Phytate Intake, Health and Disease: “Let Thy Food Be Thy Medicine and Medicine Be Thy Food”
Abstract
1. Introduction
2. Phytate and Vascular Calcification
2.1. Background and In Vitro Studies
2.2. Animal Studies
2.3. Epidemiological Studies in Humans
2.4. Clinical Trials in Humans
3. Phytate and Urolithiasis
3.1. Background and In Vitro Studies
3.2. Animal Studies
3.3. Epidemiological Studies
3.4. Clinical Trials in Humans
4. Phytate and Osteoporosis
4.1. Background and In Vitro Studies
4.2. Animal Studies
4.3. Epidemiological Studies
4.4. Clinical Trials in Humans
5. Phytate Cognitive Function and Neurodegenerative Disease
5.1. Background and In Vitro Studies
5.2. Animal Studies
5.3. Epidemiological Studies
5.4. Clinical Trials in Humans
6. Phytate Intake, Type 2 Diabetes Mellitus and Cardiovascular Health
6.1. Background and In Vitro Studies
6.2. Animal Studies
6.3. Epidemiological Studies in Humans
6.4. Clinical Trials in Humans
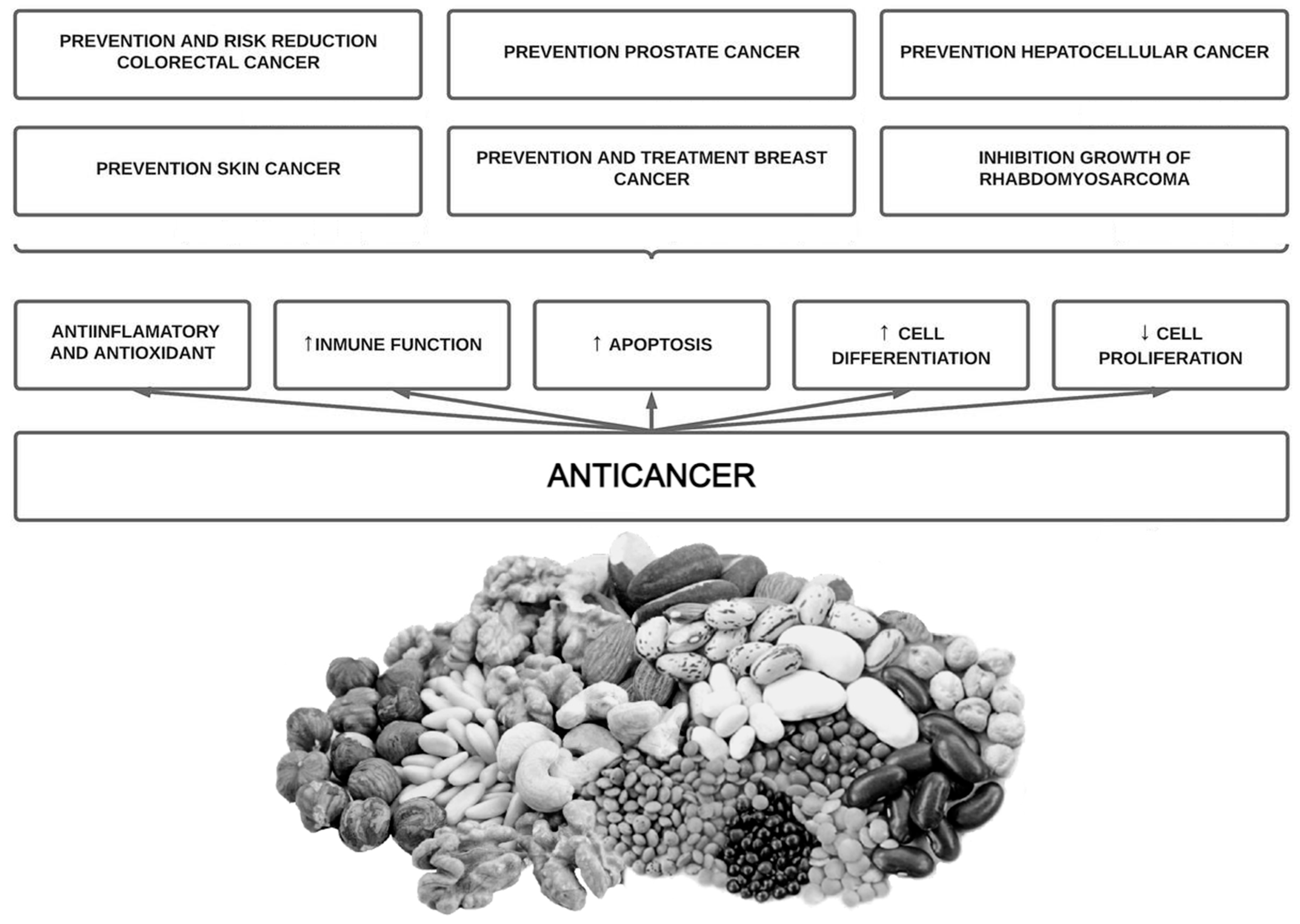
7. Phytate Intake and Cancer
7.1. Background and In Vitro Studies
7.2. Animal Studies
7.3. Epidemiological Studies in Humans
7.4. Clinical Trials in Humans
8. Other Phytate Applications
9. Controversies of Phytate Intake
10. Conclusions
- It is well known that phytate is a powerful agent for preventing calcifications in biological fluids: usefulness in renal lithiasis treatment, sialolithiasis and vascular calcifications.
- Phytate can also avoid or disturb loss of bone mass. Low doses of phytate could generate a strongly protective effect as we have seen in osteoporosis where a minimum of 307 mg/day of phytate (1–2 servings of nuts or legumes per day) reduce the risk of osteoporosis.
- Phytate has shown benefits in reducing leptin levels, increasing adiponectin, improving carbohydrate and lipid metabolism, decreasing AGEs (the potential reduction in microvascular and macrovascular diabetes-related complications and aging), improving anti-inflammatory and antioxidant effects and improving quality of life during chemotherapy.
- There is no pharmacological intervention available that directly reduces neurological decline, vascular calcifications, and urolithiasis. Phytate has shown effectivity in these regards in in vitro, animal, and epidemiological data.
- Recommending a diet high in phytate such as the Mediterranean diet or DASH diet can exert multiple health benefits with no harm.
- The effectivity of phytate intake seen in in vivo, animal and epidemiological data has revealed the need for well-designed human clinical trials to assess the effectivity of phytate intake in vascular calcifications, urolithiasis, osteoporosis, cognitive function, metabolic health and cancer. Phytate intake or phytate-based drugs/supplements should be investigated further.
Author Contributions
Funding
Conflicts of Interest
References
- Perez-Gregorio, R.; Simal-Gandara, J. A Critical Review of Bioactive Food Components, and of their Functional Mechanisms, Biological Effects and Health Outcomes. Curr. Pharm. Des. 2017, 23, 2731–2741. [Google Scholar] [CrossRef]
- Guasch-Ferré, M.; Willett, W.C. The Mediterranean diet and health: A comprehensive overview. J. Intern. Med. 2021, 290, 549–566. [Google Scholar] [CrossRef] [PubMed]
- Sanchis, P.; Molina, M.; Berga, F.; Muñoz, E.; Fortuny, R.; Costa-Bauzá, A.; Grases, F.; Buades, J.M. A Pilot Randomized Crossover Trial Assessing the Safety and Short-Term Effects of Walnut Consumption by Patients with Chronic Kidney Disease. Nutrients 2019, 12, 63. [Google Scholar] [CrossRef] [PubMed]
- Buades Fuster, J.M.; Sanchís Cortés, P.; Perelló Bestard, J.; Grases Freixedas, F. Plant phosphates, phytate and pathological calcifications in chronic kidney disease. Nefrologia 2017, 37, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Bes-Rastrollo, M.; Román-Vinas, B.; Pfrimer, K.; Sánchez-Villegas, A.; Martínez-González, M.A. Dietary patterns and nutritional adequacy in a Mediterranean country. Br. J. Nutr. 2019, 101 (Suppl. S2), S21–S28. [Google Scholar] [CrossRef]
- Demer, L.L.; Tintut, Y. Vascular Calcification: Pathobiology of a multifaceted disease. Circulation 2008, 117, 2938–2948. [Google Scholar] [CrossRef]
- Sanchis, P.; Buades, J.M.; Berga, F.; Gelabert, M.M.; Molina, M.; Íñigo, M.V.; García, S.; Gonzalez, J.; Bernabeu, M.R.; Costa-Bauzá, A.; et al. Protective Effect of Myo-Inositol Hexaphosphate (Phytate) on Abdominal Aortic Calcification in Patients With Chronic Kidney Disease. J. Ren. Nutr. 2016, 26, 226–236. [Google Scholar] [CrossRef]
- Grases, F.; Costa-Bauza, A. Key Aspects of Myo-Inositol Hexaphosphate (Phytate) and Pathological Calcifications. Molecules 2019, 24, 4434. [Google Scholar] [CrossRef]
- Sanchis, P.; López-González, Á.-A.; Costa-Bauzá, A.; Busquets-Cortés, C.; Riutord, P.; Calvo, P.; Grases, F. Understanding the Protective Effect of Phytate in Bone Decalcification Related-Diseases. Nutrients 2021, 13, 2859. [Google Scholar] [CrossRef]
- Grases, F.; Sanchis, P.; Perello, J.; Isern, B.; Prieto, R.M.; Fernandez-Palomeque, C.; Saus, C. Phytate reduces age-related cardiovascular calcification. Front. Biosci. 2008, 13, 7115–7122. [Google Scholar] [CrossRef]
- Grases, F.; Sanchis, P.; Perello, J.; Isern, B.; Prieto, R.M.; Fernandez-Palomeque, C.; Fiol, M.; Bonnin, O.; Torres, J.J. Phytate (Myo-inositol hexakisphosphate) inhibits cardiovascular calcifications in rats. Front. Biosci. 2006, 11, 136–142. [Google Scholar] [CrossRef]
- Grases, F.; Isern, B.; Sanchis, P.; Perello, J.; Torres, J.J.; Costa-Bauza, A. Phytate acts as an inhibitor in formation of renal calculi. Front. Biosci. 2007, 12, 2580–2587. [Google Scholar] [CrossRef] [PubMed]
- Perelló, J.; Salcedo, C.; Ketteler, M.; Tur, F.; Tur, E.; Isern, B.; Joubert, P.H.; Ferrer, M.D. INTRAVENOUS SNF472 INHIBITS VITAMIN D INDUCED CARDIOVASCULAR CALCIFICATION IN RATS; ASN Kidney Week: Atlanta, GA, USA, 2014; p. 96450. [Google Scholar]
- Grases, F.; Sanchis, P.; Perelló, J.; Isern, B.; Prieto, R.M.; Fernández-Palomeque, C.; Torres, J.J. Effect of Crystallization Inhibitors on Vascular Calcifications Induced by Vitamin D A Pilot Study in Sprague-Dawley Rats. Circ. J. 2007, 71, 1152–1156. [Google Scholar] [CrossRef] [PubMed]
- Ketteler, M.; Ferrer, M.D.; Tur, F.; Isern, B.; Salcedo, C.; Joubert, P.H.; Perelló, J. SNF472 INHIBITS VITAMIN D INDUCED CARDIOVASCULAR CALCIFICATION IN RATS; ASN Kidney Week: Atlanta, GA, USA, 2013. [Google Scholar]
- Grases, F.; Perello, J.; Isern, B.; Prieto, R. Study of a myo-inositol hexaphosphate-based cream to prevent dystrophic calcinosis cutis. Br. J. Dermatol. 2005, 152, 1022–1025. [Google Scholar] [CrossRef]
- Budoff, M.J.; Young, R.; Lopez, V.A.; Kronmal, R.A.; Nasir, K.; Blumenthal, R.S.; Detrano, R.C.; Bild, D.E.; Guerci, A.D.; Liu, K.; et al. Progression of Coronary Calcium and Incident Coronary Heart Disease Events. J. Am. Coll. Cardiol. 2013, 61, 1231–1239. [Google Scholar] [CrossRef]
- Vliegenthart, R.; Oudkerk, M.; Hofman, A.; Oei, H.-H.S.; Van Dijck, W.; Van Rooij, F.J.; Witteman, J.C. Coronary Calcification Improves Cardiovascular Risk Prediction in the Elderly. Circulation 2005, 112, 572–577. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Cox, A.J.; Herrington, D.M.; Jorgensen, N.W.; Xu, J.; Freedman, B.I.; Carr, J.J.; Bowden, D.W. Coronary Calcium Score Predicts Cardiovascular Mortality in Diabetes: Diabetes Heart Study. Diabetes Care 2013, 36, 972–977. [Google Scholar] [CrossRef]
