Preparation and Characterization of Ginger Peel Polysaccharide–Zn (II) Complexes and Evaluation of Anti-Inflammatory Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Material and Reagents
2.2. Extraction and Purification of Polysaccharides from Ginger Peel
2.3. Synthesis of GP–Zn (II) Derivatives
2.4. Monosaccharide Composition
2.5. Molecular Weight Distribution
2.6. SEM Analysis
2.7. UV-Vis and FTIR Spectral Analyses
2.8. XRD Analysis
2.9. CD Spectral Analysis
2.10. Thermal Analysis
2.11. Anti-Inflammatory Effects of GPs and Their Zn (II) Derivatives Based on CuSO4-Induced Zebrafish Model
2.11.1. Effect of GPs and Their Zn (II) Derivatives on the Migration of Macrophages
2.11.2. Effect of GPs and Their Zn (II) Derivatives on the Expression Levels of Inflammation-Associated Cytokines
2.12. Statistical Analysis
3. Results and Discussion
3.1. Preparation of GP–Zn (II) Complexes
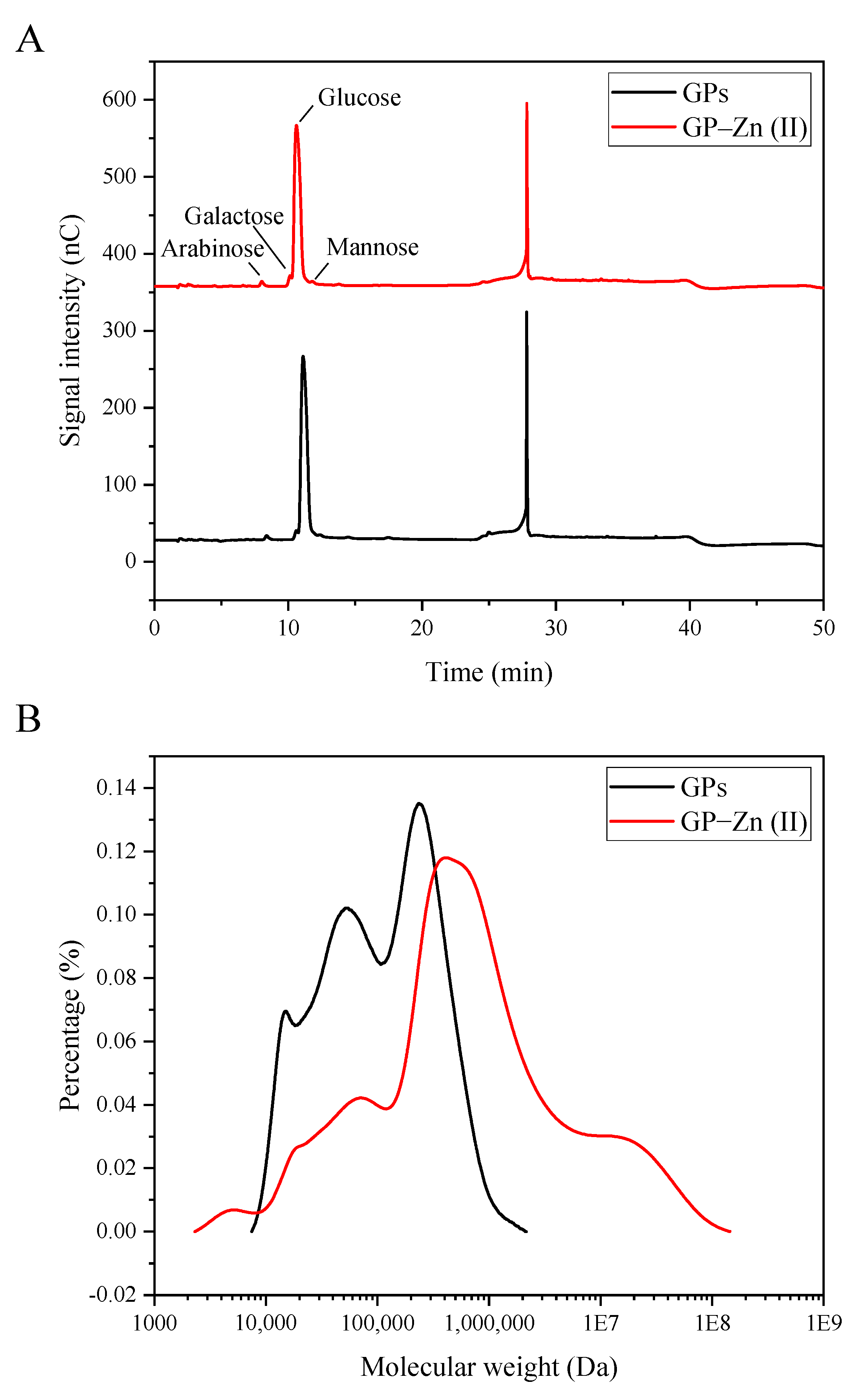
3.2. Monosaccharide Composition
3.3. Molecular Weight Distribution
3.4. Morphological Characteristics
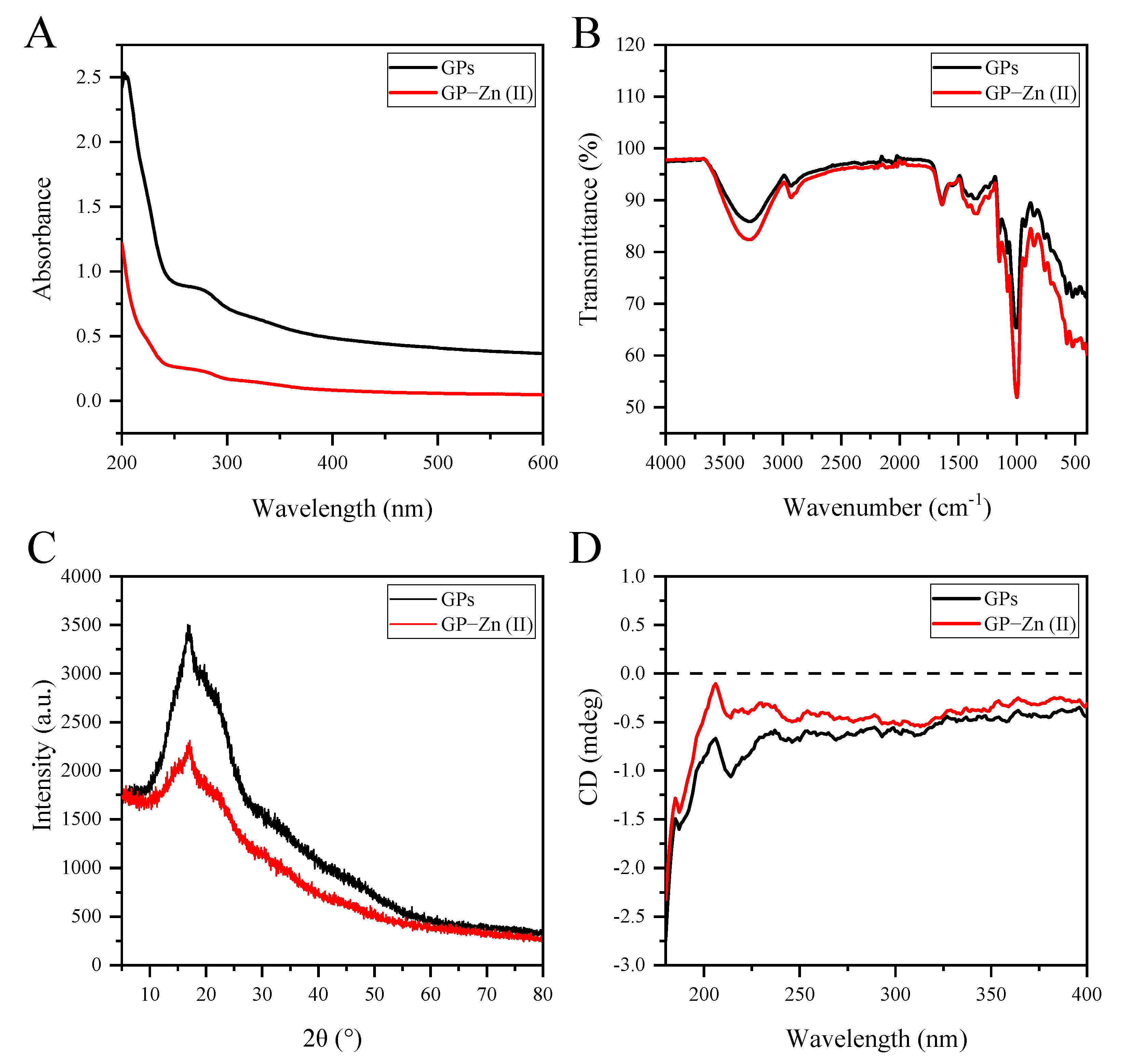
3.5. UV-Vis and FTIR Spectral Analyses
3.6. XRD Analysis
3.7. CD Spectral Analysis
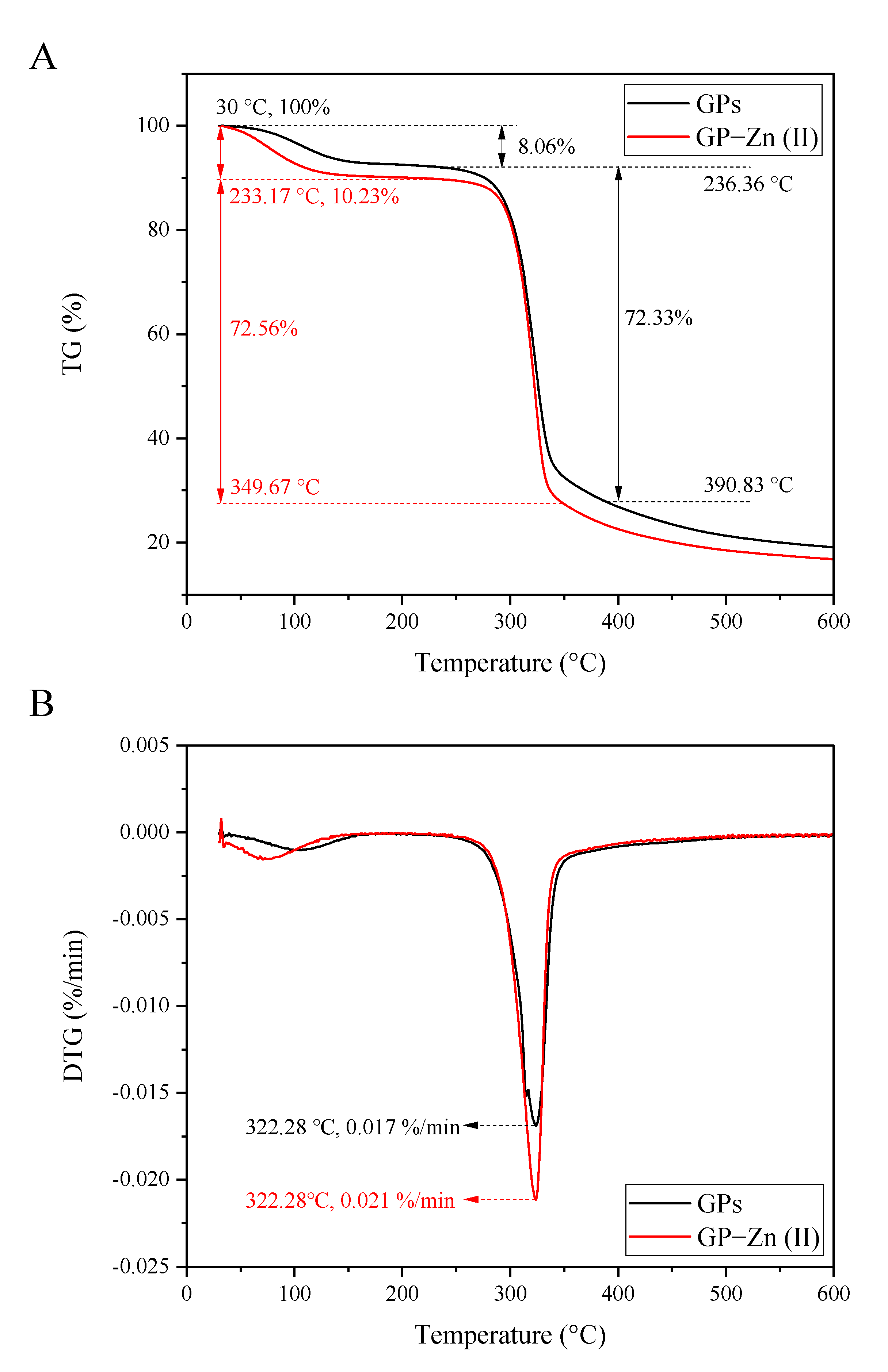
3.8. Thermal Analysis
3.9. Evaluation of the Anti-Inflammatory Activity of GPs and Their Zn (II) Derivatives and Underlying Mechanisms
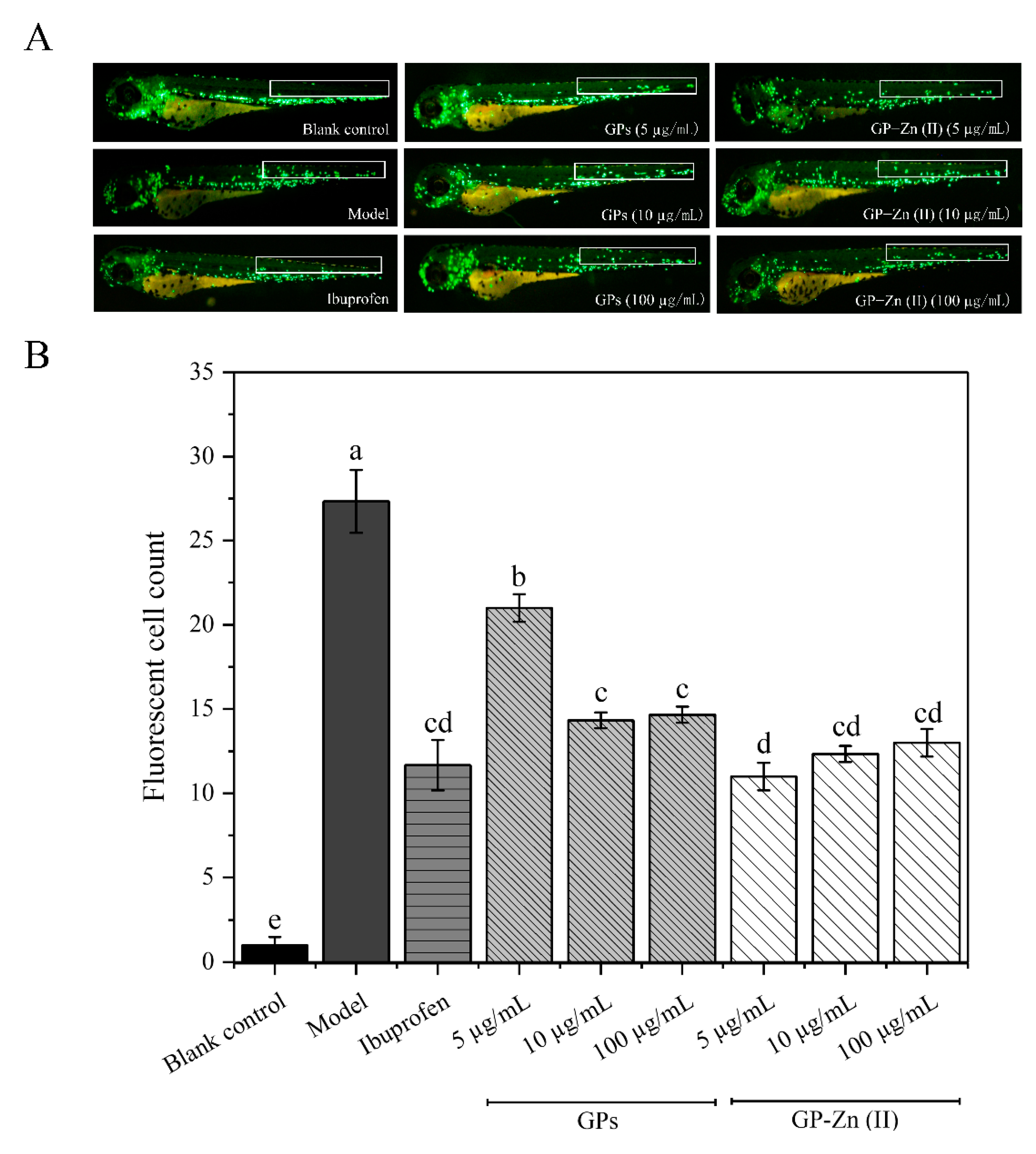
3.9.1. Anti-Inflammatory Effects Based on CuSO4-Induced Zebrafish Model
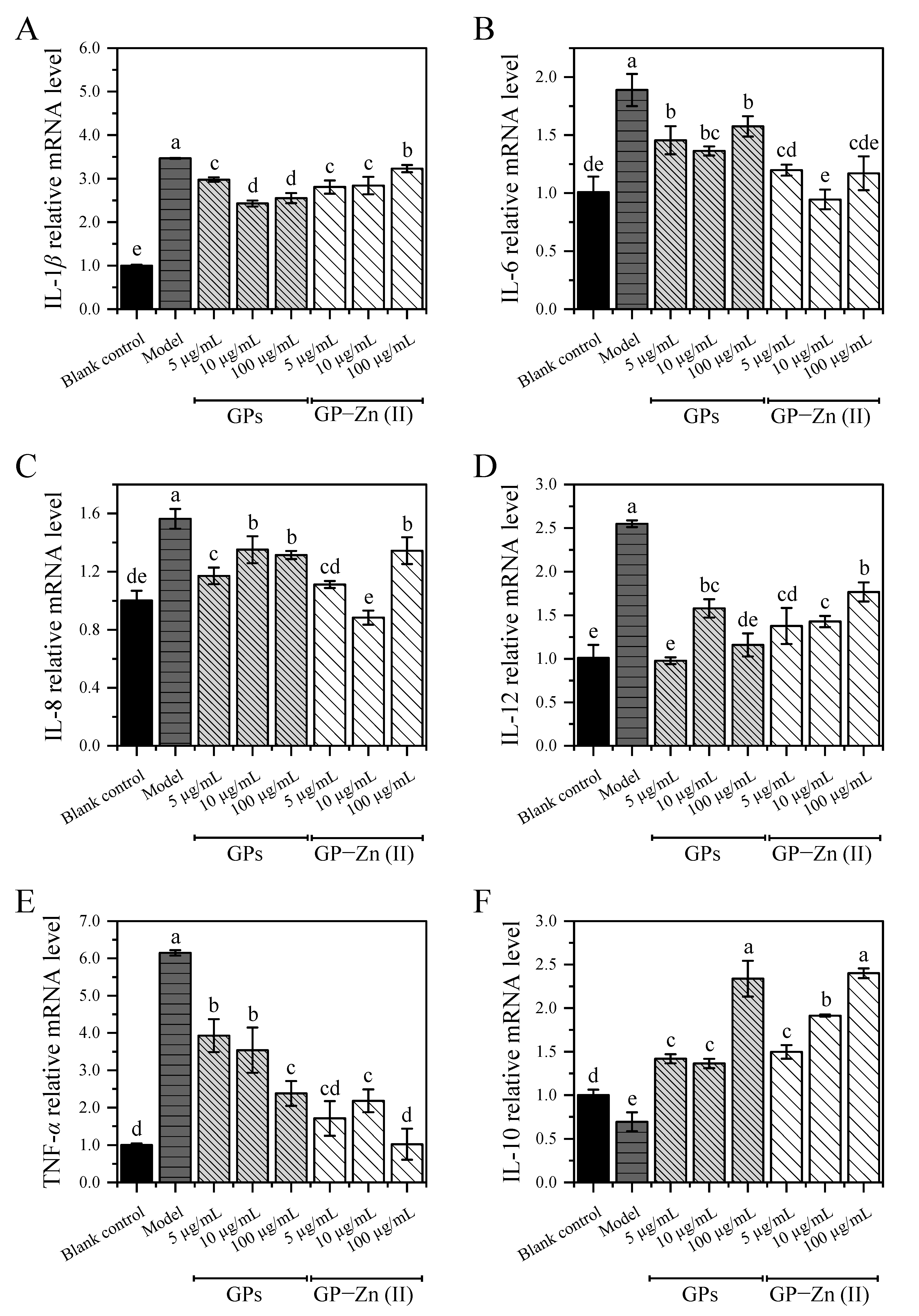
3.9.2. Effect of GPs and Their Zn (II) Derivatives on the Expression Levels of Inflammation-Associated Cytokines
