Recombinant Attenuated Edwardsiella piscicida Vaccine Displaying Regulated Lysis to Confer Biological Containment and Protect Catfish against Edwardsiellosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains, Plasmids and Growth Conditions
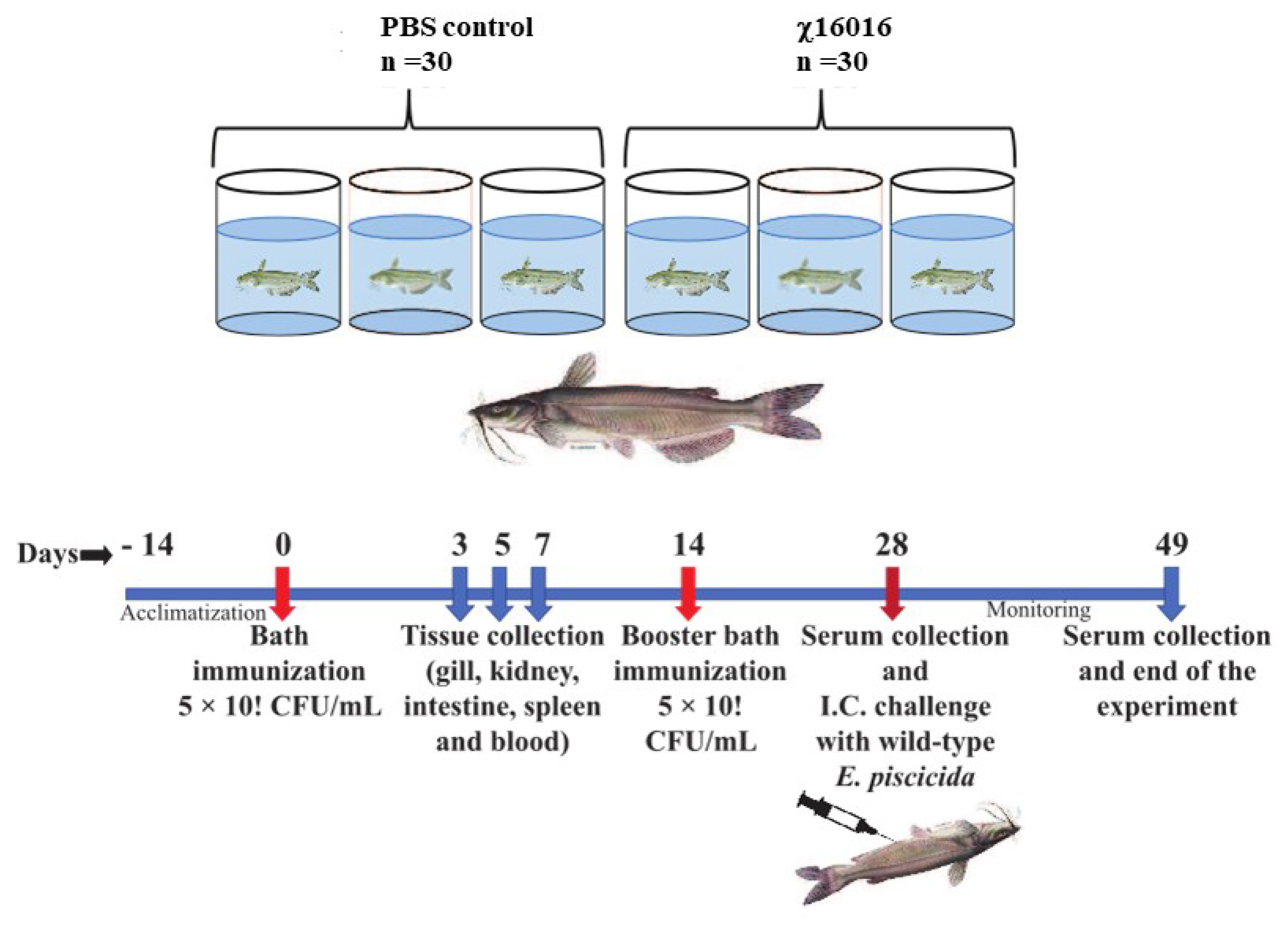
2.2. Fish Husbandry
2.3. Sequence Analysis
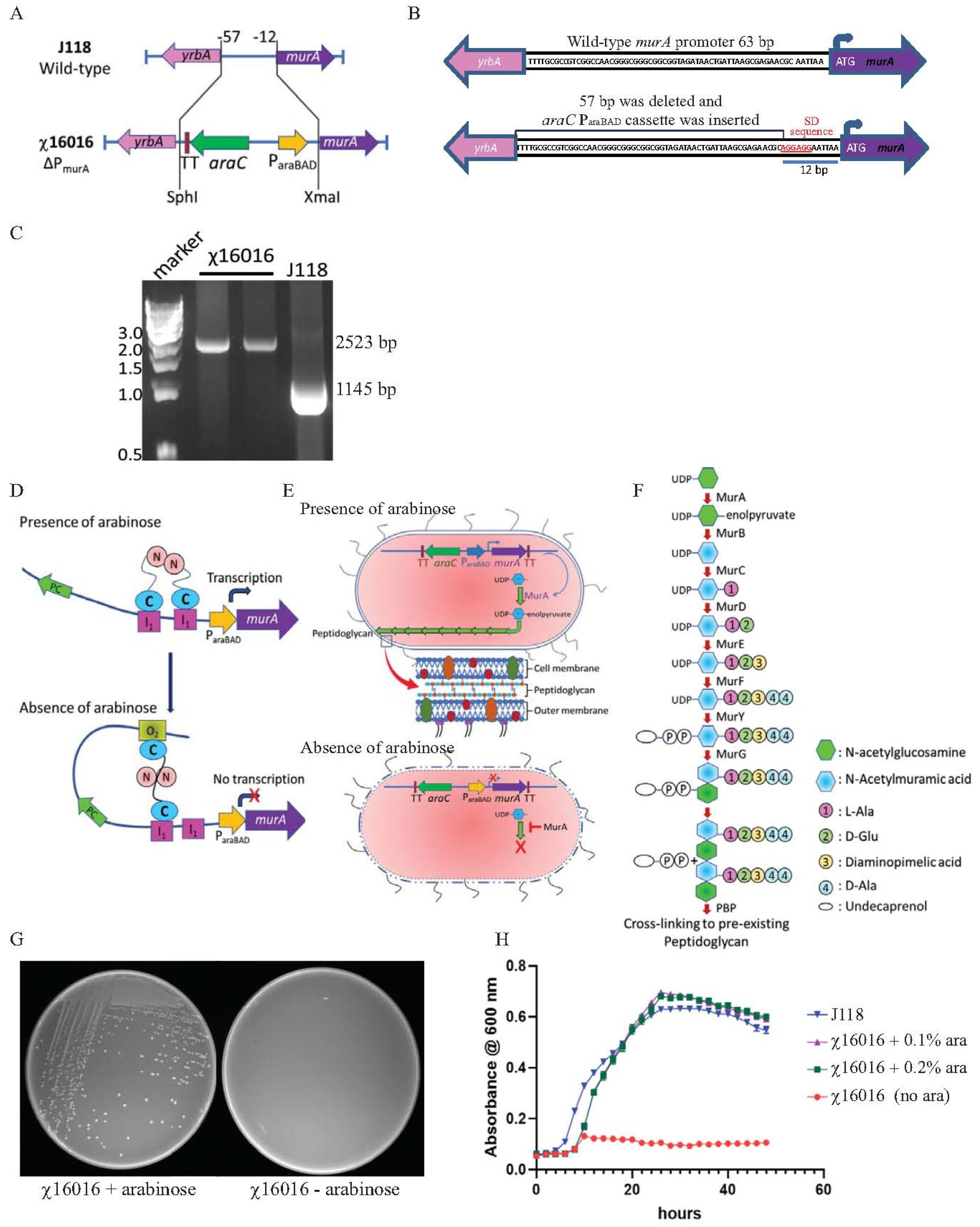
2.4. Construction of E. piscicida Deletion–Insertion Mutation of ΔPmurA
