Evaluation of Additive Neuroprotective Effect of Combination Therapy for Parkinson’s Disease Using In Vitro Models
Abstract
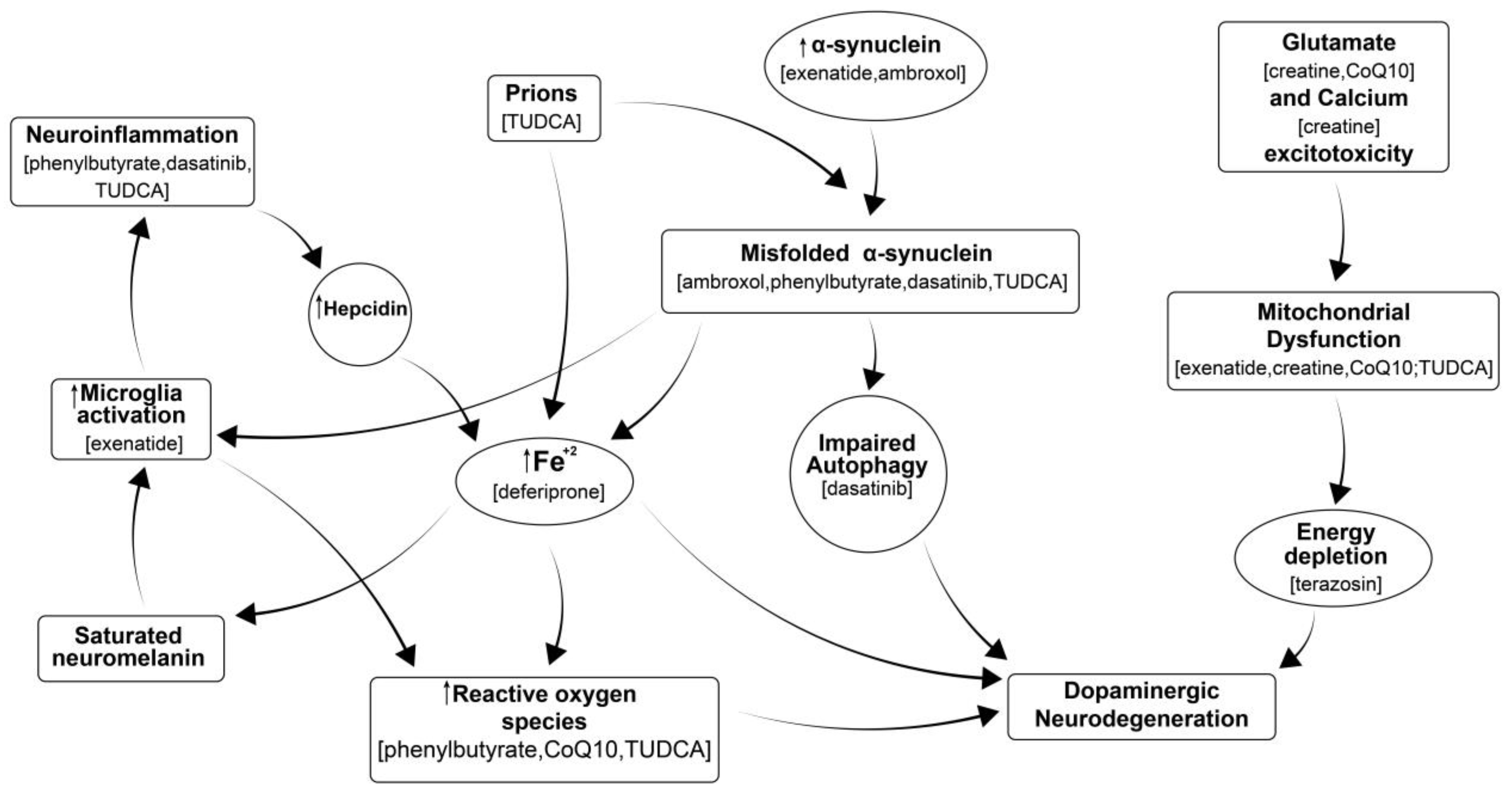
:1. Introduction
2. Materials and Methods
2.1. Study Drugs
2.2. Model 1. Wild-Type Human Dopaminergic iPSC Line Treated with 1-Methyl-4-Phenylpyridinium (MPP+)
Assay Endpoints and Image Analysis
2.3. Model 2. α-Synuclein Triplication Human Cell Line
2.4. Model 3. Human iPSC-Derived Idiopathic PD Cell Line
2.5. Dasatinib-Containing Combination Testing in iPD TD16 Cell Line
2.6. PBA/TUDCA/EXD Combo Testing in iPD TD16 Cell Line
2.7. Image Analysis
2.8. Model 4. Human Microglia Cell Line
2.9. Statistical Analysis
3. Results
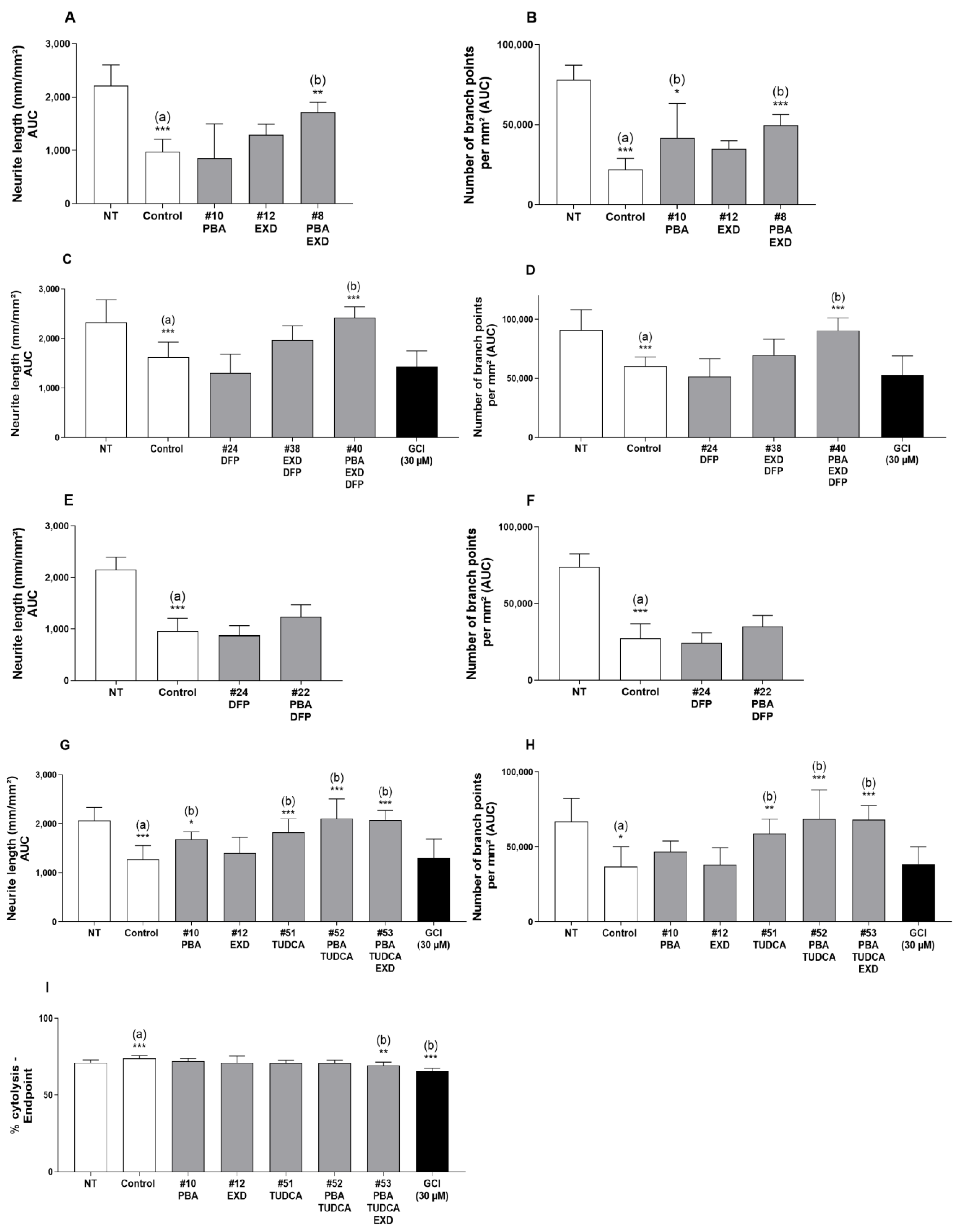
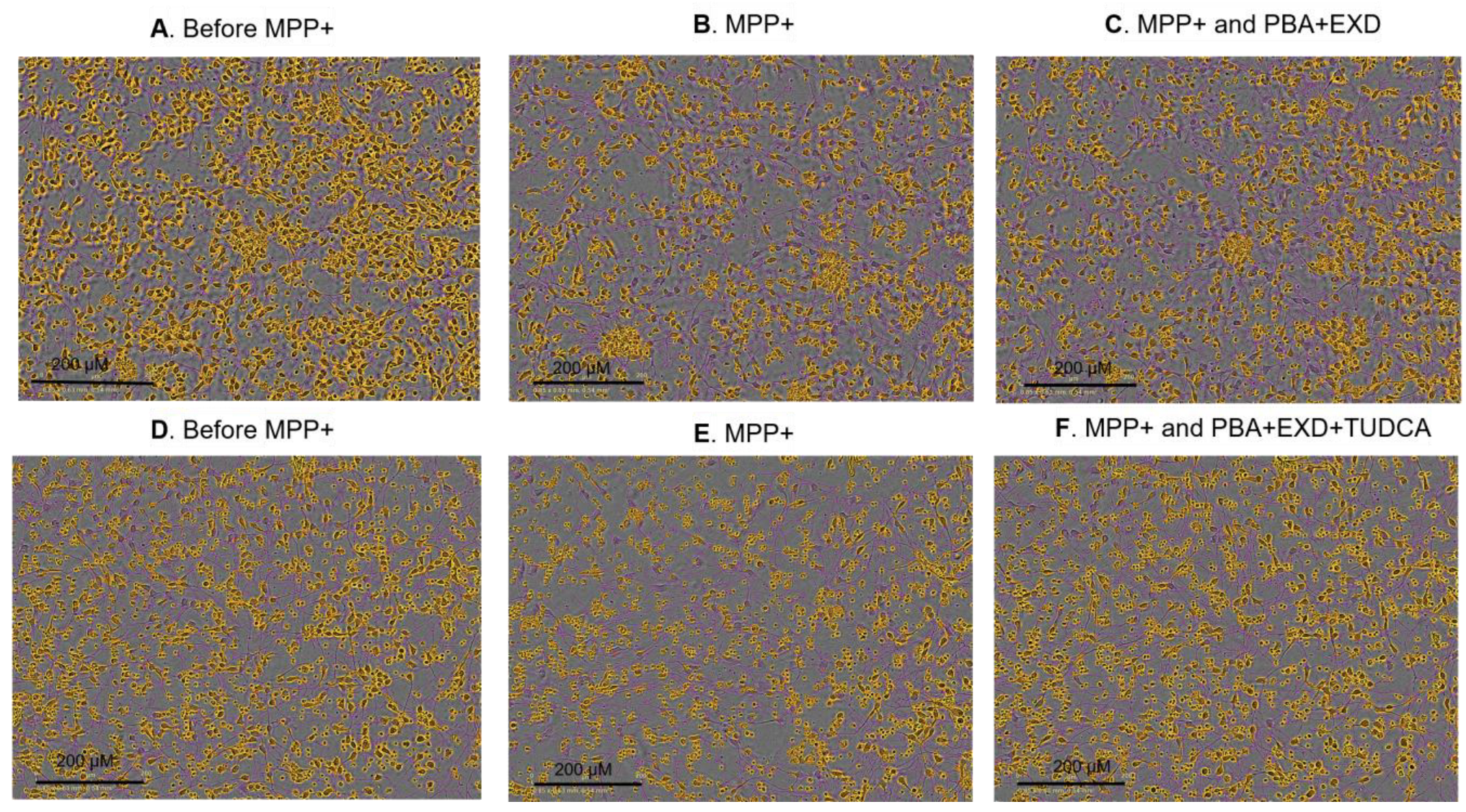
3.1. Model 1. Effect of Experimental Conditions on MPP+-Induced Toxicity in Human Dopaminergic iPSCs
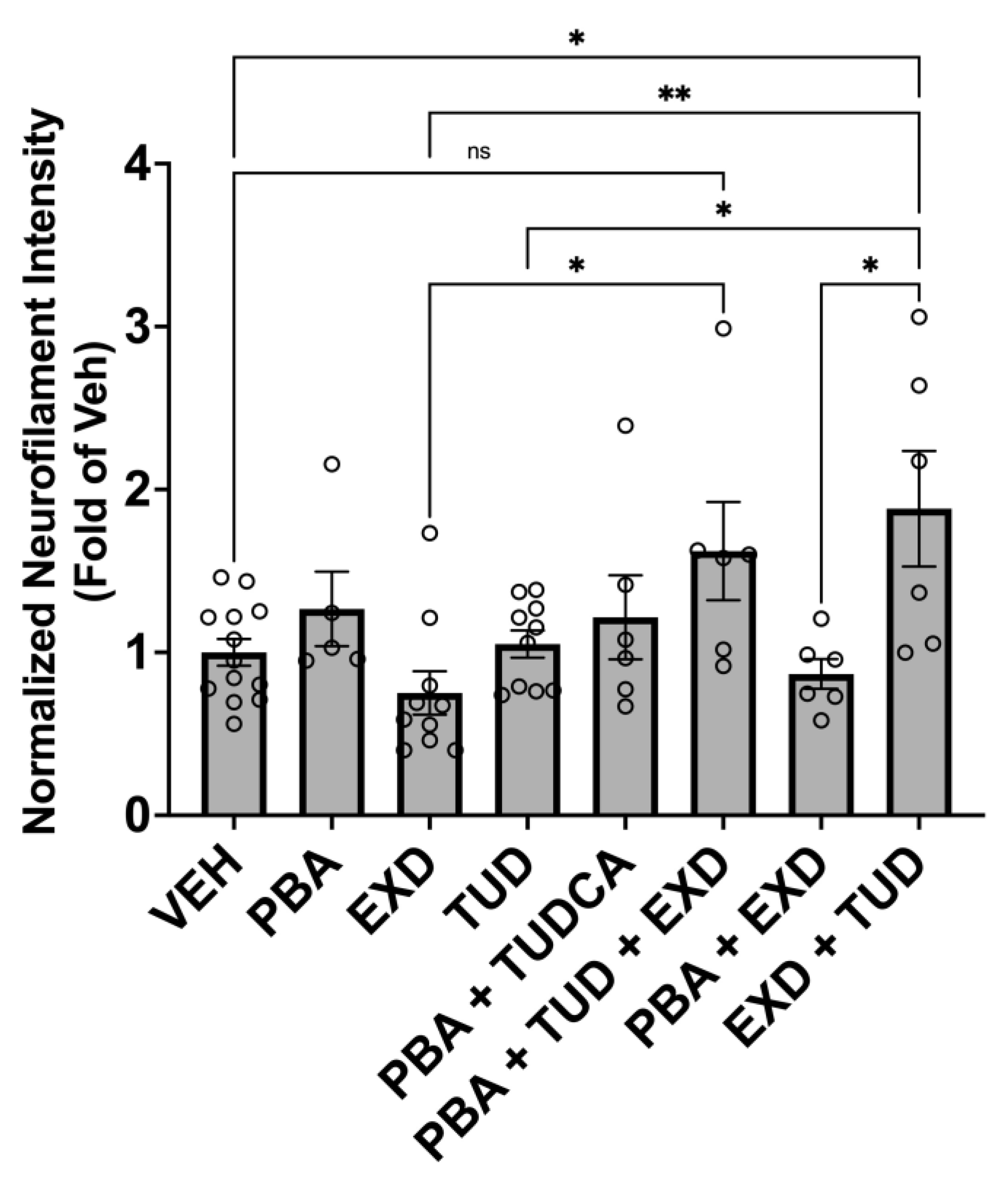
3.2. Model 2. Effect of Experimental Conditions on α-Synuclein Triplication Cell Line
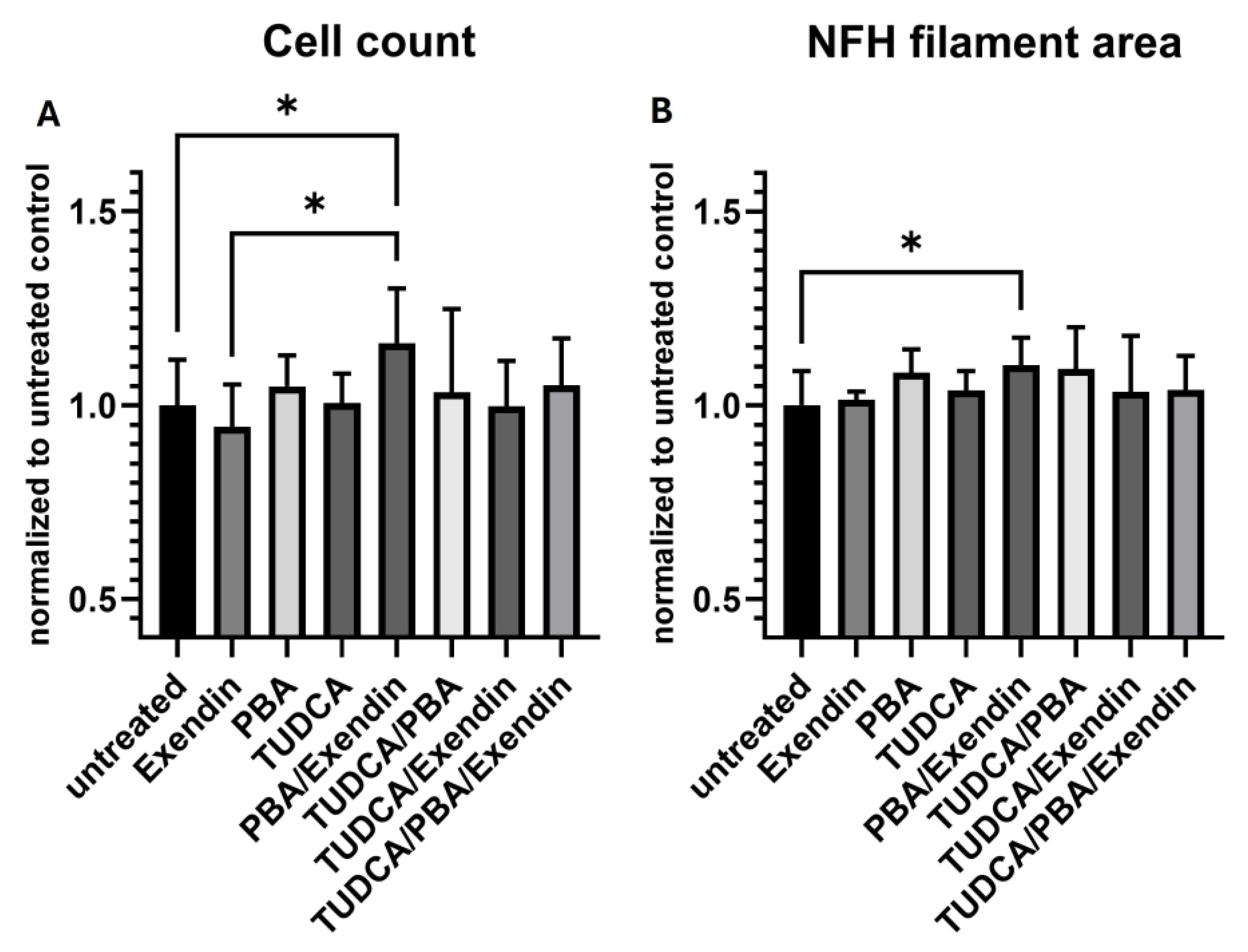
3.3. Model 3. Effect of Experimental Conditions on TD16 Idiopathic PD Cell Lines
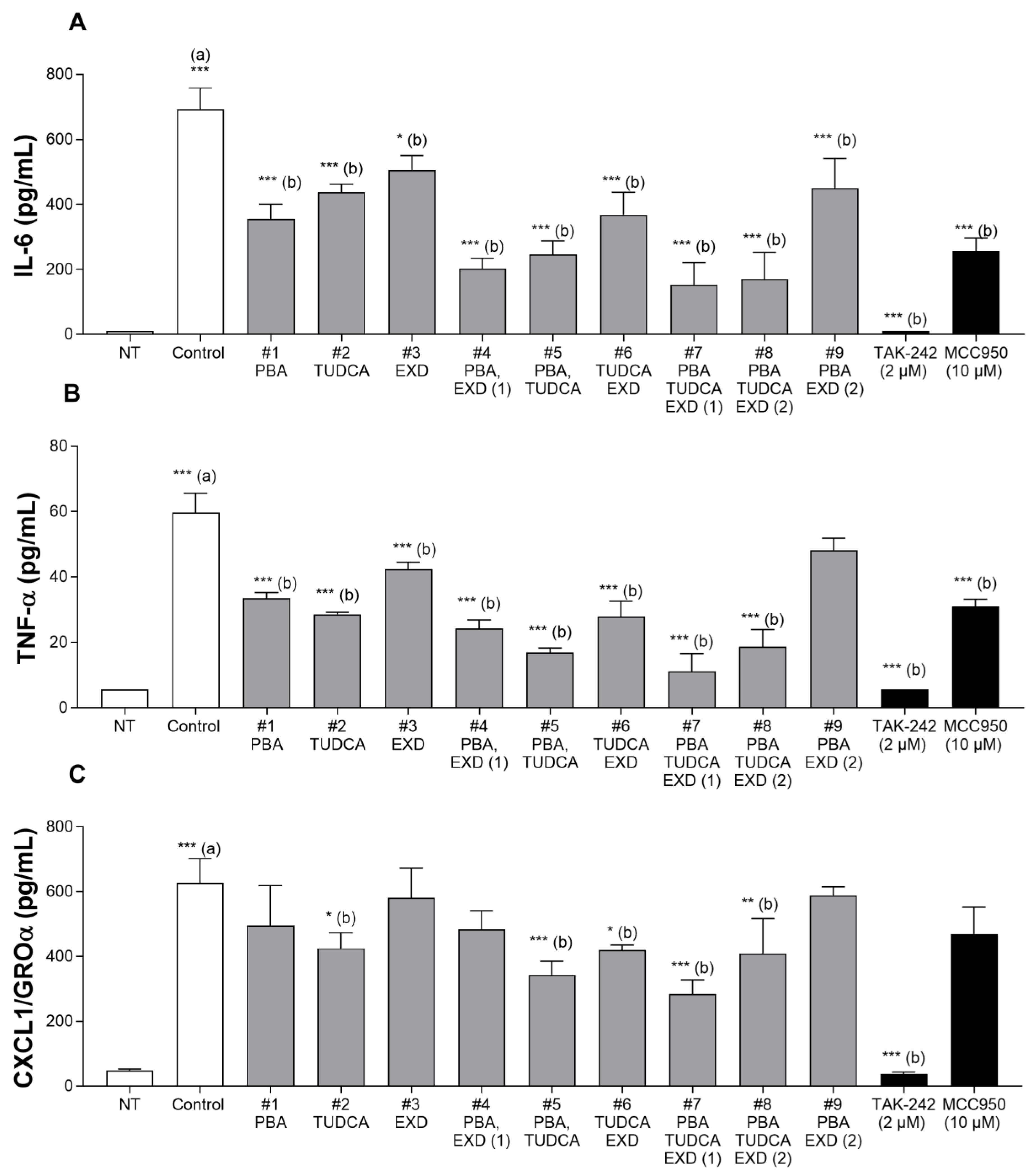
3.4. Model 4. Effect of Experimental Conditions on Human Microglia Cell Line
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, W.; Hamilton, J.L.; Kopil, C.; Beck, J.C.; Tanner, C.M.; Albin, R.L.; Ray Dorsey, E.; Dahodwala, N.; Cintina, I.; Hogan, P.; et al. Current and projected future economic burden of Parkinson’s disease in the U.S. npj Park. Dis. 2020, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Cuervo, A.M.; Stefanis, L.; Fredenburg, R.; Lansbury, P.T.; Sulzer, D. Impaired degradation of mutant alpha-synuclein by chaperone-mediated autophagy. Science 2004, 305, 1292–1295. [Google Scholar] [CrossRef] [PubMed]
- Mosharov, E.V.; Larsen, K.E.; Kanter, E.; Phillips, K.A.; Wilson, K.; Schmitz, Y.; Krantz, D.E.; Kobayashi, K.; Edwards, R.H.; Sulzer, D. Interplay between cytosolic dopamine, calcium, and alpha-synuclein causes selective death of substantia nigra neurons. Neuron 2009, 62, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S.D.; Dolatabadi, N.; Chan, S.F.; Zhang, X.; Akhtar, M.W.; Parker, J.; Soldner, F.; Sunico, C.R.; Nagar, S.; Talantova, M.; et al. Isogenic human iPSC Parkinson’s model shows nitrosative stress-induced dysfunction in MEF2-PGC1alpha transcription. Cell 2013, 155, 1351–1364. [Google Scholar] [CrossRef]
