3.1. Chemical Composition
A total of 37, 45, and 43 components were detected in VAO (0.1–25.0%), VNO (0.1–19.4%), and VTO (0.1–16.2%), respectively. A total of 22 components were found to be common in all three EOs, which were as follows: α-thujene, α-pinene, sabinene, β-pinene, myrcene, 1,8-cineole, γ-terpinene,
p-cymene, linalool,
trans-sabinenehydrate,
cis-
p-menth-2-en-1-ol, terpinen-4-ol, α-terpineol, dihydroedulan II, β-caryophyllene, α-humulene, β-iraldeine, β-caryophyllene oxide, α-muurolol, drimenol, and manool. However, they varied in their relative percentage. As summarized in
Table 1, 1,8-Cineole (25.0%), sabinene (13.3%), α-pinene (8.2%), and α-terpinyl acetate (5.5%) were the dominant compounds in
V. agnus-castus oil; sabinene (19.4%), viridiflorol (17.8%), β-caryophyllene (7.5%), and β-iraldiene (6.4%) were dominant in
V. negundo oil, while β-caryophyllene (16.2%), 5-(1-isopropenyl-4,5-dimethylbicyclo[4.3.0]nonan-5-yl)-3-methyl-2-pentenol acetate (11.7%), 13-epi-manoyl oxide (5.6%), and caryophyllene oxide (4.6%) were the abundant compounds in
V. trifolia oil. In terms of chemical class composition, VAO was dominated by oxygenated monoterpene (40.6%), followed by monoterpene hydrocarbons (31.2%) and others. The only diterpenoid present in VAO was manool (0.5%). On the other hand, VNO was mainly dominated by monoterpene hydrocarbons (29.4%), followed by oxygenated sesquiterpenes (24.8%) and oxygenated monoterpenoids (11.3%). The most abundant class found in VTO was sesquiterpene hydrocarbon (21.9%) followed by oxygenated sesquiterpene (15.8%) and oxygenated diterpenes (13.8%). For more details on chromatograms, and chemical composition mass spectra, please refer to
Supplemental Material S1.
Rezaei et al. [
42] evaluated the effects of different irrigation regimes on the essential oil composition of
V. agnus-castus under three shading levels, collected from Isfahan, Iran, and reported α-pinene (16.4–60.7%), β-terpinyl acetate (15.4–40.2%), caryophyllene (2.7–9.0%), and camphene (0.09–6%) as the main constituents. The compounds 1,8-cineole, sabinene, myrcene, α-thujene. α-terpineol, β-farnesene, spathulenol, β-caryophyllene oxide, humulane-1,6-dien-3-ol, and manool were not detected in any of the samples studied in this work, although these are present in noticeable amounts in the current study. However, in other studies, 1,8-cineole and sabinene were reported as the main EO constituents of
V. agnus castus leaves [
43,
44,
45]. These findings are generally consistent with those of the current investigation. The oxygenated sesquiterpenes identified in VAO, such as spathulenol, ledol, and
epi-α-cadinol, have previously been identified in the EO of
V. agnus castus leaves collected in Ogliastra, Sardinia, Italy [
46], however, other sesquiterpenoids such as β-eudesmol, drimenol, and flourensadiol were not detected previously in
V. agnus castus leaves essential oil. VAO also lacks compounds like limonene, viridiflorol, and globulol that are often present in most of the previous reports [
45,
46,
47,
48]. Thus, the study demonstrates different chemo-variants of
V. agnus castus both qualitatively and quantitatively.
Previous researchers have also studied the EOs of
Vitex negundo under investigation herein. For instance, the major compounds detected in VNO, sabinene (19.4%), viridiflorol (17.8%) and β-caryophyllene (7.5%), were also found to be present in the hydrodistilled
Vitex negundo leaves EO in variable amounts [
22,
49,
50]. 5-(1-Isopropenyl-4,5-dimethylbicyclo[4.3.0]nonan-5-yl)-3-methyl-2-pentenol acetate (5.2%), another major compound detected in VNO, was also found to be present in leaves essential oil of
V. negundo in notable amounts [
51]. The chemical composition of EO of
Vitex negundo extracted during the spring season from the same location (Pantnagar) revealed the presence of over 33 compounds, in which the major compounds detected were viridiflorol (23.8%), sabinene (11.2%), unidentified diterpene M
+ = 272 (11.0%), and caryophyllene (6.7%) [
50]. The composition was lacking the compounds, α-thujene, β-pinene, α-terpinene, β-phellandrene, γ-terpinene, linalool oxide,
p-cymene,
trans-sabinenehydrate, theaspirane A, β-iraldeine, β-caryophyllene oxide, ledol, humulane-1,6-dien-3-ol, and the diterpenes cubetene, phytol, manool, and sclereol, however, in the present study, these compounds are detected in noticeable amounts. Thus, the composition might vary as a result of the harvesting season. However, in another study of Indian origin, α-copaene (25.3%), β-elemene (19.2%), and camphene (21.1%) were reported as the predominant compounds in leaf essential oil of
V. negundo [
52]. Khokra et al. [
53] reported ethyl-9-hexadecenoate (28.5%), δ-guaiene (18.0%), and caryophyllene oxide (10.2%) as the major components in leaf essential oil of
