Cosmeceutical Potential of Extracts Derived from Fishery Industry Residues: Sardine Wastes and Codfish Frames
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Samples
2.3. Oxygen Radical Absorbance Capacity (ORAC) Assay
2.4. Enzymatic Assays
2.4.1. Elastase Inhibition Assay
2.4.2. Tyrosinase Inhibition Assay
2.5. Antimicrobial Susceptibility Testing
2.6. Cell-Based Assays
2.6.1. Cell Culture
2.6.2. In Vitro Cytotoxicity
2.6.3. Cellular Antioxidant Activity
2.6.4. Evaluation of IL-6 and IL-8 Secretion
2.7. Statistical Analysis
3. Results and Discussion
3.1. Antioxidant, Anti-Ageing and Anti-Hyperpigmentation Activities
3.2. Antibacterial Activity
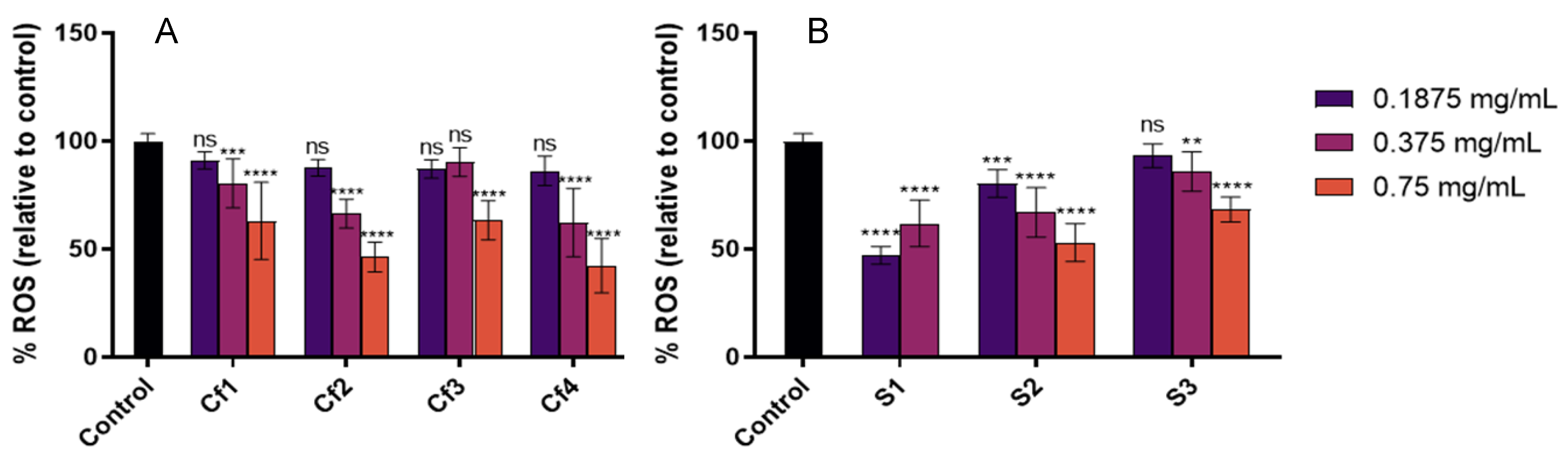
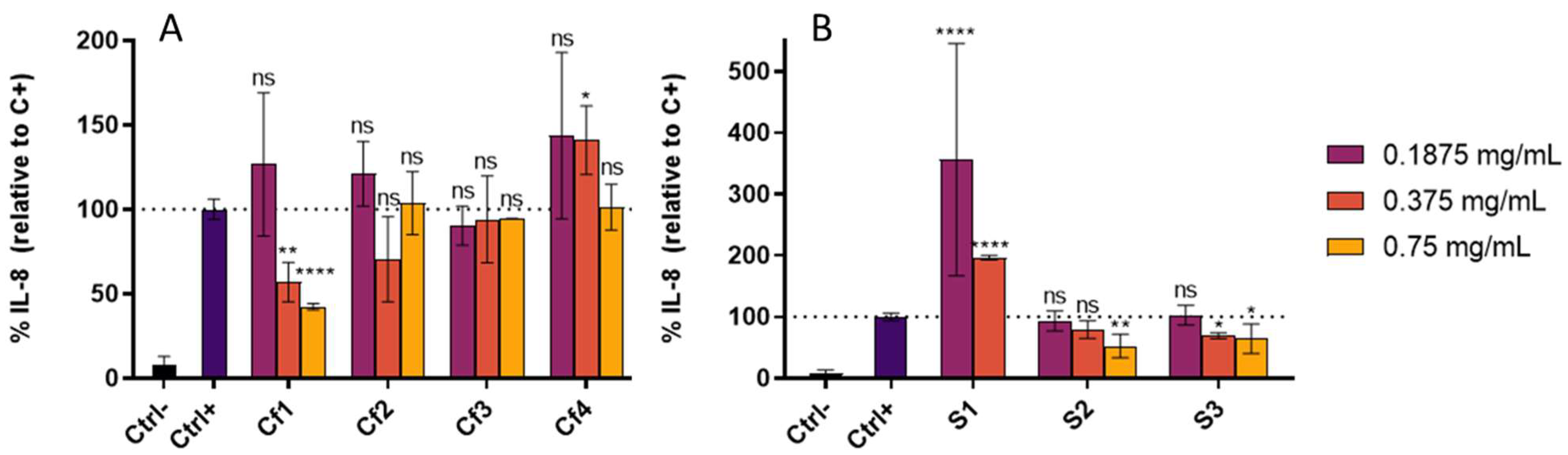
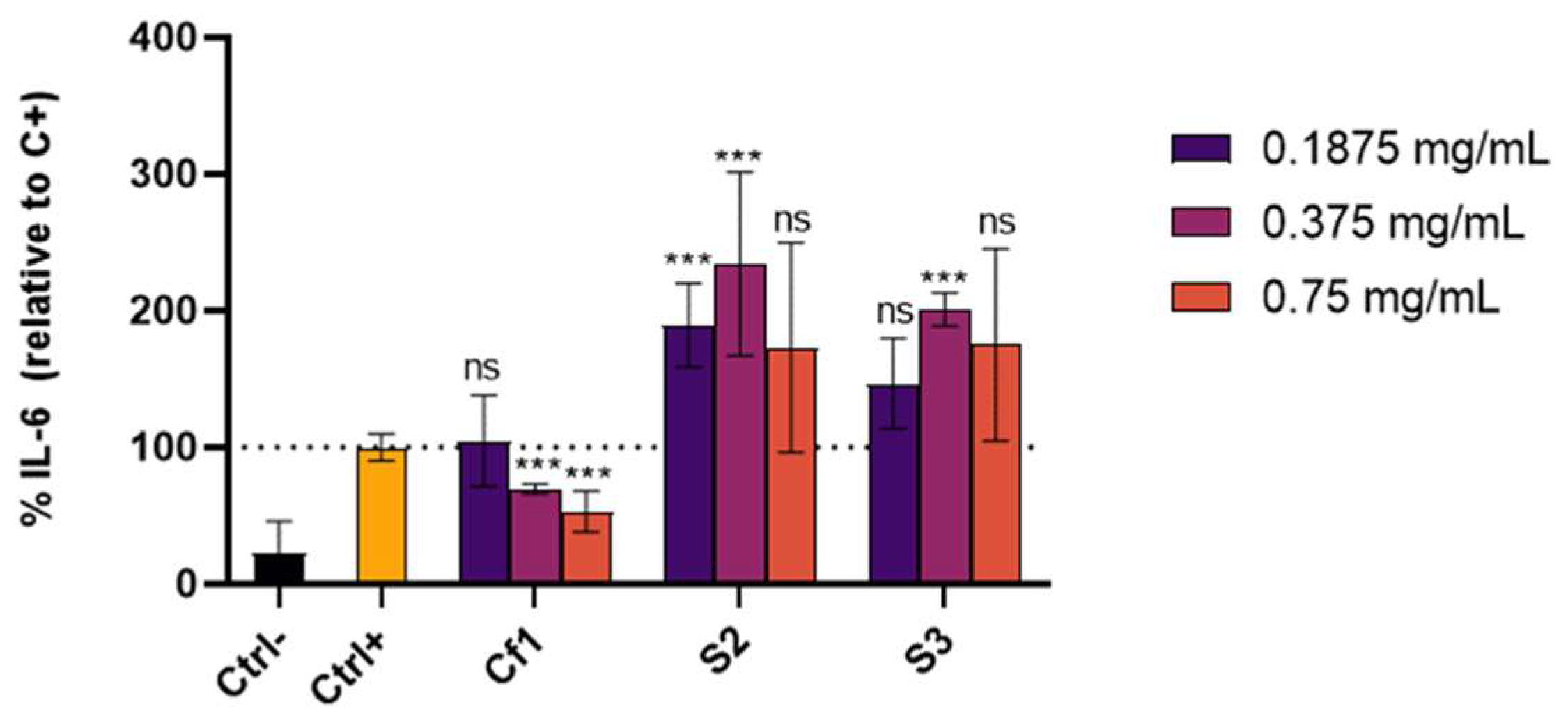
3.3. Cellular Antioxidant and Anti-Inflammatory Effect
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guillerme, J.B.; Couteau, C.; Coiffard, L. Applications for marine resources in cosmetics. Cosmetics 2017, 4, 35. [Google Scholar] [CrossRef]
- e Silva, S.A.M.; Leonardi, G.R.; Michniak-Kohn, B. An overview about oxidation in clinical practice of skin aging. An. Bras. Dermatol. 2017, 92, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Hoang, H.T.; Moon, J.Y.; Lee, Y.C. Natural antioxidants from plant extracts in skincare cosmetics: Recent applications, challenges and perspectives. Cosmetics 2021, 8, 106. [Google Scholar] [CrossRef]
- Tobin, D.J. Introduction to skin aging. J. Tissue Viability 2017, 26, 37–46. [Google Scholar] [CrossRef]
- Hashizume, H. Skin aging and dry skin. J. Dermatol. 2004, 31, 603–609. [Google Scholar] [CrossRef]
- Matos, M.S.; Romero-Díez, R.; Álvarez, A.; Bronze, M.R.; Rodríguez-Rojo, S.; Mato, R.B.; Cocero, M.J.; Matias, A.A. Polyphenol-rich extracts obtained from winemaking waste streams as natural ingredients with cosmeceutical potential. Antioxidants 2019, 8, 355. [Google Scholar] [CrossRef]
- Nordberg, J.; Arnér, E.S.J. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic. Biol. Med. 2001, 31, 1287–1312. [Google Scholar] [CrossRef]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative stress in aging human skin. Biomolecules 2015, 5, 545. [Google Scholar] [CrossRef]
- Xu, H.; Zheng, Y.-W.; Liu, Q.; Liu, L.-P.; Luo, F.-L.; Zhou, H.C.; Isoda, H.; Ohkohchi, N.; Li, Y.-M. Reactive oxygen species in skin repair, regeneration, aging, and inflammation. React. Oxyg. Species ROS Living Cells 2017, 8, 69–88. [Google Scholar] [CrossRef]