- Zucca, F.A.; Segura-Aguilar, J.; Ferrari, E.; Munoz, P.; Paris, I.; Sulzer, D.; Sarna, T.; Casella, L.; Zecca, L. Interactions of iron, dopamine and neuromelanin pathways in brain aging and Parkinson’s disease. Prog. Neurobiol. 2017, 155, 96–119. [Google Scholar] [CrossRef]
- Hurley, M.J.; Brandon, B.; Gentleman, S.M.; Dexter, D.T. Parkinson’s disease is associated with altered expression of CaV1 channels and calcium-binding proteins. Brain 2013, 136, 2077–2097. [Google Scholar] [CrossRef]
- Rocha, E.M.; De Miranda, B.; Sanders, L.H. Alpha-synuclein: Pathology, mitochondrial dysfunction and neuroinflammation in Parkinson’s disease. Neurobiol. Dis. 2018, 109, 249–257. [Google Scholar] [CrossRef]
- Shtilbans, A.; Reintsch, W.E.; Piscopo, V.E.C.; Krahn, A.I.; Durcan, T.M. Combination of tauroursodeoxycholic acid, co-enzyme Q10 and creatine demonstrates additive neuroprotective effects in in-vitro models of Parkinson’s disease. Front. Neurosci. 2024, 18, 1492028. [Google Scholar] [CrossRef]
- Ono, K.; Ikemoto, M.; Kawarabayashi, T.; Ikeda, M.; Nishinakagawa, T.; Hosokawa, M.; Shoji, M.; Takahashi, M.; Nakashima, M. A chemical chaperone, sodium 4-phenylbutyric acid, attenuates the pathogenic potency in human alpha-synuclein A30P + A53T transgenic mice. Park. Relat. Disord. 2009, 15, 649–654. [Google Scholar] [CrossRef]
- Roy, A.; Ghosh, A.; Jana, A.; Liu, X.; Brahmachari, S.; Gendelman, H.E.; Pahan, K. Sodium phenylbutyrate controls neuroinflammatory and antioxidant activities and protects dopaminergic neurons in mouse models of Parkinson’s disease. PLoS ONE 2012, 7, e38113. [Google Scholar] [CrossRef]
- Simmering, J.E.; Welsh, M.J.; Liu, L.; Narayanan, N.S.; Pottegard, A. Association of Glycolysis-Enhancing alpha-1 Blockers With Risk of Developing Parkinson Disease. JAMA Neurol. 2021, 78, 407–413. [Google Scholar] [CrossRef]