- Russo, D.; Corrao, S.; Battaglia, Y.; Andreucci, M.; Caiazza, A.; Carlomagno, A.; Lamberti, M.; Pezone, N.; Pota, A.; Russo, L.; et al. Progression of coronary artery calcification and cardiac events in patients with chronic renal disease not receiving dialysis. Kidney Int. 2011, 80, 112–118. [Google Scholar] [CrossRef]
- Shantouf, R.S.; Budoff, M.J.; Ahmadi, N.; Ghaffari, A.; Flores, F.; Gopal, A.; Noori, N.; Jing, J.; Kovesdy, C.P.; Kalantar-Zadeh, K. Total and Individual Coronary Artery Calcium Scores as Independent Predictors of Mortality in Hemodialysis Patients. Am. J. Nephrol. 2010, 31, 419–425. [Google Scholar] [CrossRef]
- Fernández-Palomeque, C.; Grau, A.; Perelló, J.; Sanchis, P.; Isern, B.; Prieto, R.M.; Costa-Bauzá, A.; Caldés, O.J.; Bonnin, O.; Garcia-Raja, A.; et al. Relationship between Urinary Level of Phytate and Valvular Calcification in an Elderly Population: A Cross-Sectional Study. PLoS ONE 2015, 10, e0136560. [Google Scholar] [CrossRef]
- Gupta, R.K.; Gangoliya, S.S.; Singh, N.K. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J. Food Sci. Technol. 2013, 52, 676–684. [Google Scholar] [CrossRef]
- Lichtenstein, A.H.; Appel, L.J.; Vadiveloo, M.; Hu, F.B.; Kris-Etherton, P.M.; Rebholz, C.M.; Sacks, F.M.; Thorndike, A.N.; Van Horn, L.; Wylie-Rosett, J.; et al. 2021 Dietary Guidance to Improve Cardiovascular Health: A Scientific Statement From the American Heart Association. Circulation 2021, 144, e472–e487. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Keogh, J.B.; Clifton, P.M. Does Nut Consumption Reduce Mortality and/or Risk of Cardiometabolic Disease? An Updated Review Based on Meta-Analyses. Int. J. Environ. Res. Public Health 2019, 16, 4957. [Google Scholar] [CrossRef] [PubMed]
- Becerra-Tomás, N.; Paz-Graniel, I.; WC Kendall, C.; Kahleova, H.; Rahelić, D.; Sievenpiper, J.L.; Salas-Salvadó, J. Nut consumption and incidence of cardiovascular diseases and cardiovascular disease mortality: A meta-analysis of prospective cohort studies. Nutr. Rev. 2019, 77, 691–709. [Google Scholar] [CrossRef] [PubMed]
- Perelló, J.; Joubert, P.; Ferrer, M.D.; Canals, A.; Sinha, S.; Salcedo, C. First-time-in-human randomized clinical trial in healthy volunteers and haemodialysis patients with SNF472, a novel inhibitor of vascular calcification. Br. J. Clin. Pharmacol. 2018, 84, 2867–2876. [Google Scholar] [CrossRef]
- Perelló, J.; Gómez, M.; Ferrer, M.D.; Rodríguez, N.Y.; Salcedo, C.; Buades, J.M.; Pérez, M.M.; Torregrosa, J.V.; Martín, E.; Maduell, F. SNF472, a novel inhibitor of vascular calcification, could be administered during hemodialysis to attain potentially therapeutic phytate levels. J. Nephrol. 2018, 31, 287–296. [Google Scholar] [CrossRef]
- Raggi, P.; Bellasi, A.; Bushinsky, D.; Bover, J.; Rodriguez, M.; Ketteler, M.; Sinha, S.; Salcedo, C.; Gillotti, K.; Padgett, C.; et al. Slowing Progression of Cardiovascular Calcification With SNF472 in Patients on Hemodialysis: Results of a Randomized Phase 2b Study. Circulation 2020, 141, 728–739. [Google Scholar] [CrossRef]
- Sinha, S.; Raggi, P.; Chertow, G.M. SNF472: Mechanism of action and results from clinical trials. Curr. Opin. Nephrol. Hypertens. 2021, 30, 424–429. [Google Scholar] [CrossRef]
- Sanchis, P.; Rivera, R.; Berga, F.; Fortuny, R.; Adrover, M.; Costa-Bauza, A.; Grases, F.; Masmiquel, L. Phytate Decreases Formation of Advanced Glycation End-Products in Patients with Type II Diabetes: Randomized Crossover Trial. Sci. Rep. 2018, 8, 9619. [Google Scholar] [CrossRef]
- Saku, K.; Tahara, N.; Takaseya, T.; Otsuka, H.; Takagi, K.; Shojima, T.; Shintani, Y.; Zaima, Y.; Kikusaki, S.; Fukuda, T.; et al. Pathological Role of Receptor for Advanced Glycation End Products in Calcified Aortic Valve Stenosis. J. Am. Heart Assoc. 2020, 9, e015261. [Google Scholar] [CrossRef]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Prieto, R.M.; Rodriguez, A.; Sanchis, P.; Morey, M.; Fiol, M.; Grases, F.; Castañer, O.; Martínez-González, M.A.; Salas-Salvadó, J.; Romaguera, D. Association of Adherence to The Mediterranean Diet with Urinary Factors Favoring Renal Lithiasis: Cross-Sectional Study of Overweight Individuals with Metabolic Syndrome. Nutrients 2019, 11, 1708. [Google Scholar] [CrossRef]
- Alelign, T.; Petros, B. Kidney Stone Disease: An Update on Current Concepts. Adv. Urol. 2018, 2018, 3068365. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; García-Ferragut, L.; Costa-Bauzá, A. Development of Calcium Oxalate Crystals on Urothelium: Effect of Free Radicals. Nephron 1998, 78, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Costa-Bauza, A. Phytate (IP6) is a powerful agent for preventing calcifications in biological fluids: Usefulness in renal lithiasis treatment. Anticancer Res. 2000, 19, 3717–3722. [Google Scholar]
- Grases, F.; Rodriguez, A.; Costa-Bauza, A. Efficacy of Mixtures of Magnesium, Citrate and Phytate as Calcium Oxalate Crystallization Inhibitors in Urine. J. Urol. 2015, 194, 812–819. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Perelló, J.; Simonet, B.; Prieto, R.; García-Raja, A. Study of Potassium Phytate Effects on Decreasing Urinary Calcium in Rats. Urol. Int. 2004, 72, 237–243. [Google Scholar] [CrossRef]
- Kim, O.-H.; Booth, C.J.; Choi, H.S.; Lee, J.; Kang, J.; Hur, J.; Jung, W.J.; Jung, Y.-S.; Choi, H.J.; Kim, H.; et al. High-phytate/low-calcium diet is a risk factor for crystal nephropathies, renal phosphate wasting, and bone loss. Elife 2020, 9, e52709. [Google Scholar] [CrossRef]
- Grases, F.; Costa-Bauza, A.; Prieto, R.M. Renal lithiasis and nutrition. Nutr. J. 2006, 5, 23. [Google Scholar] [CrossRef]
- Han, H.; Segal, A.M.; Seifter, J.L.; Dwyer, J.T. Nutritional Management of Kidney Stones (Nephrolithiasis). Clin. Nutr. Res. 2015, 4, 137–152. [Google Scholar] [CrossRef]
- Lin, B.-B.; Lin, M.-E.; Huang, R.-H.; Hong, Y.-K.; Lin, B.-L.; He, X.-J. Dietary and lifestyle factors for primary prevention of nephrolithiasis: A systematic review and meta-analysis. BMC Nephrol. 2020, 21, 267. [Google Scholar] [CrossRef]
- Curhan, G.C.; Willett, W.C.; Knight, E.L.; Stampfer, M.J. Dietary Factors and the Risk of Incident Kidney Stones in Younger Women: Nurses’ Health Study II. Arch. Intern. Med. 2004, 164, 885–891. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Saez-Torres, C.; Rodriguez, A.; Costa-Bauza, A.; Rodrigo, D.; Frontera, G.; Berga, F.; Fakier, S. Urinary Phytate (Myo-Inositol Hexaphosphate) in Healthy School Children and Risk of Nephrolithiasis. J. Ren. Nutr. 2014, 24, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.N.; Fung, T.T.; Curhan, G.C. DASH-Style Diet Associates with Reduced Risk for Kidney Stones. J. Am. Soc. Nephrol. 2009, 20, 2253–2259. [Google Scholar] [CrossRef]
- Taylor, E.N.; Stampfer, M.J.; Mount, D.B.; Curhan, G.C. DASH-Style Diet and 24-Hour Urine Composition. Clin. J. Am. Soc. Nephrol. 2010, 5, 2315–2322. [Google Scholar] [CrossRef] [PubMed]
- Conte, A.; Pizá, P.; García-Raja, A.; Grases, F.; Costa-Bauza, A.; Prieto, R.M. Urinary lithogen risk test: Usefulness in the evaluation of renal lithiasis treatment using crystallization inhibitors (citrate and phytate). Arch. Esp. Urol. 1999, 52, 305–310. [Google Scholar]
- Guimerà, J.; Martínez, A.; Bauza, J.L.; Sanchís, P.; Pieras, E.; Grases, F. Effect of phytate on hypercalciuria secondary to bone resorption in patients with urinary stones: Pilot study. Urolithiasis 2022, 50, 685–690. [Google Scholar] [CrossRef]
