Autochthonous Outbreak of SARS-CoV-2 Omicron Variant in Booster-Vaccinated (3 Doses) Healthcare Workers in Southern Italy: Just the Tip of the Iceberg?
Abstract
:1. Introduction
2. Patients and Methods
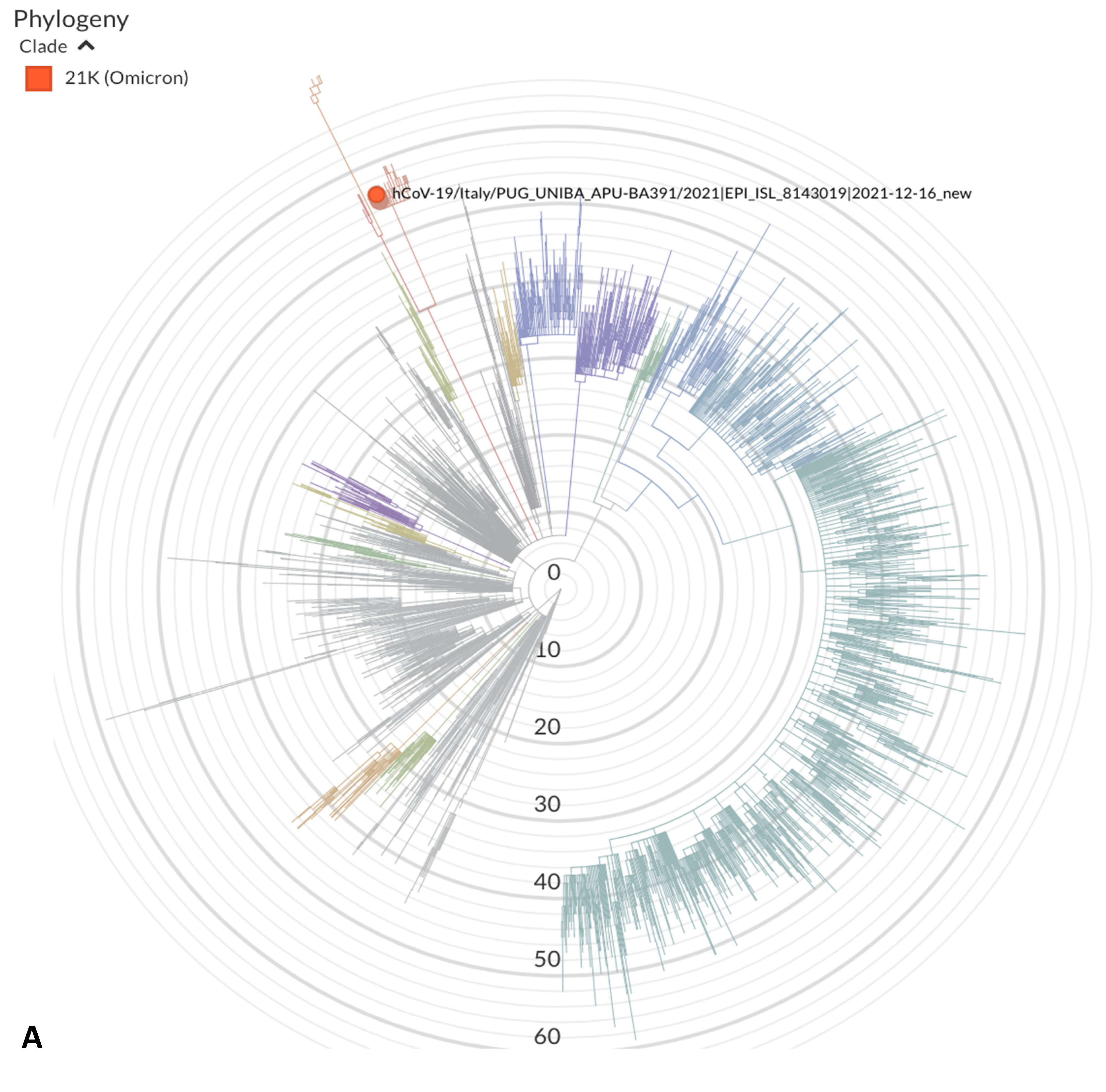
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Classification of Omicron (B.1.1.529): SARS-CoV-2 Variant of Concern. Available online: https://www.who.int/news/item/26-11-2021-classification-of-omicron-(b.1.1.529)-sars-cov-2-variant-of-concern (accessed on 20 January 2022).
