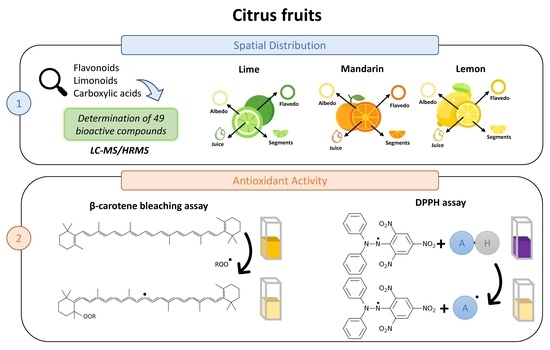
1. Introduction
Citrus fruits constitute a characteristic element of the Mediterranean diet and are extensively researched due to their bioactive composition and health benefits. Among the most known citrus fruits, lemons (Citrus limon), mandarins (Citrus reticulata), and Persian limes (Citrus latifolia) have been demonstrated to be outstanding sources of bioactive compounds such as ascorbic acid, limonoids, and flavonoids [
1].
Flavonoids are the most predominant phenolic compounds in citrus fruits and are the main sources of their flavor and coloration [
2]. These phytochemicals have been described as beneficial to human health and are used to prevent some diseases [
3] because of their anti-inflammatory, anti-cancer, or anti-obesity properties [
4,
5,
6,
7]. Citrus flavonoids’ phenolic activities, such as their free radical scavenging activity, pro-oxidant metal ion chelation, and ability to act as enzyme cofactors, explain their beneficial properties.
Limonoids have also received great attention because of their anti-inflammatory, anti-aging, anti-tumor, immunomodulatory, and antioxidant activity [
8]. Limonoids can be found as glucosides (water soluble) or aglycones (water insoluble), which contribute to the unflavored or bitter taste of citrus fruits. In a recent study, a total of 18 limonoid glucosides and 55 limonoid aglycones were reported in citrus fruits [
9]. The most representative limonoid glucoside is limonin-17-β-D-glucopyranoside, while the most abundant aglycones are limonin and nomilin [
10,
11]. The presence of limonoids has mainly been detected in essential oils obtained from citrus flavedo, and these oils have received special attention in previous years because of their high antimicrobial and preservative activity in various foods [
12,
13,
14].
Limonoids and flavonoids have been determined by different techniques, with the most extensive being liquid chromatography coupled with diode array detection (LC–DAD) [
15]; however, this technique requires the use of analytical standards for identification. For this reason, in the few last years, the use of liquid chromatography coupled with high-resolution tandem mass spectrometry (LC–MS/HRMS) has been increasing. Although this technique also requires standards for confirmation, it provides better identification capacity by comparing MS2 spectra with those included in databases. Most citrus fruits have been characterized by this technique, for example, limes, oranges, tangerines, lemons, grapefruit, etc. [
16,
17,
18,
19,
20,
21,
22].
The antioxidant properties of citrus limonoids and flavonoids have been previously described using various techniques, including 2,2-diphenyl-1-picrylhydrazyl (DPPH) antioxidant capacity and β-carotene blenching assay studies. The DPPH method relies on the strength of a 30 min radical scavenging time, which allows DPPH to react efficiently, even with weak antioxidants providing high sensitivity, while β-carotene test allows both lipophilic and hydrophilic samples to be analyzed with high reproducibility [
23]. The combination of these two assays has been used for the determination of the antioxidant activity of limonoid and flavonoid extracts from different tissues [
17,
24]. Specifically, grapefruit seeds [
25],
Rutaceae species peel [
26], albedo and flavedo from
pompia fruits [
16], kinnow peel [
27], and Spanish clementine fresh pulp [
28] have been analyzed for the presence of these high valuable compounds using both antioxidant determination assays. Moreover, changes in the limonin and nomilin concentrations in different tissues from pummelo and mandarin varieties have been studied during fruit growth and maturation [
29]. However, although citrus antioxidant activity has been widely studied, to the best of our knowledge, the spatial distribution of the antioxidant capacity in the different tissues (flavedo, albedo, pulp segments, and juice) of citrus fruits and the synergistic interaction between limonoids and flavonoids have not been studied in detail. For this reason, the aim of this research was to characterize the limonoid and flavonoid fractions of three Citrus fruits, namely, lemons, limes, and mandarins. For this purpose, three main tissues (albedo, flavedo, and pulp segment) and their respective juices were studied. A highly selective and sensitive LC–MS/HRMS approach was used for the characterization of these citrus tissues. In addition, their antioxidant activity was evaluated through the DPPH radical scavenging activity and β-carotene bleaching assays to ascertain the properties of limonoids and flavonoids and elucidate any synergistic interactions.