- The State of World Fisheries and Aquaculture 2020. Available online: https://www.fao.org/state-of-fisheries-aquaculture (accessed on 11 January 2022).
- Melgosa, R.; Marques, M.; Paiva, A.; Bernardo, A.; Fernández, N.; Sá-Nogueira, I.; Simões, P. Subcritical water extraction and hydrolysis of cod (Gadus morhua) frames to produce bioactive protein extracts. Foods 2021, 10, 1222. [Google Scholar] [CrossRef]
- Ferraro, V.; Carvalho, A.P.; Piccirillo, C.; Santos, M.M.; Paula, P.M.; Pintado, M.E. Extraction of high added value biological compounds from sardine, sardine-type fish and mackerel canning residues—A review. Mater. Sci. Eng. C 2013, 33, 3111–3120. [Google Scholar] [CrossRef] [PubMed]
- Thuanthong, M.; De Gobba, C.; Sirinupong, N.; Youravong, W.; Otte, J. Purification and characterization of angiotensin-converting enzyme-inhibitory peptides from nile tilapia (Oreochromis niloticus) skin gelatine produced by an enzymatic membrane reactor. J. Funct. Foods 2017, 36, 243–254. [Google Scholar] [CrossRef]
- Chi, C.F.; Wang, B.; Hu, F.Y.; Wang, Y.M.; Zhang, B.; Deng, S.G.; Wu, C.W. Purification and identification of three novel antioxidant peptides from protein hydrolysate of bluefin leatherjacket (Navodon septentrionalis) skin. Food Res. Int. 2015, 73, 124–129. [Google Scholar] [CrossRef]
- Seo, J.K.; Lee, M.J.; Go, H.J.; Kim, Y.J.; Park, N.G. Antimicrobial function of the GAPDH-related antimicrobial peptide in the skin of skipjack tuna, katsuwonus pelamis. Fish Shellfish. Immunol. 2014, 36, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.Y.; Hsieh, C.H.; Hung, C.C.; Jao, C.L.; Chen, M.C.; Hsu, K.C. Fish skin gelatin hydrolysates as dipeptidyl peptidase IV inhibitors and glucagon-like peptide-1 stimulators improve glycaemic control in diabetic rats: A comparison between warm- and cold-water fish. J. Funct. Foods 2015, 19, 330–340. [Google Scholar] [CrossRef]
- Cai, L.; Wu, X.; Zhang, Y.; Li, X.; Ma, S.; Li, J. Purification and characterization of three antioxidant peptides from protein hydrolysate of grass carp (Ctenopharyngodon idella) skin. J. Funct. Foods 2015, 16, 234–242. [Google Scholar] [CrossRef]
- Lu, J.; Hou, H.; Fan, Y.; Yang, T.; Li, B. Identification of MMP-1 inhibitory peptides from cod skin gelatin hydrolysates and the inhibition mechanism by MAPK signaling pathway. J. Funct. Foods 2017, 33, 251–260. [Google Scholar] [CrossRef]