V. negundo. On the other hand, the leaf essential oil of
V. negundo from Chinese origin revealed δ-guaiene (50.0%) and β-caryophyllene (38.0%) as the major constituents [
54]. Both qualitative and quantitative variations in essential oils of
V. negundo from different geographic regions might be due to the different geographical and climatic conditions.
Thomas et al. [
55], investigated the essential oil of
V. trifolia and obtained caryophyllene (38.36%) and 1,8-cineole (25.72%) as the predominant compounds. However, in the present study, the amount of 1,8-cineole is only 2.1% in VTO. β-caryophyllene is also identified as the major constituent of
V. trifolia oil by several other reports [
56,
57,
58], which is in agreement with the present study. Arpiwi et al. [
59] detected five components in
V. trifolia essential oil in which
cis-ocimene (44.57%), α-thujene (25.63%) and cyclopentene,3-isopropenyl-5,5-dimethyl (18.19%) were identified as the major constituents. However, in the present study such compounds were not detected, and the amount detected for α-thujene was also negligible (0.2%). The second major compound detected in VTO, 5-(1-isopropenyl-4,5-dimethylbicyclo[4.3.0]nonan-5-yl)-3-methyl-2-pentenolacetate (11.7%), has also been found in other
Vitex species such as
V. agnus castus and
V. negundo [
51,
60]. The noticeable diterpenes identified in VTO, 13-epi-manoyl oxide (5.6%), and 16-oxo-cleroda-3,13(14)-(e)-dien-15-oic acid (2.8%) are also being detected for the first time in
V. trifolia oil. These differences in the essential oil constituents might be due to internal and external factors and their interactions.
In addition, the compounds identified in the tested essential oils have potent biological applications. 1,8-cineole is used in cosmetic products and as a flavoring agent because of its pleasant aroma and taste. The compound has several other properties: insecticidal, antioxidant, and anti-inflammatory [
61]. Viridiflorol has prominent use as an anti-inflammatory, antioxidant, and anti-tuberculosis agent [
62]. Sabinene has antimicrobial, anti-inflammatory, and antioxidant properties described in literature [
63]. Further, the diterpene, 13-
epi-manoyl oxide, has cytotoxic antibacterial and antifungal activities [
64].
3.4. Antioxidant Activity
The antioxidant activity was determined by using different chemical-based methodologies.
Figure 3A–E depict the antioxidant activity of tested essential oil in terms of percent inhibition. Results revealed that all the antioxidant activities were in a concentration-dependent manner. The percent inhibition of free radicals (DPPH, H
2O
2, NO), reducing power, and metal chelation increased, with increasing concentration from 10 µL/mL to 50 µL/mL. Further, the percent inhibition by the tested essential oils and the standards for different antioxidant assays were plotted against concentrations, and the equation for the line was used to obtain the IC
50 (half-maximal inhibitory concentration) values.
Figure 4A–E represent the antioxidant activity of tested essential oils in terms of their IC
50 values. In the DPPH assay, the reduction of the stable radical DPPH (violet) to the yellow-colored DPPH-H is employed to measure the potential of an antioxidant molecule to act as a donor of hydrogen atoms or electrons.
Figure 4A shows that VNO reduced DPPH with an IC
50 value of 23.16 ± 0.5 µL/mL, which is close to the standard antioxidant taken for the assay, BHT (18.84 ± 0.6 µL/mL). VAO and VTO displayed moderate and weak antioxidant activity, with IC
50 25.39 ± 0.0 µL/mL and 32.49 ± 0.5 µL/mL, respectively. H
2O
2 can cross the biological membrane, and as a result it can damage the human body by forming reactive OH· radicals following Fenton reaction [
65]. In H
2O
2 radical scavenging assay, VAO (IC
50 = 24.49 ± 0.1 µL/mL) displayed good scavenging activity when compared to the standard, ascorbic acid (28.33 ± 0.5 µL/mL), followed by VNO (32.38 ± 0.5 µL/mL) and VTO (34.30 ± 0.5 µL/mL). The extent of nitrite scavenging by the samples was compared with ascorbic acid and showed IC
50 values as: ascorbic acid (24.49 ± 0.1 µL/mL) > VNO (27.58 ± 0.1 µL/mL) > VTO (32.27 ± 0.1 µL/mL) > VAO (32.95 ± 0.5 µL/mL). The reducing power of a compound is related to its ability to transfer electrons, which indicates its significant antioxidant potential. As shown in
Figure 4D, VNO displayed good reducing capability (RP
50 = 19.05 ± 0.6 μL/mL) that is very close and lower than that of the standard gallic acid (20.22 ± 0.4 μL/mL). The order of RP
50 values for different samples is in the order: VNO (19.05 ± 0.6 μL/mL) > gallic acid (20.22 ± 0.4 μL/mL) > VAO (20.97 ± 0.5 μL/mL) > VTO (22.74 ± 0.7 μL/mL). In auto-oxidation reactions, metal ion is a powerful catalyst as it can inhibit the generation of oxygen radicals. The IC
50 values of different samples and standards towards their antioxidant potentiality in terms of chelating ability were observed as: Na
2-EDTA (IC
50 = 26.23 ± 0.26 µL/mL) > VTO (IC
50 = 29.77 ± 0.2 µL/mL) > VNO (IC
50 = 31.18 ± 0.2 µL/mL) > VAO (IC
50 = 36.60 ± 0.1 µL/mL).