- Grases, F.; Perelló, J.; Sanchis, P.; Isern, B.; Prieto, R.M.; Costa-Bauza, A.; Santiago, C.; Ferragut, M.L.; Frontera, G. Anticalculus effect of a triclosan mouthwash containing phytate: A double-blind, randomized, three-period crossover trial. J. Periodontal Res. 2009, 44, 616–621. [Google Scholar] [CrossRef]
- Kanis, J.A.; McCloskey, E.V.; Johansson, H.; Cooper, C.; Rizzoli, R.; Reginster, J.-Y.; on behalf of the Scientific Advisory Board of the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) and the Committee of Scientific Advisors of the International Osteoporosis Foundation (IOF). European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos. Int. 2013, 24, 23–57. [Google Scholar] [CrossRef]
- Kontogianni, M.D.; Melistas, L.; Yannakoulia, M.; Malagaris, I.; Panagiotakos, D.B.; Yiannakouris, N. Association between dietary patterns and indices of bone mass in a sample of Mediterranean women. Nutrition 2009, 25, 165–171. [Google Scholar] [CrossRef]
- Kitchin, B.; Morgan, S. Nutritional considerations in osteoporosis. Curr. Opin. Rheumatol. 2003, 15, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Drake, M.T.; Clarke, B.L.; Khosla, S. Bisphosphonates: Mechanism of Action and Role in Clinical Practice. Mayo Clin. Proc. Mayo Clin. 2008, 83, 1032–1045. [Google Scholar] [CrossRef]
- Arriero, M.D.M.; Ramis, J.M.; Perelló, J.; Monjo, M. Inositol Hexakisphosphate Inhibits Osteoclastogenesis on RAW 264.7 Cells and Human Primary Osteoclasts. PLoS ONE 2012, 7, e43187. [Google Scholar] [CrossRef]
- Fleisch, H.; Bisaz, S. Isolation from urine of pyrophosphate, a calcification inhibitor. Am. J. Physiol. Content 1962, 203, 671–675. [Google Scholar] [CrossRef]
- Fleish, H.; Neuman, W.F. Mechanisms of calcification: Role of collagen, polyphosphates, and phosphatase. Am. J. Physiol. Content 1961, 200, 1296–1300. [Google Scholar] [CrossRef] [PubMed]
- Fleisch, H.A.; Russell, R.G.G.; Bisaz, S.; Mühlbauer, R.C.; Williams, D.A. The Inhibitory Effect of Phosphonates on the Formation of Calcium Phosphate Crystals in vitro and on Aortic and Kidney Calcification in vivo. Eur. J. Clin. Investig. 1970, 1, 12–18. [Google Scholar] [CrossRef] [PubMed]
- Fleisch, H.; Graham, R.; Russell, G.; Francis, M.D. Diphosphonates Inhibit Hydroxyapatite Dissolution in vitro and Bone Resorption in Tissue Culture and in vivo. Science 1969, 165, 1262–1264. [Google Scholar] [CrossRef]
- Grases, F.; Sanchis, P.; Prieto, R.M.; Perelló, J.; López-González, Á.A. Effect of Tetracalcium Dimagnesium Phytate on Bone Characteristics in Ovariectomized Rats. J. Med. Food 2010, 13, 1301–1306. [Google Scholar] [CrossRef]
- Gonzalez, A.A.L.; Grases, F.; Mari, B.; Tomas-Salva, M.; Rodriguez, A. Urinary phytate concentration and risk of fracture determined by the FRAX index in a group of postmenopausal women. Turk. J. Med. Sci. 2019, 49, 458–463. [Google Scholar] [CrossRef] [PubMed]
- López-González, Á.A.; Grases, F.; Monroy, N.; Marí, B.; Vicente-Herrero, M.T.; Tur, F.; Perelló, J. Protective effect of myo-inositol hexaphosphate (phytate) on bone mass loss in postmenopausal women. Eur. J. Nutr. 2012, 52, 717–726. [Google Scholar] [CrossRef]
- López-González, Á.A.; Grases, F.; Roca, P.; Mari, B.; Vicente-Herrero, M.; Costa-Bauzá, A.; Sanchis, P.; Prieto, R.M.; Perelló, J.; Freitas, T.P.; et al. Phytate (myo-Inositol Hexaphosphate) and Risk Factors for Osteoporosis. J. Med. Food 2008, 11, 747–752. [Google Scholar] [CrossRef] [PubMed]
- López-González, Á.A.; Grases, F.; Marí, B.; Vicente-Herrero, M.T.; Costa-Bauzá, A.; Monroy, N. Influencia del consumo de fitato sobre la masa ósea en mujeres posmenopáusicas de Mallorca. Reumatol. Clínica 2011, 7, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Prieto, R.M.; Fiol, M.; Perello, J.; Estruch, R.; Ros, E.; Sanchis, P.; Grases, F. Effects of Mediterranean diets with low and high proportions of phytate-rich foods on the urinary phytate excretion. Eur. J. Nutr. 2010, 49, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Rivas, A.; Romero, A.; Mariscal-Arcas, M.; Monteagudo, C.; Feriche, B.; Lorenzo, M.L.; Olea, F. Mediterranean diet and bone mineral density in two age groups of women. Int. J. Food Sci. Nutr. 2012, 64, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Rey, J.; Roncero-Martín, R.; Rico-Martín, S.; Rey-Sánchez, P.; Pedrera-Zamorano, J.D.; Pedrera-Canal, M.; López-Espuela, F.; Lavado-García, J.M. Adherence to a Mediterranean Diet and Bone Mineral Density in Spanish Premenopausal Women. Nutrients 2019, 11, 555. [Google Scholar] [CrossRef] [PubMed]
- Calderon-Garcia, J.F.; Moran, J.M.; Roncero-Martin, R.; Rey-Sanchez, P.; Rodriguez-Velasco, F.J.; Pedrera-Zamorano, J.D. Dietary Habits, Nutrients and Bone Mass in Spanish Premenopausal Women: The Contribution of Fish to Better Bone Health. Nutrients 2012, 5, 10–22. [Google Scholar] [CrossRef]
- Larvie, D.; Armah, S. Estimated Phytate Intake Is Associated with Improved Cognitive Function in the Elderly, NHANES 2013–2014. Antioxidants 2021, 10, 1104. [Google Scholar] [CrossRef]
- Bruins, M.J.; Van Dael, P.; Eggersdorfer, M. The Role of Nutrients in Reducing the Risk for Noncommunicable Diseases during Aging. Nutrients 2019, 11, 85. [Google Scholar] [CrossRef]
- Li, S.; Sun, W.; Zhang, D. Association of Zinc, Iron, Copper, and Selenium Intakes with Low Cognitive Performance in Older Adults: A Cross-Sectional Study from National Health and Nutrition Examination Survey (NHANES). J. Alzheimer’s Dis. 2019, 72, 1145–1157. [Google Scholar] [CrossRef]
- Abdulwaliyu, I.; Arekemase, S.O.; Adudu, J.A.; Batari, M.L.; Egbule, M.N.; Okoduwa, S.I.R. Investigation of the medicinal significance of phytic acid as an indispensable anti-nutrient in diseases. Clin. Nutr. Exp. 2019, 28, 42–61. [Google Scholar] [CrossRef]
- Xu, Q.; Kanthasamy, A.G.; Reddy, M.B. Phytic Acid Protects against 6-Hydroxydopamine-Induced Dopaminergic Neuron Apoptosis in Normal and Iron Excess Conditions in a Cell Culture Model. Park. Dis. 2011, 2011, 431068. [Google Scholar] [CrossRef]
- Rahmati, B.; Khalili, M.; Hamoleh-Shalali, Z.; Roghani, M.; Baluchnejadmojarad, T. Phytic Acid Mitigates Motor Asymmetry in Male Rat with Unilateral 6-Hydroxydopamine Striatal Lesion. J. Basic Clin. Pathophysiol. 2015, 3, 25–28. [Google Scholar] [CrossRef]
- Abe, T.K.; Taniguchi, M. Identification of myo-inositol hexakisphosphate (IP6) as a β-secretase 1 (BACE1) inhibitory molecule in rice grain extract and digest. FEBS Open Bio 2014, 4, 162–167. [Google Scholar] [CrossRef]
- Grases, F.; Simonet, B.M.; Prieto, R.M.; March, J. Phytate levels in diverse rat tissues: Influence of dietary phytate. Br. J. Nutr. 2001, 86, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Anekonda, T.S.; Wadsworth, T.L.; Sabin, R.; Frahler, K.; Harris, C.; Petriko, B.; Ralle, M.; Woltjer, R.; Quinn, J.F. Phytic Acid as a Potential Treatment for Alzheimer’s Pathology: Evidence from Animal and in vitro Models. J. Alzheimer’s Dis. 2011, 23, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Cherian, L.; Wang, Y.; Fakuda, K.; Leurgans, S.; Aggarwal, N.; Morris, M. Mediterranean-Dash Intervention for Neurodegenerative Delay (MIND) Diet Slows Cognitive Decline After Stroke. J. Prev. Alzheimer’s Dis. 2019, 6, 267–273. [Google Scholar] [CrossRef]