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Qiu, Z.; Qiao, Y.; Zhang, B.; Sun-Waterhouse, D.; Zheng, Z. Bioactive polysaccharides and oligosaccharides from garlic (Allium sativum L.): Production, physicochemical and biological properties, and structure-function relationships. Compr. Rev. Food Sci. F 2022, 21, 3033–3095. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Q.; Hao, H.; He, L.; Jing, Y.; Xu, T.; Chen, J.; Zhang, H.; Hu, T.; Zhang, Q.; Yang, X.; et al. Anti-inflammatory and anti-angiogenic activities of a purified polysaccharide from flesh of Cipangopaludina chinensis. Carbohydr. Polym. 2017, 176, 152–159. [Google Scholar] [CrossRef]
- Yuan, L.; Qiu, Z.; Yang, Y.; Liu, C.; Zhang, R. Preparation, structural characterization and antioxidant activity of water-soluble polysaccharides and purified fractions from blackened jujube by an activity-oriented approach. Food Chem. 2022, 385, 132637. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.K.; Chang, C.C.; Chao, C.H.; Hsu, Y.C. Structural changes, and anti-inflammatory, anti-cancer potential of polysaccharides from multiple processing of Rehmannia glutinosa. Int. J. Biol. Macromol. 2022, 206, 621–632. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Sheng, Y.; Liu, J.; Xu, G.; Yu, W.; Cui, Q.; Lu, X.; Du, P.; An, L. Hair-growth promoting effect and anti-inflammatory mechanism of Ginkgo biloba polysaccharides. Carbohydr. Polym. 2022, 278, 118811. [Google Scholar] [CrossRef]
- Sun, Y.; Huo, J.; Zhong, S.; Zhu, J.; Li, Y.; Li, X. Chemical structure and anti-inflammatory activity of a branched polysaccharide isolated from Phellinus baumii. Carbohydr. Polym. 2021, 268, 118214. [Google Scholar] [CrossRef]