Such high antioxidant activity of VNO for the DPPH and NO radical scavenging is likely due to high amount of sabinene as well as other constituents of VNO such as β-caryophyllene, terpinen-4-ol, 1,8-cineole, which already possess antioxidant potential via different parameters [
66,
67,
68]. Additionally, Kazemi [
69] showed that sabinene exhibited potent NO-scavenging effect and inhibited the expression of inducible NO synthase. Similar results were observed in previous studies in antioxidant activity of
V. negundo essential oil in which the major component was sabinene [
49]. In H
2O
2 radical scavenging assay, VAO showed good scavenging activity, which may be due to the presence of 1,8-cineole, sabinene, and β-caryophyllene as the major constituents [
69,
70]. In earlier reports, essential oil and extracts of aerial parts of
V. agnus castus have been tested for antioxidant activity as having a high amount of 1,8-cineole and β-caryophyllene in their composition, and the samples showed good antioxidant activity [
64,
71,
72]. Since essential oils are complexed mixtures of number of compounds, their whole biological activity is hard to explain. Therefore, research on the antioxidant activity of essential oils typically indicates that other minor chemical constituents that may interact synergistically or antagonistically to produce an additive and effective system against free radicals may also be responsible for the antioxidant activity [
68,
73].
3.5. Herbicidal (Phytotoxic) Activity
The tested samples demonstrated notable phytotoxic activity against seed germination and seedling growth of the wild radish (
R. raphanistrum) in a concentration-dependent manner. At the highest concentration (100 µL/mL), VAO showed inhibition of seed germination, root growth, and shoot growth of
R. raphanistrum by 66.67%, 96.66%, and 89.09%, respectively, VNO showed inhibition values of 90.0%, 89.39%, and 97.57%, respectively, while VTO showed inhibition values of 100%, 99.39%, and 92.12%, respectively (
Table 2,
Table 3 and
Table 4). Based on the IC
50 values, VAO showed IC
50 values of 82.89, 19.468, and 37.95 µL/mL regrading seed germination, root growth, and shoot growth, respectively. For VNO, the IC
50 values were 50.13, 47.06, and 16.75 µL/mL, respectively. For VTO, the IC
50 values were 29.5, 9.33, and 27.13 µL/mL, respectively (
Table 2,
Table 3 and
Table 4).
The phytotoxic potential of EOs from various
Vitex species such as
V. agnus castus,
V. negundo V. simplicifolia has also been reported previously in other plants and weeds [
15]. However, there is no study reported on phytotoxic potential of
V. trifolia. Based on the present study, it was evident that VTO was more effective against
R. raphanistrum than VNO and VAO. The suppressing effect of VTO on
R. raphanistrum could be due to high amounts of β-caryophyllene (16.2%) and the synergetic effect of β-caryophyllene with other major and minor compounds present in the oil. In previous reports, β-caryophyllene was found to be responsible for the inhibition of germination and seedling growth of several plant species such as
Brassica campestris,
Raphanus sativus,
Vigna radiata, and
Solanum lycopersicum [
22]. VNO also showed good inhibition values for seed germination and shoot growth, while VAO showed better inhibition value for root growth. The inhibition effect of samples could be due to the presence of phytotoxic compounds such as β-caryophyllene, 1,8-cineole, and sabinene, which are the main components in essential oil possessing phytotoxic activity [
22,
74]. In addition, 1,8-cineole was reported to interfere with the normal growth
Nicotiana tabacum by blocking the DNA synthesis in their cell nuclei and organelles in root apical meristem cells [
75]. Studies have also demonstrated that the terpenoids in EOs have a phytotoxic effects on plants, resulting in morphological and physiological alterations in the cells that impair plant growth [
76].