- Network for Genomic Surveillance in South Africa. NGS-SA: Updates, COVID-19 Analysis, Narratives, Reports. Durban: NGS-SA. 2021. Available online: https://www.ngs-sa.org/ngs-sa_updates_covid-19_analysis_narratives_reports/token/38 (accessed on 20 January 2022).
- World Health Organization. Enhancing Readiness for Omicron (B.1.1.529): Technical Brief and Priority Actions for Member States. 2021. Available online: https://www.who.int/publications/m/item/enhancing-readiness-for-omicron-(b.1.1.529)-technical-brief-and-priority-actions-for-member-states (accessed on 20 January 2022).
- European Center for Disease Prevention and Control. Rapid Increase of a SARS-CoV-2 Variant with Multiple Spike Protein Mutations Observed in the United Kingdom. Threat Assessment Brief. 20 December 2020. Available online: https://www.ecdc.europa.eu/en/publications-data/threat-assessment-brief-rapid-increase-sars-cov-2-variant-united-kingdom (accessed on 20 January 2022).
- Rambaut, A. Preliminary Genomic Characterisation of an Emergent SARS-CoV-2 Lineage in the UK Defined by a Novel Set of Spike Mutations—SARS-CoV-2 Coronavirus/nCoV-2019 Genomic Epidemiology, Virological. 2020. Available online: https://virological.org/t/preliminary-genomic-characterisation-of-an-emergent-sars-cov-2-lineage-in-the-uk-defined-by-a-novel-set-ofspike-mutations/563 (accessed on 30 January 2021).
- Loconsole, D.; Centrone, F.; Morcavallo, C.; Campanella, S.; Sallustio, A.; Accogli, M.; Fortunato, F.; Parisi, A.; Chironna, M. Rapid spread of the SARS-CoV-2 variant of concern 202012/01 in Southern Italy (December 2020–March 2021). Int. J. Environ. Res. Public Health 2021, 18, 4766. [Google Scholar] [CrossRef] [PubMed]
- Brandal, L.T.; MacDonald, E.; Veneti, L.; Ravlo, T.; Lange, H.; Naseer, U.; Feruglio, S.; Bragstad, K.; Hungnes, O.; Ødeskaug, L.E.; et al. Outbreak caused by the SARS-CoV-2 Omicron variant in Norway, November to December 2021. Euro Surveill. 2021, 26, 2101147. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Omicron Variant: What You Need to Know. Updated 20 December 2021. Available online: https://www.cdc.gov/coronavirus/2019-ncov/variants/omicron-variant.html (accessed on 20 January 2022).
- European Center for Disease Prevention and Control. European Weekly Epidemiological Update: Omicron Variant of Concern (VOC)—Week 50 (Data as of 19 December 2021). Available online: https://www.ecdc.europa.eu/en/news-events/weekly-epidemiological-update-omicron-variant-concern-voc-week-50-data-19-december-2021 (accessed on 20 January 2022).
- Maslo, C.; Friedland, R.; Toubkin, M.; Laubscher, A.; Akaloo, T.; Kama, B. Characteristics and Outcomes of Hospitalized Patients in South Africa During the COVID-19 Omicron Wave Compared With Previous Waves. JAMA 2022, 327, 583–584. [Google Scholar] [CrossRef] [PubMed]
- HKUMed Finds Omicron SARS-CoV-2 Can Infect Faster and Better Than Delta in Human Bronchus but with Less Severe Infection in Lung. Available online: https://www.med.hku.hk/en/news/press/20211215-omicron-sars-cov-2-infection (accessed on 20 January 2022).
- The Guardian. South Africa: Previous Infections May Explain Omicron Hospitalisation Rate. Available online: https://www.theguardian.com/world/2021/dec/14/south-africa-previous-infections-may-explain-omicron-hospitalisation-rate?fbclid=IwAR0S51uygFgpkwc4NaPnC14Sp6Azfjz-fxeLAujmZH5urFbPjsHzYbsciAM (accessed on 20 January 2022).
- GISAID Database. Available online: https://www.gisaid.org/ (accessed on 20 January 2022).
