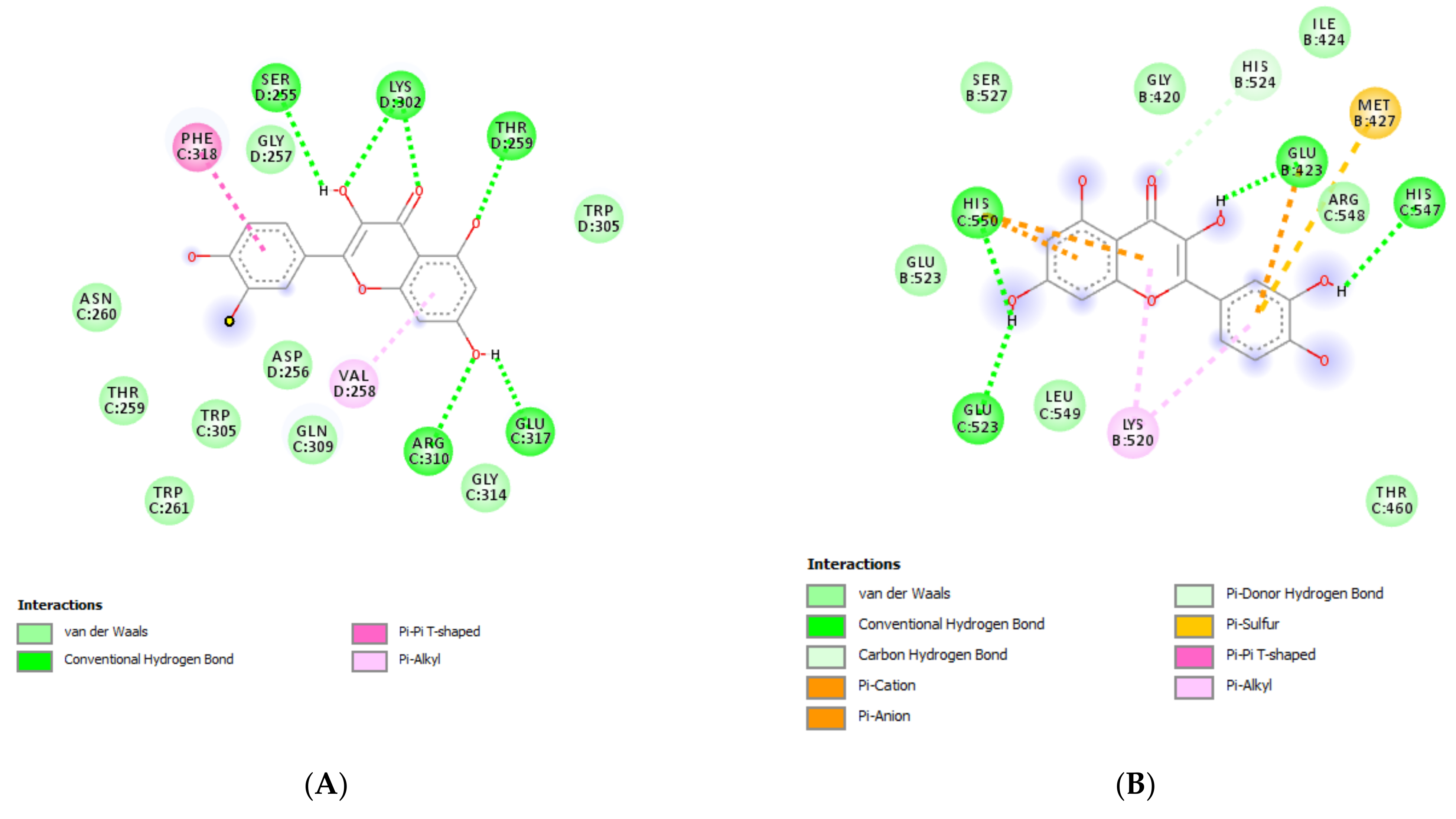
The extracts from J. curcas and J. gossypiifolia have been tested in the brine shrimp (Artemia salina) lethality test, which represents a valuable experimental model for predicting the limits of toxicity and biocompatibility in eukaryotic cells [40]. Specifically, the shrimp were exposed to scalar concentrations (0.1–20 mg/mL) of the extracts and the resulting LC50 values < 1 mg/mL indicate a high degree of toxicity in the nauplii. Although toxicological studies are still lacking for both Jatropha species, we cannot exclude that this intrinsic toxicity of the extracts could be related, at least in part, to the presence of terpenes, such as curcusones, but also flavonoids and saponins that could induce genotoxicity [41,42]. Considering the LC50 values yielded by the brine shrimp test, extract concentrations at least 10-fold lower (100 µg/mL) were chosen for the subsequent pharmacological tests [43]. Considering the intrinsic scavenging/reducing and anticholinesterase properties shown by the present extracts, the pharmacological assays were conducted using the non-tumoral hypothalamic HypoE22 cell line, which was demonstrated to be a useful experimental paradigm for investigating anti-inflammatory and neuromodulatory effects induced by herbal extracts [44]. Specifically, the HypoE22 cells were exposed to the extracts (1–100 µg/mL), and the cell viability was measured via MTT test, which showed a good tolerability of the hypothalamic cells at all tested concentrations. Indeed, the cell viability was always ≥70% (Figure 1A,B) compared to the untreated control group, and this was considered as an index of cell tolerability to the extract exposition in the 24 h following treatment [45]. Considering the results of the MTT test, the extract concentration of 100 µg/mL was chosen for the second set of experiments aiming to investigate the anti-inflammatory and neuromodulatory effects of the extracts. In this regard, the gene expression of TNFα and BDNF was measured, finding a significant reduction. Regarding the inhibition of TNFα (Figure 2), this is consistent, albeit partially, with the scavenging/reducing properties of the present extracts, but also with previous studies highlighting the capability of herbal extracts, with intrinsic antioxidant effects, to inhibit the gene expression of TNFα in HypoE22 cells [46]. However, the inhibition of the gene expression of BDNF (Figure 3), a neuropeptide playing a master role in neuroprotection [47], is discrepant with the effects of the extracts on TNFα and also with their antiradical properties. Nevertheless, we should consider that BDNF is also involved in the hypothalamic appetite-regulating network [48], with anorexigenic effects induced by its central