Potential Antiviral Activity of Lactiplantibacillus plantarum KAU007 against Influenza Virus H1N1
Abstract
:1. Introduction
2. Materials and Methods
2.1. Isolation and Identification of Probiotics from Camel Milk Samples
2.2. Preparation of Cell-Free Culture Supernatant
2.3. Mammalian Cell Culture and Its Maintenance
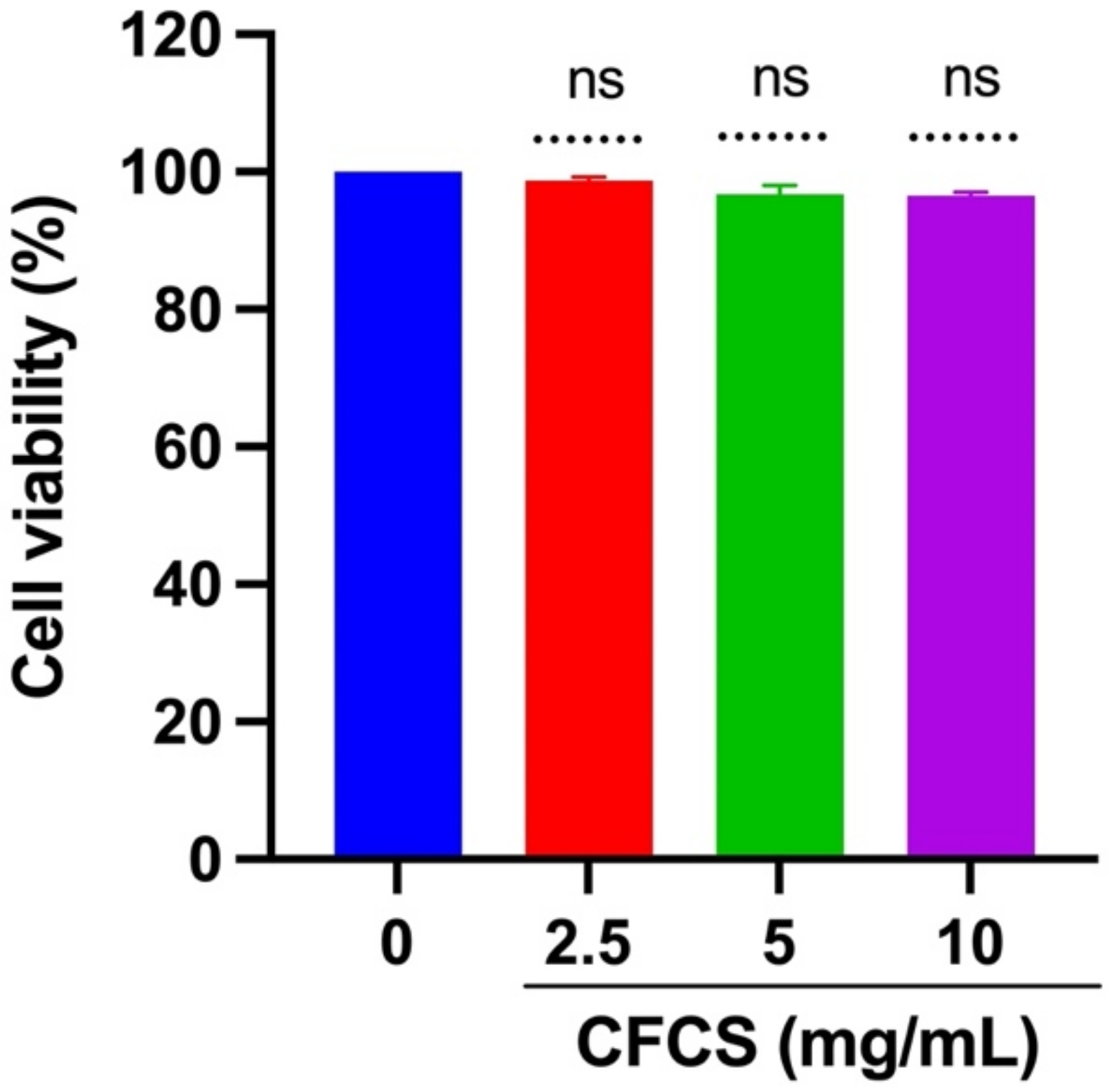
2.4. Cytotoxicity Analysis of CFCS
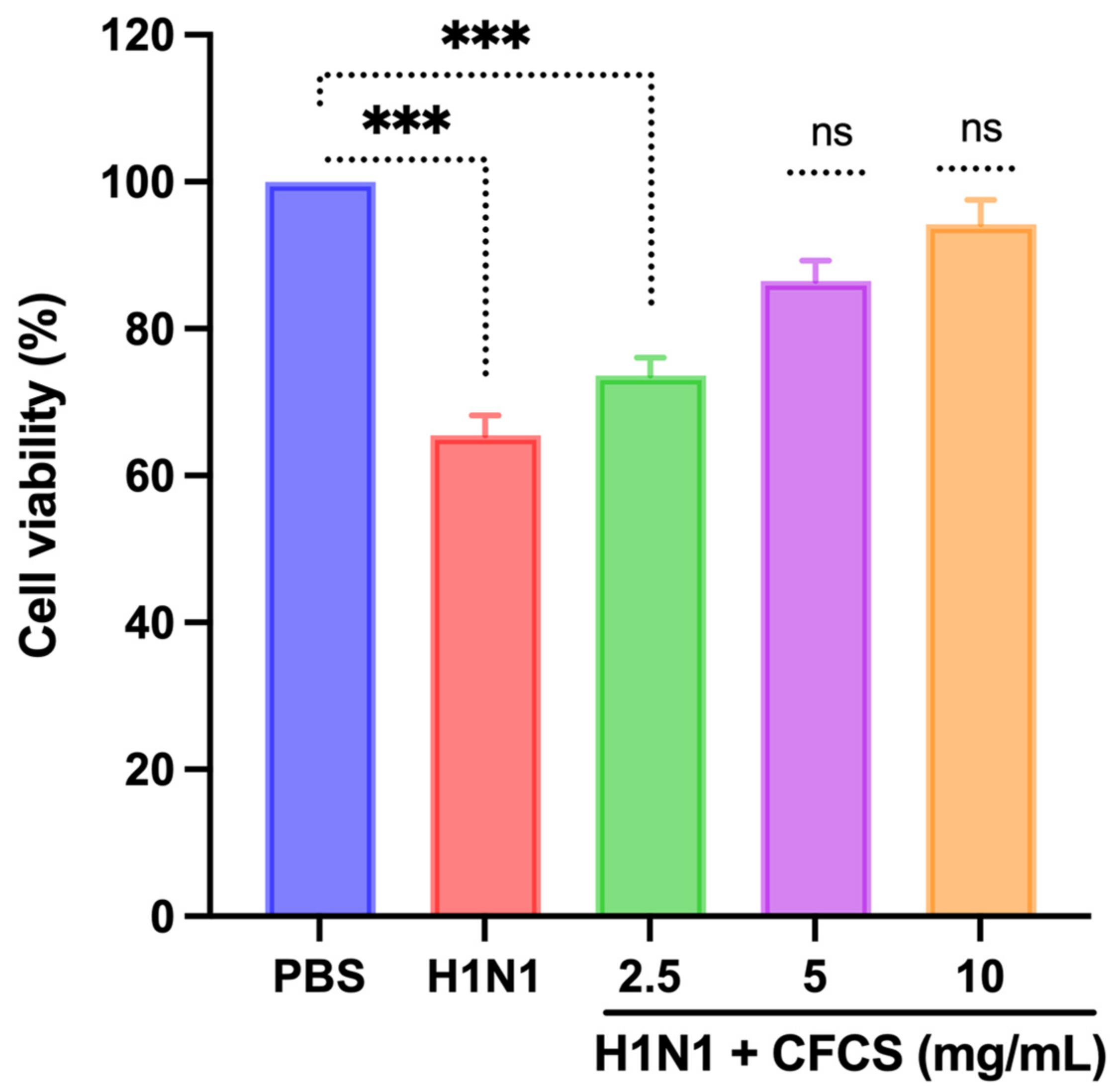
2.5. Antiviral Test of CFCS against MDCK Cells
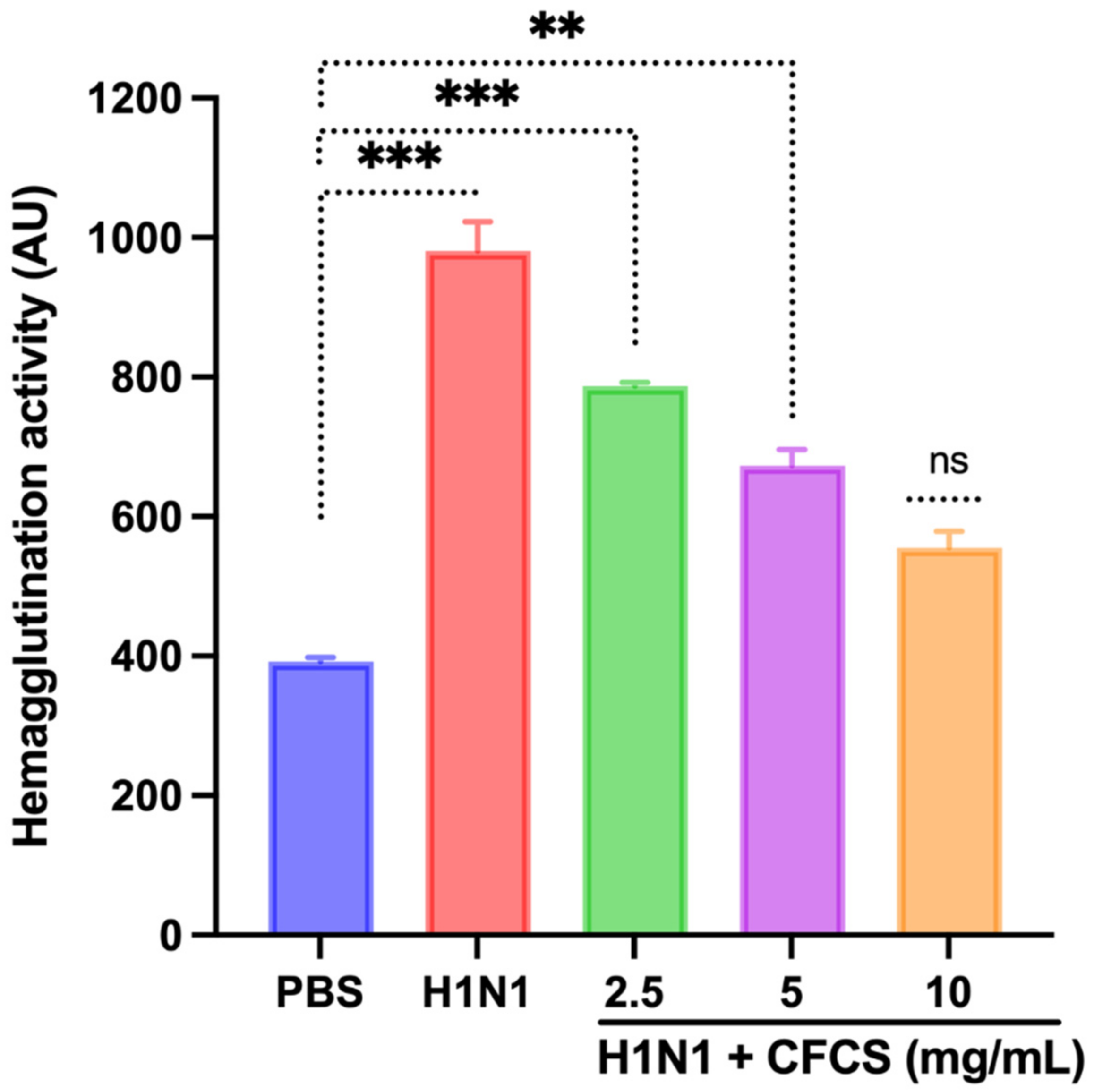
2.6. Hemagglutination Inhibition
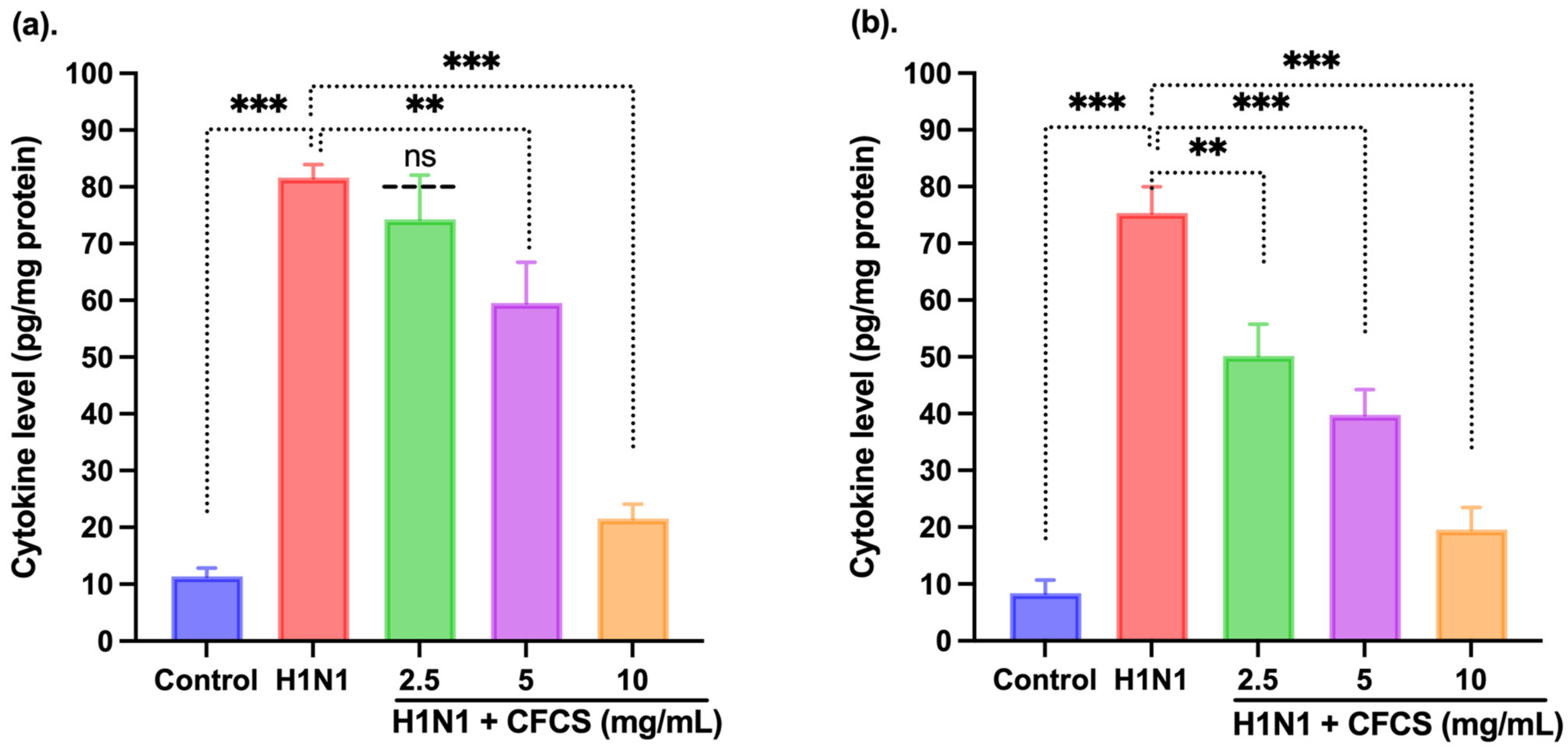
2.7. Cytokine Profile
2.8. Statistical Analysis
3. Results and Discussion
3.1. Isolation, Purification, and Identification of Probiotic LAB Isolate
3.2. Cytotoxic Assessment of CFCS against MDCK Cells
3.3. In Vitro Antiviral Activity of CFCS Was Evaluated Using MDCK Cells
3.4. Antiviral Activity of CFCS Was Evaluated Using SPF Embryonated Eggs and Hemagglutination Assay
3.5. Estimation of Cytokines by ELISA
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Drouault, S.; Corthier, G. Health Effects of Lactic Acid Bacteria Ingested in Fermented Milk. Vet. Res. 2001, 32, 101–117. [Google Scholar] [CrossRef] [Green Version]
- Nagai, T.; Makino, S.; Ikegami, S.; Itoh, H.; Yamada, H. Effects of Oral Administration of Yogurt Fermented with Lactobacillus Delbrueckii Ssp. Bulgaricus OLL1073R-1 and Its Exopolysaccharides against Influenza Virus Infection in Mice. Int. Immunopharmacol. 2011, 11, 2246–2250. [Google Scholar] [CrossRef] [PubMed]
- Thompson, W.W.; Shay, D.K.; Weintraub, E.; Brammer, L.; Bridges, C.B.; Cox, N.J.; Fukuda, K. Influenza-Associated Hospitalizations in the United States. J. Am. Med. Assoc. 2004, 292, 1333–1340. [Google Scholar] [CrossRef]
- Boubk, T.A.F.; Sabir, J.S.M.; Abo-Aba, S.E.M.; Sabir, M.J.; Hajrah, N.H.; Alhibshi, A.M.; Abu-Zeid, M.; Omri, A.E.; Sabir, M.J.; Mattar, E.H.; et al. Molecular Generation and Characterization of an Efficient Recombinant Vaccine for Avian Influenza A/H5N8 in Saudi Arabia. Trop. J. Pharm. Res. 2019, 18, 2279–2285. [Google Scholar] [CrossRef]