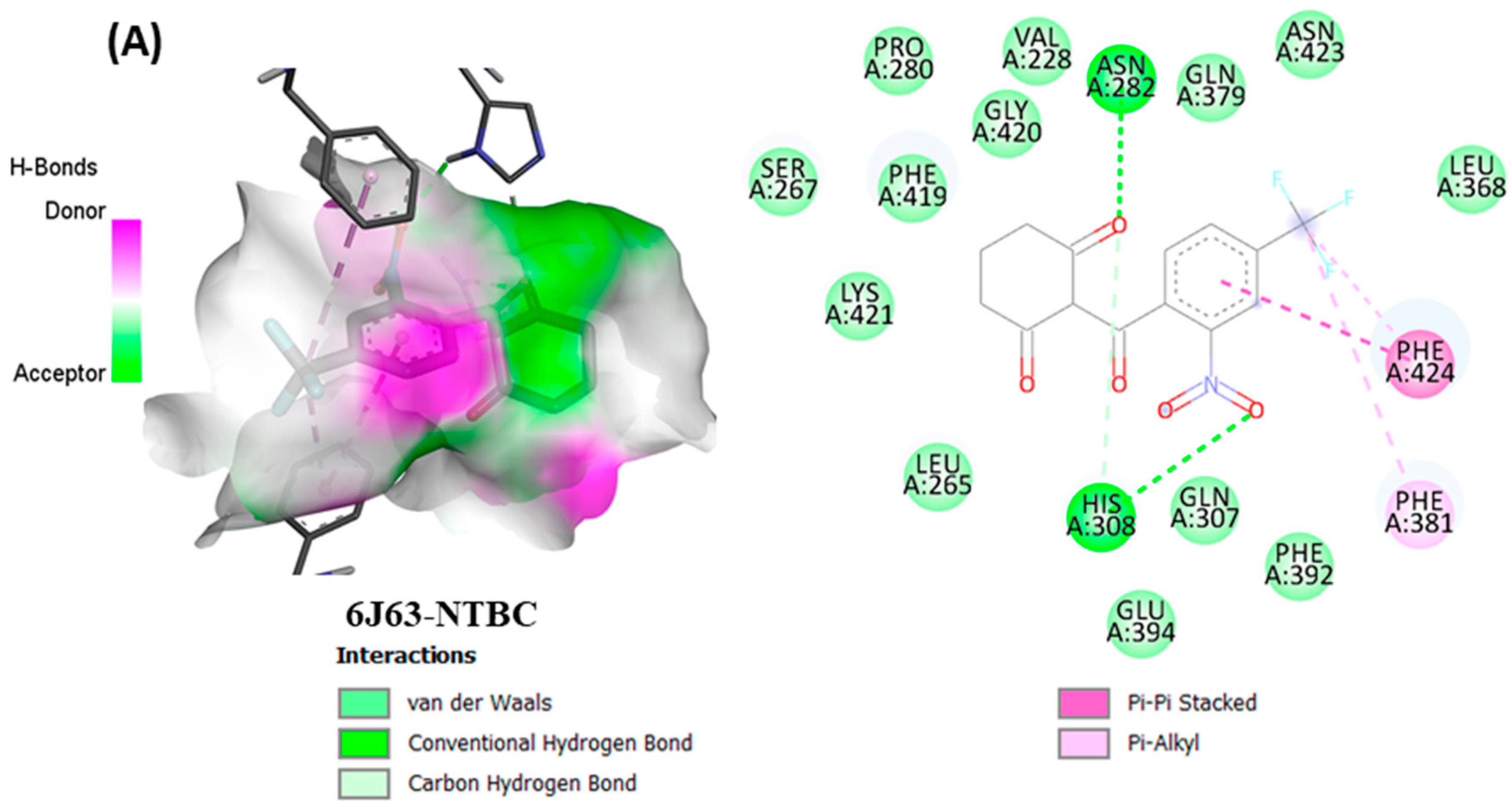
3.7. Molecular Docking
From in vitro studies, it was found that essential oils have potent antioxidant and phytotoxic activity. We also examined whether the major phytoconstituents from VAO, VNO, and VTO physically bind with antioxidant protein (human peroxiredoxin 5, PDB: 1HD2) and 4-hydroxyphenylpyruvate dioxygenase (HPPD, PDB: 6J63) receptors. The tested essential oils displayed good inhibition of the free radicals, for which the enzyme human peroxiredoxin 5 was selected, as it has broader activity against the reactive oxygen species (ROS) and is mostly involved in the stress protection mechanism [
80,
81]. The reason for selecting HPPD is that it is known to be the target protein for compounds with post-emergence herbicidal activity. In our results, the tested essential oils were found to have good post-emergence herbicidal activity against the receptor species, for which HPPD was selected as a target enzyme [
18,
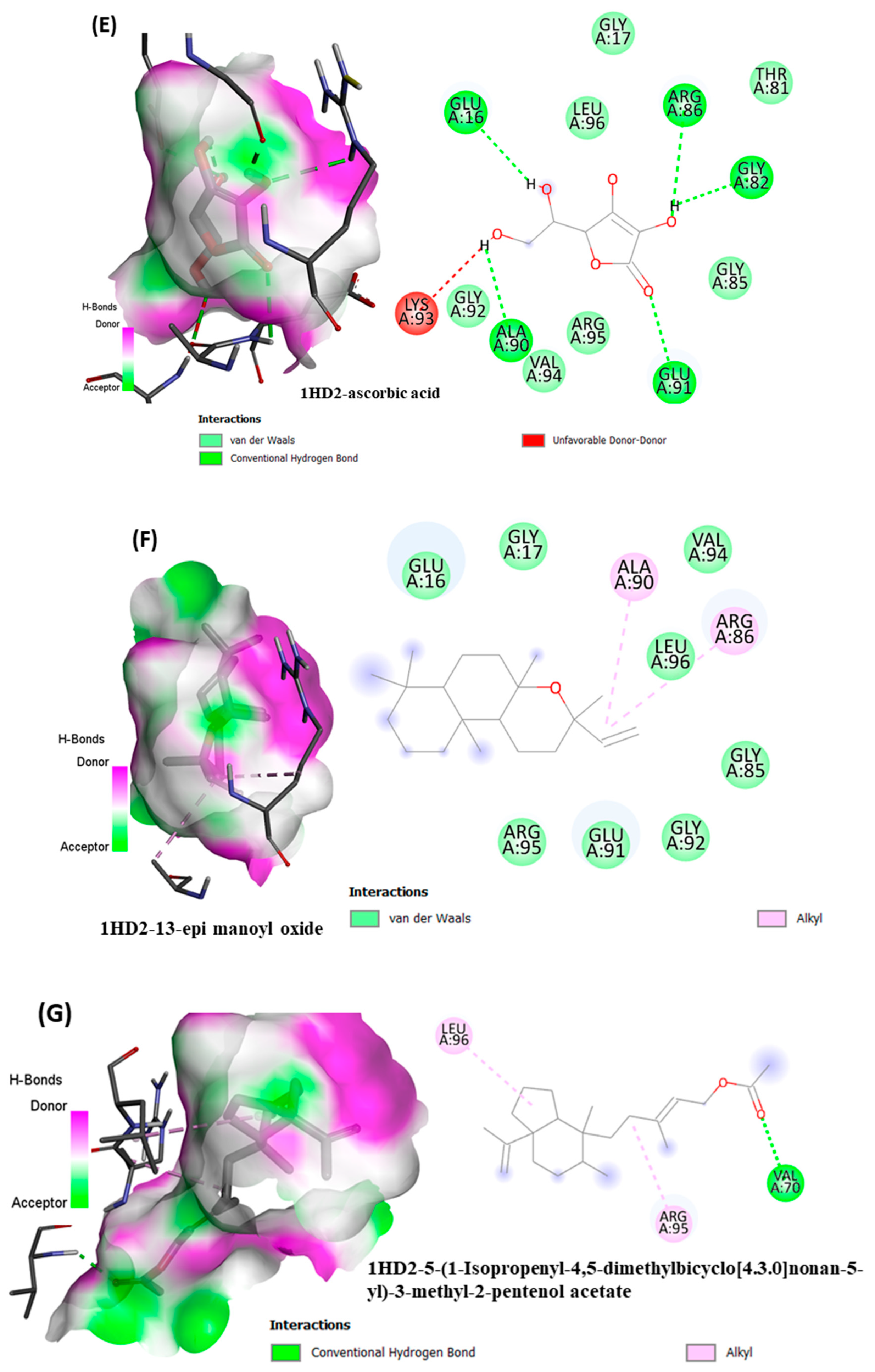
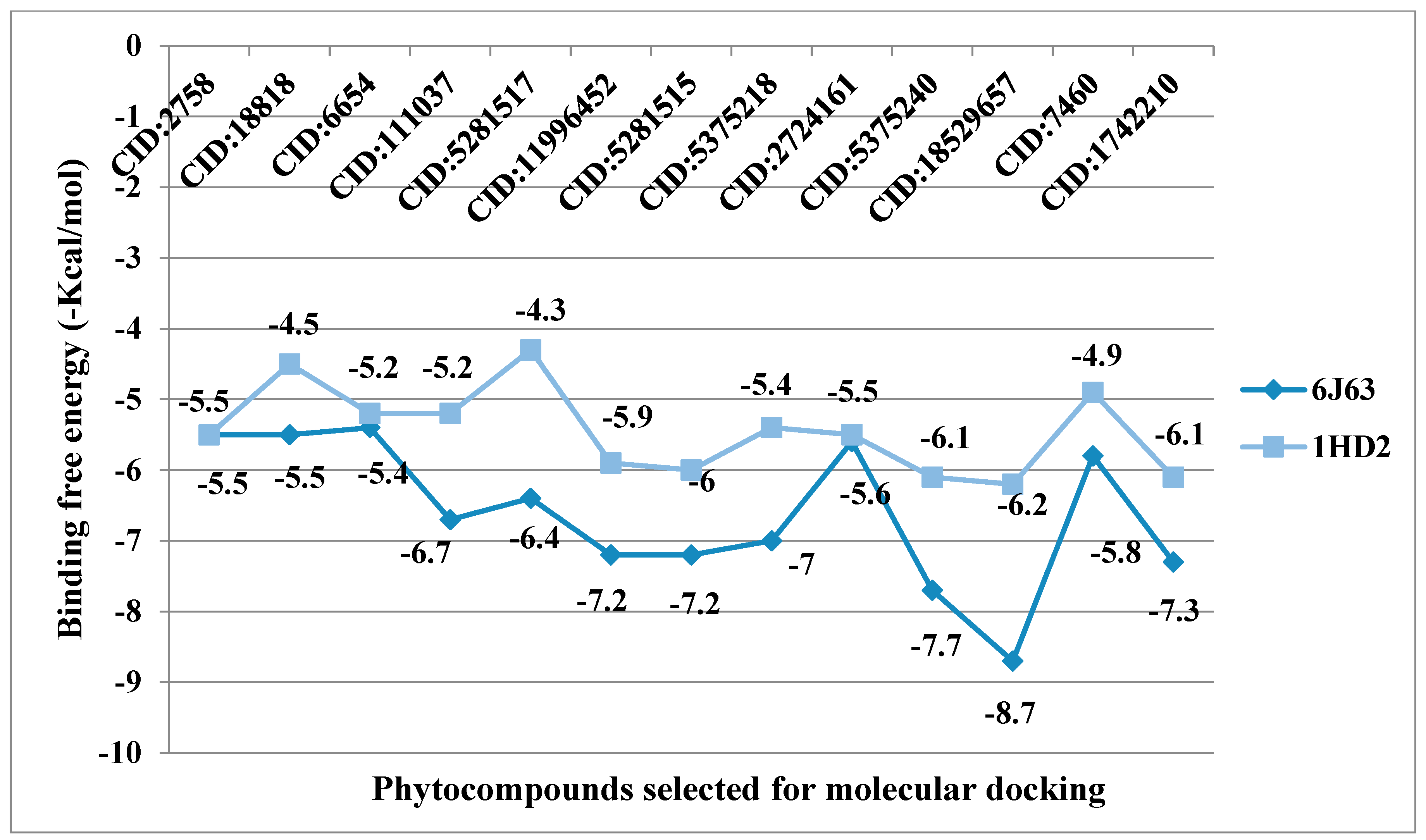
38]. Among all selected phytocompounds, 13-
epi-manoyl oxide demonstrated the best binding affinity with human peroxiredoxin 5 (−6.2 kcal mol) and HPPD (−8.7 kcal/mol). By introspecting the multiple dock poses, the best docked pose was selected as having the lowest binding energy. The best docked pose of 13-