- European Centre for Disease Prevention and Control. Threat Assessment Brief: Implications of the Further Emergence and Spread of the SARS CoV 2 B.1.1.529 Variant of Concern (Omicron) for the EU/EEA First Update. Available online: https://www.ecdc.europa.eu/en/publications-data/covid-19-threat-assessment-spread-omicron-first-update (accessed on 20 January 2022).
- Ministero Della Salute. Comunicazione dei casi Confermati e Probabili di Infezione da SARS CoV-2 VOC Omicron; Circolare Ministeriale: 0056808-10/12/2021-DGPRE-DGPRE-P. Available online: https://www.seremi.it/sites/default/files/Sorveglianza%20casi%20VOC%20Omicron.pdf (accessed on 11 February 2022).
- Istituto Superiore di Sanità. Stima Della Prevalenza Delle Varianti VOC (Variants of Concern) in Italia: Beta, Gamma, Delta, Omicron e Altre Varianti di SARS-CoV-2 (Indagine del 06/12/2021). Available online: https://www.epicentro.iss.it/coronavirus/pdf/sars-cov-2-monitoraggio-varianti-indagini-rapide-6-dicembre-2021.pdf (accessed on 11 February 2022).
- Istituto Superiore di Sanità. Stima della prevalenza delle varianti VOC (Variant Of Concern) e di altre varianti di SARS-CoV-2 in Italia (Indagine del 03/01/2022). Available online: https://www.iss.it/documents/20126/0/Report_flashVarianti_14gennaio22.pdf/b44b1a7d-a0c1-67fd-44b7-34c8b775c088?t=1642162662435 (accessed on 11 February 2022).
- National Institute of Health (NIH). Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. Available online: https://files.covid19treatmentguidelines.nih.gov/guidelines/covid19treatmentguidelines.pdf (accessed on 20 January 2022).
- Singanayagam, A.; Patel, M.; Charlett, A.; Bernal, J.L.; Saliba, V.; Ellis, J.; Ladhani, S.; Zambon, M.; Gopal, R. Duration of infectiousness and correlation with RT-PCR cycle threshold values in cases of COVID-19, England, January to May 2020. Euro Surveill. 2020, 25, 2001483. [Google Scholar] [CrossRef] [PubMed]
- Loconsole, D.; Sallustio, A.; Accogli, M.; Leaci, A.; Sanguedolce, A.; Parisi, A.; Chironna, M. Investigation of an outbreak of symptomatic SARS-CoV-2 VOC 202012/01-lineage B.1.1.7 infection in healthcare workers, Italy. Clin. Microbiol. Infect. 2021, 27, 1174.e1–1174.e4. [Google Scholar] [CrossRef] [PubMed]
- Loconsole, D.; Sallustio, A.; Accogli, M.; Centrone, F.; Casulli, D.; Madaro, A.; Tedeschi, E.; Parisi, A.; Chironna, M. Symptomatic SARS-CoV-2 reinfection in a healthy healthcare worker in Italy confirmed by whole-genome sequencing. Viruses 2021, 13, 899. [Google Scholar] [CrossRef] [PubMed]
- Ministero Della Salute. Diffusione Nuova Variante VOC B.1.1.529 Omicron. Circolare Ministeriale: 0054258-26/11/2021-DGPRE-DGPRE-P. Available online: https://www.trovanorme.salute.gov.it/norme/renderNormsanPdf?anno=2021&codLeg=84134&parte=1%20&serie=null (accessed on 11 February 2022).
- Espenhain, L.; Funk, T.; Overvad, M.; Edslev, S.M.; Fonager, J.; Ingham, A.C.; Rasmussen, M.; Madsen, S.L.; Espersen, C.H.; Sieber, R.N.; et al. Epidemiological characterisation of the first 785 SARS-CoV-2 Omicron variant cases in Denmark, December 2021. Euro Surveill. 2021, 26, 2101146. [Google Scholar] [CrossRef]
- Jansen, L.; Tegomoh, B.; Lange, K.; Showalter, K.; Figliomeni, J.; Abdalhamid, B.; Iwen, P.C.; Fauver, J.; Buss, B.; Donahue, M. Investigation of a SARS-CoV-2 B.1.1.529 (Omicron) variant cluster—Nebraska, November–December 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1782–1784. [Google Scholar] [CrossRef] [PubMed]
- UK Health Security Agency. SARS-CoV-2 Variants of Concern and Variants under Investigation in England. Technical Briefing: Update on Hospitalisation and Vaccine Effectiveness for Omicron VOC-21NOV-01 (B.1.1.529); 31 December 2021. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1044481/Technical-Briefing-31-Dec-2021-Omicron_severity_update.pdf (accessed on 20 January 2022).