- Kim, S.; Moon, M.; Park, S. Exendin-4 protects dopaminergic neurons by inhibition of microglial activation and matrix metalloproteinase-3 expression in an animal model of Parkinson’s disease. J. Endocrinol. 2009, 202, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, L.; Li, L.; Holscher, C. Semaglutide is Neuroprotective and Reduces alpha-Synuclein Levels in the Chronic MPTP Mouse Model of Parkinson’s Disease. J. Park. Dis. 2019, 9, 157–171. [Google Scholar] [CrossRef]
- Igoillo-Esteve, M.; Oliveira, A.F.; Cosentino, C.; Fantuzzi, F.; Demarez, C.; Toivonen, S.; Hu, A.; Chintawar, S.; Lopes, M.; Pachera, N.; et al. Exenatide induces frataxin expression and improves mitochondrial function in Friedreich ataxia. JCI Insight 2020, 5, e134221. [Google Scholar] [CrossRef]
- Chang, G.; Liu, J.; Qin, S.; Jiang, Y.; Zhang, P.; Yu, H.; Lu, K.; Zhang, N.; Cao, L.; Wang, Y.; et al. Cardioprotection by exenatide: A novel mechanism via improving mitochondrial function involving the GLP-1 receptor/cAMP/PKA pathway. Int. J. Mol. Med. 2018, 41, 1693–1703. [Google Scholar] [CrossRef]
- Mishra, A.; Chandravanshi, L.P.; Trigun, S.K.; Krishnamurthy, S. Ambroxol modulates 6-Hydroxydopamine-induced temporal reduction in Glucocerebrosidase (GCase) enzymatic activity and Parkinson’s disease symptoms. Biochem. Pharmacol. 2018, 155, 479–493. [Google Scholar] [CrossRef]
- Devos, D.; Moreau, C.; Devedjian, J.C.; Kluza, J.; Petrault, M.; Laloux, C.; Jonneaux, A.; Ryckewaert, G.; Garcon, G.; Rouaix, N.; et al. Targeting chelatable iron as a therapeutic modality in Parkinson’s disease. Antioxid. Redox Signal. 2014, 21, 195–210. [Google Scholar] [CrossRef] [PubMed]
- Devos, D.; Labreuche, J.; Rascol, O.; Corvol, J.C.; Duhamel, A.; Guyon Delannoy, P.; Poewe, W.; Compta, Y.; Pavese, N.; Ruzicka, E.; et al. Trial of Deferiprone in Parkinson’s Disease. N. Engl. J. Med. 2022, 387, 2045–2055. [Google Scholar] [CrossRef]
- Burklen, T.S.; Schlattner, U.; Homayouni, R.; Gough, K.; Rak, M.; Szeghalmi, A.; Wallimann, T. The creatine kinase/creatine connection to Alzheimer’s disease: CK-inactivation, APP-CK complexes and focal creatine deposits. J. Biomed. Biotechnol. 2006, 2006, 35936. [Google Scholar] [CrossRef]