- Hancock, K.; Veguilla, V.; Lu, X.; Zhong, W.; Butler, E.N.; Sun, H.; Liu, F.; Dong, L.; DeVos, J.R.; Gargiullo, P.M.; et al. Cross-Reactive Antibody Responses to the 2009 Pandemic H1N1 Influenza Virus. N. Engl. J. Med. 2009, 361, 1945–1952. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, L.R.; Wloch, M.K.; Ye, M.; Reyes, L.R.; Boutsaboualoy, S.; Dunne, C.E.; Chaplin, J.A.; Rusalov, D.; Rolland, A.P.; Fisher, C.L.; et al. Phase 1 Clinical Trials of the Safety and Immunogenicity of Adjuvanted Plasmid DNA Vaccines Encoding Influenza A Virus H5 Hemagglutinin. Vaccine 2010, 28, 2565–2572. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.; Zhu, X.; Li, Y.; Shi, M.; Zhang, J.; Bourgeois, M.; Yang, H.; Chen, X.; Recuenco, S.; Gomez, J.; et al. New World Bats Harbor Diverse Influenza A Viruses. PLoS Pathog. 2013, 9, e1003657. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Long, J.K.; Mossad, S.B.; Goldman, M.P. Antiviral Agents for Treating Influenza. Clevel. Clin. J. Med. 2000, 67, 92–95. [Google Scholar] [CrossRef]
- Beigel, J.; Bray, M. Current and Future Antiviral Therapy of Severe Seasonal and Avian Influenza. Antivir. Res. 2008, 78, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Díaz, D.A.; Muñoz, A.L.; Tavera-Rodríguez, P.; Herrera-Sepúlveda, M.T.; Ruiz-Moreno, H.A.; Laiton-Donato, K.; Franco-Muñoz, C.; Pelaez-Carvajal, D.; Cuellar, D.; Muñoz-Suarez, A.M.; et al. Low Neutralizing Antibody Titers against the Mu Variant of SARS-CoV-2 in 31 BNT162b2 Vaccinated Individuals in Colombia. Vaccines 2022, 10, 180. [Google Scholar] [CrossRef]
- Khong, K.-W.; Liu, D.; Leung, K.-Y.; Lu, L.; Lam, H.-Y.; Chen, L.; Chan, P.-C.; Lam, H.-M.; Xie, X.; Zhang, R.; et al. Antibody Response of Combination of BNT162b2 and CoronaVac Platforms of COVID-19 Vaccines against Omicron Variant. Vaccines 2022, 10, 160. [Google Scholar] [CrossRef] [PubMed]