- Collie, S.; Champion, J.; Moultrie, H.; Bekker, L.G.; Gray, G. Effectiveness of BNT162b2 vaccine against Omicron variant in South Africa. N. Engl. J. Med. 2021, 386, 494–496. [Google Scholar] [CrossRef]
- Barda, N.; Dagan, N.; Cohen, C.; Hernan MA Lipsitch, M.; Kohane, I.S.; Reis, B.Y.; Balicer, R.D. Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: An observational study. Lancet 2021, 398, 2093–2100. [Google Scholar] [CrossRef]
- Garcia-Beltran, W.F.; St Denis, K.J.; Hoelzemer, A.; Lam, E.C.; Nitido, A.D.; Sheehan, M.L.; Berrios, C.; Ofoman, O.; Chang, C.C.; Hauser, B.M.; et al. mRNA-based COVID-19 vaccine boosters induce neutralizing immunity against SARS-CoV-2 Omicron variant. Cell 2022, 185, 457–466.e4. [Google Scholar] [CrossRef] [PubMed]
- Azzi, L.; Dalla Gasperina, D.; Veronesi, G.; Shallak, M.; Ietto, G.; Iovino, D.; Baj, A.; Gianfagna, F.; Maurino, V.; Focosi, D.; et al. Mucosal immune response in BNT162b2 COVID-19 vaccine recipients. EBioMedicine 2021, 75, 103788. [Google Scholar] [CrossRef] [PubMed]
- Helmsdal, G.; Hansen, O.K.; Møller, L.F.; Christiansen, D.H.; Skaalum Petersen, M.; Kristiansen, M.F. Omicron outbreak at a private gathering in the Faroe Islands, infecting 21 of 33 triple-vaccinated healthcare workers. medRxiv, 2021; preprint. [Google Scholar] [CrossRef] [PubMed]
- Karim, S.S.A.; Karim, Q.A. Omicron SARS-CoV-2 variant: A new chapter in the COVID-19 pandemic. Lancet 2021, 398, 2126–2128. [Google Scholar] [CrossRef]
- Roberts, S.C.; Palacios, C.F.; Grubaugh, N.D.; Alpert, T.; Ott, I.M.; Breban, M.I.; Martinello, R.A.; Smith, C.; Davis, M.W.; Mcmanus, D.; et al. An outbreak of SARS-CoV-2 on a transplant unit in the early vaccination era. Transpl. Infect. Dis, 2021; Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Callaway, E.; Ledford, H. How bad is Omicron? What scientists know so far. Nature 2021, 600, 197–199. [Google Scholar] [CrossRef] [PubMed]
- European Center for Disease Prevention and Control. Rapid Risk Assessment. Assessment of the Further Emergence and Potential Impact of the SARS-CoV-2 Omicron Variant of Concern in the Context of Ongoing Transmission of the Delta Variant of Concern in the EU/EEA, 18th Update. 15 December 2021. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/covid-19-assessment-further-emergence-omicron-18th-risk-assessment-december-2021.pdf (accessed on 29 December 2021).