- Genius, J.; Geiger, J.; Bender, A.; Moller, H.J.; Klopstock, T.; Rujescu, D. Creatine protects against excitoxicity in an in vitro model of neurodegeneration. PLoS ONE 2012, 7, e30554. [Google Scholar] [CrossRef]
- Writing Group for the NINDS Exploratory Trials in Parkinson Disease (NET-PD) Investigators; Kieburtz, K.; Tilley, B.C.; Elm, J.J.; Babcock, D.; Hauser, R.; Ross, G.W.; Augustine, A.H.; Augustine, E.U.; Aminoff, M.J.; et al. Effect of creatine monohydrate on clinical progression in patients with Parkinson disease: A randomized clinical trial. JAMA 2015, 313, 584–593. [Google Scholar] [CrossRef]
- Lee, D.; Shim, M.S.; Kim, K.Y.; Noh, Y.H.; Kim, H.; Kim, S.Y.; Weinreb, R.N.; Ju, W.K. Coenzyme Q10 inhibits glutamate excitotoxicity and oxidative stress-mediated mitochondrial alteration in a mouse model of glaucoma. Investig. Ophthalmol. Vis. Sci. 2014, 55, 993–1005. [Google Scholar] [CrossRef]
- Yang, L.; Calingasan, N.Y.; Wille, E.J.; Cormier, K.; Smith, K.; Ferrante, R.J.; Beal, M.F. Combination therapy with coenzyme Q10 and creatine produces additive neuroprotective effects in models of Parkinson’s and Huntington’s diseases. J. Neurochem. 2009, 109, 1427–1439. [Google Scholar] [CrossRef]
- Hebron, M.L.; Lonskaya, I.; Moussa, C.E. Nilotinib reverses loss of dopamine neurons and improves motor behavior via autophagic degradation of alpha-synuclein in Parkinson’s disease models. Hum. Mol. Genet. 2013, 22, 3315–3328. [Google Scholar] [CrossRef]
- Simuni, T.; Fiske, B.; Merchant, K.; Coffey, C.S.; Klingner, E.; Caspell-Garcia, C.; Lafontant, D.E.; Matthews, H.; Wyse, R.K.; Brundin, P.; et al. Efficacy of Nilotinib in Patients With Moderately Advanced Parkinson Disease: A Randomized Clinical Trial. JAMA Neurol. 2021, 78, 312–320. [Google Scholar] [CrossRef]
- Karuppagounder, S.S.; Wang, H.; Kelly, T.; Rush, R.; Nguyen, R.; Bisen, S.; Yamashita, Y.; Sloan, N.; Dang, B.; Sigmon, A.; et al. The c-Abl inhibitor IkT-148009 suppresses neurodegeneration in mouse models of heritable and sporadic Parkinson’s disease. Sci. Transl. Med. 2023, 15, eabp9352. [Google Scholar] [CrossRef]