- Abdallah, M.M.; Leonardo, I.C.; Krstić, L.; Enríquez-De-salamanca, A.; Diebold, Y.; González-García, M.J.; Gaspar, F.B.; Matias, A.A.; Bronze, M.R.; Fernández, N. Potential ophthalmological application of extracts obtained from tuna vitreous humor using lactic acid-based deep eutectic systems. Foods 2022, 11, 342. [Google Scholar] [CrossRef]
- Rodrigues, L.A.; Pereira, C.V.; Carvalho Partidario, A.M.; Gouveia, L.F.; Simoes, P.; Paiva, A.; Matias, A.A. Supercritical CO2 extraction of bioactive lipids from canned sardine waste streams. J. CO2 Util. 2021, 43, 101359. [Google Scholar] [CrossRef]
- Šližyte, R.; Mozuraityte, R.; Martínez-Alvarez, O.; Falch, E.; Fouchereau-Peron, M.; Rustad, T. Functional, bioactive and antioxidative properties of hydrolysates obtained from cod (Gadus morhua) backbones. Process Biochem. 2009, 44, 668–677. [Google Scholar] [CrossRef]
- Melgosa, R.; Trigueros, E.; Sanz, M.T.; Cardeira, M.; Rodrigues, L.; Fernández, N.; Matias, A.A.; Bronze, M.R.; Marques, M.; Paiva, A.; et al. Supercritical CO2 and subcritical water technologies for the production of bioactive extracts from sardine (Sardina pilchardus) waste. J. Supercrit. Fluids 2020, 164, 104943. [Google Scholar] [CrossRef]
- Venkatesan, J.; Anil, S.; Kim, S.K.; Shim, M.S. Marine fish proteins and peptides for cosmeceuticals: A review. Mar. Drugs 2017, 15, 143. [Google Scholar] [CrossRef] [PubMed]
- Pedras, B.; Salema-Oom, M.; Sá-Nogueira, I.; Simões, P.; Paiva, A.; Barreiros, S. Valorization of white wine grape pomace through application of subcritical water: Analysis of extraction, hydrolysis, and biological activity of the extracts obtained. J. Supercrit. Fluids 2017, 128, 138–144. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Hampsch-Woodill, M.; Flanagan, J.A.; Prior, R.L. High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J. Agric. Food Chem. 2002, 50, 4437–4444. [Google Scholar] [CrossRef]
- Oliveira-Alves, S.C.; Andrade, F.; Prazeres, I.; Silva, A.B.; Capelo, J.; Duarte, B.; Caçador, I.; Coelho, J.; Serra, A.T.; Bronze, M.R. Impact of Drying Processes on the Nutritional Composition, Volatile Profile, Phytochemical Content and Bioactivity of Salicornia ramosissima J. Woods. Antioxidants 2021, 10, 1312. [Google Scholar] [CrossRef]
- Wittenauer, J.; MäcKle, S.; Sußmann, D.; Schweiggert-Weisz, U.; Carle, R. Inhibitory effects of polyphenols from grape pomace extract on collagenase and elastase activity. Fitoterapia 2015, 101, 179–187. [Google Scholar] [CrossRef]
- Chan, E.W.C.; Lim, Y.Y.; Wong, L.F.; Lianto, F.S.; Wong, S.K.; Lim, K.K.; Joe, C.E.; Lim, T.Y. Antioxidant and tyrosinase inhibition properties of leaves and rhizomes of ginger species. Food Chem. 2008, 109, 477–483. [Google Scholar] [CrossRef]
