A Phase I/II Clinical Trial of Intradermal, Controllable Self-Replicating Ribonucleic Acid Vaccine EXG-5003 against SARS-CoV-2
Abstract
:1. Introduction
2. Materials and Methods
2.1. EXG-5003 Vaccine
2.2. Trial Design and Participants
2.3. Study Procedures
2.4. Humoral Immunity Assays: Anti-RBD Antibody
2.5. Humoral Immunity Assays: Cell-Free SARS-CoV-2 Neutralization Assay
2.6. Humoral Immunity Assays: SARS-CoV-2 Spike Pseudotyped Lentivirus Assay
2.7. Cellular Immunity: FluoroSpot Assay
2.8. Safety Assessment
2.9. Statistical Analysis
3. Results
3.1. Participants
3.2. Safety
3.3. Short-Term Humoral Immunity Elicited by EXG-5003

3.4. Short-Term Cellular Immunity Elicited by EXG-5003
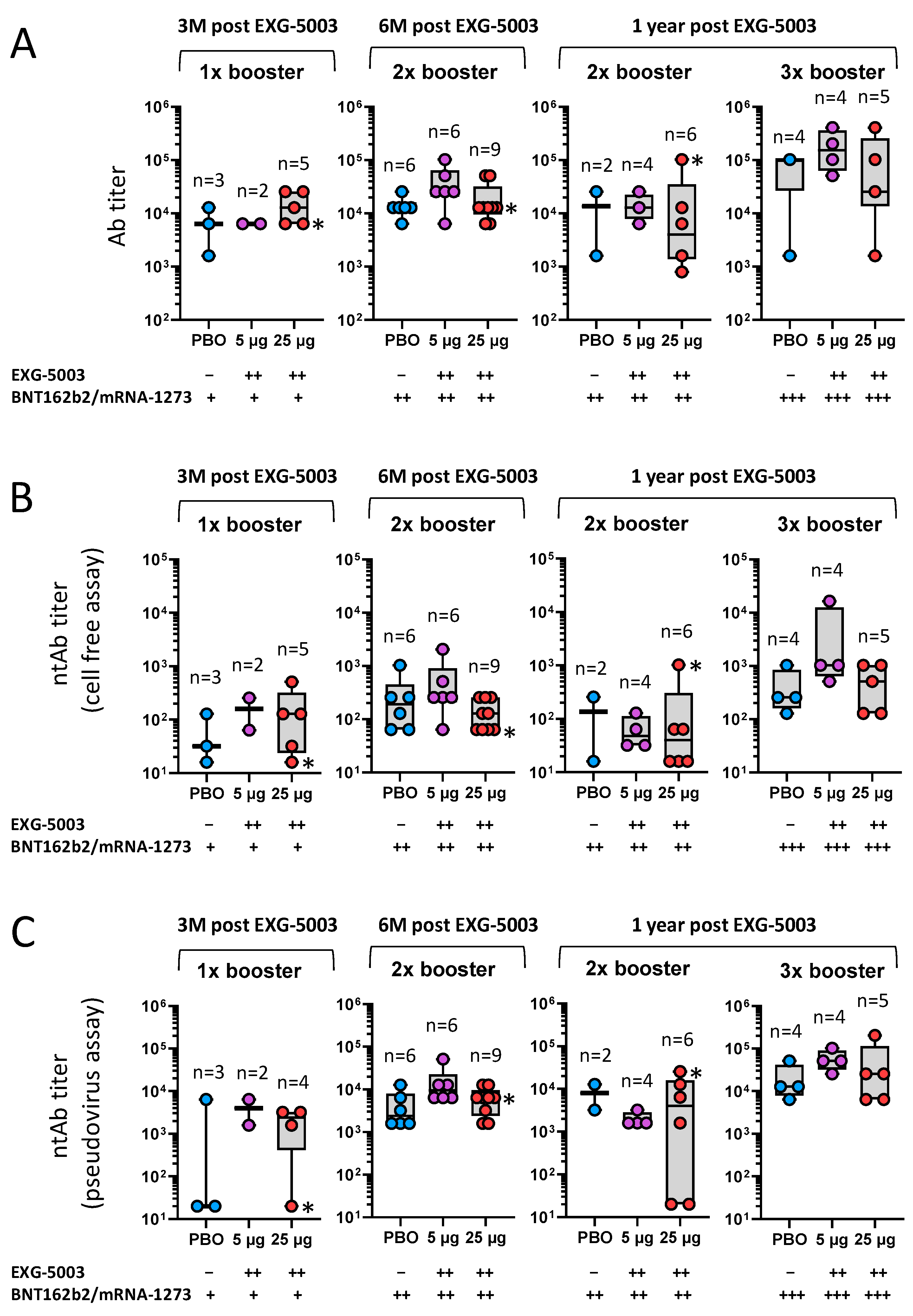
3.5. Long-Term Humoral Immunity Elicited by EXG-5003 following Administration of Licensed Vaccines
3.6. Long-Term Cellular Immunity Elicited by EXG-5003 following Administration of Licensed Vaccines
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Coronavirus Dashboard. Available online: https://covid19.who.int/ (accessed on 17 September 2023).