- Duan, W.M.; Rodrigues, C.M.; Zhao, L.R.; Steer, C.J.; Low, W.C. Tauroursodeoxycholic acid improves the survival and function of nigral transplants in a rat model of Parkinson’s disease. Cell Transplant. 2002, 11, 195–205. [Google Scholar] [CrossRef]
- Castro-Caldas, M.; Carvalho, A.N.; Rodrigues, E.; Henderson, C.J.; Wolf, C.R.; Rodrigues, C.M.; Gama, M.J. Tauroursodeoxycholic acid prevents MPTP-induced dopaminergic cell death in a mouse model of Parkinson’s disease. Mol. Neurobiol. 2012, 46, 475–486. [Google Scholar] [CrossRef]
- Cuevas, E.; Burks, S.; Raymick, J.; Robinson, B.; Gomez-Crisostomo, N.P.; Escudero-Lourdes, C.; Lopez, A.G.G.; Chigurupati, S.; Hanig, J.; Ferguson, S.A.; et al. Tauroursodeoxycholic acid (TUDCA) is neuroprotective in a chronic mouse model of Parkinson’s disease. Nutr. Neurosci. 2020, 25, 1374–1391. [Google Scholar] [CrossRef]
- Cai, R.; Zhang, Y.; Simmering, J.E.; Schultz, J.L.; Li, Y.; Fernandez-Carasa, I.; Consiglio, A.; Raya, A.; Polgreen, P.M.; Narayanan, N.S.; et al. Enhancing glycolysis attenuates Parkinson’s disease progression in models and clinical databases. J. Clin. Investig. 2019, 129, 4539–4549. [Google Scholar] [CrossRef] [PubMed]
- Baden, M.Y.; Fukui, K.; Hosokawa, Y.; Iwahashi, H.; Imagawa, A.; Shimomura, I. Examination of a Viral Infection Mimetic Model in Human iPS Cell-Derived Insulin-Producing Cells and the Anti-Apoptotic Effect of GLP-1 Analogue. PLoS ONE 2015, 10, e0144606. [Google Scholar] [CrossRef]
- Magalhaes, J.; Gegg, M.E.; Migdalska-Richards, A.; Schapira, A.H. Effects of ambroxol on the autophagy-lysosome pathway and mitochondria in primary cortical neurons. Sci. Rep. 2018, 8, 1385. [Google Scholar] [CrossRef]
- Aguirre, P.; Mena, N.P.; Carrasco, C.M.; Munoz, Y.; Perez-Henriquez, P.; Morales, R.A.; Cassels, B.K.; Mendez-Galvez, C.; Garcia-Beltran, O.; Gonzalez-Billault, C.; et al. Iron Chelators and Antioxidants Regenerate Neuritic Tree and Nigrostriatal Fibers of MPP+/MPTP-Lesioned Dopaminergic Neurons. PLoS ONE 2015, 10, e0144848. [Google Scholar] [CrossRef]