2.5. Growth Curve Analysis
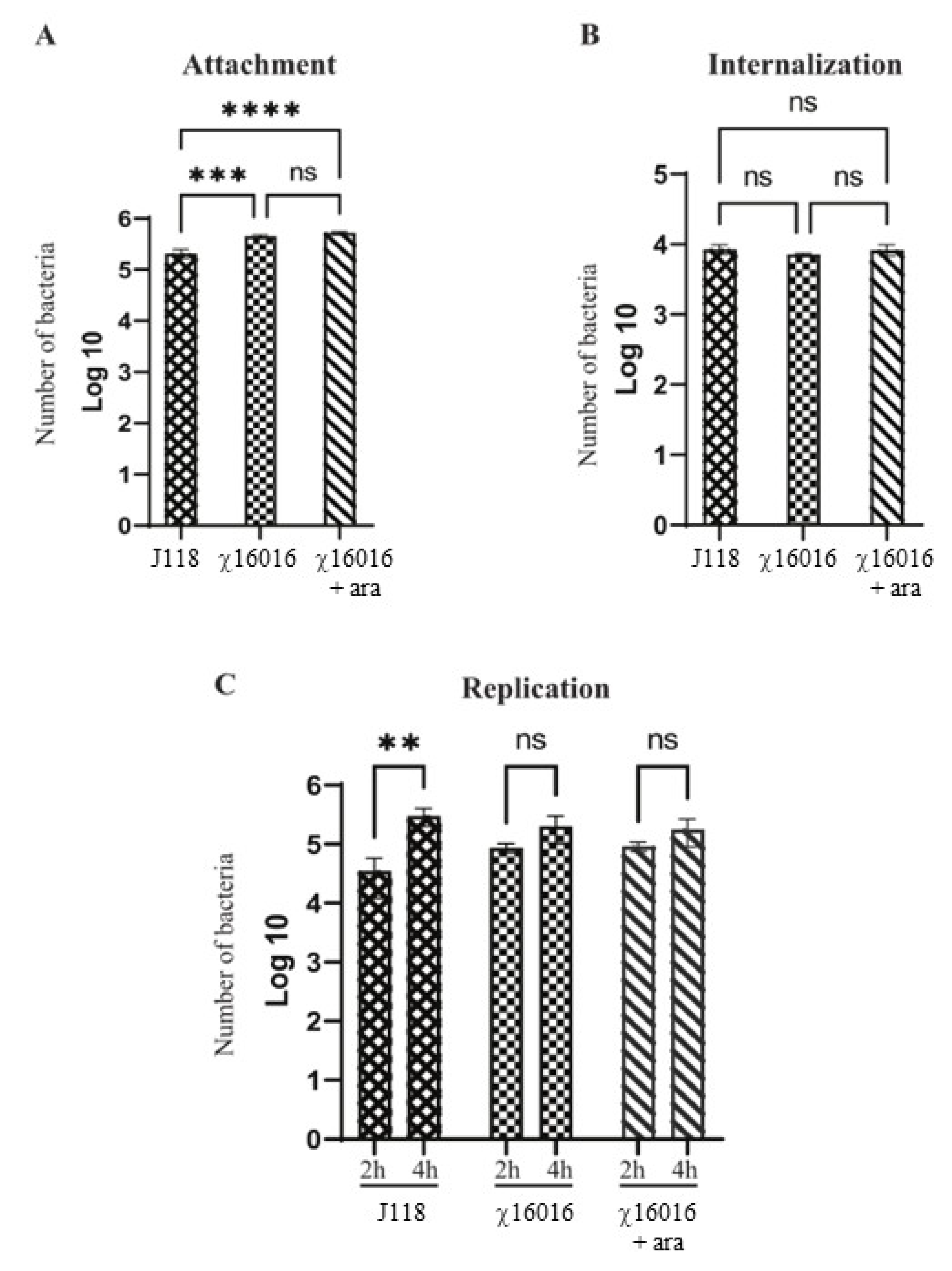
2.6. Attachment, Internalization and Intracellular Replication Assays
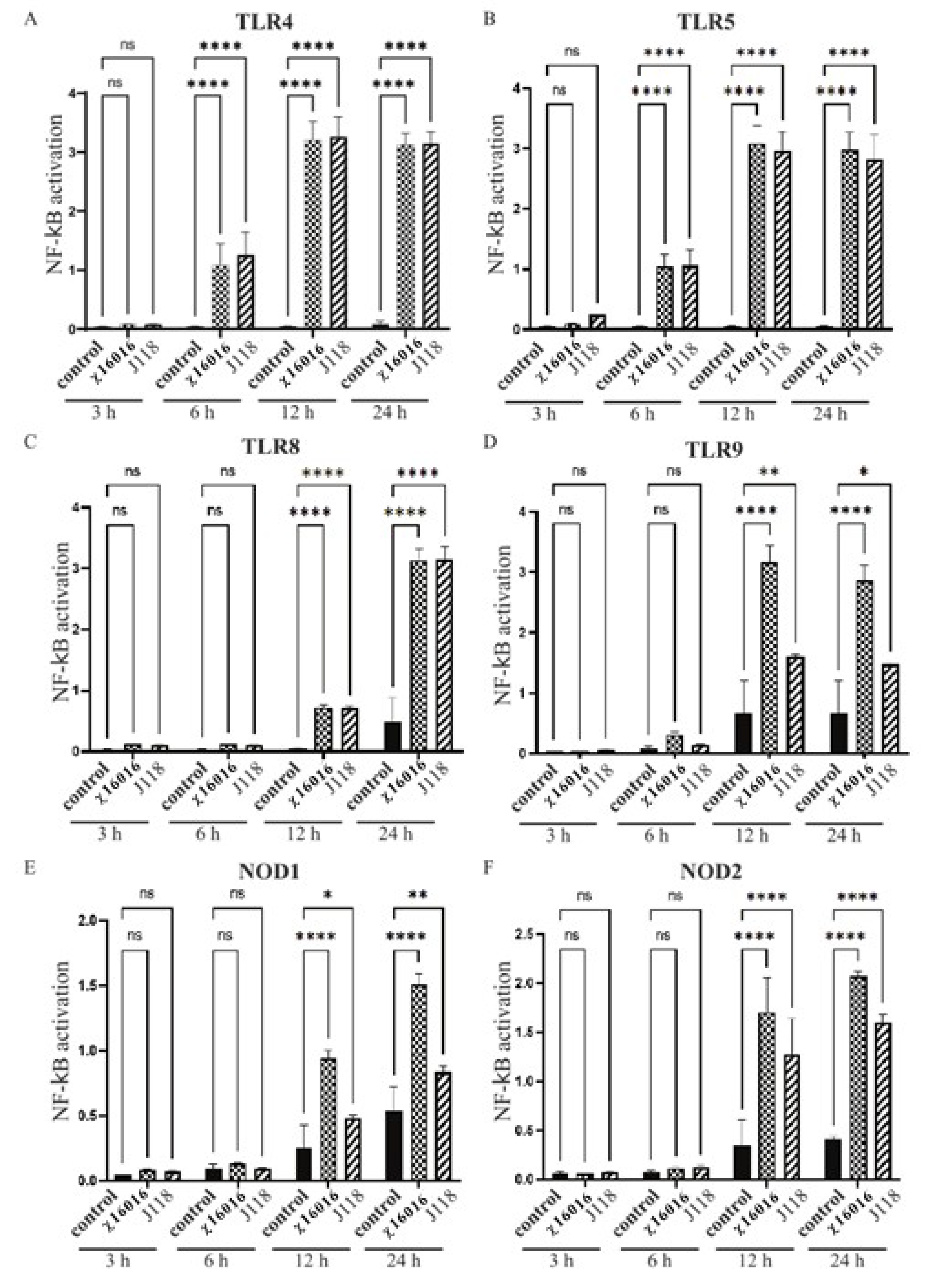
2.7. Activation of TLRs and NLRs by E. piscicida ΔPmurA Strain
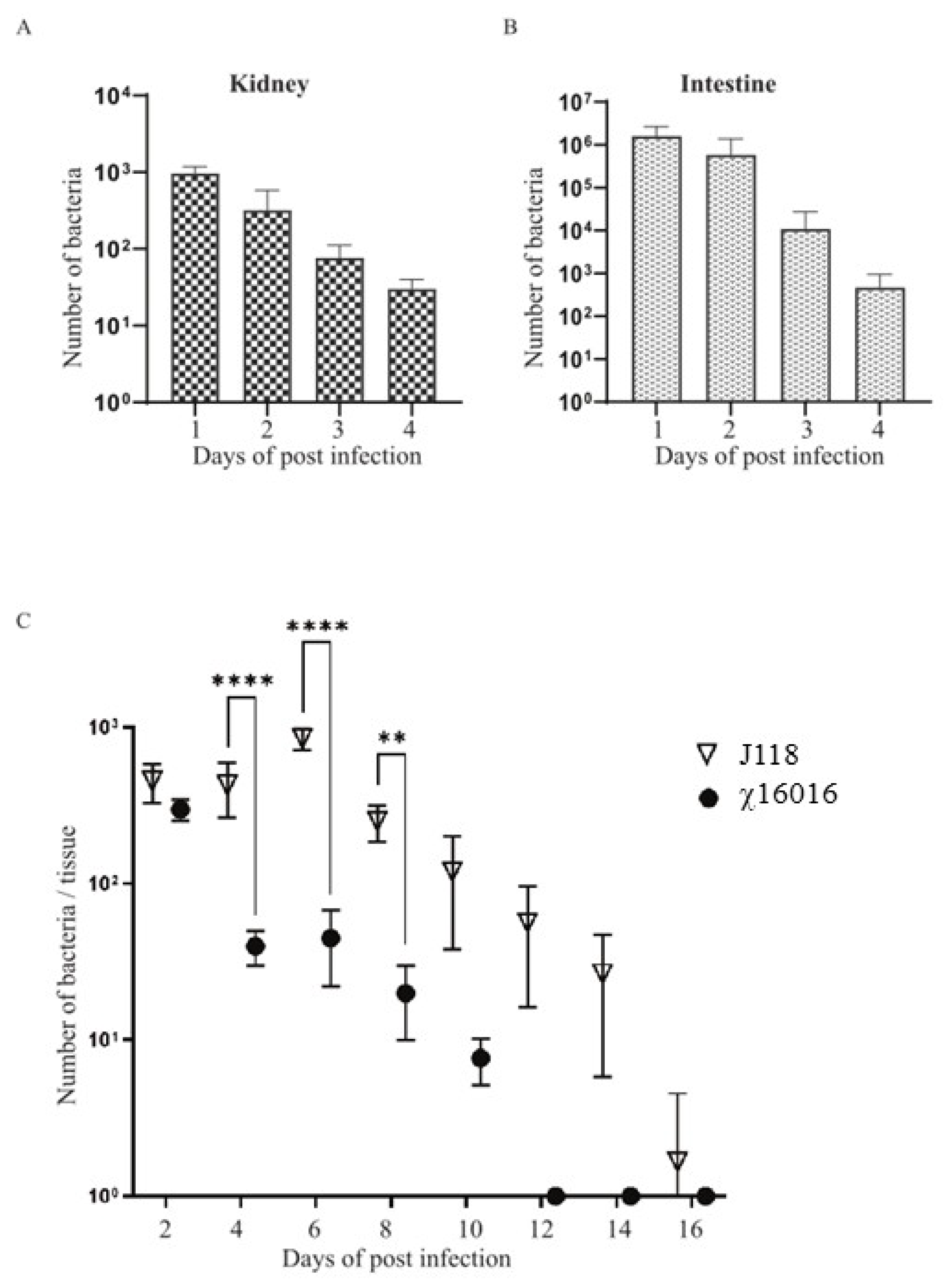
2.8. Colonization and Lysis of χ16016 in Catfish Tissues
2.9. Determination of Lethal Dose 50 (LD50) χ16016 via Bath Immersion and IC Injection