- Sonabend, R.; Whittles, L.K.; Imai, N.; Perez-Guzman, P.N.; Knock, E.S.; Rawson, T.; Gaythorpe, K.A.M.; Djaafara, B.A.; Hinsley, W.; FitzJohn, R.G.; et al. Non-pharmaceutical interventions, vaccination, and the SARS-CoV-2 delta variant in England: A mathematical modelling study. Lancet 2021, 398, 1825–1835. [Google Scholar] [CrossRef]

| Case | Sex | Age (Years) | Clinical Status | Symptom Onset | Date of Sample Collection | Date of Negative Sample | Real-Time PCR (Ct *) | Vaccine (BNT162b2 mRNA COVID-19 Vaccine) | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N Gene | ORF1ab Gene | S Gene | Dose I | Dose II | Dose III | |||||||
| 1 (HCW) | F | 29 | Mild | 15/12/2021 | 16/12/2021 | - | 20 | 21 | - | 07/01/2021 | 29/01/2021 | 26/10/2021 |
| 2 (HCW) | F | 40 | Asymptomatic | 16/12/2021 | 04/01/2022 | 19 | 20 | - | 11/01/2021 | 03/02/2021 | 02/11/2021 | |
| 3 (HCW) | F | 31 | Asymptomatic | 18/12/2021 | 29/12/2021 | 26 | 27 | - | 07/01/2021 | 29/01/2021 | 19/11/2021 | |
| 4 (HCW) | F | 32 | Asymptomatic | 16/12/2021 | 04/01/2022 | 14 | 15 | - | 07/01/2021 | 29/01/2021 | 27/10/2021 | |
| 5 (HCW) | F | 43 | Asymptomatic | 17/12/2021 | - | 15 | 16 | - | 11/01/2021 | 03/02/2021 | 25/10/2021 | |
| 6 (HCW) | F | 32 | Mild | 15/12/2021 | 17/12/2021 | 04/01/2022 | 20 | 20 | - | 11/01/2021 | 03/02/2021 | 06/12/2021 |
| 7 (HCW) | M | 29 | Asymptomatic | 20/12/2021 | - | 15 | 16 | - | 12/01/2021 | 04/02/2021 | 16/11/2021 | |
| 8 (HCW) | M | 30 | Asymptomatic | 17/12/2021 | 04/01/2022 | 29 | 29 | - | 09/02/2021 | 03/03/2021 | 11/11/2021 | |
| 9 (HCW) | M | 29 | Asymptomatic | 20/12/2021 | 30/12/2021 | 25 | 25 | - | 11/01/2021 | 03/02/2021 | 03/11/2021 | |
| 10 (HCW) | F | 29 | Mild | 16/12/2021 | 17/12/2021 | 31/12/2021 | 20 | 21 | - | 05/01/2021 | 27/01/2021 | 22/10/2021 |
| 11 (HCW) | F | 29 | Mild | 18/12/2021 | 17/12/2021 | - | 18 | 19 | - | 07/01/2021 | 29/01/2021 | 28/10/2021 |
| 12 (HCW) | M | 41 | Asymptomatic | 16/12/2021 | 04/01/2022 | 17 | 17 | - | 12/01/2021 | 04/02/2021 | 03/11/2021 | |
| 13 (HCW) | M | 25 | Asymptomatic | 16/12/2021 | 04/01/2022 | 31 | 31 | - | 05/01/2021 | 27/01/2021 | 03/11/2021 | |
| 14 (HCW) | F | 28 | Mild | 15/12/2021 | 16/12/2021 | 30/12/2021 | 26 | 28 | - | 05/01/2021 | 27/01/2021 | 14/10/2021 |
| 15 (HCW) | F | 29 | Asymptomatic | 20/12/2021 | - | 20 | 20 | - | 09/01/2021 | 01/02/2021 | 03/11/2021 | |
| 16 (Patient) | F | 49 | Severe | 21/12/2021 | 21/12/2021 | 03/01/2022 | 26 | 26 | - | 30/03/2021 | 20/04/2021 | 21/09/2021 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loconsole, D.; Bisceglia, L.; Centrone, F.; Sallustio, A.; Accogli, M.; Dalfino, L.; Brienza, N.; Chironna, M. Autochthonous Outbreak of SARS-CoV-2 Omicron Variant in Booster-Vaccinated (3 Doses) Healthcare Workers in Southern Italy: Just the Tip of the Iceberg? Vaccines 2022, 10, 283. https://doi.org/10.3390/vaccines10020283
Loconsole D, Bisceglia L, Centrone F, Sallustio A, Accogli M, Dalfino L, Brienza N, Chironna M. Autochthonous Outbreak of SARS-CoV-2 Omicron Variant in Booster-Vaccinated (3 Doses) Healthcare Workers in Southern Italy: Just the Tip of the Iceberg? Vaccines. 2022; 10(2):283. https://doi.org/10.3390/vaccines10020283
Chicago/Turabian StyleLoconsole, Daniela, Lucia Bisceglia, Francesca Centrone, Anna Sallustio, Marisa Accogli, Lidia Dalfino, Nicola Brienza, and Maria Chironna. 2022. "Autochthonous Outbreak of SARS-CoV-2 Omicron Variant in Booster-Vaccinated (3 Doses) Healthcare Workers in Southern Italy: Just the Tip of the Iceberg?" Vaccines 10, no. 2: 283. https://doi.org/10.3390/vaccines10020283