administration [49]. The plasma levels of BDNF were lower in people suffering from anorexia, compared to healthy subjects, whereas the BDNF concentration tends to arise after normalization of body weight [50]. In this context, we hypothesize that BDNF modulation could be involved in the anorexigenic effect induced by J. curcas administration in rats [51]. Considering the results of the qualitative fingerprint analysis, a bioinformatics approach was conducted with the aim to identify the putative targets underlying the observed effects. In the case of J. curcas, the bioinformatics analysis, carried out on the platform STITCH, considered the following phytochemicals: loliolide, orientin, soorientin, vitexin, isovitexin, isoquercitrin, quercetin, jasmonic acid, luteolin, sebacic acid, and apigenin, present in the extracts from all J. curcas plant materials tested in the present study (Figure 4). While in the case of J. gossypifolia, the selected phytochemicals were quinic acid, catechin, epiatechin, scopoletin, ferulic acid, loliolide, vicenin-1, orientin, vicenin-3, vitexin, isoorientin, dihydrokaempferol, isovitexin, luteolin-7-O-glucoside, isoquercitrin, quercetin, isorhamnetin, apigenin, sebacic acid, naringenin, jasmonic acid, and luteolin (Figure 5). The bioinformatics predictions indicated, among the selected phytochemicals, prominent positions of quercetin, apigenin and naringenin in the scenario of putative interactions. Specifically, all of them were predicted to interact with estrogen receptor 1 (ESR1), whereas the sole apigenin displayed putative interactions with tyrosine-protein kinase HCK (HCK), playing a key role in regulating the innate immune response and with the apoptosis marker myeloid cell leukemia 1 (MCL1). Both ESR1 and MCL1 are expressed in the hypothalamus [52,53], whereas the bioinformatics platform STRINGH highlighted putative interactions with BDNF and TNFα (Figure 6). Therefore, the present bioinformatics analysis suggests that ESR1 and MCL1 could be targets of the selected phenolic compounds for mediating, at least in part, the inhibition of the gene expression of both BDNF and TNFα in the hypothalamus. In this regard, docking runs were also conducted to calculate the putative affinities of quercetin towards ESR1 and MCL1. The results of docking experiments (Figure 7A,B) showed identical micromolar affinities (1.9 µM) of quercetin towards the selected proteins. In the case of MCL1, the quercetin affinity is mainly due to the formation of hydrogen bonds with the protein, whereas pi-interactions also seem to be involved in the binding of quercetin with ESR1. Overall, these results further suggest that the present target proteins are crucial for mediating the observed pharmacological effects in the hypothalamus.