- Cunha, M.P.; Martin-de-Saavedra, M.D.; Romero, A.; Parada, E.; Egea, J.; Del Barrio, L.; Rodrigues, A.L.; Lopez, M.G. Protective effect of creatine against 6-hydroxydopamine-induced cell death in human neuroblastoma SH-SY5Y cells: Involvement of intracellular signaling pathways. Neuroscience 2013, 238, 185–194. [Google Scholar] [CrossRef]
- Cooper, O.; Seo, H.; Andrabi, S.; Guardia-Laguarta, C.; Graziotto, J.; Sundberg, M.; McLean, J.R.; Carrillo-Reid, L.; Xie, Z.; Osborn, T.; et al. Pharmacological rescue of mitochondrial deficits in iPSC-derived neural cells from patients with familial Parkinson’s disease. Sci. Transl. Med. 2012, 4, 141ra190. [Google Scholar] [CrossRef] [PubMed]
- Stojkovska, I.; Wani, W.Y.; Zunke, F.; Belur, N.R.; Pavlenko, E.A.; Mwenda, N.; Sharma, K.; Francelle, L.; Mazzulli, J.R. Rescue of alpha-synuclein aggregation in Parkinson’s patient neurons by synergistic enhancement of ER proteostasis and protein trafficking. Neuron 2022, 110, 436–451.e11. [Google Scholar] [CrossRef]
- Chen, C.X.; Abdian, N.; Maussion, G.; Thomas, R.A.; Demirova, I.; Cai, E.; Tabatabaei, M.; Beitel, L.K.; Karamchandani, J.; Fon, E.A.; et al. A Multistep Workflow to Evaluate Newly Generated iPSCs and Their Ability to Generate Different Cell Types. Methods Protoc. 2021, 4, 50. [Google Scholar] [CrossRef]
- Krahn, A.; Reintsch, W.; Cai, E.; Durcan, T.M. Uptake of a-Synuclein Preformed Fibrils (PFFs) in Dopaminergic Neural Progenitor Cells. Zenodo 2020. [Google Scholar] [CrossRef]
- Chen, E.; Lauinger, N.; Rocha, C.; Rao, T.; Durcan, T. Generation of dopaminergic or cortical neurons from neuronal progenitors. Zenodo 2019. [Google Scholar] [CrossRef]
- Chen, C.X.-Q.; Deneault, E.; Abdian, N.; You, Z.; Sirois, J.; Nicouleau, M.; Shlaifer, I.; Villegas, L.; Boivin, M.-N.; Gaborieau, L.; et al. Generation of patient-derived pluripotent stem cell-lines and CRISPR modified isogenic controls with mutations in the Parkinson’s associated GBA gene. Stem Cell Res. 2022, 64, 102919. [Google Scholar] [CrossRef]
- Gullo, F.; Ceriani, M.; D’Aloia, A.; Wanke, E.; Constanti, A.; Costa, B.; Lecchi, M. Plant Polyphenols and Exendin-4 Prevent Hyperactivity and TNF-alpha Release in LPS-Treated In vitro Neuron/Astrocyte/Microglial Networks. Front. Neurosci. 2017, 11, 500. [Google Scholar] [CrossRef] [PubMed]