- Rodrigues, L.A.; Pereira, C.V.; Leonardo, I.C.; Fernández, N.; Gaspar, F.B.; Silva, J.M.; Reis, R.L.; Duarte, A.R.C.; Paiva, A.; Matias, A.A. Terpene-based natural deep eutectic systems as efficient solvents to recover astaxanthin from brown crab shell residues. ACS Sustain. Chem. Eng. 2020, 8, 2246–2259. [Google Scholar] [CrossRef]
- Serra, A.T.; Matias, A.A.; Frade, R.F.M.; Duarte, R.O.; Feliciano, R.P.; Bronze, M.R.; Figueira, M.E.; de Carvalho, A.; Duarte, C.M.M. Characterization of traditional and exotic apple varieties from portugal. Part 2—Antioxidant and antiproliferative activities. J. Funct. Foods 2010, 2, 46–53. [Google Scholar] [CrossRef]
- Di Caprio, R.; Lembo, S.; Di Costanzo, L.; Balato, A.; Monfrecola, G. Anti-inflammatory properties of low and high doxycycline doses: An in vitro study. Mediat. Inflamm. 2015, 2015, 329418. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, P.K.; Maity, N.; Nema, N.K.; Sarkar, B.K. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011, 19, 64–73. [Google Scholar] [CrossRef]
- Chalamaiah, M.; Dinesh Kumar, B.; Hemalatha, R.; Jyothirmayi, T. Fish protein hydrolysates: Proximate composition, amino acid composition, antioxidant activities and applications: A review. Food Chem. 2012, 135, 3020–3038. [Google Scholar] [CrossRef] [PubMed]
- Asaduzzaman, A.K.M.; Chun, B.S. Hydrolyzates produced from mackerel Scomber japonicus skin by the pressurized hydrothermal process contain amino acids with antioxidant activities and functionalities. Fish. Sci. 2014, 80, 369–380. [Google Scholar] [CrossRef]
- Vázquez, J.A.; Rodríguez-Amado, I.; Sotelo, C.G.; Sanz, N.; Pérez-Martín, R.I.; Valcárcel, J. Production, characterization, and bioactivity of fish protein hydrolysates from aquaculture turbot (Scophthalmus maximus) wastes. Biomolecules 2020, 10, 310. [Google Scholar] [CrossRef] [PubMed]
- Upata, M.; Siriwoharn, T.; Makkhun, S.; Yarnpakdee, S.; Regenstein, J.M.; Wangtueai, S. Tyrosinase inhibitory and antioxidant activity of enzymatic protein hydrolysate from jellyfish (Lobonema smithii). Foods 2022, 11, 615. [Google Scholar] [CrossRef]
- Fan, Y.F.; Zhu, S.X.; Hou, F.B.; Zhao, D.F.; Pan, Q.S.; Xiang, Y.W.; Qian, X.K.; Ge, G.B.; Wang, P. Spectrophotometric assays for sensing tyrosinase activity and their applications. Biosensors 2021, 11, 290. [Google Scholar] [CrossRef] [PubMed]