- Excler, J.L.; Saville, M.; Privor-Dumm, L.; Gilbert, S.; Hotez, P.J.; Thompson, D.; Abdool-Karim, S.; Kim, J.H. Factors, enablers and challenges for COVID-19 vaccine development. BMJ Glob. Health 2023, 8, e011879. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Perez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Moss, P. The T cell immune response against SARS-CoV-2. Nat. Immunol. 2022, 23, 186–193. [Google Scholar] [CrossRef]
- Sette, A.; Sidney, J.; Crotty, S. T Cell Responses to SARS-CoV-2. Annu. Rev. Immunol. 2023, 41, 343–373. [Google Scholar] [CrossRef] [PubMed]
- Wherry, E.J.; Barouch, D.H. T cell immunity to COVID-19 vaccines. Science 2022, 377, 821–822. [Google Scholar] [CrossRef]
- Anderson, E.J.; Rouphael, N.G.; Widge, A.T.; Jackson, L.A.; Roberts, P.C.; Makhene, M.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; Pruijssers, A.J.; et al. Safety and Immunogenicity of SARS-CoV-2 mRNA-1273 Vaccine in Older Adults. N. Engl. J. Med. 2020, 383, 2427–2438. [Google Scholar] [CrossRef] [PubMed]
- Nathan, A.; Rossin, E.J.; Kaseke, C.; Park, R.J.; Khatri, A.; Koundakjian, D.; Urbach, J.M.; Singh, N.K.; Bashirova, A.; Tano-Menka, R.; et al. Structure-guided T cell vaccine design for SARS-CoV-2 variants and sarbecoviruses. Cell 2021, 184, 4401–4413.e4410. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA Vaccine against SARS-CoV-2—Preliminary Report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef]
- Munro, A.P.S.; Feng, S.; Janani, L.; Cornelius, V.; Aley, P.K.; Babbage, G.; Baxter, D.; Bula, M.; Cathie, K.; Chatterjee, K.; et al. Safety, immunogenicity, and reactogenicity of BNT162b2 and mRNA-1273 COVID-19 vaccines given as fourth-dose boosters following two doses of ChAdOx1 nCoV-19 or BNT162b2 and a third dose of BNT162b2 (COV-BOOST): A multicentre, blinded, phase 2, randomised trial. Lancet Infect. Dis. 2022, 22, 1131–1141. [Google Scholar] [CrossRef]
- Ozbay Kurt, F.G.; Lepper, A.; Gerhards, C.; Roemer, M.; Lasser, S.; Arkhypov, I.; Bitsch, R.; Bugert, P.; Altevogt, P.; Gouttefangeas, C.; et al. Booster dose of mRNA vaccine augments waning T cell and antibody responses against SARS-CoV-2. Front. Immunol. 2022, 13, 1012526. [Google Scholar] [CrossRef]
- Hernandez, S.P.A.; Hersby, D.S.; Munk, K.K.; Tamhane, T.; Trubach, D.; Tagliamonte, M.; Buonaguro, L.; Gang, A.O.; Hadrup, S.R.; Saini, S.K. Three doses of BNT162b2 COVID-19 mRNA vaccine establish long-lasting CD8(+) T cell immunity in CLL and MDS patients. Front. Immunol. 2022, 13, 1035344. [Google Scholar] [CrossRef] [PubMed]
- Reinscheid, M.; Luxenburger, H.; Karl, V.; Graeser, A.; Giese, S.; Ciminski, K.; Reeg, D.B.; Oberhardt, V.; Roehlen, N.; Lang-Meli, J.; et al. COVID-19 mRNA booster vaccine induces transient CD8+ T effector cell responses while conserving the memory pool for subsequent reactivation. Nat. Commun. 2022, 13, 4631. [Google Scholar] [CrossRef] [PubMed]