- van den Brink, A.C.; Brouwer-Brolsma, E.M.; Berendsen, A.A.M.; Van De Rest, O. The Mediterranean, Dietary Approaches to Stop Hypertension (DASH), and Mediterranean-DASH Intervention for Neurodegenerative Delay (MIND) Diets Are Associated with Less Cognitive Decline and a Lower Risk of Alzheimer’s Disease—A Review. Adv. Nutr. 2019, 10, 1040–1065. [Google Scholar] [CrossRef]
- McCormick, B.; Caulfield, L.; Richard, S.; Pendergast, L.; Murray-Kolb, L.; MAL-ED Network Investigators. Nurturing Environments and Nutrient-Rich Diets May Improve Cognitive Development: Analysis of Cognitive Trajectories from Six to Sixty Months from the MAL-ED Study (OR10-01-19). Curr. Dev. Nutr. 2019, 3, nzz034–OR10. [Google Scholar] [CrossRef]
- American Diabetes Association Standards of Medical Care in Diabetes—2022 Abridged for Primary Care Providers. Clin. Diabetes 2022, 40, 10–38. [CrossRef]
- Visseren, F.L.J.; Mach, F.; Smulders, Y.M.; Carballo, D.; Koskinas, K.C.; Bäck, M.; Benetos, A.; Biffi, A.; Boavida, J.-M.; Capodanno, D.; et al. ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 2021, 42, 3227–3337. [Google Scholar] [CrossRef]
- Zavodnik, I.B.; Piasecka, A.; Szosland, K.; Bryszewska, M. Human red blood cell membrane potential and fluidity in glucose solutions. Scand. J. Clin. Lab. Investig. 1997, 57, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Zheng, F.; Lu, W.; Jia, C.; Li, H.; Wang, Z.; Jia, W. Relationships between glucose excursion and the activation of oxidative stress in patients with newly diagnosed type 2 diabetes or impaired glucose regulation. Endocrine 2010, 37, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Peppa, M.; Vlassara, H. Advanced glycation end products and diabetic complications: A General overview. Hormones 2005, 4, 28–37. [Google Scholar] [CrossRef]
- Negre-Salvayre, A.; Salvayre, R.; Augé, N.; Pamplona, R.; Portero-Otín, M. Hyperglycemia and Glycation in Diabetic Complications. Antioxid. Redox Signal. 2009, 11, 3071–3109. [Google Scholar] [CrossRef]
- Sözmen, E.Y.; Sözmen, B.; Delen, Y.; Onat, T. Catalase/Superoxide Dismutase (SOD) and Catalase/Paraoxonase (PON) Ratios May Implicate Poor Glycemic Control. Arch. Med. Res. 2001, 32, 283–287. [Google Scholar] [CrossRef]
- Dilworth, L.; Omoruyi, F.; Simon, O.; Morrison, E.S.A.; Asemota, H. The effect of phytic acid on the levels of blood glucose and some enzymes of carbohydrate and lipid metabolism. West Indian Med. J. 2005, 54, 102–106. [Google Scholar] [CrossRef]
- Berridge, M.J.; Irvine, R.F. Inositol trisphosphate, a novel second messenger in cellular signal transduction. Nature 1984, 312, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, S.S.; Cheryan, M. Effects of Phytic Acid, Divalent Cations, and Their Interactions on ?-Amylase Activity. J. Food Sci. 1984, 49, 516–519. [Google Scholar] [CrossRef]
- Omoruyi, F.; Stennett, D.; Foster, S.; Dilworth, L. New Frontiers for the Use of IP6 and Inositol Combination in Treating Diabetes Mellitus: A Review. Molecules 2020, 25, 1720. [Google Scholar] [CrossRef] [PubMed]
- Yuangklang, C.; Wensing, T.; Lemmens, A.G.; Jittakhot, S.; Beynen, A.C. Effect of sodium phytate supplementation on fat digestion and cholesterol metabolism in female rats. J. Anim. Physiol. Anim. Nutr. 2005, 89, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Luthra, M.; Balasubramanian, D. Nonenzymatic glycation alters protein structure and stability. A study of two eye lens crystallins. J. Biol. Chem. 1993, 268, 18119–18127. [Google Scholar] [CrossRef]
- Liu, J.; Ru, Q.; Ding, Y. Glycation a promising method for food protein modification: Physicochemical properties and structure, a review. Food Res. Int. 2012, 49, 170–183. [Google Scholar] [CrossRef]
- Adrover, M.; Mariño, L.; Sanchis, P.; Pauwels, K.; Kraan, Y.; Lebrun, P.; Vilanova, B.; Muñoz, F.; Broersen, K.; Donoso, J. Mechanistic Insights in Glycation-Induced Protein Aggregation. Biomacromolecules 2014, 15, 3449–3462. [Google Scholar] [CrossRef]
- Mariño, L.; Maya-Aguirre, C.A.; Pauwels, K.; Vilanova, B.; Ortega-Castro, J.; Frau, J.; Donoso, J.; Adrover, M. Glycation of Lysozyme by Glycolaldehyde Provides New Mechanistic Insights in Diabetes-Related Protein Aggregation. ACS Chem. Biol. 2017, 12, 1152–1162. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, L.M.; Lages, A.; Gomes, R.A.; Neves, H.; Família, C.; Coelho, A.V.; Quintas, A. Insulin glycation by methylglyoxal results in native-like aggregation and inhibition of fibril formation. BMC Biochem. 2011, 12, 41. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, L.M.; Gomes, R.A.; Yang, D.; Dennison, S.R.; Família, C.; Lages, A.; Coelho, A.V.; Murphy, R.M.; Phoenix, D.A.; Quintas, A. Insights into the molecular mechanism of protein native-like aggregation upon glycation. Biochim. Biophys. Acta (BBA) Proteins Proteom. 2013, 1834, 1010–1022. [Google Scholar] [CrossRef] [PubMed]
- Weiss, G.; Goodnough, L.T. Anemia of chronic disease. N. Engl. J. Med. 2005, 352, 1011–1023. [Google Scholar] [CrossRef] [PubMed]
- Ikenaga, T.; Kakumoto, K.; Kohda, N.; Yamamoto, T. Effect of Inositol Hexaphosphate (IP6) on Serum Uric Acid in Hyperuricemic Subjects: A Randomized, Double-Blind, Placebo-Controlled, Crossover Study. Plant Foods Hum. Nutr. 2019, 74, 316–321. [Google Scholar] [CrossRef]
- Onomi, S.; Okazaki, Y.; Katayama, T. Effect of Dietary Level of Phytic Acid on Hepatic and Serum Lipid Status in Rats Fed a High-sucrose Diet. Biosci. Biotechnol. Biochem. 2004, 68, 1379–1381. [Google Scholar] [CrossRef]
- Lee, S.-H.; Park, H.-J.; Chun, H.-K.; Cho, S.-Y.; Cho, S.-M.; Lillehoj, H.S. Dietary phytic acid lowers the blood glucose level in diabetic KK mice. Nutr. Res. 2006, 26, 474–479. [Google Scholar] [CrossRef]
- Kuppusamy, A.; Muthusamy, U.; Thirumalaisamy, S.A.; Varadharajan, S.; Ramasamy, K.; Ramanathan, S. In vitro (α-glucosidase and α-amylase inhibition) and in vivo antidiabetic property of phytic acid (IP6) in streptozotocin- nicotinamide-induced type 2 diabetes mellitus (NIDDM) in rats. J. Complement. Integr. Med. 2011, 8, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Omoruyi, F.O.; Budiaman, A.; Eng, Y.; Olumese, F.E.; Hoesel, J.L.; Ejilemele, A.; Okorodudu, A.O. The Potential Benefits and Adverse Effects of Phytic Acid Supplement in Streptozotocin-Induced Diabetic Rats. Adv. Pharmacol. Pharm. Sci. 2013, 2013, 172494. [Google Scholar] [CrossRef] [PubMed]
- Foster, S.R.; Omoruyi, F.O.; Bustamante, J.; Lindo, R.L.A.; Dilworth, L.L. The effect of combined inositol hexakisphosphate and inositol supplement in streptozotocin-induced type 2 diabetic rats. Int. J. Exp. Pathol. 2016, 97, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Panagiotakos, D.B.; Tzima, N.; Pitsavos, C.; Chrysohoou, C.; Zampelas, A.; Toussoulis, D.; Stefanadis, C. The Association between Adherence to the Mediterranean Diet and Fasting Indices of Glucose Homoeostasis: The ATTICA Study. J. Am. Coll. Nutr. 2007, 26, 32–38. [Google Scholar] [CrossRef]
- Martín-Peláez, S.; Fito, M.; Castaner, O. Mediterranean Diet Effects on Type 2 Diabetes Prevention, Disease Progression, and Related Mechanisms. A Review. Nutrients 2020, 12, 2236. [Google Scholar] [CrossRef]
- Chandalia, M.; Garg, A.; Lutjohann, D.; Von Bergmann, K.; Grundy, S.M.; Brinkley, L.J. Beneficial Effects of High Dietary Fiber Intake in Patients with Type 2 Diabetes Mellitus. N. Engl. J. Med. 2000, 342, 1392–1398. [Google Scholar] [CrossRef]
- Venn, B.J.; Mann, J.I. Cereal grains, legumes and diabetes. Eur. J. Clin. Nutr. 2004, 58, 1443–1461. [Google Scholar] [CrossRef]
- Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvadó, J.; Ruiz-Gutiérrez, V.; Covas, M.I.; Fiol, M.; Gómez-Gracia, E.; López-Sabater, M.C.; Vinyoles, E.; et al. Effects of a Mediterranean-Style Diet on Cardiovascular Risk Factors: A randomized trial. Ann. Intern. Med. 2006, 145, 1–11. [Google Scholar] [CrossRef]
- Toobert, D.J.; Glasgow, R.E.; Strycker, L.A.; Barrera, M.; Radcliffe, J.L.; Wander, R.C.; Bagdade, J.D. Biologic and Quality-of-Life Outcomes From the Mediterranean Lifestyle Program. Diabetes Care 2003, 26, 2288–2293. [Google Scholar] [CrossRef]
- Elhayany, A.; Lustman, A.; Abel, R.; Attal-Singer, J.; Vinker, S. A low carbohydrate Mediterranean diet improves cardiovascular risk factors and diabetes control among overweight patients with type 2 diabetes mellitus: A 1-year prospective randomized intervention study. Diabetes Obes. Metab. 2010, 12, 204–209. [Google Scholar] [CrossRef]
- Mirmiran, P.; Hosseini, S.; Hosseinpour-Niazi, S.; Azizi, F. Legume consumption increase adiponectin concentrations among type 2 diabetic patients: A randomized crossover clinical trial. Endocrinol. Diabetes Y Nutr. 2018, 66, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Communication DIABETES MELLITUS|Endocrinología, Diabetes y Nutrición|Endocrinología, Diabetes y Nutrición. Available online: https://www.elsevier.es/en-revista-endocrinologia-diabetes-nutricion-13-congresos-63-congreso-nacional-sociedad-espanola-148-sesion-diabetes-mellitus-7167-comunicacion-el-consumo-diario-de-fitato-86211 (accessed on 3 December 2022).
- Avgerinos, K.I.; Spyrou, N.; Mantzoros, C.S.; Dalamaga, M. Obesity and cancer risk: Emerging biological mechanisms and perspectives. Metabolism 2019, 92, 121–135. [Google Scholar] [CrossRef]
- Proietti, S.; Pasta, V.; Cucina, A.; Aragona, C.; Palombi, E.; Vucenik, I.; Bizzarri, M. Inositol hexaphosphate (InsP6) as an effective topical treatment for patients receiving adjuvant chemotherapy after breast surgery. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 43–50. [Google Scholar]
- Vucenik, I.; Shamsuddin, A.M. Cancer Inhibition by Inositol Hexaphosphate (IP6) and Inositol: From Laboratory to Clinic. J. Nutr. 2003, 133, 3778S–3784S. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Ferry, S.; Matsuda, M.; Yoshida, H.; Hirata, M. Inositol hexakisphosphate blocks tumor cell growth by activating apoptotic machinery as well as by inhibiting the Akt/NFkappaB-mediated cell survival pathway. Carcinogenesis 2002, 23, 2031–2041. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, C.; Dhanalakshmi, S.; Singh, R.P.; Agarwal, R. Inositol Hexaphosphate Inhibits Growth and Induces G1 Arrest and Apoptotic Death of Androgen-Dependent Human Prostate Carcinoma LNCaP Cells. Neoplasia 2004, 6, 646–659. [Google Scholar] [CrossRef]
- Sakamoto, K.; Venkatraman, G.; Shamsuddin, A.M. Growth inhibition and differentiation of HT-29 cells in vitro by inositol hexaphosphate (phytic acid). Carcinogenesis 1993, 14, 1815–1819. [Google Scholar] [CrossRef]
- Yang, G.Y.; Shamsuddin, A.M. IP6-induced growth inhibition and differentiation of HT-29 human colon cancer cells: Involvement of intracellular inositol phosphates. Anticancer Res. 1995, 15, 2479–2487. [Google Scholar]
- Shamsuddin, A.M.; Baten, A.; Lalwani, N.D. Effects of inositol hexaphosphate on growth and differentiation in K-562 erythroleukemia cell line. Cancer Lett. 1992, 64, 195–202. [Google Scholar] [CrossRef]
- Deliliers, G.L.; Servida, F.; Fracchiolla, N.S.; Ricci, C.; Borsotti, C.; Colombo, G.; Soligo, D. Effect of inositol hexaphosphate (IP6) on human normal and leukaemic haematopoietic cells. Br. J. Haematol. 2002, 117, 577–587. [Google Scholar] [CrossRef] [PubMed]
- Shamsuddin, A.M.; Yang, G.Y.; Vucenik, I. Novel anti-cancer functions of IP6: Growth inhibition and differentiation of human mammary cancer cell lines in vitro. Anticancer Res. 1996, 16, 3287–3292. [Google Scholar] [PubMed]
- Zi, X.; Singh, R.P.; Agarwal, R. Impairment of erbB1 receptor and fluid-phase endocytosis and associated mitogenic signaling by inositol hexaphosphate in human prostate carcinoma DU145 cells. Carcinogenesis 2000, 21, 2225–2235. [Google Scholar] [CrossRef] [PubMed]
- Shamsuddin, A.M.; Yang, G.-Y. Inositol hexaphosphate inhibits growth and induces differentiation of PC-3 human prostate cancer cells. Carcinogenesis 1995, 16, 1975–1979. [Google Scholar] [CrossRef]
- Singh, R.P.; Agarwal, C.; Agarwal, R. Inositol hexaphosphate inhibits growth, and induces G1 arrest and apoptotic death of prostate carcinoma DU145 cells: Modulation of CDKI-CDK-cyclin and pRb-related protein-E2F complexes. Carcinogenesis 2003, 24, 555–563. [Google Scholar] [CrossRef]
- Vucenik, I.; Tantivejkul, K.; Zhang, Z.S.; Cole, K.E.; Saied, I.; Shamsuddin, A.M. IP6 in treatment of liver cancer. I. IP6 inhibits growth and reverses transformed phenotype in HepG2 human liver cancer cell line. Anticancer Res. 1999, 18, 4083–4090. [Google Scholar]
- Vucenik, I.; Tomazic, V.J.; Fabian, D.; Shamsuddin, A.M. Antitumor activity of phytic acid (inositol hexaphosphate) in murine transplanted and metastatic fibrosarcoma, a pilot study. Cancer Lett. 1992, 65, 9–13. [Google Scholar] [CrossRef]
- Vucenik, I.; Kalebic, T.; Tantivejkul, K.; Shamsuddin, A.M. Novel anticancer function of inositol hexaphosphate: Inhibition of human rhabdomyosarcoma in vitro and in vivo. Anticancer Res. 1998, 18, 1377–1384. [Google Scholar]
- Markiewicz, L.; Ogrodowczyk, A.; Wiczkowski, W.; Wróblewska, B. Phytate and Butyrate Differently Influence the Proliferation, Apoptosis and Survival Pathways in Human Cancer and Healthy Colonocytes. Nutrients 2021, 13, 1887. [Google Scholar] [CrossRef]
- Shamsuddin, A.M.; Elsayed, A.M.; Ullah, A. Suppression of large intestinal cancer in F344 rats by inositol hexaphosphate. Carcinogenesis 1988, 9, 577–580. [Google Scholar] [CrossRef]
- Shamsuddin, A.M.; Ullah, A.; Chakravarthy, A.K. Inositol and inositol hexaphosphate suppress cell proliferation and tumor formation in CD-1 mice. Carcinogenesis 1989, 10, 1461–1463. [Google Scholar] [CrossRef]
- Shamsuddin, A.M.; Ullah, A. Inositol hexaphosphate inhibits large intestinal cancer in F344 rats 5 months after induction by azoxymethane. Carcinogenesis 1989, 10, 625–626. [Google Scholar] [CrossRef]
- Nelson, R.L.; Yoo, S.J.; Tanure, J.C.; Andrianopoulos, G.; Misumi, A. The effect of iron on experimental colorectal carcinogenesis. Anticancer Res. 1989, 9, 1477–1482. [Google Scholar] [PubMed]
- Pretlow, T.P.; O’Riordan, M.A.; Somich, G.A.; Amini, S.B.; Pretlow, T.G. Aberrant crypts correlate with tumor incidence in F344 rats treated with azoxymethane and phytate. Carcinogenesis 1992, 13, 1509–1512. [Google Scholar] [CrossRef]
- Shivapurkar, N.; Tang, Z.; Frost, A.; Alabaster, O. A rapid dual organ rat carcinogenesis bioassay for evaluating the chemoprevention of breast and colon cancer. Cancer Lett. 1996, 100, 169–179. [Google Scholar] [CrossRef] [PubMed]
- Challa, A.; Rao, D.R.; Reddy, B.S. Interactive suppression of aberrant crypt foci induced by azoxymethane in rat colon by phytic acid and green tea. Carcinogenesis 1997, 18, 2023–2026. [Google Scholar] [CrossRef] [PubMed]
- Jenab, M.; Thompson, L.U. Phytic acid in wheat bran affects colon morphology, cell differentiation and apoptosis. Carcinogenesis 2000, 21, 1547–1552. [Google Scholar] [CrossRef]
- Thompson, L.U.; Zhang, L. Phytic acid and minerals: Effect on early markers of risk for mammary and colon carcinogenesis. Carcinogenesis 1991, 12, 2041–2045. [Google Scholar] [CrossRef]