- Promden, W.; Viriyabancha, W.; Monthakantirat, O.; Umehara, K.; Noguchi, H.; De-Eknamkul, W. Correlation between the potency of flavonoids on mushroom tyrosinase inhibitory activity and melanin synthesis in melanocytes. Molecules 2018, 23, 1403. [Google Scholar] [CrossRef]
- Zolghadri, S.; Bahrami, A.; Hassan Khan, M.T.; Munoz-Munoz, J.; Garcia-Molina, F.; Garcia-Canovas, F.; Saboury, A.A. A comprehensive review on tyrosinase inhibitors. J. Enzym. Inhib. Med. Chem. 2019, 34, 279–309. [Google Scholar] [CrossRef]
- Strzępek-Gomółka, M.; Gaweł-Bęben, K.; Angelis, A.; Antosiewicz, B.; Sakipova, Z.; Kozhanova, K.; Głowniak, K.; Kukula-Koch, W. Identification of mushroom and murine tyrosinase inhibitors from Achillea biebersteinii afan. Extract. Molecules 2021, 26, 964. [Google Scholar] [CrossRef]
- Ji, S.; Qi, X.; Ma, S.; Liu, X.; Min, Y. Effects of dietary threonine levels on intestinal immunity and antioxidant capacity based on cecal metabolites and transcription sequencing of broiler. Animals 2019, 9, 739. [Google Scholar] [CrossRef] [Green Version]
- Cojocaru, E.; Filip, N.; Ungureanu, C.; Filip, C.; Danciu, M. Effects of valine and leucine on some antioxidant enzymes in hypercholesterolemic rats. Health 2014, 6, 2313–2321. [Google Scholar] [CrossRef]
- Nayak, B.N.; Buttar, H.S. Evaluation of the antioxidant properties of tryptophan and its metabolites in in vitro assay. J. Complement. Integr. Med. 2016, 13, 129–136. [Google Scholar] [CrossRef] [PubMed]
- Jones, S. Permeability rules for antibiotic design. Nat. Biotechnol. 2017, 35, 639. [Google Scholar] [CrossRef] [PubMed]
- Mayslich, C.; Grange, P.A.; Dupin, N. Cutibacterium acnes as an opportunistic pathogen: An update of its virulence-associated factors. Microorganisms 2021, 9, 303. [Google Scholar] [CrossRef]
- Huan, Y.; Kong, Q.; Mou, H.; Yi, H. Antimicrobial peptides: Classification, design, application and research progress in multiple fields. Front. Microbiol. 2020, 11, 2559. [Google Scholar] [CrossRef]
- Rodrigues, L.A.; Leonardo, I.C.; Gaspar, F.B.; Roseiro, L.C.; Duarte, A.R.C.; Matias, A.A.; Paiva, A. Unveiling the potential of betaine/polyol-based deep eutectic systems for the recovery of bioactive protein derivative-rich extracts from sardine processing residues. Sep. Purif. Technol. 2021, 276, 119267. [Google Scholar] [CrossRef]
- Essien, S.O.; Young, B.; Baroutian, S. The antibacterial and antiproliferative ability of kānuka, Kunzea ericoides, leaf extracts obtained by subcritical water extraction. J. Chem. Technol. Biotechnol. 2021, 96, 1308–1315. [Google Scholar] [CrossRef]