- Agrati, C.; Castilletti, C.; Goletti, D.; Sacchi, A.; Bordoni, V.; Mariotti, D.; Notari, S.; Matusali, G.; Meschi, S.; Petrone, L.; et al. Persistent Spike-specific T cell immunity despite antibody reduction after 3 months from SARS-CoV-2 BNT162b2-mRNA vaccine. Sci. Rep. 2022, 12, 6687. [Google Scholar] [CrossRef] [PubMed]
- Blakney, A.K.; Ip, S.; Geall, A.J. An Update on Self-Amplifying mRNA Vaccine Development. Vaccines 2021, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- McKay, P.F.; Hu, K.; Blakney, A.K.; Samnuan, K.; Brown, J.C.; Penn, R.; Zhou, J.; Bouton, C.R.; Rogers, P.; Polra, K.; et al. Self-amplifying RNA SARS-CoV-2 lipid nanoparticle vaccine candidate induces high neutralizing antibody titers in mice. Nat. Commun. 2020, 11, 3523. [Google Scholar] [CrossRef] [PubMed]
- de Alwis, R.; Gan, E.S.; Chen, S.; Leong, Y.S.; Tan, H.C.; Zhang, S.L.; Yau, C.; Low, J.G.H.; Kalimuddin, S.; Matsuda, D.; et al. A single dose of self-transcribing and replicating RNA-based SARS-CoV-2 vaccine produces protective adaptive immunity in mice. Mol. Ther. 2021, 29, 1970–1983. [Google Scholar] [CrossRef] [PubMed]
- Erasmus, J.H.; Khandhar, A.P.; O’Connor, M.A.; Walls, A.C.; Hemann, E.A.; Murapa, P.; Archer, J.; Leventhal, S.; Fuller, J.T.; Lewis, T.B.; et al. An Alphavirus-derived replicon RNA vaccine induces SARS-CoV-2 neutralizing antibody and T cell responses in mice and nonhuman primates. Sci. Transl. Med. 2020, 12, eabc9396. [Google Scholar] [CrossRef]
- Langereis, M.A.; Albulescu, I.C.; Stammen-Vogelzangs, J.; Lambregts, M.; Stachura, K.; Miller, S.; Bosco-Lauth, A.M.; Hartwig, A.E.; Porter, S.M.; Allen, M.; et al. An alphavirus replicon-based vaccine expressing a stabilized Spike antigen induces protective immunity and prevents transmission of SARS-CoV-2 between cats. NPJ Vaccines 2021, 6, 122. [Google Scholar] [CrossRef]
- Szurgot, I.; Hanke, L.; Sheward, D.J.; Vidakovics, L.P.; Murrell, B.; McInerney, G.M.; Liljestrom, P. DNA-launched RNA replicon vaccines induce potent anti-SARS-CoV-2 immune responses in mice. Sci. Rep. 2021, 11, 3125. [Google Scholar] [CrossRef]
- Rappaport, A.R.; Hong, S.J.; Scallan, C.D.; Gitlin, L.; Akoopie, A.; Boucher, G.R.; Egorova, M.; Espinosa, J.A.; Fidanza, M.; Kachura, M.A.; et al. Low-dose self-amplifying mRNA COVID-19 vaccine drives strong protective immunity in non-human primates against SARS-CoV-2 infection. Nat. Commun. 2022, 13, 3289. [Google Scholar] [CrossRef]
- Komori, M.; Nogimori, T.; Morey, A.L.; Sekida, T.; Ishimoto, K.; Hassett, M.R.; Masuta, Y.; Ode, H.; Tamura, T.; Suzuki, R.; et al. saRNA vaccine expressing membrane-anchored RBD elicits broad and durable immunity against SARS-CoV-2 variants of concern. Nat. Commun. 2023, 14, 2810. [Google Scholar] [CrossRef] [PubMed]