- Romero-Ramirez, L.; Garcia-Rama, C.; Wu, S.; Mey, J. Bile acids attenuate PKM2 pathway activation in proinflammatory microglia. Sci. Rep. 2022, 12, 1459. [Google Scholar] [CrossRef]
- Yanguas-Casas, N.; Barreda-Manso, M.A.; Nieto-Sampedro, M.; Romero-Ramirez, L. TUDCA: An Agonist of the Bile Acid Receptor GPBAR1/TGR5 With Anti-Inflammatory Effects in Microglial Cells. J. Cell. Physiol. 2017, 232, 2231–2245. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Ko, W.K.; Jo, M.J.; Arai, Y.; Choi, H.; Kumar, H.; Han, I.B.; Sohn, S. Anti-inflammatory effect of Tauroursodeoxycholic acid in RAW 264.7 macrophages, Bone marrow-derived macrophages, BV2 microglial cells, and spinal cord injury. Sci. Rep. 2018, 8, 3176. [Google Scholar] [CrossRef]
- Meissner, W.G.; Remy, P.; Giordana, C.; Maltete, D.; Derkinderen, P.; Houeto, J.L.; Anheim, M.; Benatru, I.; Boraud, T.; Brefel-Courbon, C.; et al. Trial of Lixisenatide in Early Parkinson’s Disease. N. Engl. J. Med. 2024, 390, 1176–1185. [Google Scholar] [CrossRef]
- Salameh, T.S.; Rhea, E.M.; Talbot, K.; Banks, W.A. Brain uptake pharmacokinetics of incretin receptor agonists showing promise as Alzheimer’s and Parkinson’s disease therapeutics. Biochem. Pharmacol. 2020, 180, 114187. [Google Scholar] [CrossRef]
| # | Drugs in Combination | # | Drugs in Combination |
|---|---|---|---|
| 1 | PBA, TZ, EXD, CR, CoQ10, Medium | 23 | AMB, MPP+ |
| 2 | PBA, TZ, EXD, CR, CoQ10, MPP+ | 24 | DFP, MPP+ |
| 3 | TZ, EXD, CR, CoQ10, MPP+ | 25 | PBA, TZ, EXD, AMB, DFP, Medium |
| 4 | EXD, CR, CoQ10, MPP+ | 26 | PBA, TZ, EXD, AMB, DFP, MPP+ |
| 5 | PBA, TZ, CR, CoQ10, MPP+ | 27 | PBA, TZ, AMB, DFP, MPP+ |
| 6 | PBA, TZ, EXD, MPP+ | 28 | PBA, TZ, DFP, MPP+ |
| 7 | TZ, EXD, MPP+ | 29 | TZ, EXD, AMB, DFP, MPP+ |
| 8 | PBA, EXD, MPP+ | 30 | EXD, AMB, DFP, MPP+ |
| 9 | PBA, TZ, MPP+ | 31 | PBA, EXD, AMB, DFP, MPP+ |
| 10 | PBA, MPP+ | 32 | PBA, TZ, EXD, DFP, MPP+ |
| 11 | TZ, MPP+ | 33 | TZ, AMB, DFP, MPP+ |
| 12 | EXD, MPP+ | 34 | TZ, EXD, AMB, MPP+ |
| 13 | CR, CoQ10, MPP+ | 35 | TZ, DFP, MPP+ |