Figure 1.
Null effect induced by the extracts (1–100 µg/mL) of J. curcas (A) and J. gossypiifolia (B) on HypoE22 cell viability.

Figure 2.
Inhibitory effects of J. curcas and J. gossypiifolia (100 µg/mL) on TNFα gene expression, in HypoE22 cells. ANOVA, p < 0.0001; *** p < 0.001, ** p < 0.01, * p < 0.05 vs. Control.

Figure 3.
Inhibitory effects of J. curcas and J. gossypiifolia (100 µg/mL) on BDNF gene expression, in HypoE22 cells. ANOVA, p < 0.0001; *** p < 0.001 vs. Control.

Figure 4.
Components-targets analysis conducted through the bioinformatics platform STITCH for unravelling putative targets underlying the pharmacological effects on the extracts of J. curcas. The network pharmacology approach considered the most representative phytocompounds of the extracts, namely loliolide, orientin, soorientin, vitexin, isovitexin, isoquercitrin, quercetin, jasmonic acid, luteolin, sebacic acid, and apigenin.
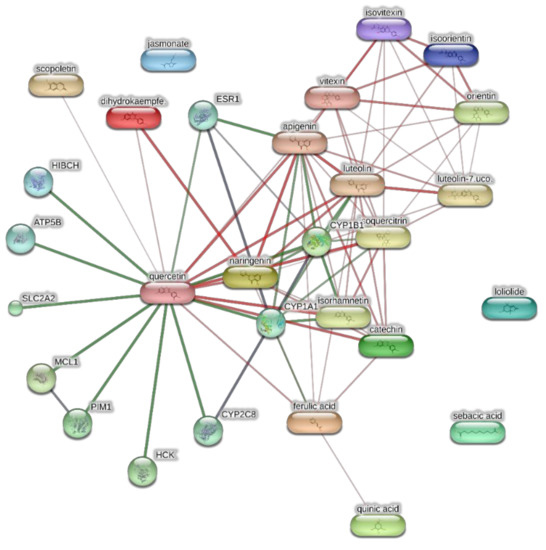
Figure 5.
Components-targets analysis conducted through the bioinformatics platform STITCH for unravelling putative targets underlying the pharmacological effects on the extracts of J. gossypiifolia. The network pharmacology approach considered the most representative phytocompounds of the extracts, namely quinic acid, catechin, epiatechin, scopoletin, ferulic acid, loliolide, vicenin-1, orientin, vicenin-3, vitexin, isoorientin, dihydrokaempferol, isovitexin, luteolin-7-O-glucoside, isoquercitrin, quercetin, isorhamnetin, apigenin, sebacic acid, naringenin, jasmonic acid, and luteolin.

Figure 6.
Protein–protein interactions predicted through the bioinformatics platform STRINGH. The bioinformatics resource showed interactions of BDNF with ESR1. While TNFα was predicted to interact with both HCK and MCL1. Considering the expression of ESR1 and MCL1 in the hypothalamus, the present bioinformatics prediction suggests that ESR1 and MCL1 could be targets underlying the modulation of BDNF and TNFα induced by the extracts, in HypoE22 cells.

Figure 7.