2. Materials and Methods
2.1. Samples
Limes, mandarins, and lemons were purchased in two local markets in Cordoba and Murcia (La Corredera and Veronicas local markets, Spain) in October 2022. Fruits were washed and processed to separate the albedo, flavedo, pulp segments, and juice. The solid samples were lyophilized and subsequently ground. The powder was stored in the dark at −20 °C.
2.2. Reagents
Ethanol (EtOH), methanol (MeOH), ethyl acetate (EA), acetone, chloroform, acetonitrile (ACN), and formic acid were purchased from Scharlab (Barcelona, Spain). n-Hexane was obtained from Sigma−Aldrich (St. Louis, MO, USA). All solvents were LC- or MS-grade. Simplicity® UV Millipore equipment (Burlington, MA, USA) was used to generate purified Milli-Q-water. Syringaldehyde (SA), used as external standard, was acquired from Sigma−Aldrich. For antioxidant assays, we used 1,1-diphenyl-2-picrylhydrazyl (DPPH), linoleic acid, Tween-20, and β-carotene standards obtained from Sigma-Aldrich. Silica gel 60 (0.06–0.2 mm particle size) from Merck (Darmstadt, Germany) was used for the fractionation of extracts.
2.3. Instrumentation and Software
The analytical equipment consisted of a 1200 series LC system from Agilent Technologies (Palo Alto, CA, USA) coupled with a dual electrospray ionization source and a high-resolution Agilent 6540 quadrupole–time of flight detector QTOF (LC–MS/HRMS).
Agilent MassHunter Workstation (version B6.00 Profinder, Agilent Technologies, Santa Clara, CA, USA) was used for data acquisition. MassHunter Qualitative v7.0 software and MetaboAnalyst 5.0 (
https://metaboanalyst.ca/, accessed on 9 January 2023) were used for targeted extraction of MS/MS information, metabolomics data analysis, and interpretation.
2.4. Metabolites Extraction
Albedo, flavedo, and pulp segments of mandarins, limes, and lemons were crushed and homogenized after lyophilization. A total of 1 g of each tissue was extracted in 10 mL of n-hexane while stirring at 1000 rpm for 30 min at 30 °C to remove the lipidic fraction. The n-hexane extract was discarded, and the solids were recovered and dried under gentle nitrogen flow prior to the triplicate extraction of bioactive compounds with 10 mL of (50:25:25 v/v/v) EA/acetone/EtOH for 30 min at 30 °C. The three fractions were mixed, evaporated under vacuum, and then reconstituted in 1 mL of MeOH containing SA at 1 µg/mL. Six extracts were obtained from each tissue (three extracts for triplicate analysis of the antioxidant activity and three extracts for fractionation).
Lyophilized juice samples of the three fruits were solubilized in water at 150 mg of total solids/mL. A total of 10 mL of reconstituted juice was extracted with 5 mL of EA for 10 min at 30 °C. The EA fraction was dried under vacuum and reconstituted in 1 mL of MeOH with SA at 1 µg/mL.