- Wu, D.T.; He, Y.; Fu, M.X.; Gan, R.Y.; Hu, Y.C.; Peng, L.X.; Zhao, G.; Zou, L. Structural characteristics and biological activities of a pectic-polysaccharide from okra affected by ultrasound assisted metal-free Fenton reaction. Food Hydrocoll. 2022, 122, 107085. [Google Scholar] [CrossRef]
- Chen, D.; Sun, H.; Shen, Y.; Luo, M.; Xin, X.; Xu, Z. Selenium bio-absorption and antioxidant capacity in mice treated by selenium modified rice germ polysaccharide. J. Funct. Foods 2019, 61, 103492. [Google Scholar] [CrossRef]
- Gong, H.; Li, W.; Sun, J.; Jia, L.; Guan, Q.; Guo, Y.; Wang, Y. A review on plant polysaccharide based on drug delivery system for construction and application, with emphasis on traditional Chinese medicine polysaccharide. Int. J. Biol. Macromol. 2022, 211, 711–728. [Google Scholar] [CrossRef]
- Zhang, W.; Li, L.; Ma, Y.; Chen, X.; Lan, T.; Chen, L.; Zheng, Z. Structural characterization and hypoglycemic activity of a novel pumpkin peel polysaccharide-chromium (III) complex. Foods 2022, 11, 1821. [Google Scholar] [CrossRef]
- Bao, B.; Prasad, A.S.; Beck, F.W.; Fitzgerald, J.T.; Snell, D.; Bao, G.W.; Singh, T.; Cardozo, L.J. Zinc decreases C-reactive protein, lipid peroxidation, and inflammatory cytokines in elderly subjects: A potential implication of zinc as an atheroprotective agent. Am. J. Clin. Nutr. 2010, 91, 1634–1641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chasapis, C.T.; Loutsidou, A.C.; Spiliopoulou, C.A.; Stefanidou, M.E. Zinc and human health: An update. Arch. Toxicol. 2012, 86, 521–534. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, S.; Wang, N.; Xu, X.; Liu, H.; Wang, D.; Zhang, Y. Preparation and characterization of Auricularia cornea Ehrenb polysaccharide-Zn complex and its hypoglycemic activity through regulating insulin resistance in HepG2 cells. J. Food Qual. 2021, 2021, 4497128. [Google Scholar] [CrossRef]
- Zhang, M.; Zhao, H.; Shen, Y.; Wang, Y.; Zhao, Z.; Zhang, Y. Preparation, characterization and antioxidant activity evaluation in vitro of Fritillaria ussuriensis polysaccharide-zinc complex. Int. J. Biol. Macromol. 2020, 146, 462–474. [Google Scholar] [CrossRef]
- Zhao, S.; Li, B.; Chen, G.; Hu, Q.; Zhao, L. Preparation, characterization, and anti-inflammatory effect of the chelate of Flammulina velutipes polysaccharide with Zn. Food Agric. Immunol. 2017, 28, 162–177. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.H.; Zhang, W.D.; Yin, F.; Liu, J.; Zhao, X.L.; Liu, S.Q.; Xu, L.; Chen, Y.B.; Yang, H. Effects of different treatments on properties of biogas fermentation with ginger skin. Adv. Mat. Res. 2014, 953, 284–289. [Google Scholar] [CrossRef]
- Feng, X.; Xia, Y.; Chen, G.T.; Xu, J.J.; Liao, X.J.; Zhao, L.Y. Purification and structural analysis of polysaccharides from ginger peels. Food Sci. 2017, 38, 185–190. [Google Scholar] [CrossRef]
- Zhou, S.; Huang, G.; Chen, G. Extraction, structural analysis, derivatization and antioxidant activity of polysaccharide from Chinese yam. Food Chem. 2021, 361, 130089. [Google Scholar] [CrossRef]
- Li, W.; Wang, D.; Dong, S.; Hou, L.; Zhang, X.; Zheng, Z. Preparation of ginger peel polysaccharidein-zinc complex and in vitro simulated study. Sci. Technol. Food Ind. 2022, 43, 1–12. [Google Scholar] [CrossRef]