- Meggiolaro, A.; Sane Schepisi, M.; Nikolaidis, G.F.; Mipatrini, D.; Siddu, A.; Rezza, G. Effectiveness of Vaccination against SARS-CoV-2 Infection in the Pre-Delta Era: A Systematic Review and Meta-Analysis. Vaccines 2022, 10, 157. [Google Scholar] [CrossRef]
- Bahreini-Esfahani, N.; Moravejolahkami, A.R. Can Synbiotic Dietary Pattern Predict Lactobacillales Strains in Breast Milk? Breastfeed. Med. 2020, 15, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Parvez, S.; Malik, K.A.; Ah Kang, S.; Kim, H.Y. Probiotics and Their Fermented Food Products Are Beneficial for Health. J. Appl. Microbiol. 2006, 100, 1171–1185. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Lone, J.B.; Bajpai, V.K.; Paek, W.K.; Lim, J. ZIKA Virus: An Emerging Worldwide Threat. Front. Microbiol. 2017, 8, 1417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rather, I.A.; Choi, K.-H.; Bajpai, V.K.; Park, Y.-H. Antiviral Mode of Action of Lactobacillus Plantarum Yml009 on Influenza Virus H1n1. Bangladesh J. Pharmacol. 2015, 10, 475–482. [Google Scholar] [CrossRef] [Green Version]
- Bajpai, V.K.; Chandra, V.; Kim, N.-H.; Rai, R.; Kumar, P.; Kim, K.; Aeron, A.; Kang, S.C.; Maheshwari, D.K.; Na, M.; et al. Ghost Probiotics with a Combined Regimen: A Novel Therapeutic Approach against the Zika Virus, an Emerging World Threat. Crit. Rev. Biotechnol. 2018, 38, 438–454. [Google Scholar] [CrossRef] [PubMed]
- Alqazlan, N.; Alizadeh, M.; Boodhoo, N.; Taha-Abdelaziz, K.; Nagy, E.; Bridle, B.; Sharif, S. Probiotic Lactobacilli Limit Avian Influenza Virus Subtype H9N2 Replication in Chicken Cecal Tonsil Mononuclear Cells. Vaccines 2020, 8, 605. [Google Scholar] [CrossRef] [PubMed]
- Paknahad, Z.; Moravejolahkami, A.R. Probiotics Against Viruses; COVID-19 Is a Paper Tiger: A Systematic Review. Endocr. Metab. Immune Disord. Drug Targets 2020, 21, 1252–1260. [Google Scholar] [CrossRef] [PubMed]
- Mohsin, M.; Zhang, Z.; Yin, G. Effect of Probiotics on the Performance and Intestinal Health of Broiler Chickens Infected with Eimeria Tenella. Vaccines 2022, 10, 97. [Google Scholar] [CrossRef]