- Lu, C.; Li, C.; Chen, B.; Shen, Y. Composition and antioxidant, antibacterial, and anti-HepG2 cell activities of polyphenols from seed coat of amygdalus pedunculata pall. Food Chem. 2018, 265, 111–119. [Google Scholar] [CrossRef]
- Wolfe, K.L.; Rui, H.L. Cellular antioxidant activity (CAA) assay for assessing antioxidants, foods, and dietary supplements. J. Agric. Food Chem. 2007, 55, 8896–8907. [Google Scholar] [CrossRef]
- Csekes, E.; Račková, L. Skin Aging, Cellular senescence and natural polyphenols. Int. J. Mol. Sci. 2021, 22, 12641. [Google Scholar] [CrossRef]
- Li, S.; Xie, R.; Jiang, C.; Liu, M. Schizandrin A Alleviates LPS-Induced Injury in human keratinocyte cell hacat through a microRNA-127-dependent regulation. Cell. Physiol. Biochem. 2018, 49, 2229–2239. [Google Scholar] [CrossRef] [PubMed]
- Zampetti, A.; Mastrofrancesco, A.; Flori, E.; Maresca, V.; Picardo, M.; Amerio, P.; Feliciani, C. Proinflammatory cytokine production in HaCaT cells treated by eosin: Implications for the topical treatment of psoriasis. Int. J. Immunopathol. Pharmacol. 2009, 22, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- Colombo, I.; Sangiovanni, E.; Maggio, R.; Mattozzi, C.; Zava, S.; Corbett, Y.; Fumagalli, M.; Carlino, C.; Corsetto, P.A.; Scaccabarozzi, D.; et al. HaCaT cells as a reliable in vitro differentiation model to dissect the inflammatory/repair response of human keratinocytes. Mediat. Inflamm. 2017, 2017, 7435621. [Google Scholar] [CrossRef]
- Jeong, S.J.; Lim, H.S.; Seo, C.S.; Jin, S.E.; Yoo, S.R.; Lee, N.; Shin, H.K. Anti-inflammatory actions of herbal formula Gyejibokryeong-hwan regulated by inhibiting chemokine production and STAT1 activation in HaCaT cells. Biol. Pharm. Bull. 2015, 38, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Rock, K.L.; Kono, H. The inflammatory response to cell death. Annu. Rev. Pathol. 2008, 3, 99–126. [Google Scholar] [CrossRef]
- Ngo, D.H.; Vo, T.S.; Ngo, D.N.; Wijesekara, I.; Kim, S.K. Biological activities and potential health benefits of bioactive peptides derived from marine organisms. Int. J. Biol. Macromol. 2012, 51, 378–383. [Google Scholar] [CrossRef]
- Song, R.; Wei, R.B.; Luo, H.Y.; Wang, D.F. Isolation and characterization of an antibacterial peptide fraction from the pepsin hydrolysate of half-fin anchovy (Setipinna taty). Molecules 2012, 17, 2980. [Google Scholar] [CrossRef]
- Ahmed, R.; Chun, B.S. Subcritical water hydrolysis for the production of bioactive peptides from tuna skin collagen. J. Supercrit. Fluids 2018, 141, 88–96. [Google Scholar] [CrossRef]