- Vucenik, I.; Zhang, Z.S.; Shamsuddin, A.M. IP6 in treatment of liver cancer. II. Intra-tumoral injection of IP6 regresses pre-existing human liver cancer xenotransplanted in nude mice. Anticancer Res. 1999, 18, 4091–4096. [Google Scholar]
- Hirose, M.; Ozaki, K.; Takaba, K.; Fukushima, S.; Shirai, T.; Ito, N.; Nobuyuki, I. Modifying effects of the naturally occurring antioxidants γ-oryzanol, phytic acid, tannic acid and n-tritriacontane-16,18-dione in a rat wide-spectrum organ carcinogenesis model. Carcinogenesis 1991, 12, 1917–1921. [Google Scholar] [CrossRef]
- Estensen, R.D.; Wattenberg, L.W. Studies of chemopreventive effects of myo-inositol on benzo(a)pyrene-induced neoplasia of the lung and forestomach of female A/J mice. Carcinogenesis 1993, 14, 1975–1977. [Google Scholar] [CrossRef] [PubMed]
- Wattenberg, L. Chalcones, myo-inositol and other novel inhibitors of pulmonary carcinogenesis. J. Cell. Biochem. 1995, 59, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Vucenik, I.; Sakamoto, K.; Bansal, M.; Shamsuddin, A.M. Inhibition of rat mammary carcinogenesis by inositol hexaphosphate (phytic acid). A pilot study. Cancer Lett. 1993, 75, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Vucenik, I.; Yang, G.-Y.; Shamsuddin, A.M. Inositol hexaphosphate and inositol inhibit DMBA-induced rat mammary cancer. Carcinogenesis 1995, 16, 1055–1058. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Nakatsuru, Y.; Zarkovic, M.; Shamsuddin, A.M. Inhibition of skin cancer by IP6 in vivo: Initiation-promotion model. Anticancer Res. 2000, 19, 3749–3752. [Google Scholar]
- Jagadeesh, S.; Banerjee, P.P. Inositol hexaphosphate represses telomerase activity and translocates TERT from the nucleus in mouse and human prostate cancer cells via the deactivation of Akt and PKCα. Biochem. Biophys. Res. Commun. 2006, 349, 1361–1367. [Google Scholar] [CrossRef]
- Vucenik, I.; Yang, G.; Shamsuddin, A.M. Comparison of pure inositol hexaphosphate and high-bran diet in the prevention of DMBA-induced rat mammary carcinogenesis. Nutr. Cancer 1997, 28, 7–13. [Google Scholar] [CrossRef]
- Mentella, M.C.; Scaldaferri, F.; Ricci, C.; Gasbarrini, A.; Miggiano, G.A.D. Cancer and Mediterranean Diet: A Review. Nutrients 2019, 11, 2059. [Google Scholar] [CrossRef]
- Minihan, A.K.; Patel, A.V.; Flanders, W.D.; Sauer, A.G.; Jemal, A.; Islami, F. Proportion of Cancer Cases Attributable to Physical Inactivity by US State, 2013–2016. Med. Sci. Sports Exerc. 2021, 54, 417–423. [Google Scholar] [CrossRef]
- Lauby-Secretan, B.; Scoccianti, C.; Loomis, D.; Grosse, Y.; Bianchini, F.; Straif, K. Body Fatness and Cancer—Viewpoint of the IARC Working Group. N. Engl. J. Med. 2016, 375, 794–798. [Google Scholar] [CrossRef]
- Key, T.J.; Bradbury, K.E.; Perez-Cornago, A.; Sinha, R.; Tsilidis, K.K.; Tsugane, S. Diet, nutrition, and cancer risk: What do we know and what is the way forward? BMJ 2020, 368, m511. [Google Scholar] [CrossRef]
- Papadimitriou, N.; Markozannes, G.; Kanellopoulou, A.; Critselis, E.; Alhardan, S.; Karafousia, V.; Kasimis, J.C.; Katsaraki, C.; Papadopoulou, A.; Zografou, M.; et al. An umbrella review of the evidence associating diet and cancer risk at 11 anatomical sites. Nat. Commun. 2021, 12, 4579. [Google Scholar] [CrossRef]
- Buckland, G.; Travier, N.; Cottet, V.; González, C.A.; Lujan-Barroso, L.; Agudo, A.; Trichopoulou, A.; Lagiou, P.; Trichopoulos, D.; Peeters, P.; et al. Adherence to the mediterranean diet and risk of breast cancer in the European prospective investigation into cancer and nutrition cohort study. Int. J. Cancer 2012, 132, 2918–2927. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean Diet and Survival in a Greek Population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Castelló, A.; Researchers, O.B.O.M.-S.; Amiano, P.; de Larrea, N.F.; Martín, V.; Alonso, M.H.; Castaño-Vinyals, G.; Pérez-Gómez, B.; Olmedo-Requena, R.; Guevara, M.; et al. Low adherence to the western and high adherence to the mediterranean dietary patterns could prevent colorectal cancer. Eur. J. Nutr. 2018, 58, 1495–1505. [Google Scholar] [CrossRef] [PubMed]
- Schneider, L.; Su, L.J.; Arab, L.; Bensen, J.T.; Farnan, L.; Fontham, E.T.H.; Song, L.; Hussey, J.; Merchant, A.T.; Mohler, J.L.; et al. Dietary patterns based on the Mediterranean diet and DASH diet are inversely associated with high aggressive prostate cancer in PCaP. Ann. Epidemiol. 2019, 29, 16–22.e1. [Google Scholar] [CrossRef] [PubMed]
- Kenfield, S.A.; Dupre, N.; Richman, E.L.; Stampfer, M.J.; Chan, J.M.; Giovannucci, E.L. Mediterranean Diet and Prostate Cancer Risk and Mortality in the Health Professionals Follow-up Study. Eur. Urol. 2014, 65, 887–894. [Google Scholar] [CrossRef]
- Praud, D.; Bertuccio, P.; Bosetti, C.; Turati, F.; Ferraroni, M.; La Vecchia, C. Adherence to the Mediterranean diet and gastric cancer risk in Italy. Int. J. Cancer 2013, 134, 2935–2941. [Google Scholar] [CrossRef]
- Bravi, F.; Spei, M.-E.; Polesel, J.; Di Maso, M.; Montella, M.; Ferraroni, M.; Serraino, D.; Libra, M.; Negri, E.; La Vecchia, C.; et al. Mediterranean Diet and Bladder Cancer Risk in Italy. Nutrients 2018, 10, 1061. [Google Scholar] [CrossRef]
- Filomeno, M.; Bosetti, C.; Garavello, W.; Levi, F.; Galeone, C.; Negri, E.; La Vecchia, C. The role of a Mediterranean diet on the risk of oral and pharyngeal cancer. Br. J. Cancer 2014, 111, 981–986. [Google Scholar] [CrossRef] [PubMed]
- Rosato, V.; Polesel, J.; Bosetti, C.; Serraino, D.; Negri, E.; La Vecchia, C. Population Attributable Risk for Pancreatic Cancer in Northern Italy. Pancreas 2015, 44, 216–220. [Google Scholar] [CrossRef]
- Anic, G.M.; Park, Y.; Subar, A.F.; Schap, T.E.; Reedy, J. Index-based dietary patterns and risk of lung cancer in the NIH–AARP diet and health study. Eur. J. Clin. Nutr. 2015, 70, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Onstad, M.A.; Schmandt, R.E.; Lu, K.H. Addressing the Role of Obesity in Endometrial Cancer Risk, Prevention, and Treatment. J. Clin. Oncol. 2016, 34, 4225–4230. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, J.M. The food matrix: Implications in processing, nutrition and health. Crit. Rev. Food Sci. Nutr. 2018, 59, 3612–3629. [Google Scholar] [CrossRef]
- Graf, E.; Eaton, J.W. Dietary suppression of colonic cancer. Fiber or phytate? Cancer 1985, 56, 717–718. [Google Scholar] [CrossRef]
- Wiśniewski, K.; Jozwik, M.; Wojtkiewicz, J. Cancer Prevention by Natural Products Introduced into the Diet—Selected Cyclitols. Int. J. Mol. Sci. 2020, 21, 8988. [Google Scholar] [CrossRef] [PubMed]
- Lam, S.; McWilliams, A.; Leriche, J.; MacAulay, C.; Wattenberg, L.; Szabo, E. A Phase I Study of myo-Inositol for Lung Cancer Chemoprevention. Cancer Epidemiol. Biomark. Prev. 2006, 15, 1526–1531. [Google Scholar] [CrossRef]
- Lam, S.; Mandrekar, S.J.; Gesthalter, Y.; Ziegler, K.L.A.; Seisler, D.K.; Midthun, D.E.; Mao, J.T.; Aubry, M.C.; McWilliams, A.; Sin, D.D.; et al. A Randomized Phase IIb Trial of myo-Inositol in Smokers with Bronchial Dysplasia. Cancer Prev. Res. 2016, 9, 906–914. [Google Scholar] [CrossRef]
- Bačić, I.; Družijanić, N.; Karlo, R.; Škifić, I.; Jagić, S. Efficacy of IP6 + inositol in the treatment of breast cancer patients receiving chemotherapy: Prospective, randomized, pilot clinical study. J. Exp. Clin. Cancer Res. 2010, 29, 12–15. [Google Scholar] [CrossRef]