epi-manoyl oxide exhibited 2 pi-alkyl interaction, 1 pi-sigma interaction, and other Van der Waal interactions with 6J63 containing amino acid residues such as Phe A:424, Phe A:419, and Phe A:381, as represented in
Figure 6B. Similarly, the best docked pose of 13-
epi-manoyl oxide exhibited alkyl interaction with 1HD2 containing amino acid Ala A:90, Arg A:86, and exhibited Van der Waal interaction. For comparison purposes, a docking study of Nitisinone (CID:115355) was also performed with HPPD. Nitisinone (2-[2-nitro-4-(trifluoromethyl)benzoyl]cyclohexane-1,3-dione, (NTBC)) is a known inhibitor of HPPD. The docking study of ascorbic acid (CID:54670067), a known antioxidant, was performed with 1HD2. The binding energy for NTBC complexed with 6J63 was −8.9 kcal/mol, which is very close to that of 13-
epi-manoyl oxide (−8.7 kcal/mol). On the other hand, binding energy of ascorbic acid complexed with 1HD2 came out to be −5.7 kcal/mol, which was higher than most of the compounds such as 13-
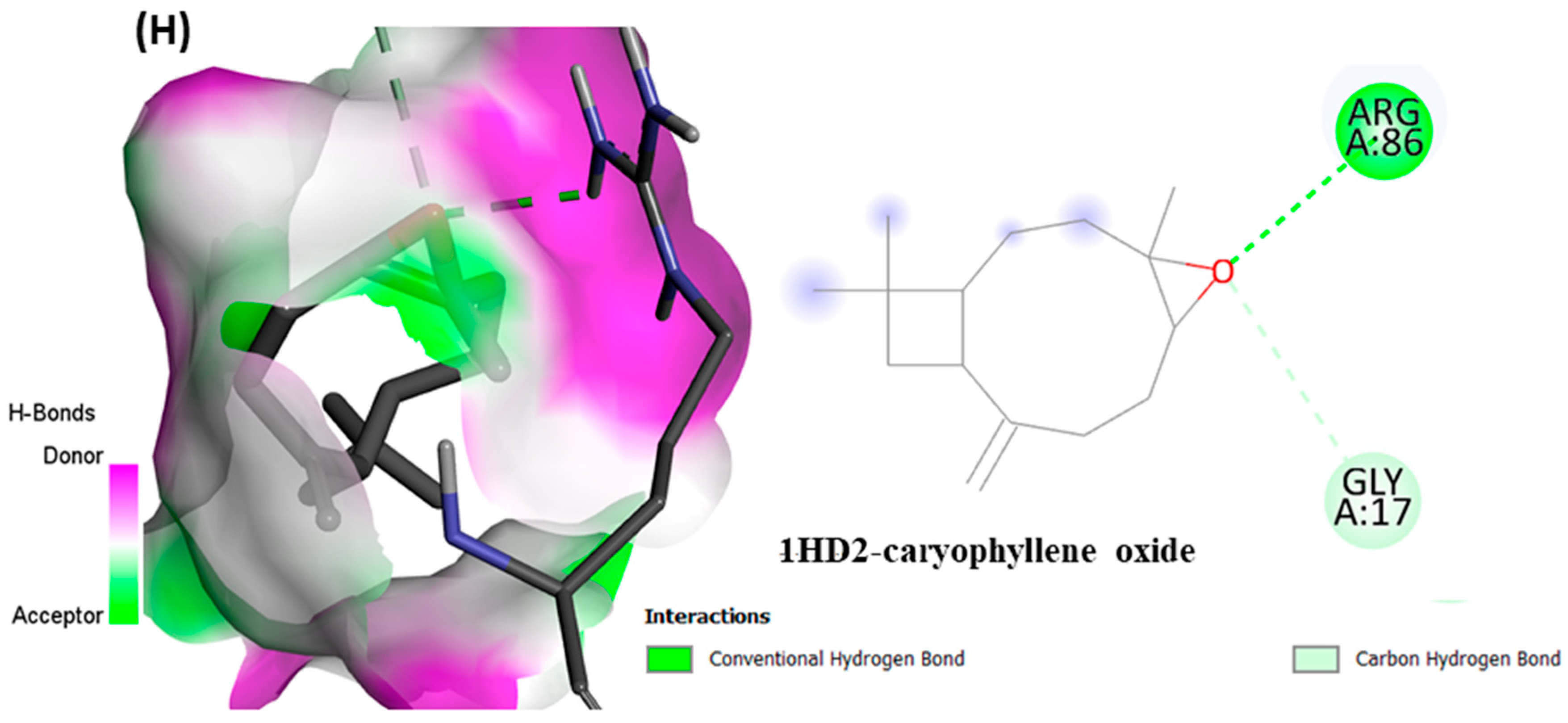
epi-manoyl oxide (−6.2 kcal/mol), caryophyllene oxide (−6.1 kcal/mol), 5-(1-isopropenyl-4,5-dimethylbicyclo [4.3.0]nonan-5-yl)-3-methyl-2-pentenol acetate (−6.1 kcal/mol), β-caryophyllene (−6.0 kcal/mol), and viridiflorol (−5.9 kcal/mol), as shown in
Figure 7. The lower values of binding free energy demonstrate more significant interaction between the receptor and the ligand. Our results were consistent with previous in silico studies reported by Alminderej et al. [
73], where a phenylpropanoid-rich