(A) Putative interactions between quercetin and myeloid cell leukemia 1 (MCL1; PDBID: 5FDR). Free energy of binding (ΔG) and affinity (Ki) are −7.8 kcal/mol and 1.9 µM, respectively. (B) Putative interactions between quercetin and estrogen receptor 1 (ESR1; PDBID: 1QKU). Free energy of binding (ΔG) and affinity (Ki) are −7.8 kcal/mol and 1.9 µM, respectively.
4. Conclusions
This study demonstrated the multifunctional potential of two Jatropha species, J. curcas and J. gossypiifolia as antioxidant, antidiabetic, anti-neurodegenerative and anti-hyperpigmenting agents. Moreover, the spectrophotometric coupled with HPLC-MS analysis revealed the plants to contain notable bioactive compounds that could have resulted in the biological properties demonstrated herein. This was most apparent for the antioxidant capacity whereby the leaf extract of J. curcas, while the stem bark extract of J. gossypiifolia, both obtained by homogenizer assisted extraction showed the most significant free radical scavenging and reducing activity, and were also found to contain higher TPC and TFC. Furthermore, the homogenizer assisted extraction could be considered as a better extraction method than maceration to extract antioxidant compounds. The extracts were also tested in hypothalamic HypoE22 cells, and the pattern of gene expression coupled to bioinformatics analysis indicated anti-inflammatory and neuromodulatory effects, thus supporting further investigations, especially in experimental models of obesity. The data retained from the present study suggest the use of these two species as therapeutically important plants. Nevertheless, more intense investigations in vivo and under clinical settings could help to assess their respective safety profile.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/antiox10050792/s1, Table S1: Chemical composition of J. curcas leaves (HAE), Table S2: Chemical composition of J. curcas stem bark (HAE), Table S3: Chemical composition of J. gossypifolia leaves (HAE).
Author Contributions
Conceptualization, G.Z., G.O., C.F. and L.M.; methodology, G.Z., G.O., C.F. and L.M.; software, G.Z., G.O., C.F. and L.M.; validation, G.Z., G.O., C.F. and L.M.; formal analysis, G.Z., G.O., C.F. and L.M.; investigation, K.I.S., G.A., O.K.E., J.B.S., S.L., S.C.D.S., L.R., A.C., J.J. and Z.C.; resources, G.Z., G.O., C.F. and L.M.; data curation, G.Z., G.O., C.F. and L.M.; writing—original draft preparation, M.F.M. and C.F.; writing—review and editing, M.F.M., G.Z., G.O., C.F. and L.M.; visualization, L.B.; supervision, L.B.; project administration, G.Z., G.O., C.F. and L.M.; funding acquisition, G.O., C.F. and L.M. All authors have read and agreed to the published version of the manuscript.
Funding
The study was supported by Italian Ministry funds (FAR 2020), granted to Giustino Orlando, Luigi Menghini and Claudio Ferrante.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Acknowledgments
The present article is part of the third mission activities of the Botanic Garden “Giardino dei Semplici” planned for the 20th anniversary of the establishment.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kumar, A.; Sharma, S. An evaluation of multipurpose oil seed crop for industrial uses (Jatropha curcas L.): A review. Ind. Crop. Prod. 2008, 28, 1–10. [Google Scholar] [CrossRef]