- Akahata, W.; Sekida, T.; Nogimori, T.; Ode, H.; Tamura, T.; Kono, K.; Kazami, Y.; Washizaki, A.; Masuta, Y.; Suzuki, R.; et al. Safety and immunogenicity of SARS-CoV-2 self-amplifying RNA vaccine expressing an anchored RBD: A randomized, observer-blind phase 1 study. Cell Rep. Med. 2023, 4, 101134. [Google Scholar] [CrossRef]
- Elliott, T.; Cheeseman, H.M.; Evans, A.B.; Day, S.; McFarlane, L.R.; O’Hara, J.; Kalyan, M.; Amini, F.; Cole, T.; Winston, A.; et al. Enhanced immune responses following heterologous vaccination with self-amplifying RNA and mRNA COVID-19 vaccines. PLoS Pathog. 2022, 18, e1010885. [Google Scholar] [CrossRef] [PubMed]
- Low, J.G.; de Alwis, R.; Chen, S.; Kalimuddin, S.; Leong, Y.S.; Mah, T.K.L.; Yuen, N.; Tan, H.C.; Zhang, S.L.; Sim, J.X.Y.; et al. A phase I/II randomized, double-blinded, placebo-controlled trial of a self-amplifying COVID-19 mRNA vaccine. NPJ Vaccines 2022, 7, 161. [Google Scholar] [CrossRef] [PubMed]
- Amano, T.; Yu, H.; Amano, M.; Leyder, E.; Badiola, M.; Ray, P.; Kim, J.; Ko, A.C.; Achour, A.; Weng, N.P.; et al. Controllable self-replicating RNA vaccine delivered intradermally elicits predominantly cellular immunity. iScience 2023, 26, 106335. [Google Scholar] [CrossRef]
- Dugan, H.L.; Henry, C.; Wilson, P.C. Aging and influenza vaccine-induced immunity. Cell Immunol. 2020, 348, 103998. [Google Scholar] [CrossRef]
- Hickling, J.K.; Jones, K.R.; Friede, M.; Zehrung, D.; Chen, D.; Kristensen, D. Intradermal delivery of vaccines: Potential benefits and current challenges. Bull. World Health Organ. 2011, 89, 221–226. [Google Scholar] [CrossRef]
- Kiecker, F.; Streitz, M.; Ay, B.; Cherepnev, G.; Volk, H.D.; Volkmer-Engert, R.; Kern, F. Analysis of antigen-specific T-cell responses with synthetic peptides—What kind of peptide for which purpose? Hum. Immunol. 2004, 65, 523–536. [Google Scholar] [CrossRef]
- Pollock, K.M.; Cheeseman, H.M.; Szubert, A.J.; Libri, V.; Boffito, M.; Owen, D.; Bern, H.; O’Hara, J.; McFarlane, L.R.; Lemm, N.M.; et al. Safety and immunogenicity of a self-amplifying RNA vaccine against COVID-19: COVAC1, a phase I, dose-ranging trial. eClinicalMedicine 2022, 44, 101262. [Google Scholar] [CrossRef]
- Mansanguan, S.; Charunwatthana, P.; Piyaphanee, W.; Dechkhajorn, W.; Poolcharoen, A.; Mansanguan, C. Cardiovascular Manifestation of the BNT162b2 mRNA COVID-19 Vaccine in Adolescents. Trop. Med. Infect. Dis. 2022, 7, 196. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.N.; Chen, Y.S.; Hsu, C.C.; Hua, Y.C.; Tseng, W.C.; Lu, C.W.; Lin, M.T.; Chen, C.A.; Wu, M.H.; Chen, Y.T.; et al. Changes of ECG parameters after BNT162b2 vaccine in the senior high school students. Eur. J. Pediatr. 2023, 182, 1155–1162. [Google Scholar] [CrossRef] [PubMed]