Piper cubeba EO gave similar results in terms of a proposed in vitro antioxidant activity by targeting human periredoxin 5. In this study, the compounds viridiflorol and caryophyllene oxide showed significant interaction with 1HD2 receptor as in the present study. In a recent study, focusing on the phytotoxic potential of
Calycolpus goetheanus EO, it was found that the major components of the specimen, 1,8-cineole and β-caryophyllene interacted favorably with the HPPD protein [
18]. These results are in general agreement with those obtained in the present study.
The listed binding energies of the volatiles docked with human peroxiredoxin 5 and HPPD (
Figure 7) were found to be in the range −6.2 to −4.3 kcal/mol and −8.7 to −5.4 kcal/mol, respectively. Based on the study, it was observed that the major constituents interacted favorably with the receptors—most of which are the Van der Waal interactions. The analysis of ligand recognition reveals that the compounds can be good antioxidant and phytotoxic agents.
Figure 6A–H shows the interaction of selected volatiles with the receptors (6J63 and 1HD2) having the least binding energies (higher docking scores), along with their 2D interaction with amino acid residues.
3.8. ADMET Analysis
The forecasting of ADME (absorption, distribution, metabolism, and excretion) properties of the selected compounds, including their pharmacokinetic and drug-like properties, have been estimated using SwissADME online server (