- Félix-Silva, J.; Giordani, R.B.; Silva, A.A.D., Jr.; Zucolotto, S.M.; Fernandes-Pedrosa, M.D.F. Jatropha gossypiifolia L. (Euphorbiaceae): A Review of Traditional Uses, Phytochemistry, Pharmacology, and Toxicology of This Medicinal Plant. Evid. Based Complement. Altern. Med. 2014, 2014, 369204. [Google Scholar] [CrossRef]
- Sabandar, C.W.; Ahmat, N.; Jaafar, F.M.; Sahidin, I. Medicinal property, phytochemistry and pharmacology of several Jatropha species (Euphorbiaceae): A review. Phytochemistry 2013, 85, 7–29. [Google Scholar] [CrossRef]
- Abdelgadir, H.; Van Staden, J. Ethnobotany, ethnopharmacology and toxicity of Jatropha curcas L. (Euphorbiaceae): A review. S. Afr. J. Bot. 2013, 88, 204–218. [Google Scholar] [CrossRef]
- Devappa, R.K.; Makkar, H.P.S.; Becker, K. Jatropha Diterpenes: A Review. J. Am. Oil Chem. Soc. 2010, 88, 301–322. [Google Scholar] [CrossRef]
- Cavalcante, N.B.; Santos, A.D.; Almeida, J.R.G. The genus Jatropha (Euphorbiaceae): A review on secondary chemical metabolites and biological aspects. Chem. Interact. 2020, 318, 108976. [Google Scholar] [CrossRef]
- Oduola, T.; Adeosun, G.O.; Oduola, T.A.; Avwioro, G.O.; Oyeniyi, M.A. Mechanism of Action of Jatropha Gossypifolia Stem Latex as a Haemostatic Agent. Electron. J. Gen. Med. 2005, 2, 140–143. [Google Scholar] [CrossRef]
- Sukohar, A.; Herawati, H.; Puspasari, G. Anticancer activity of Jatrophone an isolated compound from jatropha gossypifolia plant against hepatocellular cancer cell hep G2 1886. Biomed. Pharmacol. J. 2017, 10, 667–673. [Google Scholar]
- Dahake, R.; Roy, S.; Patil, D.; Rajopadhye, S.; Chowdhary, A. Potential anti-HIV activity of Jatropha curcas Linn. Leaf extracts. J. Antivir. Antiretrovir. 2013, 5, 160–165. [Google Scholar] [CrossRef]
- Warra, A. Cosmetic potentials of physic nut (Jatropha curcas Linn.) seed oil: A review. Am. J. Sci. Ind. Res. 2012, 3, 358–366. [Google Scholar] [CrossRef]
- Shetty, S.; Udupa, S.L.; Udupa, A.L.; Vollala, V.R. Wound healing activities of Bark Extract of Jatropha curcas Linn in albino rats. Saudi Med. J. 2006, 27, 1473–1476. [Google Scholar] [PubMed]
- Sangeetha, J.; Divya, K.; Vamsikrishna, M.P.A.; Rani, G.L. Anti-Inflammatory and Antibacterial Activity of Jatropha curcas Linn. Asian J. Pharm. Res. Health Care 2010, 2, 3. [Google Scholar]
- Slinkard, K.; Singleton, V.L. Total phenol analysis: Automation and comparison with manual methods. Am. J. Enol. Vitic. 1977, 28, 49–55. [Google Scholar]
- Zengin, G.; Nithiyanantham, S.; Locatelli, M.; Ceylan, R.; Uysal, S.; Aktumsek, A.; Selvi, P.K.; Maskovic, P. Screening of in vitro antioxidant and enzyme inhibitory activities of different extracts from two uninvestigated wild plants: Centranthus longiflorus subsp. longiflorus and Cerinthe minor subsp. auriculata. Eur. J. Integr. Med. 2016, 8, 286–292. [Google Scholar] [CrossRef]
- Zengin, G.; Uysal, A.; Diuzheva, A.; Gunes, E.; Jekő, J.; Cziáky, Z.; Picot-Allain, C.M.N.; Mahomoodally, M.F. Characterization of phytochemical components of Ferula halophila extracts using HPLC-MS/MS and their pharmacological po-tentials: A multi-functional insight. J. Pharm. Biomed. Anal. 2018, 160, 374–382. [Google Scholar] [CrossRef]