2.5. LC–MS/HRMS Analysis
Chromatographic separation of the extracts was performed by using a Zorbax Eclipse Plus C18 chromatographic column (1.8 μm particle size, 150 × 3.0 mm i.d., Agilent Technologies). The injection volume was 2 μL, and the mobile phases were composed of 0.1% of formic acid in both deionized water (phase A) and acetonitrile (phase B). Flow rate was constant at 0.25 mL/min. Chromatographic gradient was programmed as follows: 4% phase B was selected as initial composition; then, from min 0 to 1, mobile phase B was increased to 25%; from min 1 to 6, phase B was changed to 40%; from min 6 to 8, phase B was modified to 60%; and from min 8 to 10, mobile phase B was increased from 60 to 100%. The latter composition was maintained for 12 min to guarantee the elution of metabolites and the sterilization of the column. A post-time of 13 min was set to equilibrate the initial conditions for the next analysis.
The dual electrospray ionization source (ESI) was operated in both positive and negative ionization modes. ESI parameters were as follows: nebulizer gas at 50 psi and drying gas flow rate and temperature at 10 L/min and 325 °C, respectively. The capillary voltage was set at 3500 V, while the fragmentor, skimmer, and octapole voltages were 130, 65, and 750 V, respectively. Data were acquired in centroid mode in the extended dynamic range (2 GHz). Full scan was carried out at 6 spectra/s within the m/z range of 60–1200, with subsequent activation of the three most intense precursor ions (allowed charge: single or double) by MS/MS using a collision energy of 20 eV and 40 eV at 3 spectra/s within the m/z range of 30–1200. An active exclusion window was programmed for 0.75 min to avoid repetitive fragmentation of the most intense precursor ions, thus increasing the detection coverage. To assure the desired mass accuracy of recorded ions, continuous internal calibration was performed during analyses with the use of signals at m/z 121.0509 (protonated purine) and m/z 922.0098 (protonated hexakis (1H, 1H, 3H-tetrafluoropropoxy) phosphazene or HP-921) in the positive ionization mode and m/z of 112.9856 (trifluoroacetic acid anion) and m/z 1033.9881 (HP-921) in the negative ionization mode. A quality control sample (QC) obtained by mixing aliquots of extracts was used to ensure the reproducibility and robustness of the methodology and chemometric results.
2.6. Data processing and Statistical Analysis
MassHunter Profinder (version B10.00; Agilent Technologies, Santa Clara, CA, USA) was used to process the data obtained by LC–MS/HRMS. Treatment of the raw data file started with the extraction of potential molecular features (MFs) by the applicable algorithm included in the software. The recursive extraction algorithm considered all ions exceeding 5000 counts as cut-off value. Additionally, the isotopic distribution used to determine whether a molecular feature is valid should be defined by two or more ions (with a peak spacing tolerance of m/z 0.0025 plus 10.0 ppm in mass accuracy). Apart from [M + H]+ and [M−H]− ions, adduct formation in the positive (Na+) and negative ionization (HCOO−, Cl−) modes and neutral loss by dehydration were included to identify features corresponding to the same potential metabolite. Thus, ions with identical elution profiles and related m/z values (representing different adducts or isotopes of the same compound) were extracted as entities characterized by their retention time (RT), the intensity at the apex chromatographic peak, and accurate mass. Then, a recursion step assured correct integration of the entities in all analyses.
Identification was achieved following the methodology previously described by Ledesma-Escobar et al. [
17]. After extraction and alignment of all MFs, the software MassHunter Qualitative v.7.0 was used for targeted extraction of MS2 information associated with the monitored MFs in the whole dataset. This information was used for tentative identification of metabolites by searching the MoNA-MassBank of North America (
https://mona.fiehnlab.ucdavis.edu/spectra/search, accessed on 13 December 2022) database and others developed by the research group. Additionally, some compounds were confirmed according to both their MS2 information and retention time by using commercially available standards. Finally, the compounds that were not found in the databases, or whose commercial standards were unavailable, were identified via an analysis of neutral mass losses combined with the characteristic fragmentation patterns of their derivatives confirmed by commercial standards.