2.10. Evaluation of Anti-E. piscicida IgM in Catfish Serum and Skin Mucus via Enzyme-Linked Immunosorbent Assay (ELISA)
2.11. Immunization and Challenge
2.12. Isolation of RNA and Synthesis of First-Strand cDNA & Quantitative Real-Time PCR Analysis
2.13. Statistical Analysis
3. Results
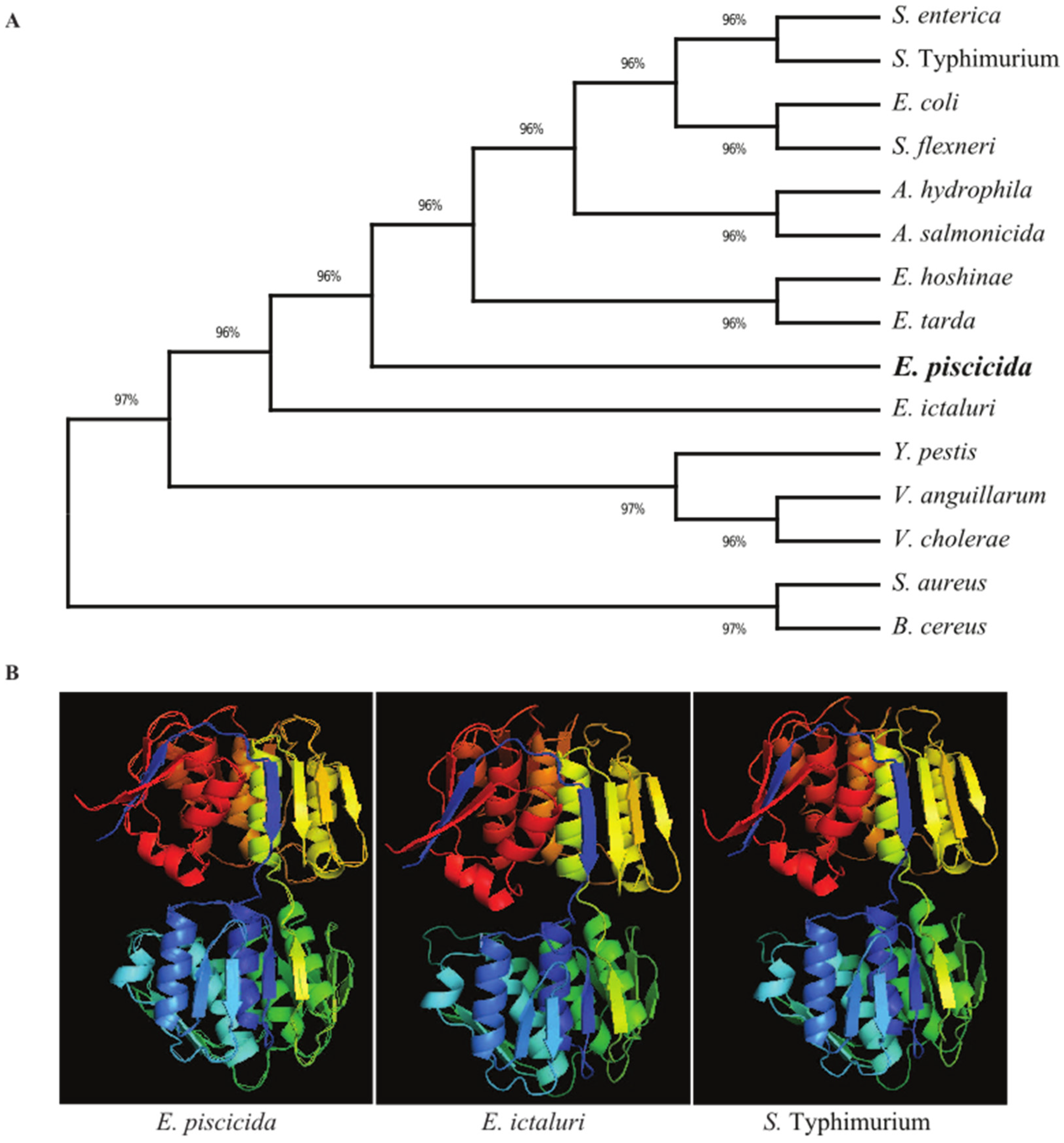
3.1. Analysis of Sequences, Construction of Phylogenetic Tree and Generation of Homology 3D Structure of MurA