- Mollica, A.; Stefanucci, A.; Zengin, G.; Locatelli, M.; Macedonio, G.; Orlando, G.; Ferrante, C.; Menghini, L.; Recinella, L.; Leone, S. Polyphenolic composition, enzyme inhibitory effects ex-vivo and in-vivo studies on two Brassicaceae of north-central Italy. Biomed. Pharmacother. 2018, 107, 129–138. [Google Scholar] [CrossRef]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for Analysis of Plant Phenolic Compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef]
- Pereira, G.A.; Molina, G.; Arruda, H.S.; Pastore, G.M. Optimizing the Homogenizer-Assisted Extraction (HAE) of Total Phenolic Compounds from Banana Peel. J. Food Process. Eng. 2016, 40, e12438. [Google Scholar] [CrossRef]
- Sultana, B.; Anwar, F.; Ashraf, M. Effect of Extraction Solvent/Technique on the Antioxidant Activity of Selected Medicinal Plant Extracts. Molecules 2009, 14, 2167–2180. [Google Scholar] [CrossRef]
- Pietrzak, W.; Nowak, R.; Olech, M. Effect of extraction method on phenolic content and antioxidant activity of mistletoe extracts from Viscum album subsp. abietis. Chem. Pap. 2014, 68, 976–982. [Google Scholar] [CrossRef]
- Islam, M.; Hossain, M.S.; Rokeya, B. Biological Investigation of Jatropha gossypiifolia: A Stiff Medicinal Plant in Bangladesh. Iran. J. Pharm. Sci. 2017, 13, 35–48. [Google Scholar]
- Saosoong, K.; Litthanapongsatorn, I.; Ruangviriyachai, C. Antioxidant Activity of the Extracts from Jatropha curcas Fruit and Its Correlation with Total Phenolic Content. Orient. J. Chem. 2015, 32, 1121–1127. [Google Scholar] [CrossRef][Green Version]
- Igbinosa, O.O.; Igbinosa, I.H.; Chigor, V.N.; Uzunuigbe, O.E.; Oyedemi, S.O.; Odjadjare, E.E.; Okoh, A.I.; Igbinosa, E.O. Polyphenolic Contents and Antioxidant Potential of Stem Bark Extracts from Jatropha curcas (Linn). Int. J. Mol. Sci. 2011, 12, 2958–2971. [Google Scholar] [CrossRef] [PubMed]
- Abd-Alla, H.I.; Moharram, F.A.; Gaara, A.H.; El-Safty, M.M. Phytoconstituents of Jatropha curcas L. Leaves and their Immunomodulatory Activity on Humoral and Cell-Mediated Immune Response in Chicks. Z. Für Naturforschung C 2009, 64, 495–501. [Google Scholar] [CrossRef]
- Mitra, C.; Bhatnagar, S.; Sinha, M. Chemical examination of Jatropha curcas. Indian J. Chem. 1970, 8, 1047. [Google Scholar]
- Martínez-Herrera, J.; Siddhuraju, P.; Francis, G.; Dávila-Ortíz, G.; Becker, K. Chemical composition, toxic/antimetabolic constituents, and effects of different treatments on their levels, in four provenances of Jatropha curcas L. from Mexico. Food Chem. 2006, 96, 80–89. [Google Scholar] [CrossRef]
- Naengchomnong, W.; Thebtaranonth, Y.; Wiriyachitra, P.; Okamoto, K.; Clardy, J. Isolation and structure determination of four novel diterpenes from jatropha curcus. Tetrahedron Lett. 1986, 27, 2439–2442. [Google Scholar] [CrossRef]
- Vega-Ruiz, Y.C.; Hayano-Kanashiro, C.; Gámez-Meza, N.; Medina-Juárez, L.A. Determination of Chemical Constituents and Antioxidant Activities of Leaves and Stems from Jatropha cinerea (Ortega) Müll. Arg and Jatropha cordata (Ortega). Müll. Arg. Plants 2021, 10, 212. [Google Scholar]