- Yang, K.X.; Swami, K. Determination of metals in marine species by microwave digestion and inductively coupled plasma mass spectrometry analysis. Spectrochim. Acta B 2007, 62, 1177–1181. [Google Scholar] [CrossRef]
- Li, L.; Qiu, Z.; Dong, H.; Ma, C.; Qiao, Y.; Zheng, Z. Structural characterization and antioxidant activities of one neutral polysaccharide and three acid polysaccharides from the roots of Arctium lappa L.: A comparison. Int. J. Biol. Macromol. 2021, 182, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.S.; Qiu, Z.C.; Qiao, X.G.; Waterhouse, G.I.N.; Zhu, W.Q.; Zhao, W.Q.; He, Q.X.; Zheng, Z.J. Creating burdock polysaccharide-oleanolic acid-ursolic acid nanoparticles to deliver enhanced anti-inflammatory effects: Fabrication, structural characterization and property evaluation. Food Sci. Hum. Wellness 2022, 11, 1–25. [Google Scholar] [CrossRef]
- Jia, Y.; Li, N.; Wang, Q.; Zhou, J.; Liu, J.; Zhang, M.; He, C.; Chen, H. Effect of Fe (III), Zn (II), and Cr (III) complexation on the physicochemical properties and bioactivities of corn silk polysaccharide. Int. J. Biol. Macromol. 2021, 189, 847–856. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wei, S.; Lu, X.; Qiao, X.; Simal-Gandara, J.; Capanoglu, E.; Woźniak, Ł.; Zou, L.; Cao, H.; Xiao, J.; et al. A neutral polysaccharide with a triple helix structure from ginger: Characterization and immunomodulatory activity. Food Chem. 2021, 350, 129261. [Google Scholar] [CrossRef] [PubMed]