3.2. Construction and Evaluation of E. piscicida murA Mutant
3.3. Principle of Arabinose-Regulated MurA Synthesis
3.4. Attachment, Internalization and Persistence of E. piscicida Wild-Type and ΔPmurA (χ16016) Strains in Fish Cell Line
3.5. Activation of NF-κB Pathway through TLRs and NLRs by E. piscicida Strains
3.6. Determination of Lethal Dose 50 (LD50) Dose of Wild-Type and Vaccine Candidate via IC Injection and Bath Immersion
3.7. Colonization and Lysis of χ16016 in Catfish Tissues
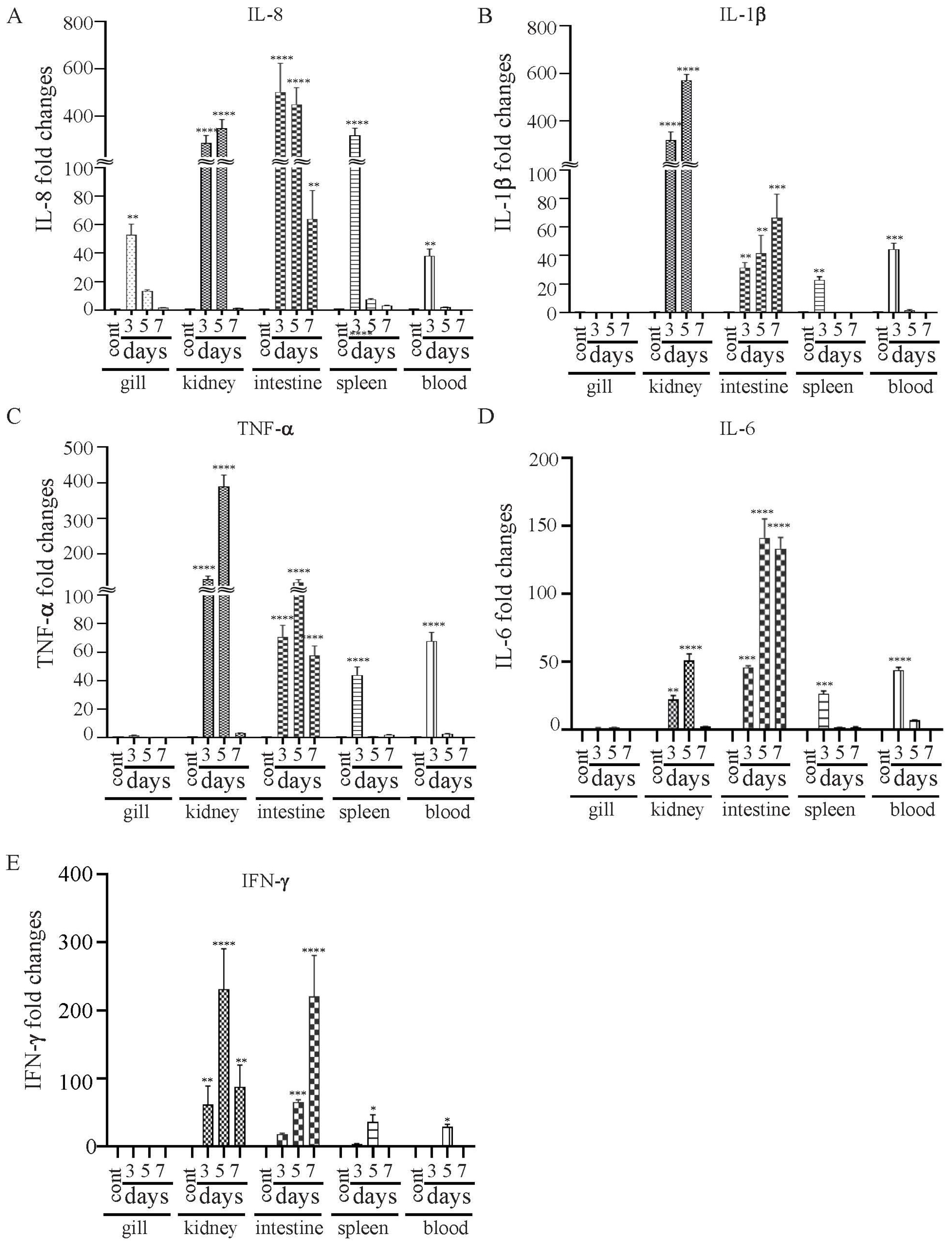
3.8. Expressional Modulation of il-8, il-1β, tnf-a, il-6 and ifn-γ Genes in χ16016-Vaccinated and Control Catfish
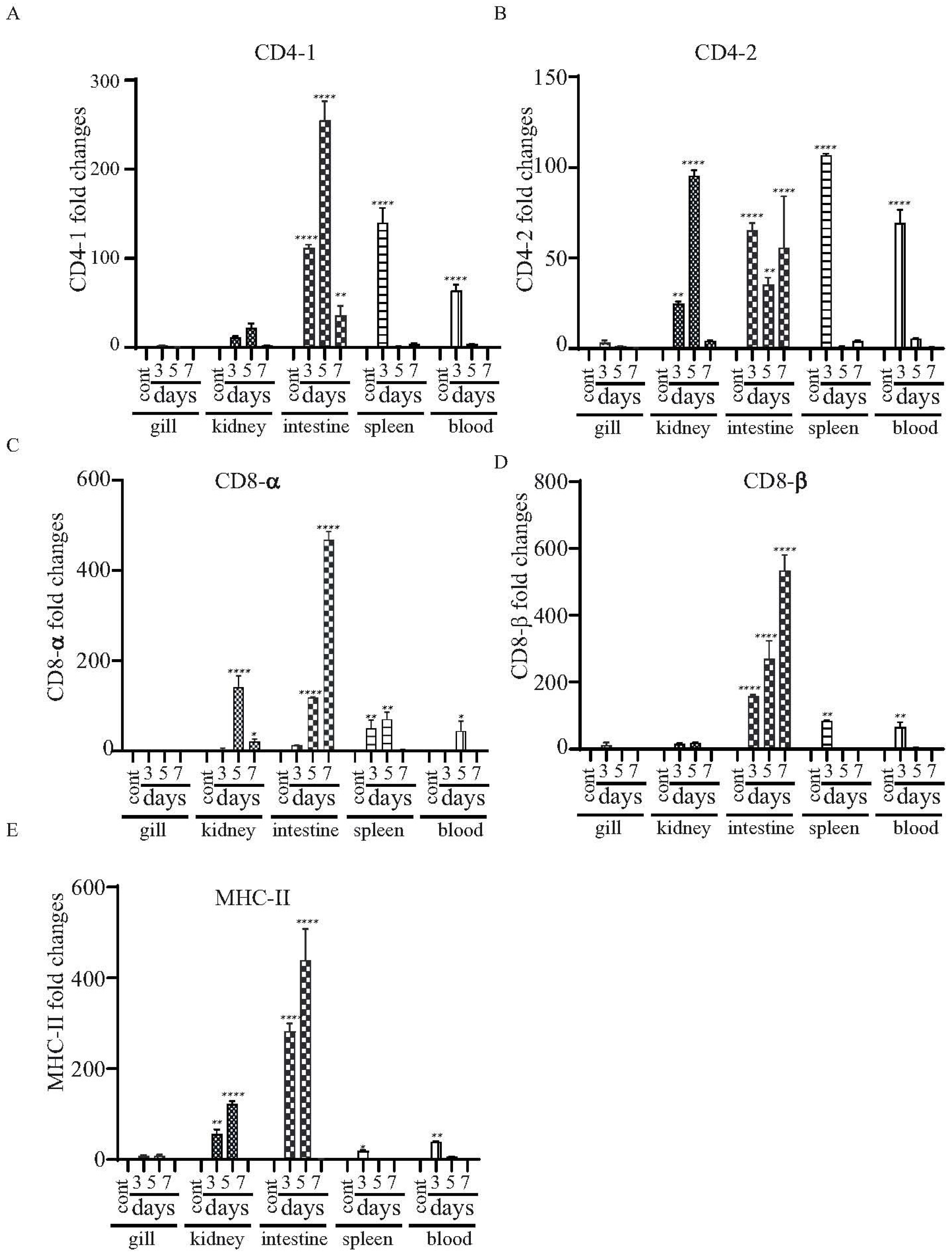
3.9. Expressional Modulation of the T Cell-Related Genes in χ16016 Vaccinated and Control Catfish
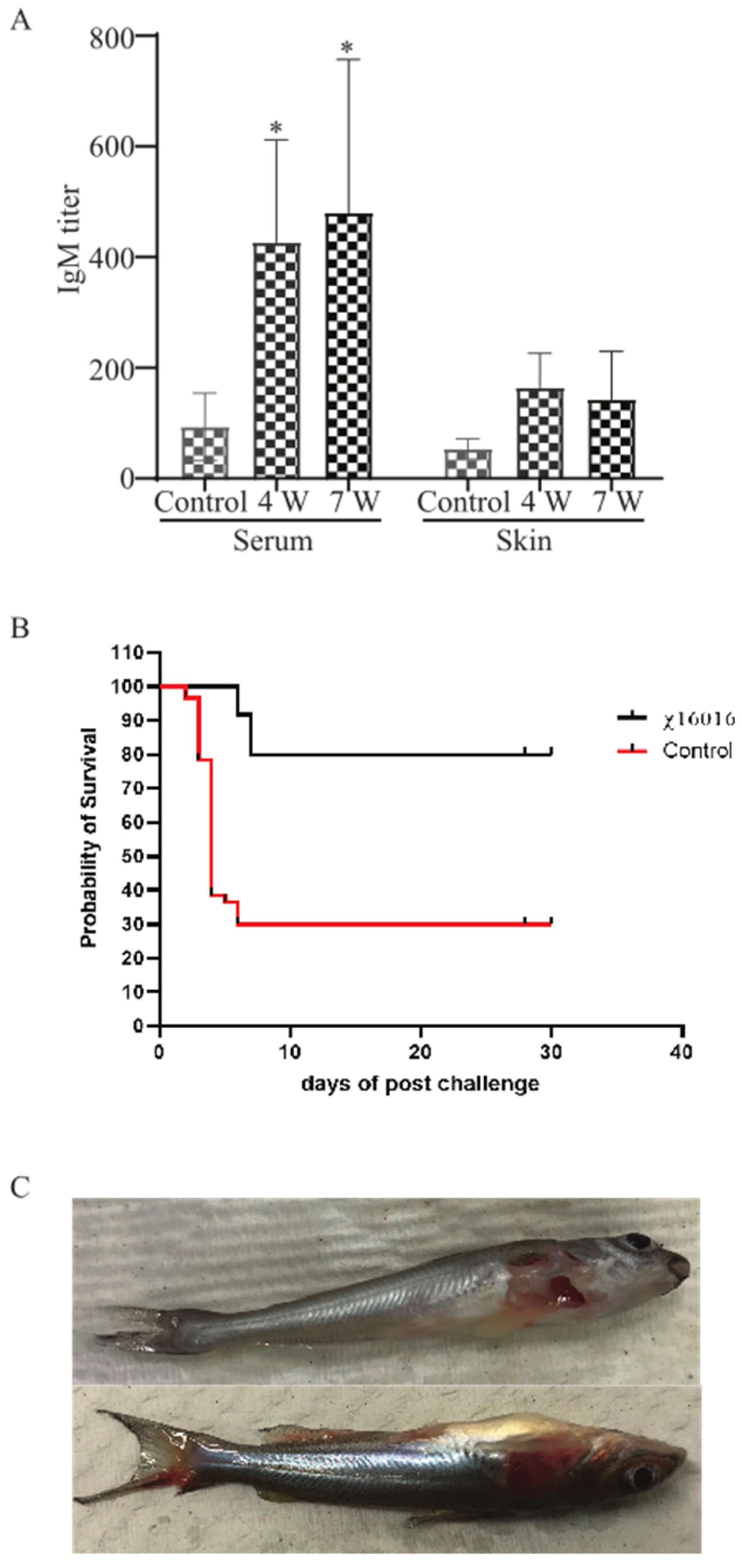
3.10. Analysis of E. piscicida Anti-LPS Antibody Responses in Control and Immunized Catfish
3.11. Survival Rate of Vaccinated Fish after Wild-Type E. piscicida Challenge
3.12. The Clinical Observation of Catfish after Challenge with E. piscicida
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- USDA. Catfish Production: Value of Sales Up 12% from 2020. AgFax 2022. Available online: https://www.agfax.com/2022/02/11/catfish-production-value-of-sales-up-12-from-2020/ (accessed on 1 July 2023).