- Mangrolia, U.; Osborne, J.W. Probiotics in Counteracting the Role of Neutrophils in Cancer Metastasis. Vaccines 2021, 9, 1306. [Google Scholar] [CrossRef] [PubMed]
- Taghinezhad-S, S.; Mohseni, A.H.; Bermúdez-Humarán, L.G.; Casolaro, V.; Cortes-Perez, N.G.; Keyvani, H.; Simal-Gandara, J. Probiotic-Based Vaccines May Provide Effective Protection against COVID-19 Acute Respiratory Disease. Vaccines 2021, 9, 466. [Google Scholar] [CrossRef]
- Dunne, C.; O’Mahony, L.; Murphy, L.; Thornton, G.; Morrissey, D.; O’Halloran, S.; Feeney, M.; Flynn, S.; Fitzgerald, G.; Daly, C.; et al. In Vitro Selection Criteria for Probiotic Bacteria of Human Origin: Correlation with in Vivo Findings. Am. J. Clin. Nutr. 2001, 73 (Suppl. 2), 386S–392S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nomura, M.; Kobayashi, M.; Narita, T.; Kimoto-Nira, H.; Okamoto, T. Phenotypic and Molecular Characterization of Lactococcus Lactis from Milk and Plants. J. Appl. Microbiol. 2006, 101, 396–405. [Google Scholar] [CrossRef] [PubMed]
- Seo, B.J.; Rather, I.A.; Kumar, V.J.R.; Choi, U.H.; Moon, M.R.; Lim; Park, Y.H. Evaluation of Leuconostoc Mesenteroides YML003 as a Probiotic against Low-Pathogenic Avian Influenza (H9N2) Virus in Chickens. J. Appl. Microbiol. 2012, 113, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.N.; Lee, D.H.; Lee, Y.N.; Park, J.K.; Yuk, S.S.; Yang, S.Y.; Lee, H.J.; Woo, S.H.; Kim, H.M.; Lee, J.B.; et al. Intranasal Administration of Live Lactobacillus Species Facilitates Protection against Influenza Virus Infection in Mice. Antivir. Res. 2012, 93, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, V.K.; Rather, I.A.; Majumder, R.; Alshammari, F.H.; Nam, G.-J.; Park, Y.-H. Characterization and Antibacterial Mode of Action of Lactic Acid Bacterium Leuconostoc Mesenteroides HJ69 from Kimchi. J. Food Biochem. 2017, 41, e12290. [Google Scholar] [CrossRef]