- Wang, M.; Zhou, J.; Pallarés, N.; Bäuerl, C.; Collado, M.C.; Dar, B.N.; Barba, F.J. Role of extracts obtained from rainbow trout and sole side streams by accelerated solvent extraction and pulsed electric fields on modulating bacterial and anti-inflammatory activities. Separations 2021, 8, 187. [Google Scholar] [CrossRef]
| Sample | Defatting Conditions | SW Extraction Conditions | Extraction Yield (g/100 g Feed) [11,22] | Protein Content (wt %) [11,22] |
|---|---|---|---|---|
| Cf1 | - | 90 °C, 100 bar | 13.2 ± 0.5 | 81.6 ± 0.3 |
| Cf2 | - | 140 °C, 100 bar | 27.7 ± 0.5 | 93.6 ± 0.3 |
| Cf3 | - | 190 °C, 100 bar | 41.4 ± 0.5 | 95 ± 0.3 |
| Cf4 | - | 250 °C, 100 bar | 53.9 ± 0.5 | 84.4 ± 0.3 |
| S1 | ScCO2 (40 °C, 250 bar) | 190 °C, 100 bar | 45.7 ± 2.8 | 87.5 ± 2.7 |
| S2 | - | 250 °C, 100 bar | 58.5 ± 0.4 | 57.5 ± 1.8 |
| S3 | ScCO2 (40 °C, 250 bar) | 250 °C, 100 bar | 61.7 ± 2.0 | 85.2 ± 0.6 |
| Sample | Antioxidant Activity | Anti-Ageing Activity | Anti-Hyperpigmentation Activity |
|---|---|---|---|
| ORAC | Elastase Inhibition | Tyrosinase Inhibition | |
| (µmol TEAC/mg extract) | (IC50, mg extract/mL) | (IC50, mg extract/mL) | |
| Cf1 | 0.64 ± 0.18 | 42.58 (37.38, 49.88) | >100 ** |
| Cf2 | 0.54 ± 0.22 | 60.35 (50.71, 73.43) | 78.59 (61.41, 100.70) |
| Cf3 | 0.59 ± 0.26 | 34.74 (24.57, 49.16) | >100 ** |
| Cf4 | 1.29 ± 0.26 | 38.11 (30.42, 49.52) | 40.89 (30.26, 54.43) |
| S1 | 1.94 ± 0.08 | 44.29 (39.10, 53.44) | 82.51 (57.12, 108.39) |
| S2 | 1.19 ± 0.10 | >300 * | 3.70 (3.26, 4.42) |
| S3 | 1.24 ± 0.06 | 17.96 (12.36, 25.83) | 10.40 (5.02, 18.15) |
| Sample | MIC* Median | MIC Median | MBC Median |
|---|---|---|---|
| (mg/mL) | (mg/mL) | (mg/mL) | |
| (n = 1/n = 2/n = 3) | (n = 1/n = 2/n = 3) | (n = 1/n = 2/n = 3) | |
| Cf1 | 0.39 | >50.00 | >50.00 |
| (0.39/0.39/0.20) | (>50.00/>50.00/>50.00) | (>50.00/>50.00/>50.00) | |
| Cf2 | 0.78 | >50.00 | >50.00 |
| (0.78/0.78/0.39) | (>50.00/>50.00/>50.00) | (>50.00/>50.00/>50.00) | |
| Cf3 | 0.39 | >50.00 | >50.00 |
| (0.39/0.39/0.20) | (>50.00/>50.00/>50.00) | (>50.00/>50.00/>50.00) | |
| Cf4 | 0.10 | >50.00 | >50.00 |
| (0.10/0.10/0.10) | (>50.00/>50.00/>50.00) | (>50.00/>50.00/>50.00) | |
| S1 | 1.56 | 25.00 | 50.00 |
| (1.56/1.56/0.78) | (25.00/25.00/25.00) | (50.00/50.00/50.00) | |
| S2 | 0.07 | 1.17 | 9.38 |
| (0.07/0.07/0.07) | (1.17/2.34/1.17) | (9.38/9.38/9.38) | |
| S3 | 0.27 | 68.75 | 68.75 |
| (0.27/0.54/0.27) | (68.75/68.75/34.38) | (68.75/68.75/68.75) |