- Baten, A.; Ullah, A.; Tomazic, V.J.; Shamsuddin, A.M. Inositol-phosphate-induced enhancement of natural killer cell activity correlates with tumor suppression. Carcinogenesis 1989, 10, 1595–1598. [Google Scholar] [CrossRef]
- Zhang, Z.; Song, Y.; Wang, X.-L. Inositol hexaphosphate-induced enhancement of natural killer cell activity correlates with suppression of colon carcinogenesis in rats. World J. Gastroenterol. 2005, 11, 5044–5046. [Google Scholar] [CrossRef] [PubMed]
- Khurana, S.; Baldeo, C.; Joseph, R.W. Inositol hexaphosphate plus inositol induced complete remission in stage IV melanoma: A case report. Melanoma Res. 2019, 29, 322–324. [Google Scholar] [CrossRef]
- Nassar, R.I.; Nassar, M. Antimicrobial effect of phytic acid on Enterococcus faecalis. Int. Arab. J. Antimicrob. Agents 2016, 6, 1–7. [Google Scholar] [CrossRef]
- Kim, N.H.; Rhee, M.S. Phytic Acid and Sodium Chloride Show Marked Synergistic Bactericidal Effects against Nonadapted and Acid-Adapted Escherichia coli O157:H7 Strains. Appl. Environ. Microbiol. 2016, 82, 1040–1049. [Google Scholar] [CrossRef]
- Yadav, A.K.; Sirohi, P.; Saraswat, S.; Rani, M.; Singh, M.P.; Srivastava, S.; Singh, N.K. Inhibitory Mechanism on Combination of Phytic Acid with Methanolic Seed Extract of Syzygium cumini and Sodium Chloride over Bacillus subtilis. Curr. Microbiol. 2018, 75, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Otake, T.; Shimonaka, H.; Kanai, M.; Miyano, K.; Ueba, N.; Kunita, N.; Kurimura, T. Inhibitory Effect of Inositol Hexasulfate and Inositol Hexaphosphoric Acid (Phytic acid) on the Proliferation of the Human Immunodeficiency Virus (HIV) in vitro. Kansenshogaku Zasshi 1989, 63, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Rungratanawanich, W.; Qu, Y.; Wang, X.; Essa, M.M.; Song, B.-J. Advanced glycation end products (AGEs) and other adducts in aging-related diseases and alcohol-mediated tissue injury. Exp. Mol. Med. 2021, 53, 168–188. [Google Scholar] [CrossRef] [PubMed]
- Stodolak, B.; Starzyńska, A.; Czyszczoń, M.; Żyła, K. The effect of phytic acid on oxidative stability of raw and cooked meat. Food Chem. 2007, 101, 1041–1045. [Google Scholar] [CrossRef]
- Trela, B.C. Iron Stabilization with Phytic Acid in Model Wine and Wine. Am. J. Enol. Vitic. 2010, 61, 253–259. [Google Scholar] [CrossRef]
- Xie, Y.; Luo, H.; Duan, J.; Hong, C.; Ma, P.; Li, G.; Zhang, T.; Wu, T.; Ji, G. Phytic acid enhances the oral absorption of isorhamnetin, quercetin, and kaempferol in total flavones of Hippophae rhamnoides L. Fitoterapia 2014, 93, 216–225. [Google Scholar] [CrossRef]
- Nordbö, H.; Rölla, G. Desorption of Salivary Proteins from Hydroxyapatite by Phytic Acid and Glycerophosphate and the Plaque-Inhibiting Effect of the Two Compounds In Vivo. J. Dent. Res. 1972, 51, 800–802. [Google Scholar] [CrossRef] [PubMed]
- Nassar, R.; Nassar, M.; Vianna, M.E.; Naidoo, N.; Alqutami, F.; Kaklamanos, E.G.; Senok, A.; Williams, D. Antimicrobial Activity of Phytic Acid: An Emerging Agent in Endodontics. Front. Cell. Infect. Microbiol. 2021, 11, 753649. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Guo, S. Phytic acid and its interactions: Contributions to protein functionality, food processing, and safety. Compr. Rev. Food Sci. Food Saf. 2021, 20, 2081–2105. [Google Scholar] [CrossRef]
- Brouns, F. Phytic Acid and Whole Grains for Health Controversy. Nutrients 2021, 14, 25. [Google Scholar] [CrossRef] [PubMed]
- López-Moreno, M.; Garcés-Rimón, M.; Miguel, M. Antinutrients: Lectins, goitrogens, phytates and oxalates, friends or foe? J. Funct. Foods 2022, 89, 104938. [Google Scholar] [CrossRef]
- Reddy, M.B.; Hurrell, R.F.; Juillerat, M.A.; Cook, J.D. The influence of different protein sources on phytate inhibition of nonheme-iron absorption in humans. Am. J. Clin. Nutr. 1996, 63, 203–207. [Google Scholar] [CrossRef]
- Hurrell, R.; Egli, I. Iron bioavailability and dietary reference values. Am. J. Clin. Nutr. 2010, 91, 1461S–1467S. [Google Scholar] [CrossRef]
- Castro-Alba, V.; Lazarte, C.E.; Bergenståhl, B.; Granfeldt, Y. Phytate, iron, zinc, and calcium content of common Bolivian foods and their estimated mineral bioavailability. Food Sci. Nutr. 2019, 7, 2854–2865. [Google Scholar] [CrossRef]
- Fredlund, K.; Isaksson, M.; Rossander-Hulthén, L.; Almgren, A.; Sandberg, A.-S. Absorption of zinc and retention of calcium: Dose-dependent inhibition by phytate. J. Trace Elements Med. Biol. 2006, 20, 49–57. [Google Scholar] [CrossRef]
- Miller, L.V.; Hambidge, K.M.; Krebs, N.F. Zinc Absorption Is Not Related to Dietary Phytate Intake in Infants and Young Children Based on Modeling Combined Data from Multiple Studies. J. Nutr. 2015, 145, 1763–1769. [Google Scholar] [CrossRef]
- Hoppe, M.; Ross, A.B.; Svelander, C.; Sandberg, A.-S.; Hulthén, L. Low-phytate wholegrain bread instead of high-phytate wholegrain bread in a total diet context did not improve iron status of healthy Swedish females: A 12-week, randomized, parallel-design intervention study. Eur. J. Nutr. 2018, 58, 853–864. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, C.; Viteri, F.E.; Lönnerdal, B.; Young, K.A.; Raboy, V.; Brown, K.H. Effect of genetically modified, low-phytic acid maize on absorption of iron from tortillas. Am. J. Clin. Nutr. 1998, 68, 1123–1127. [Google Scholar] [CrossRef] [PubMed]
- Mendoza, C.; Viteri, F.E.; Lönnerdal, B.; Raboy, V.; Young, K.A.; Brown, K.H. Absorption of iron from unmodified maize and genetically altered, low-phytate maize fortified with ferrous sulfate or sodium iron EDTA. Am. J. Clin. Nutr. 2001, 73, 80–85. [Google Scholar] [CrossRef]
- Grases, F.; Simonet, B.M.; Prieto, R.M.; March, J.G. Dietary phytate and mineral bioavailability. J. Trace Elements Med. Biol. 2001, 15, 221–228. [Google Scholar] [CrossRef]
- Cullumbine, H.; Basnayake, V.; Wickramanayake, T.W. Mineral Metabolism on Rice Diets. Br. J. Nutr. 1950, 4, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.R.P.; Fox, F.W.; Irving, J.T. Studies in human mineral metabolism: 1. The effect of bread rich in phytate phosphorus on the metabolism of certain mineral salts with special reference to calcium. Biochem. J. 1948, 42, 452–462. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Costa-Bauzá, A.; Berga, F.; Rodríguez, A.; Gomila, R.; Martorell, G.; Martínez-Cignoni, M. Evaluation of inositol phosphates in urine after topical administration of myo-inositol hexaphosphate to female Wistar rats. Life Sci. 2018, 192, 33–37. [Google Scholar] [CrossRef]
- Grases, F.; Simonet, B.; March, J.; Prieto, R. Inositol hexakisphosphate in urine: The relationship between oral intake and urinary excretion. Br. J. Urol. 2000, 85, 138–142. [Google Scholar] [CrossRef]
- Grases, F.; Simonet, B.M.; Vucenik, I.; Prieto, R.M.; Costa-Bauzá, A.; March, J.G.; Shamsuddin, A.M. Absorption and excretion of orally administered inositol hexaphosphate (IP6or phytate) in humans. BioFactors 2001, 15, 53–61. [Google Scholar] [CrossRef]
- Schlemmer, U.; Frølich, W.; Prieto, R.M.; Grases, F. Phytate in foods and significance for humans: Food sources, intake, processing, bioavailability, protective role and analysis. Mol. Nutr. Food Res. 2009, 53, S330–S375. [Google Scholar] [CrossRef]
- Grases, F.; Simonet, B.M.; Prieto, R.M.; March, J.G. Variation of InsP4,InsP5 and InsP6 levels in tissues and biological fluids depending on dietary phytate. J. Nutr. Biochem. 2001, 12, 595–601. [Google Scholar] [CrossRef] [PubMed]