- Rather, I.A.; Bajpai, V.K.; Huh, Y.S.; Han, Y.-K.; Bhat, E.A.; Lim, J.; Paek, W.K.; Park, Y.-H. Probiotic Lactobacillus Sakei ProBio-65 Extract Ameliorates the Severity of Imiquimod Induced Psoriasis-like Skin Inflammation in a Mouse Model. Front. Microbiol. 2018, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Rather, I.A.; Kim, H.; Kim, S.; Kim, T.; Jang, J.; Seo, J.; Lim, J.; Park, Y.-H. A Double-Blind, Placebo Controlled-Trial of a Probiotic Strain Lactobacillus Sakei Probio-65 for the Prevention of Canine Atopic Dermatitis. J. Microbiol. Biotechnol. 2015, 25, 1966–1969. [Google Scholar] [CrossRef]
- Bajpai, V.K.; Rather, I.A.; Park, Y.-H. Partially Purified Exo-Polysaccharide from Lactobacillus Sakei Probio 65 with Antioxidant, α-Glucosidase and Tyrosinase Inhibitory Potential. J. Food Biochem. 2016, 40, 264–274. [Google Scholar] [CrossRef]
- Rather, I.A.; Choi, S.-B.; Kamli, M.R.; Hakeem, K.R.; Sabir, J.S.M.; Park, Y.-H.; Hor, Y.-Y. Potential Adjuvant Therapeutic Effect of Lactobacillus Plantarum Probio-88 Postbiotics against SARS-CoV-2. Vaccines 2021, 9, 1067. [Google Scholar] [CrossRef] [PubMed]
- Hanniffy, S.; Wiedermann, U.; Repa, A.; Mercenier, A.; Daniel, C.; Fioramonti, J.; Tlaskolova, H.; Kozakova, H.; Israelsen, H.; Madsen, S.; et al. Potential and Opportunities for Use of Recombinant Lactic Acid Bacteria in Human Health. Adv. Appl. Microbiol. 2004, 56, 1–64. [Google Scholar] [CrossRef] [PubMed]
- Fujiwara, D.; Inoue, S.; Wakabayashi, H.; Fujii, T. The Anti-Allergic Effects of Lactic Acid Bacteria Are Strain Dependent and Mediated by Effects on Both Th1/Th2 Cytokine Expression and Balance. Int. Arch. Allergy Immunol. 2004, 135, 205–215. [Google Scholar] [CrossRef]
- Nagao, F.; Nakayama, M.; Muto, T.; Okumura, K. Effects of a Fermented Milk Drink Containing Lactobacillus Casei Strain Shirota on the Immune System in Healthy Human Subjects. Biosci. Biotechnol. Biochem. 2000, 64, 2706–2708. [Google Scholar] [CrossRef] [PubMed]
- Bohlouli, J.; Namjoo, I.; Borzoo-Isfahani, M.; Hojjati Kermani, M.A.; Balouch Zehi, Z.; Moravejolahkami, A.R. Effect of Probiotics on Oxidative Stress and Inflammatory Status in Diabetic Nephropathy: A Systematic Review and Meta-Analysis of Clinical Trials. Heliyon 2021, 7, e05925. [Google Scholar] [CrossRef]