| Sample | MIC* Median | MIC Median | MBC Median |
|---|---|---|---|
| (mg/mL) | (mg/mL) | (mg/mL) | |
| (n = 1/n = 2/n = 3) | (n = 1/n = 2/n = 3) | (n = 1/n = 2/n = 3) | |
| Cf1 | 12.50 | >50.00 | > 50.00 |
| (12.50/12.50/12.50) | (>50.00/>50.00/>50.00) | (>50.00/>50.00/>50.00) | |
| Cf2 | 25.00 | >50.00 | >50.00 |
| (25.00/25.00/50.00) | (>50.00/>50.00/>50.00) | (>50.00/>50.00/>50.00) | |
| Cf3 | 0.78 | >50.00 | >50.00 |
| (0.39/0.78/0.78) | (>50.00/>50.00/>50.00) | (>50.00/>50.00/>50.00) | |
| Cf4 | 0.39 | >50.00 | >50.00 |
| (0.39/0.39/0.20) | (>50.00/>50.00/>50.00) | (>50.00/>50.00/>50.00) | |
| S1 | 3.13 | 50.00 | 50.00 |
| (3.13/3.13/6.25) | (50.00/50.00/50.00) | (50.00/50.00/50.00) | |
| S2 | 0.07 | 4.69 | 18.75 |
| (0.07/0.07/0.07) | (4.69/4.69/9.38) | (18.75/18.75/18.75) | |
| S3 | 0.54 | 68.75 | 68.75 |
| (0.54/1.07/0.54) | (68.75/68.75/68.75) | (68.75/>68.75/68.75) |
| Sample | MIC* Median | MIC Median | MBC Median |
|---|---|---|---|
| (mg/mL) | (mg/mL) | (mg/mL) | |
| (n = 1/n = 2/n = 3) | (n = 1/n = 2/n = 3) | (n = 1/n = 2/n = 3) | |
| S2 | 0.29 | 0.59 | 2.34 |
| (0.29/0.29/0.29) | (0.59/0.29/0.59) | (4.69/1.17/2.34) | |
| S3 | 2.15 | 17.19 | > 68.75 |
| (2.15/2.15/2.15) | (34.38/17.19/17.19) | (>68.75/68.75/>68.75) |
| Sample | IC50 (mg Extract/mL) |
|---|---|
| Cf1 | 9.7 (9.5, 9.9) |
| Cf2 | 3.6 (3.5, 3.7) |
| Cf3 | 17.3 (15.2, 19.6) |
| Cf4 | 2.0 (1.9, 2.2) |
| S1 | 0.6 (0.5, 0.7) |
| S2 | 3.4 (3.3, 3.6) |
| S3 | >5.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardeira, M.; Bernardo, A.; Leonardo, I.C.; Gaspar, F.B.; Marques, M.; Melgosa, R.; Paiva, A.; Simões, P.; Fernández, N.; Serra, A.T. Cosmeceutical Potential of Extracts Derived from Fishery Industry Residues: Sardine Wastes and Codfish Frames. Antioxidants 2022, 11, 1925. https://doi.org/10.3390/antiox11101925
Cardeira M, Bernardo A, Leonardo IC, Gaspar FB, Marques M, Melgosa R, Paiva A, Simões P, Fernández N, Serra AT. Cosmeceutical Potential of Extracts Derived from Fishery Industry Residues: Sardine Wastes and Codfish Frames. Antioxidants. 2022; 11(10):1925. https://doi.org/10.3390/antiox11101925
Chicago/Turabian StyleCardeira, Martim, Ana Bernardo, Inês C. Leonardo, Frédéric B. Gaspar, Marta Marques, Rodrigo Melgosa, Alexandre Paiva, Pedro Simões, Naiara Fernández, and Ana Teresa Serra. 2022. "Cosmeceutical Potential of Extracts Derived from Fishery Industry Residues: Sardine Wastes and Codfish Frames" Antioxidants 11, no. 10: 1925. https://doi.org/10.3390/antiox11101925