Quantitative analysis was carried out in relative terms by preparing calibration models for limonin, nomilin, hesperidin, and quercetin as standard compounds (determination coefficients > 0.98; calibration range between 50 ng/mL and 20 μg/mL). Syringaldehyde (1 μg/mL) was used as external standard. Flavonoid glucosides and aglycones were quantitated with models prepared with hesperidin or quercetin, respectively, while limonoids were determined using their corresponding aglycone standards.
2.7. Silica Gel Column Fractionation of Crude Extracts
Crude extracts (1 mL) were fractioned by being passed through a normal-phase silica gel (5 g) column. The solid phase was conditioned with hexane and the metabolites were eluted with 2 mL of EA (fraction 1), 2 mL of 1:1 (v/v) EtOH/acetone (fraction 2), and 2 mL of 1:1 (v/v) EtOH/H2O (fraction 3). Each fraction was evaporated under vacuum and dissolved with 1 mL of MeOH. The solutions were stored at −80 °C in darkness until use.
2.8. Antioxidant Assays
2.8.1. DPPH Radical Scavenging Activity
DPPH radical scavenging activity was evaluated by mixing 100 µL of sample (either crude extract or fraction) with 900 µL of 20 mg/L DPPH solution in MeOH. The mixture was stirred for 30 min at room temperature and the absorbance was measured at 517 nm. Reference solution was prepared following the same procedure but using MeOH instead of sample. The antiradical scavenging potential was expressed as the percentage of decoloration of DPPH solution according to the following equation:
where:
As, absorbance of the sample;
AR, absorbance of the reference solution.
2.8.2. β-Carotene Bleaching Assay
A stock solution for this test was prepared as follows: 500 µL of Tween-20 was dissolved in 50 mL of water previously saturated with O2 by air bubbling and mixed with 25 µL of linoleic acid and 500 µL of 1 mg/mL β-carotene solution in chloroform. The mixture was subjected to rotary evaporator under reduced pressure at 30 °C to remove chloroform. Then, the mixture was shaken vigorously until a crystalline emulsion was obtained (approximately 5 min) with the characteristic orange color of β-carotene. Stock solution was immediately used after preparation.
For the measurement of antioxidant activity, 100 µL of the crude extract or fraction was added to 900 µL of the β-carotene/linoleic acid stock solution and stirred for 2 h at 50 °C. The absorbance of the samples was measured at 470 nm immediately after preparation (time 0 min) and after 2 h (time 120 min). A control sample was prepared with MeOH instead of sample. The blank was composed of water, Tween-20, and linoleic acid at the same proportions as those used in the stock solution. The antioxidant activity was calculated as the ability to inhibit the discoloration of β-carotene using the following equation:
where:
As, Absorbance of the sample;
Ac, absorbance of control; 0 and 120 refer to time (min).
4. Discussion
In this report, the spatial distribution and antioxidant activity of bioactive compounds in limes, lemons, and mandarin tissues were studied. We observed that flavonoids were predominant in the flavedo and albedo of the three fruits (97.1% and 67.8%, respectively, in limes; 97.0% and 92.5% in mandarins; and 92.4% and 56.9% in lemons). However, hesperidin, diosmin, and eriocitrin were the most concentrated flavonoids in limes; hesperidin and naringin were predominant in mandarins; and apigenin and diosmetin derivatives and hesperidin were highly concentrated in lemons. The presence of these functional ingredients has been previously described in other citrus limon varieties but not distributed in the different tissues of the fruit [
16]. Similarly, limonin derivatives were the most concentrated limonoids in limes and mandarins, while nomilin derivatives were the most abundant in lemons.