- Félix-Silva, J.; Gomes, J.A.; Fernandes, J.M.; Moura, A.K.; Menezes, Y.A.; Santos, E.C.; Tambourgi, D.V.; Silva-Junior, A.A.; Zucolotto, S.M.; Fernandes-Pedrosa, M.F. Comparison of two Jatropha species (Euphorbiaceae) used popularly to treat snakebites in Northeastern Brazil: Chemical profile, inhibitory activity against Bothrops erythromelas venom and antibacterial activity. J. Ethnopharmacol. 2018, 213, 12–20. [Google Scholar] [CrossRef]
- Rofida, S. Antioxidant activity of Jatropha curcas and Jatropha gossypifolia by DPPH method. Pharm. J. Indonesia 2015, 2, 281–284. [Google Scholar]
- Saishri, R.; Ravichandran, N.; Vadivel, V.; Brindha, P. Pharmacognostic Studies on Leaf of Jatropha gossypifolia L. Int. J. Pharm. Sci. Res. 2016, 7, 163. [Google Scholar]
- Tay, Y.N.; A Bakar, M.H.; Azmi, M.N.; Saad, N.A.; Awang, K.; Litaudon, M.; Kassim, M.A. Inhibition of Carbohydrate Hydrolysing Enzymes, Antioxidant Activity and Polyphenolic Content of Beilschmiedia Species Extracts. In Proceedings of the IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2020; Volume 716, p. 12007. [Google Scholar]
- Murray, A.P.; Faraoni, M.B.; Castro, M.J.; Alza, N.P.; Cavallaro, V. Natural AChE Inhibitors from Plants and their Contribution to Alzheimer’s Disease Therapy. Curr. Neuropharmacol. 2013, 11, 388–413. [Google Scholar] [CrossRef] [PubMed]
- Santos, T.C.D.; Gomes, T.M.; Pinto, B.A.S.; Camara, A.L.; Paes, A.M.D.A. Naturally occurring acetylcholinesterase inhibitors and their potential use for Alzheimer’s disease therapy. Front. Pharmacol. 2018, 9, 1192. [Google Scholar] [CrossRef]
- Feitosa, C.M.; Freitas, R.M.; Luz, N.N.N.; Bezerra, M.Z.B.; Trevisan, M.T.S. Acetylcholinesterase inhibition by somes promising Brazilian medicinal plants. Braz. J. Biol. 2011, 71, 783–789. [Google Scholar] [CrossRef]
- Saleem, H.; Ahmad, I.; Shahid, M.N.; Gill, M.S.A.; Nadeem, M.F.; Mahmood, W.; Rashid, I. In Vitro Acetylcholinesterase and Butyrylcholinesterase Inhibitory Potentials of Jatropha gossypifolia Plant Extracts. Acta Pol. Pharm. Drug Res. 2016, 73, 419–423. [Google Scholar]
- Cui, H.-X.; Duan, F.-F.; Jia, S.-S.; Cheng, F.-R.; Yuan, K. Antioxidant and Tyrosinase Inhibitory Activities of Seed Oils from Torreya grandis Fort. ex Lindl. BioMed Res. Int. 2018, 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hao, M.-M.; Sun, Y.; Wang, L.-F.; Wang, H.; Zhang, Y.-J.; Li, H.-Y.; Zhuang, P.-W.; Yang, Z. Synergistic promotion on tyrosinase inhibition by antioxidants. Molecules 2018, 23, 106. [Google Scholar] [CrossRef]
- Fu, R.; Zhang, Y.; Guo, Y.; Chen, F. Antioxidant and tyrosinase inhibition activities of the ethanol-insoluble fraction of water extract of Sapium sebiferum (L.) Roxb. leaves. S. Afr. J. Bot. 2014, 93, 98–104. [Google Scholar] [CrossRef]
- Radulović, N.S.; Mladenović, M.Z.; Randjelovic, P.J.; Stojanović, N.M.; Dekić, M.S.; Blagojević, P.D. Toxic essential oils. Part IV: The essential oil of Achillea falcata L. as a source of biologically/pharmacologically active trans-sabinyl esters. Food Chem. Toxicol. 2015, 80, 114–129. [Google Scholar] [CrossRef]
- Liu, J.-Q.; Yang, Y.-F.; Li, X.-Y.; Liu, E.-Q.; Li, Z.-R.; Zhou, L.; Li, Y.; Qiu, M.-H. Cytotoxicity of naturally occurring rhamnofolane diterpenes from Jatropha curcas. Phytochemistry 2013, 96, 265–272. [Google Scholar] [CrossRef]