- Rather, I.A.; Bajpai, V.K.; Kumar, S.; Lim, J.; Paek, W.K.; Park, Y.-H. Probiotics and Atopic Dermatitis: An Overview. Front. Microbiol. 2016, 7, 507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rather, I.A.; Kim, B.-C.; Lew, L.-C.; Cha, S.-K.; Lee, J.H.; Nam, G.-J.; Majumder, R.; Lim, J.; Lim, S.-K.; Seo, Y.-J.; et al. Oral Administration of Live and Dead Cells of Lactobacillus Sakei ProBio65 Alleviated Atopic Dermatitis in Children and Adolescents: A Randomized, Double-Blind, and Placebo-Controlled Study. Probiotics Antimicrob. Proteins 2021, 13, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Majumder, R.; Alshammari, F.H.; Park, J.G.; Bajpai, V.K. Review-Ulcerative Colitis and Probiotics: An Overview. Pak. J. Pharm. Sci. 2016, 29, 1877–1880. [Google Scholar]
- Majumder, R.; Rather, I.A.; Bajpai, V.K.; Park, Y.-H. In Vitro Antiviral Activity of Lactobacillus Plantarum Using SPF Embryonated Eggs and Hemagglutination Assay. Bangladesh J. Pharmacol. 2015, 10, 688. [Google Scholar] [CrossRef] [Green Version]
- Koh, W.Y.; Utra, U.; Ahmad, R.; Rather, I.A.; Park, Y.-H. Evaluation of Probiotic Potential and Anti-Hyperglycemic Properties of a Novel Lactobacillus Strain Isolated from Water Kefir Grains. Food Sci. Biotechnol. 2018, 27, 1369–1376. [Google Scholar] [CrossRef]
- Paray, B.A.; Rather, I.A.; Al-Sadoon, M.K.; Fanar Hamad, A.-S. Pharmaceutical Significance of Leuconostoc Mesenteroides KS-TN11 Isolated from Nile Tilapia, Oreochromis Niloticus. Saudi Pharm. J. 2018, 26, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, V.K.; Majumder, R.; Rather, I.A.; Nam, G.-J.; Park, Y.-H. Molecular Characterization of Lactobacillus Plantarum YML016 with Anti-Diabetic, Anti-Melanogenic and Anti-Viral Efficacy. Natl. Acad. Sci. Lett. 2018, 41, 301–305. [Google Scholar] [CrossRef]