- Nie, C.; Zhu, P.; Ma, S.; Wang, M.; Hu, Y. Purification, characterization and immunomodulatory activity of polysaccharides from stem lettuce. Carbohydr. Polym. 2018, 188, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Yu, X.; Yang, X.; Li, Y.; Yao, Y.; Lui, E.M.K.; Ren, G. Structural and anti-inflammatory characterization of a novel neutral polysaccharide from North American ginseng (Panax quinquefolius). Int. J. Biol. Macromol. 2015, 74, 12–17. [Google Scholar] [CrossRef]
- Yang, Y.; Qiu, Z.; Li, L.; Vidyarthi, S.K.; Zheng, Z.; Zhang, R. Structural characterization and antioxidant activities of one neutral polysaccharide and three acid polysaccharides from Ziziphus jujuba cv. Hamidazao: A comparison. Carbohyd. Polym. 2021, 261, 117879. [Google Scholar] [CrossRef]
- Ji, X.; Hou, C.; Yan, Y.; Shi, M.; Liu, Y. Comparison of structural characterization and antioxidant activity of polysaccharides from jujube (Ziziphus jujuba Mill.) fruit. Int. J. Biol. Macromol. 2020, 149, 1008–1018. [Google Scholar] [CrossRef]
- Zhu, Z.Y.; Dong, F.; Liu, X.; Lv, Q.; Liu, F.; Chen, L.; Wang, T.; Wang, Z.; Zhang, Y. Effects of extraction methods on the yield, chemical structure and anti-tumor activity of polysaccharides from Cordyceps gunnii mycelia. Carbohydr. Polym. 2016, 140, 461–471. [Google Scholar] [CrossRef]
- Wang, L.; Wang, L.; Su, C.; Wen, C.; Gong, Y.; You, Y.; Zhao, J.; Han, Y.; Song, S.; Xiao, H. Characterization and digestion features of a novel polysaccharide-Fe (III) complex as an iron supplement. Carbohydr. Polym. 2020, 249, 116812. [Google Scholar] [CrossRef]
- Bai, X.; Qiu, Z.; Zheng, Z.; Song, S.; Zhao, R.; Lu, X.; Qiao, X. Preparation and characterization of garlic polysaccharide-Zn (II) complexes and their bioactivities as a zinc supplement in Zn-deficient mice. Food Chem. X 2022, 15, 100361. [Google Scholar] [CrossRef]