| 14 | PBA, CR, CoQ10, MPP+ | 36 | TZ, AMB, MPP+ |
| 15 | TZ, CR, CoQ10, MPP+ | 37 | EXD, AMB, MPP+ |
| 16 | EXD, CR, CoQ10, MPP+ | 38 | EXD, DFP, MPP+ |
| 17 | PBA, TZ, EXD, AMB, MPP+ | 39 | TZ, EXD, DFP, MPP+ |
| 18 | PBA, TZ, AMB, MPP+ | 40 | PBA, EXD, DFP, MPP+ |
| 19 | AMB, DFP, MPP+ | 41 | PBA, EXD, AMB, MPP+ |
| 20 | PBA, AMB, MPP+ | 51 | TUDCA, MPP+ |
| 21 | PBA, AMB, DFP, MPP+ | 52 | PBA, TUDCA, MPP+ |
| 22 | PBA, DFP, MPP+ | 53 | PBA, TUDCA, EXD, MPP+ |
| Drugs in Combination | Drugs in Combination |
|---|---|
| Dasatinib, TUDCA | Dasatinib, Creatine, CoQ10 |
| Dasatinib, EXD | Dasatinib, Terazosin |
| Dasatinib, PBA | PBA, EXD |
| Dasatinib, TUDCA, PBA | PBA, TUDCA |
| Dasatinib, PBA, EXD | TUDCA, EXD |
| Dasatinib, TUDCA, EXD | PBA, TUDCA, EXD |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shtilbans, A.; Esneault, E.; Simon, F.; Mazzulli, J.R.; Quiriconi, D.J.; Rom, D.; Reintsch, W.E.; Krahn, A.I.; Durcan, T.M. Evaluation of Additive Neuroprotective Effect of Combination Therapy for Parkinson’s Disease Using In Vitro Models. Antioxidants 2025, 14, 396. https://doi.org/10.3390/antiox14040396
Shtilbans A, Esneault E, Simon F, Mazzulli JR, Quiriconi DJ, Rom D, Reintsch WE, Krahn AI, Durcan TM. Evaluation of Additive Neuroprotective Effect of Combination Therapy for Parkinson’s Disease Using In Vitro Models. Antioxidants. 2025; 14(4):396. https://doi.org/10.3390/antiox14040396
Chicago/Turabian StyleShtilbans, Alexander, Elise Esneault, Florian Simon, Joseph R. Mazzulli, Drew J. Quiriconi, Dror Rom, Wolfgang E. Reintsch, Andrea I. Krahn, and Thomas M. Durcan. 2025. "Evaluation of Additive Neuroprotective Effect of Combination Therapy for Parkinson’s Disease Using In Vitro Models" Antioxidants 14, no. 4: 396. https://doi.org/10.3390/antiox14040396
APA StyleShtilbans, A., Esneault, E., Simon, F., Mazzulli, J. R., Quiriconi, D. J., Rom, D., Reintsch, W. E., Krahn, A. I., & Durcan, T. M. (2025). Evaluation of Additive Neuroprotective Effect of Combination Therapy for Parkinson’s Disease Using In Vitro Models. Antioxidants, 14(4), 396. https://doi.org/10.3390/antiox14040396