http://www.swissadme.ch/, accessed on 12 August 2022). The collective laws of Lipinski’s [
82], Egan’s [
83], and Veber’s [
84], which determine the properties of a drug, were followed. According to the rule that the compound should not violate more than 1 Lipinski rule, molecular weight (MW) < 500, topological surface area (TPSA) < 140, number of H-bond acceptors (nOHA) ≤ 5, number of H-bond donors (nOHD) ≤ 5, water partition coefficient (WLOGP) ≤ 5.88, number of rotatable bonds (nRB) ≤ 10. Based on the current findings, 12 out of 13 compounds selected followed the Lipinski’s, Egan’s, and Verber’s rule, indicating the good drug-like properties of the compounds. The bioavailability score was found to be 0.55 for all the compounds selected, indicating higher bioactivity of the molecule. The compounds share TPSA values less than 30 Å
2, indicating good brain penetration and good lipophilicity behavior, with the consensus Log Po/w coming in the range 2.60–5.14 (
Table 5). There was no P-glycoprotein (P-gp) substrate found, suggesting the good intestinal absorption of compounds. Except sabinene, α-pinene, β-farnesene, β-caryophyllene, 13-
epi-manoyl acetate, and α-phellandrene, all compounds showed high gastrointestinal absorption. The compounds that were predicted to not cross the blood–brain barrier (BBB) were β-farnesene, β-caryophyllene, 5-(1-isopropenyl-4,5-dimethylbicyclo[4.3.0]nonan-5-yl)-3-methyl-2-pentenol acetate, and 13-
epi-manoyl oxide.