- d’Alençon, C.A.; Peña, O.A.; Wittmann, C.; Gallardo, V.E.; Jones, R.A.; Loosli, F.; Liebel, U.; Grabher, C.; Allende, M.L. A high-throughput chemically induced inflammation assay in zebrafish. BMC Biol. 2010, 8, 151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gui, Y.H.; Liu, L.; Wu, W.; Zhang, Y.; Jia, Z.L.; Shi, Y.P.; Kong, H.T.; Liu, K.C.; Jiao, W.H.; Lin, H.W. Discovery of nitrogenous sesquiterpene quinone derivatives from sponge Dysidea septosa with anti-inflammatory activity in vivo zebrafish model. Bioorg. Chem. 2020, 94, 103435. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Qiu, Z.; Gu, D.; Wang, F.; Zhang, R. A novel anti-inflammatory polysaccharide from blackened jujube: Structural features and protective effect on dextran sulfate sodium-induced colitic mice. Food Chem. 2022, 405, 134869. [Google Scholar] [CrossRef]
- Wang, Q.; Huang, J.; Zheng, Y.; Guan, X.; Lai, C.; Gao, H.; Ho, C.T.; Lin, B. Selenium-enriched oolong tea (Camellia sinensis) extract exerts anti-inflammatory potential via targeting NF-κB and MAPK pathways in macrophages. Food Sci. Hum. Wellness 2022, 11, 635–642. [Google Scholar] [CrossRef]
- Zhou, Y.; Hu, Z.; Ye, F.; Guo, T.; Luo, Y.; Zhou, W.; Qin, D.; Tang, Y.; Cao, F.; Luo, F.; et al. Mogroside V exerts anti-inflammatory effect via MAPK-NF-κB/AP-1 and AMPK-PI3K/Akt/mTOR pathways in ulcerative colitis. J. Funct. Foods 2021, 87, 104807. [Google Scholar] [CrossRef]