- Corr, S.C.; Gahan, C.G.M.; Hill, C. Impact of Selected Lactobacillus and Bifidobacterium Species on Listeria Monocytogenes Infection and the Mucosal Immune Response. FEMS Immunol. Med. Microbiol. 2007, 50, 380–388. [Google Scholar] [CrossRef] [Green Version]
- Ağagündüz, D.; Yılmaz, B.; Şahin, T.Ö.; Güneşliol, B.E.; Ayten, Ş.; Russo, P.; Spano, G.; Rocha, J.M.; Bartkiene, E.; Özogul, F. Dairy Lactic Acid Bacteria and Their Potential Function in Dietetics: The Food–Gut-Health Axis. Foods 2021, 10, 3099. [Google Scholar] [CrossRef]
- Makino, S.; Ikegami, S.; Kume, A.; Horiuchi, H.; Sasaki, H.; Orii, N. Reducing the Risk of Infection in the Elderly by Dietary Intake of Yoghurt Fermented with Lactobacillus Delbrueckii Ssp. Bulgaricus OLL1073R-1. Br. J. Nutr. 2010, 104, 998–1006. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kanauchi, O.; Andoh, A.; AbuBakar, S.; Yamamoto, N. Probiotics and Paraprobiotics in Viral Infection: Clinical Application and Effects on the Innate and Acquired Immune Systems. Curr. Pharm. Des. 2018, 24, 710–717. [Google Scholar] [CrossRef]
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; di Napoli, R. Features, Evaluation and Treatment Coronavirus (COVID-19); StatPearls-NCBI Bookshelf: Treasure Island, FL, USA, 2020. [Google Scholar]
- Park, M.K.; NGO, V.; Kwon, Y.M.; Lee, Y.T.; Yoo, S.; Cho, Y.H.; Hong, S.M.; Hwang, H.S.; Ko, E.J.; Jung, Y.J.; et al. Lactobacillus Plantarum DK119 as a Probiotic Confers Protection against Influenza Virus by Modulating Innate Immunity. PLoS ONE 2013, 8, e75368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reed, L.J.; Muench, H. A Simple Method of Estimating Fifty per Cent Endpoints. Am. J. Epidemiol. 1938, 27. [Google Scholar] [CrossRef]
- Sunmola, A.A.; Ogbole, O.O.; Faleye, T.O.C.; Adetoye, A.; Adeniji, J.A.; Ayeni, F.A. Antiviral Potentials of Lactobacillus Plantarum, Lactobacillus Amylovorus, and Enterococcus Hirae against Selected Enterovirus. Folia Microbiol. 2019, 64, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-Y.; Tsao, S.-P. Long Term Supplementation of Lactobacillus Plantarum PS128 Attenuated High-Intensity Exercise Induced Acute-Phase Inflammation in Triathletes. Curr. Dev. Nutr. 2020, 4, 1564. [Google Scholar] [CrossRef]