Some of the compounds interacted mainly with two isoenzymes of the cytochrome (CYP) family, namely CYP2C19 and CYP2C9, suggesting their efficiency while having minimal toxicity. Drug-like properties and GI absorption of selected compounds from VAO, VNO, and VTO were also represented by the boiled-egg prediction (
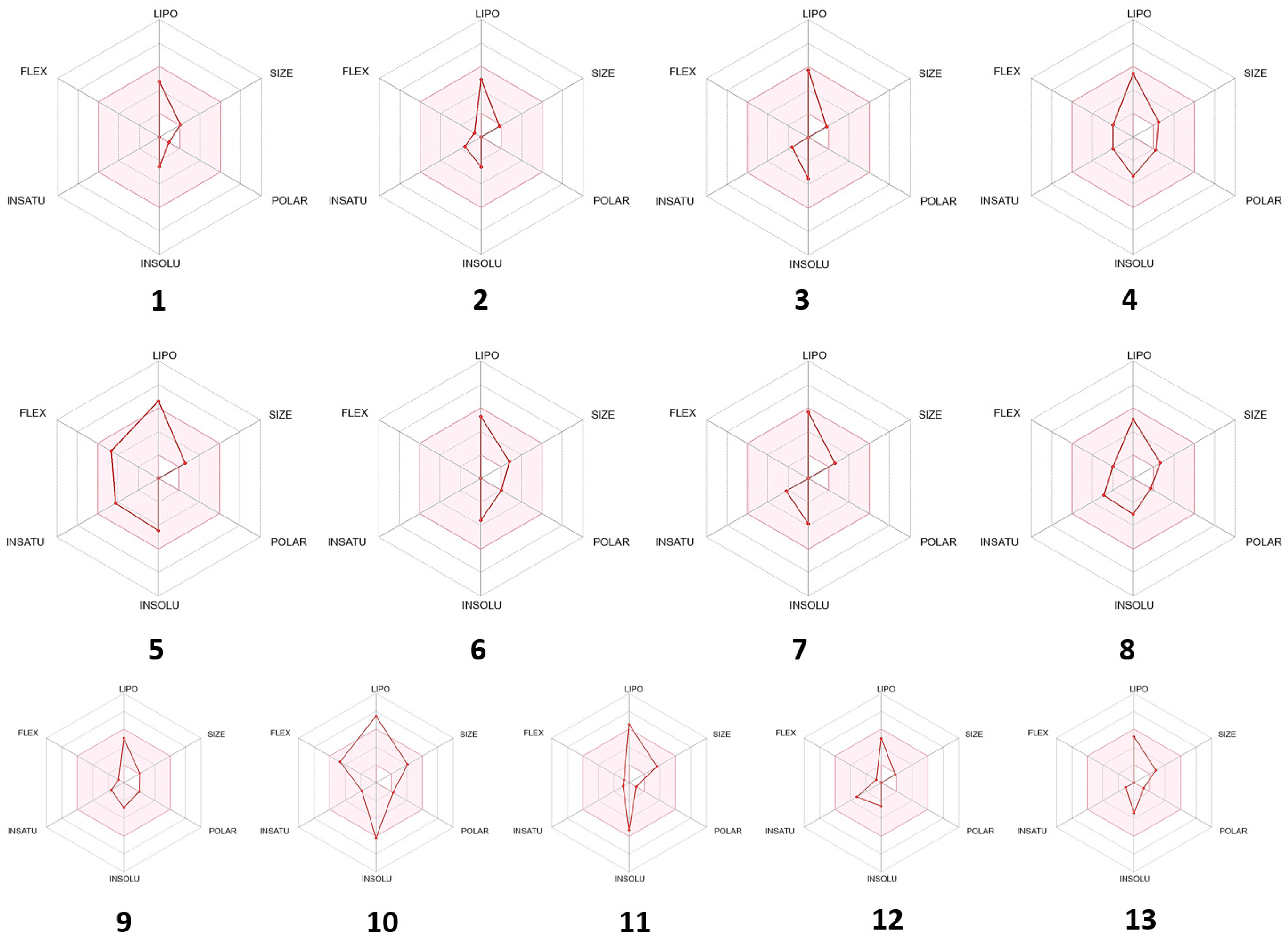
Figure 8) and bioavailability radar graph (
Figure 9). The compounds present in the yellow zone in the boiled-egg graph can permeate through the blood–brain barrier (BBB), and the pink area of the bioavailability radar graphs shows the drug-likeness of the compounds.
The toxicity parameters of selected phytocompounds were predicted using web server ProTox II (
Table 6). All the selected compounds were predicted not to be hepatotoxic, carcinogenic, cytotoxic, immunotoxic, and mutagenic, except α-terpinyl acetate (hepatotoxic), 5-(1-isopropenyl-4,5-dimethylbic clo[4.3.0]nonan-5-yl)-3-methyl-2-pentenol acetate (carcinogenic), β-iraldiene, and caryophyllene oxide (immunotoxic). The LD
50 values were also calculated to ensure the safety of the selected compounds as shown in
Table 6. The compounds with LD
50 > 2000 mg/kg, suggesting their safety for biological administration and as potential drugs.