- Lewis, D.H.; Tarpley, R.J.; Marks, J.E.; Sis, R.F. Drug induced structural changes in olfactory organ of channel catfish Ictalurus punctatus, Rafinesque. J. Fish Biol. 1985, 26, 355–358. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Veterinary Services, Center for Emerging Issues. Assessing Infectious Disease Emergence Potential in the U.S. Aquaculture Industry [Electronic Resource]: Phase 1, U.S. Aquaculture Industry Profile; U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Veterinary Services, Center for Emerging Issues: Fort Collins, CO, USA, 2007.
- Wise, A.L.; LaFrentz, B.R.; Kelly, A.M.; Khoo, L.H.; Xu, T.; Liles, M.R.; Bruce, T.J. A Review of Bacterial Co-Infections in Farmed Catfish: Components, Diagnostics, and Treatment Directions. Animals 2021, 11, 3240. [Google Scholar] [CrossRef] [PubMed]
- Preena, P.G.; Dharmaratnam, A.; Swaminathan, T.R. A peek into mass mortality caused by antimicrobial resistant Edwardsiella tarda in goldfish, Carassius auratus in Kerala. Biologia 2022, 77, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Griffin, M.; Ware, C.; Quiniou, S.; Steadman, J.; Gaunt, P.; Khoo, L.; Soto, E. Edwardsiella piscicida identified in the southeastern USA by gyrB sequence, species-specific and repetitive sequence-mediated PCR. Dis. Aquat. Org. 2014, 108, 23–35. [Google Scholar] [CrossRef]
- Hawke, J. Enteric Septicemia of Catfish. 2015. Available online: https://fisheries.tamu.edu/files/2019/01/SRAC_0477.pdf (accessed on 1 July 2023).
- Tsheten, T.; Tshering, D.; Gyem, K.; Dorji, S.; Wangchuk, S.; Tenzin, T.; Norbu, L.; Jamtsho, T. An Outbreak of Aeromonas hydrophila Food Poisoning in Deptsang Village, Samdrup Jongkhar, Bhutan, 2016. J. Res. Health Sci. 2016, 16, 224–227. [Google Scholar] [PubMed]
- Swain, B.; Powell, C.T.; Curtiss, R., 3rd. Construction and Evaluation of Recombinant Attenuated Edwardsiella piscicida Vaccine (RAEV) Vector System Encoding Ichthyophthirius multifiliis (Ich) Antigen IAG52B. Front. Immunol. 2021, 12, 802760. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Bruce, T.J.; Jones, E.M.; Cain, K.D. A Review of Fish Vaccine Development Strategies: Conventional Methods and Modern Biotechnological Approaches. Microorganisms 2019, 7, 569. [Google Scholar] [CrossRef]
- Yadav, D.K.; Yadav, N.; Khurana, S.M.P. Vaccines: Present Status and Applications. In Animal Biotechnology; Verma, A.S., Singh, A., Eds.; Academic Press: San Diego, CA, USA, 2014; Chapter 26; pp. 491–508. [Google Scholar]
- Bensen, D.; Rodriguez, S.; Nix, J.; Cunningham, M.; Tari, L. Structure of MurA (UDP-N-acetylglucosamine enolpyruvyl transferase), from Vibrio fischeri in complex with substrate UDP-N-acetylglucosamine and the drug fosfomycin. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2012, 68 Pt 4, 382–385. [Google Scholar] [CrossRef]
- Baum, E.Z.; Montenegro, D.A.; Licata, L.; Turchi, I.; Webb, G.C.; Foleno, B.D.; Bush, K. Identification and Characterization of New Inhibitors of the Escherichia coli MurA Enzyme. Antimicrob. Agents Chemother. 2001, 45, 3182–3188. [Google Scholar] [CrossRef]
- Kong, W.; Wanda, S.-Y.; Zhang, X.; Bollen, W.; Tinge, S.A.; Roland, K.L.; Curtiss, R., III. Regulated programmed lysis of recombinant Salmonella in host tissues to release protective antigens and confer biological containment. Proc. Natl. Acad. Sci. USA 2008, 105, 9361–9366. [Google Scholar] [CrossRef]
- Xu, D.-H.; Pridgeon, J.W.; Klesius, P.H.; Shoemaker, C.A. Parasitism by protozoan Ichthyophthirius multifiliis enhanced invasion of Aeromonas hydrophila in tissues of channel catfish. Veter Parasitol. 2012, 184, 101–107. [Google Scholar] [CrossRef]
- Pridgeon, J.; Klesius, P.; Lewbart, G.; Daniels, H.; Jacob, M. Edwardsiella tarda and Aeromonas hydrophila isolated from diseased Southern flounder (Paralichthys lethostigma) are virulent to channel catfish and Nile tilapia. J. Coast. Life Med. 2014, 2, 337–343. [Google Scholar]
- Hoelzer, K.; Bielke, L.; Blake, D.P.; Cox, E.; Cutting, S.M.; Devriendt, B.; Erlacher-Vindel, E.; Goossens, E.; Karaca, K.; Lemiere, S.; et al. Vaccines as alternatives to antibiotics for food producing animals. Part 2: New approaches and potential solutions. Vet. Res. 2018, 49, 70. [Google Scholar] [CrossRef]
- Roland, K.; Curtiss, R., 3rd; Sizemore, D. Construction and evaluation of a delta cya delta crp Salmonella typhimurium strain expressing avian pathogenic Escherichia coli O78 LPS as a vaccine to prevent airsacculitis in chickens. Avian Dis. 1999, 43, 429–441. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, M.; Xiao, J.; Wu, H.; Wang, X.; Lv, Y.; Xu, L.; Zheng, H.; Wang, S.; Zhao, G.; et al. Genome Sequence of the Versatile Fish Pathogen Edwardsiella tarda Provides Insights into Its Adaptation to Broad Host Ranges and Intracellular Niches. PLoS ONE 2009, 4, e7646. [Google Scholar] [CrossRef]