| Year of Publication and Authors | Type of Clinical Trial | Studied Population | Phytate Formulation and Dose Administrated | Duration of the Intervention | Route of Phytate Administration | Outcomes | References |
|---|---|---|---|---|---|---|---|
| Perelló et al., 2018 | Double-blind, randomized, placebo-controlled Phase I study | Healthy and hemodialysis patients | In healthy volunteers the dose was 0.5 mg/kg and ascended to 12.5 mg/kg. In hemodialysis patients was 9 mg/kg | 28 days | Parenteral | Safety and tolerability, and lack of significant dialysability of IV SNF472. | [27] |
| Raggi et al., 2020 | Double-blind, placebo-controlled Phase 2b trial | Hemodialysis patients | Patients were randomized 1:1:1 to SNF472 300 mg (n = 92), SNF472 600 mg (n = 91), or placebo (n = 91) by infusion in the hemodialysis lines thrice weekly during hemodialysis sessions | 52 weeks | Parenteral | SNF472 significantly reduced the progression of coronary artery calcium and aortic valve calcification in patients receiving hemodialysis | [29] |
| Sanchis et al., 2018 | Single-center, randomized, crossover, open-label study. | Patients living with type 2 diabetes | Diet plan with InsP6 supplementation (1 capsule of 380 mg of calcium–magnesium InsP6 thrice daily) or the same diet plan without IP6 supplementation. | 48 weeks | Oral | InsP6 supplements for 3 months significantly reduced levels of circulating AGEs and HbA1c | [31] |
| Estruch et al., 2018 | Multicenter trial randomly assigned (in a 1:1:1 ratio) | High cardiovascular risk patients without cardiovascular disease | A Mediterranean diet supplemented with extra-virgin olive oil, a Mediterranean diet supplemented with mixed nuts, or a control diet (advice to reduce dietary fat. | Median follow-up of 4.8 years | Oral | Mediterranean diet supplemented with nuts (high phytate content) reduces the incidence of major cardiovascular events was lower compared to a reduced-fat diet. | [33] |
| Conte et al., 1999 | Single center randomized trial. | Active calcium oxalate stone-formers. | Potassium citrate, phytate-rich dietary complement or placebo. | 15 days | Oral | Reduction in the lithogen urinary risk | [48] |
| Guimerà et al., 2022 | Single center randomized trial. | Patients with hypercalciuria (> 250 mg/24 h) and osteopenia or osteoporosis in the femur and/or spine (determined by densitometry) | 1 capsule of 380 mg of calcium–magnesium InsP6 versus placebo. | 12 weeks. | Oral | Phytate group had significantly lower calciuria and ß-Crosslaps levels. | [49] |
| Grases et al., 2009 | Randomized, double-blind, three-period crossover clinical | Healthy dental plaque-forming volunteers | Control period (no mouthwash treat- ment), a placebo period (mouthwash with 0.001% zinc) and a phytate-treatment period (mouthwash with 0.001% zinc and 0.1% phytate). | 3 weeks each period for 9 weeks total. | Topical mouthwash | High efficacy exhibited by phytate in reducing dental calculus formation. | [50] |
| Ikenaga et al., 2019 | Randomized, double-blind placebo-controlled crossover study. | Asymptomatic hyperuricemic subjects | Placebo or InsP6 drinks (600 mg twice daily). | 2 weeks | Oral | Significantly reduced uric acid levels in comparation with placebo. | [100] |
| Estruch et al., 2006 | Substudy of a multicenter, randomized, primary prevention trial of cardiovascular disease. | Asymptomatic at high cardiovascular risk. | A Mediterranean diet supplemented with extra-virgin olive oil, a Mediterranean diet supplemented with mixed nuts, or a control diet (advice to reduce dietary fat. | 3 months | Oral | Improved fasting blood glucose, reduced blood pressure and increased HDL/cholesterol ratios in the high phytate diet in comparison with a low-fat diet. | [110] |
| Toobert et al., 2003 | Randomized clinical trial | Postmenopausal women living with type 2 diabetes | Mediterranean Lifestyle Program (Mediterranean low-saturated fat diet, stress management training, exercise, group support, and smoking cessation) versus with usual care. | 6 months duration | Oral | Reduction in HbA1c by the high phytate diet. | [111] |
| Elhayany et al., 2010 | Prospective randomized clinical trial | Patients living with overweight and type 2 diabetes | A low carbohydrate Mediterranean, a traditional Mediterranean and the 2003 American Diabetic Association diet were compared. | 12-month period | Oral | Both high phytate diets showed better weight loss effects and better reduction in HbA1c compared to other diet | [112] |
| Mirmiran et al., 2019 | Randomized crossover clinical trial | Patients living with type 2 diabetes | Compared a legume-free diet or a legume-based diet. | 8 weeks | Oral | Legumes (which are high in phytate) increased serum adiponectin concentrations | [113] |
| Lam et al., 2006 | A phase I, open-label, multiple dose, dose-escalation clinical study | Smokers | A dose escalation study ranging from 12 to 30 g/d of myo-inositol for a month | 1 month | Oral | Myo-Inositol in a daily dose of 18 g for 3 months is safe and well tolerated. | [178] |
| Lam et al., 2016 | A randomized, double blind, placebo-controlled phase IIb study | Smokers with ≥1 site of dysplasia identified by autofluorescence bronchoscopy-directed biopsy | Placebo or myo-inositol 9 g once a day for 2 weeks, and then twice a day. | 6 months | Oral | Reduction in progressive disease rates and IL-6 levels. | [169] |
| Bacic et al., 2010 | Prospective, randomized, pilot clinical study. | Patients with invasive ductal breast cancer who received polychemotherapy. | InsP6 + Inositol (6 g) in comparation with placebo (vitamin C) | 6 months | Oral | Significantly improved patients quality of life and protected patients from the reduction in the number of leukocytes and platelets. | [170] |
| Proietti et al., 2017 | Double-blind and randomized controlled trial | Patients with breast cancer 6 weeks after lumpectomy. | 4% topical skin formulation of InsP6 once a day versus placebo (gel containing hyaluronic acid). | 6 months | Topical skin | Topical InsP6 significantly improved quality of life and functional status reducing side effects compared to control group. Moreover, white blood cells and platelets count values where higher in the treated group. | [171] |
| Sanchis et al., 2022 | Randomized and crossover trial | Patients living with T2DM | 1 capsule of 380 mg of calcium–magnesium InsP6 thrice daily | 12 weeks | Oral | Significant decrease serum levels of HbA1c and increase adiponectin levels. | [114] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pujol, A.; Sanchis, P.; Grases, F.; Masmiquel, L. Phytate Intake, Health and Disease: “Let Thy Food Be Thy Medicine and Medicine Be Thy Food”. Antioxidants 2023, 12, 146. https://doi.org/10.3390/antiox12010146
Pujol A, Sanchis P, Grases F, Masmiquel L. Phytate Intake, Health and Disease: “Let Thy Food Be Thy Medicine and Medicine Be Thy Food”. Antioxidants. 2023; 12(1):146. https://doi.org/10.3390/antiox12010146
Chicago/Turabian StylePujol, Antelm, Pilar Sanchis, Felix Grases, and Luis Masmiquel. 2023. "Phytate Intake, Health and Disease: “Let Thy Food Be Thy Medicine and Medicine Be Thy Food”" Antioxidants 12, no. 1: 146. https://doi.org/10.3390/antiox12010146
APA StylePujol, A., Sanchis, P., Grases, F., & Masmiquel, L. (2023). Phytate Intake, Health and Disease: “Let Thy Food Be Thy Medicine and Medicine Be Thy Food”. Antioxidants, 12(1), 146. https://doi.org/10.3390/antiox12010146