- Almeida, P.; Araújo, S.; Santos, I.; Marin-Morales, M.; Benko-Iseppon, A.; Santos, A.; Randau, K.; Brasileiro-Vidal, A. Genotoxic potential of leaf extracts of Jatropha gossypiifolia L. Genet. Mol. Res. 2016, 15, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ferrante, C.; Recinella, L.; Ronci, M.; Orlando, G.; Di Simone, S.; Brunetti, L.; Chiavaroli, A.; Leone, S.; Politi, M.; Tirillini, B.; et al. Protective effects induced by alcoholic Phlomis fruticosa and Phlomis herba-venti extracts in isolated rat colon: Focus on antioxidant, anti-inflammatory, and antimicrobial activities in vitro. Phytother. Res. 2019, 33, 2387–2400. [Google Scholar] [CrossRef]
- Recinella, L.; Chiavaroli, A.; di Giacomo, V.; Antolini, M.D.; Acquaviva, A.; Leone, S.; Brunetti, L.; Menghini, L.; Ak, G.; Zengin, G. Anti-Inflammatory and Neuromodulatory Effects Induced by Tanacetum parthenium Water Extract: Results from In Silico, In Vitro and Ex Vivo Studies. Molecules 2021, 26, 22. [Google Scholar] [CrossRef]
- Zengin, G.; Menghini, L.; Di Sotto, A.; Mancinelli, R.; Sisto, F.; Carradori, S.; Cesa, S.; Fraschetti, C.; Filippi, A.; Angiolella, L. Chromatographic analyses, in vitro biological activities, and cytotoxicity of Cannabis sativa L. essential oil: A multi-disciplinary study. Molecules 2018, 23, 3266. [Google Scholar] [CrossRef]
- Ferrante, C.; Recinella, L.; Locatelli, M.; Guglielmi, P.; Secci, D.; Leporini, L.; Chiavaroli, A.; Leone, S.; Martinotti, S.; Brunetti, L.; et al. Protective Effects Induced by Microwave-Assisted Aqueous Harpagophytum Extract on Rat Cortex Synaptosomes Challenged with Amyloid β-Peptide. Phytother. Res. 2017, 31, 1257–1264. [Google Scholar] [CrossRef]
- Brigadski, T.; Leßmann, V. The physiology of regulated BDNF release. Cell Tissue Res. 2020, 382, 15–45. [Google Scholar] [CrossRef]
- Xu, B.; Goulding, E.H.; Zang, K.; Cepoi, D.; Cone, R.D.; Jones, K.R.; Tecott, L.H.; Reichardt, L.F. Brain-derived neu-rotrophic factor regulates energy balance downstream of melanocortin-4 receptor. Nat. Neurosci. 2003, 6, 736–742. [Google Scholar] [CrossRef]
- Wang, C.; Bomberg, E.; Billington, C.; Levine, A.; Kotz, C.M. Brain-derived neurotrophic factor in the hypothalamic paraventricular nucleus reduces energy intake. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R1003–R1012. [Google Scholar] [CrossRef]
- Piotrowicz, Z.; Chalimoniuk, M.; Czuba, M.; Langfort, J. Rola neurotroficznego czynnika pochodzenia mózgowego w kontroli łaknienia. Postępy Biochem. 2020, 66, 205–212. [Google Scholar]
- Rakshit, K.; Darukeshwara, J.; Raj, K.R.; Narasimhamurthy, K.; Saibaba, P.; Bhagya, S. Toxicity studies of detoxified Jatropha meal (Jatropha curcas) in rats. Food Chem. Toxicol. 2008, 46, 3621–3625. [Google Scholar] [CrossRef]
- Garcia-Galiano, D.; Cara, A.L.; Tata, Z.; Allen, S.J.; Myers, M.G.; Schipani, E.; Elias, C.F. ERα Signaling in GHRH/Kiss1 Dual-Phenotype Neurons Plays Sex-Specific Roles in Growth and Puberty. J. Neurosci. 2020, 40, 9455–9466. [Google Scholar] [CrossRef]
- Bazhanova, E. Apoptosis of the hypothalamus neurosecretory cells in stress and ageing: The role of immune modulators. Rossiiskii fiziologicheskii zhurnal imeni IM Sechenova 2012, 98, 542–550. [Google Scholar] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).