The highest proportion of carboxylic acids was found in lemons, followed by mandarins and limes, with citric acid being the most concentrated in all cases, followed by malic and ascorbic acids. However, it is worth noting that lemons were the richest fruit in terms of carboxylic acids (
Figure 1), especially ascorbic acid, which was found at a twofold higher concentration in lemons than in limes and mandarins. The presence of the identified carboxylic acids along with others was examined in lemons prior to this study and thus confirms this statement [
17].
The antioxidant activity and radical scavenging capacity of citrus fruits have been widely discussed in the literature, but most studies have targeted two main sources: juice and industrial residue extracts [
24]. Generally, citrus fruit’s antioxidant activity is mainly attributed to ascorbic acid, phenols, and, to a lesser extent, other minor compounds such as limonoids [
24]. Measuring the antioxidant activity of complex mixtures such as citrus juices or extracts is not an easy task since this act is affected by diverse aspects such as the structural [
30], pH [
31], or synergistic effects precipitated by different chemical families [
32,
33]. In this study, we evaluated the antioxidant activity in extracts from different citrus fruits to discern the contribution of the studied compounds to bioactivity. For this purpose, we used the β-carotene bleaching assay and the radical scavenging potential test (DPPH assay).
The β-carotene assay, which accounted for the composition of the extracts, showed that limonoids are crucial for antioxidant capacity given that this class of metabolites is particularly concentrated in mandarin juice and in the extracts from flavedo, albedo, and segments of lemons. Our DPPH assay revealed that despite their flavonoid content, carboxylic acids are believed to influence antioxidant capacity. Altunkaya et al. [
31] demonstrated that the antioxidant activity of lettuce extracts also depended on the pH and level of synergism with added phenols. Hence, the content of carboxylic acids alters the total antioxidant activity of extracts.
Finally, the purification of the extracts was carried out using a column packed with silica defining three fractions (less, mid, and polar). Concerning the limonoid fraction (L), we observed that the antioxidant activity was like that of the extract, with only an approximate decrease of 20% for all samples. This result reinforces the hypothesis that limonoids play a key role in reducing the formation of hydroperoxides from linoleic acid and thus lipidic peroxidation. In addition, the antioxidant activity was generally lower in the juices compared to the extracts from fruit tissues. On the contrary, the radical scavenging activity in juice was reduced by more than 50% compared to the extract. In this case, juices demonstrated higher capacity than the extracts from fruit tissues. These results agree with those observed by Yu et al. [
25], who showed that limonin had reduced radical scavenging capacity. On the other hand, the fractions rich in flavonoids (F) presented an antioxidant activity around 30% lower than that measured in the extracts, with the highest levels observed in lemon flavedo and mandarin and lime segments. Complementarily, the radical scavenging potential of this fraction was only reduced by around 10%. The maximum DPPH activity was found in flavedo from limes and mandarins and in lemon albedo, while minimum values were measured in juices. Finally, the carboxylic acids fraction (A) was particularly affected with respect to antioxidant capacity as determined in both assays. Thus, juice provided better values than those found in fruit tissues except for lime and lemon segments, which presented an antioxidant capacity similar to that obtained from juice.
5. Conclusions
This study revealed that the spatial distribution of bioactive compounds is constant in the three citrus fruits. Thus, flavonoids are mostly located in the citrus peel (in both the flavedo and albedo); on the contrary, carboxylic acids are mainly found in segments and, therefore, are extracted in the juice. Regarding limonoids, they are quantitatively distributed in the albedo and segments and, consequently, are also extracted in the juice. Additionally, the composition of bioactive compounds allows for the discrimination of both citrus tissues and fruits. Our determination of these fruits’ antioxidant properties demonstrated that limonoid content can be correlated with the antioxidant activity measured by the β-carotene bleaching assay, for which there is a synergistic effect caused by other families. However, the radical scavenging activity was explained by flavonoids and ascorbic acid. Finally, these results do not enable us to draw plausible conclusions about the contributions of individual compounds to the bioactive properties of the complex extracts. However, the fractionation of the extracts provided information about the antioxidant capacity of the three main chemical families.