| Cohort 1 | Cohort 1 | Cohort 2 | Cohort 2 | |
|---|---|---|---|---|
| EXG-5003 5 µg | Placebo | EXG-5003 25 µg | Placebo | |
| (n = 16) | (n = 4) | (n = 16) | (n = 4) | |
| Race | ||||
| Asian | 16 (100%) | 4 (100%) | 16 (100%) | 4 (100%) |
| Gender | ||||
| Male | 10 (62.5%) | 3 (75%) | 7 (43.8%) | 4 (100%) |
| Female | 6 (37.5%) | 1 (25%) | 9 (56.2%) | 0 (0%) |
| Age (year) | ||||
| Mean | 39.375 | 46.75 | 39.562 | 42.75 |
| SD | 8.405 | 7.932 | 10.354 | 4.272 |
| Median | 39.5 | 48.0 | 43.0 | 44.0 |
| Min–Max | 22–53 | 36–55 | 22–55 | 37–46 |
| Weight (kg) | ||||
| Mean | 66.862 | 64.8 | 63.094 | 74.625 |
| SD | 18.756 | 4.721 | 12.837 | 10.633 |
| Median | 61.5 | 65.85 | 63.05 | 72.9 |
| Min–Max | 46.9–107.8 | 58.2–69.3 | 47.5–84.7 | 65–87.7 |
| Height (cm) | ||||
| Mean | 169.200 | 169.375 | 164.162 | 169.175 |
| SD | 8.791 | 6.874 | 8.804 | 5.142 |
| Median | 170.550 | 172.55 | 164.550 | 170.35 |
| Min–Max | 154.2–183.1 | 159.1–173.3 | 152–180 | 162–174 |
| BMI (kg/m2) | ||||
| Mean | 23.265 | 22.575 | 23.301 | 26.173 |
| SD | 6.072 | 0.534 | 3.680 | 4.374 |
| Median | 21.484 | 22.593 | 22.637 | 26.269 |
| Min–Max | 17.849–39.789 | 22.039–23.075 | 16.553–29.938 | 22.13–30.026 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koseki, T.; Teramachi, M.; Koga, M.; Ko, M.S.H.; Amano, T.; Yu, H.; Amano, M.; Leyder, E.; Badiola, M.; Ray, P.; et al. A Phase I/II Clinical Trial of Intradermal, Controllable Self-Replicating Ribonucleic Acid Vaccine EXG-5003 against SARS-CoV-2. Vaccines 2023, 11, 1767. https://doi.org/10.3390/vaccines11121767
Koseki T, Teramachi M, Koga M, Ko MSH, Amano T, Yu H, Amano M, Leyder E, Badiola M, Ray P, et al. A Phase I/II Clinical Trial of Intradermal, Controllable Self-Replicating Ribonucleic Acid Vaccine EXG-5003 against SARS-CoV-2. Vaccines. 2023; 11(12):1767. https://doi.org/10.3390/vaccines11121767
Chicago/Turabian StyleKoseki, Takenao, Mayumi Teramachi, Minako Koga, Minoru S. H. Ko, Tomokazu Amano, Hong Yu, Misa Amano, Erica Leyder, Maria Badiola, Priyanka Ray, and et al. 2023. "A Phase I/II Clinical Trial of Intradermal, Controllable Self-Replicating Ribonucleic Acid Vaccine EXG-5003 against SARS-CoV-2" Vaccines 11, no. 12: 1767. https://doi.org/10.3390/vaccines11121767
APA StyleKoseki, T., Teramachi, M., Koga, M., Ko, M. S. H., Amano, T., Yu, H., Amano, M., Leyder, E., Badiola, M., Ray, P., Kim, J., Ko, A. C., Achour, A., Weng, N.-p., Imai, T., Yoshida, H., Taniuchi, S., Shintani, A., Fujigaki, H., ... Doi, Y. (2023). A Phase I/II Clinical Trial of Intradermal, Controllable Self-Replicating Ribonucleic Acid Vaccine EXG-5003 against SARS-CoV-2. Vaccines, 11(12), 1767. https://doi.org/10.3390/vaccines11121767