- Prakoeswa, C.R.S.; Herwanto, N.; Prameswari, R.; Astari, L.; Sawitri, S.; Hidayati, A.N.; Indramaya, D.M.; Kusumowidagdo, E.R.; Surono, I.S. Lactobacillus Plantarum IS-10506 Supplementation Reduced SCORAD in Children with Atopic Dermatitis. Benef. Microbes 2017, 8, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Seo, B.J.; Kumar, V.J.R.; Choi, U.-H.; Choi, K.-H.; Lim, J.; Park, Y.-H. Biopreservative Potential of Lactobacillus Plantarum YML007 and Efficacy as a Replacement for Chemical Preservatives in Animal Feed. Food Sci. Biotechnol. 2014, 23, 195–200. [Google Scholar] [CrossRef]
- Ahmad Rather, I.; Seo, B.J.; Rejish Kumar, V.J.; Choi, U.H.; Choi, K.H.; Lim, J.H.; Park, Y.H. Isolation and Characterization of a Proteinaceous Antifungal Compound from Lactobacillus Plantarum YML007 and Its Application as a Food Preservative. Lett. Appl. Microbiol. 2013, 57, 69–76. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rather, I.A.; Kamli, M.R.; Sabir, J.S.M.; Paray, B.A. Potential Antiviral Activity of Lactiplantibacillus plantarum KAU007 against Influenza Virus H1N1. Vaccines 2022, 10, 456. https://doi.org/10.3390/vaccines10030456
Rather IA, Kamli MR, Sabir JSM, Paray BA. Potential Antiviral Activity of Lactiplantibacillus plantarum KAU007 against Influenza Virus H1N1. Vaccines. 2022; 10(3):456. https://doi.org/10.3390/vaccines10030456
Chicago/Turabian StyleRather, Irfan A., Majid Rasool Kamli, Jamal S. M. Sabir, and Bilal Ahmad Paray. 2022. "Potential Antiviral Activity of Lactiplantibacillus plantarum KAU007 against Influenza Virus H1N1" Vaccines 10, no. 3: 456. https://doi.org/10.3390/vaccines10030456
APA StyleRather, I. A., Kamli, M. R., Sabir, J. S. M., & Paray, B. A. (2022). Potential Antiviral Activity of Lactiplantibacillus plantarum KAU007 against Influenza Virus H1N1. Vaccines, 10(3), 456. https://doi.org/10.3390/vaccines10030456