- Hao, W.; Chen, Z.; Yuan, Q.; Ma, M.; Gao, C.; Zhou, Y.; Zhou, H.; Wu, X.; Wu, D.; Farag, M.A.; et al. Ginger polysaccharides relieve ulcerative colitis via maintaining intestinal barrier integrity and gut microbiota modulation. Int. J. Biol. Macromol. 2022, 219, 730–739. [Google Scholar] [CrossRef] [PubMed]
- Li, J.J.; Zhang, Y.; Han, L.W.; Tian, Q.P.; He, Q.X.; Wang, X.M.; Sun, C.; Han, J.; Liu, K.C. Tenacissoside H exerts an anti-inflammatory effect by regulating the NF-κb and p38 pathways in zebrafish. Fish Shellfish Immun. 2018, 83, 205–212. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, W.; Qiu, Z.; Ma, Y.; Zhang, B.; Li, L.; Li, Q.; He, Q.; Zheng, Z. Preparation and Characterization of Ginger Peel Polysaccharide–Zn (II) Complexes and Evaluation of Anti-Inflammatory Activity. Antioxidants 2022, 11, 2331. https://doi.org/10.3390/antiox11122331
Li W, Qiu Z, Ma Y, Zhang B, Li L, Li Q, He Q, Zheng Z. Preparation and Characterization of Ginger Peel Polysaccharide–Zn (II) Complexes and Evaluation of Anti-Inflammatory Activity. Antioxidants. 2022; 11(12):2331. https://doi.org/10.3390/antiox11122331
Chicago/Turabian StyleLi, Wenwen, Zhichang Qiu, Yue Ma, Bin Zhang, Lingyu Li, Qiulin Li, Qiuxia He, and Zhenjia Zheng. 2022. "Preparation and Characterization of Ginger Peel Polysaccharide–Zn (II) Complexes and Evaluation of Anti-Inflammatory Activity" Antioxidants 11, no. 12: 2331. https://doi.org/10.3390/antiox11122331
APA StyleLi, W., Qiu, Z., Ma, Y., Zhang, B., Li, L., Li, Q., He, Q., & Zheng, Z. (2022). Preparation and Characterization of Ginger Peel Polysaccharide–Zn (II) Complexes and Evaluation of Anti-Inflammatory Activity. Antioxidants, 11(12), 2331. https://doi.org/10.3390/antiox11122331