- Edwards, R.A.; Keller, L.H.; Schifferli, D.M. Improved allelic exchange vectors and their use to analyze 987P fimbria gene expression. Gene 1998, 207, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Wang, S.; Curtiss, R. Highly Efficient Method for Introducing Successive Multiple Scarless Gene Deletions and Markerless Gene Insertions into the Yersinia pestis Chromosome. Appl. Environ. Microbiol. 2008, 74, 4241–4245. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Zhang, Q.; He, T.T.; Li, D.Y.; Liu, L.Y.; Nie, P.; Xie, H.X. The Edwardsiella piscicida Type III Effector EseJ Suppresses Expression of Type 1 Fimbriae, Leading to Decreased Bacterial Adherence to Host Cells. Infect. Immun. 2019, 87, e00187-19. [Google Scholar] [CrossRef]
- Swain, B.; Powell, C.T.; Curtiss, R. Virulence, immunogenicity and live vaccine potential of aroA and phoP mutants of Edwardsiella piscicida in zebrafish. Microb. Pathog. 2022, 162, 105355. [Google Scholar] [CrossRef]
- Swain, B.; Basu, M.; Samanta, M. NOD1 and NOD2 receptors in mrigal (Cirrhinus mrigala): Inductive expression and downstream signalling in ligand stimulation and bacterial infections. J. Biosci. 2013, 38, 533–548. [Google Scholar] [CrossRef]
- Samanta, M.; Swain, B.; Basu, M.; Panda, P.; Mohapatra, G.B.; Sahoo, B.R.; Maiti, N.K. Molecular characterization of toll-like receptor 2 (TLR2), analysis of its inductive expression and associated down-stream signaling molecules following ligands exposure and bacterial infection in the Indian major carp, rohu (Labeo rohita). Fish Shellfish. Immunol. 2012, 32, 411–425. [Google Scholar] [CrossRef]
- Arpaia, N.; Godec, J.; Lau, L.; Sivick, K.E.; McLaughlin, L.M.; Jones, M.B.; Dracheva, T.; Peterson, S.N.; Monack, D.M.; Barton, G.M. TLR Signaling Is Required for Salmonella typhimurium Virulence. Cell 2011, 144, 675–688. [Google Scholar] [CrossRef] [PubMed]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Swain, B.; Powell, C.T.; Curtiss, R. Pathogenicity and immunogenicity of Edwardsiella piscicida ferric uptake regulator (fur) mutations in zebrafish. Fish Shellfish Immunol. 2020, 107, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Oluwole, A.O.; Corey, R.A.; Brown, C.M.; Hernández-Rocamora, V.M.; Stansfeld, P.J.; Vollmer, W.; Bolla, J.R.; Robinson, C.V. Peptidoglycan biosynthesis is driven by lipid transfer along enzyme-substrate affinity gradients. Nat. Commun. 2022, 13, 2278. [Google Scholar] [CrossRef]
- Mondal, H.; Thomas, J. A review on the recent advances and application of vaccines against fish pathogens in aquaculture. Aquac. Int. 2022, 30, 1971–2000. [Google Scholar] [CrossRef] [PubMed]
- Cain, K. The many challenges of disease management in aquaculture. J. World Aquac. Soc. 2022, 53, 1080–1083. [Google Scholar] [CrossRef]
- Plant, K.P.; LaPatra, S.E. Advances in fish vaccine delivery. Dev. Comp. Immunol. 2011, 35, 1256–1262. [Google Scholar] [CrossRef]
- Ledesma-Feliciano, C.; Chapman, R.; Hooper, J.W.; Elma, K.; Zehrung, D.; Brennan, M.B.; Spiegel, E.K. Improved DNA Vaccine Delivery with Needle-Free Injection Systems. Vaccines 2023, 11, 280. [Google Scholar] [CrossRef]
- Lee, S.; Nguyen, M.T. Recent Advances of Vaccine Adjuvants for Infectious Diseases. Immune Netw. 2015, 15, 51–57. [Google Scholar] [CrossRef]
- Davison, J. Towards safer vectors for the field release of recombinant bacteria. Environ. Biosaf. Res. 2002, 1, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Kotton, C.N.; Hohmann, E.L. Enteric Pathogens as Vaccine Vectors for Foreign Antigen Delivery. Infect. Immun. 2004, 72, 5535–5547. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Kong, Q.; Curtiss, R., 3rd. New technologies in developing recombinant attenuated Salmonella vaccine vectors. Microb. Pathog. 2013, 58, 17–28. [Google Scholar] [CrossRef]
- Juárez-Rodríguez, M.D.; Yang, J.; Kader, R.; Alamuri, P.; Curtiss, R., 3rd; Clark-Curtiss, J.E. Live attenuated Salmonella vaccines displaying regulated delayed lysis and delayed antigen synthesis to confer protection against Mycobacterium tuberculosis. Infect. Immun. 2012, 80, 815–831. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.D.; Vivas, E.I.; Walsh, C.T.; Kolter, R. MurA (MurZ), the enzyme that catalyzes the first committed step in peptidoglycan biosynthesis, is essential in Escherichia coli. J. Bacteriol. 1995, 177, 4194–4197. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Brown, J.R.; Sylvester, D.R.; Huang, J.; Chalker, A.F.; So, C.Y.; Holmes, D.J.; Payne, D.J.; Wallis, N.G. Two Active Forms of UDP-N-Acetylglucosamine Enolpyruvyl Transferase in Gram-Positive Bacteria. J. Bacteriol. 2000, 182, 4146–4152. [Google Scholar] [CrossRef]
- Barreteau, H.; Kovač, A.; Boniface, A.; Sova, M.; Gobec, S.; Blanot, D. Cytoplasmic steps of peptidoglycan biosynthesis. FEMS Microbiol. Rev. 2008, 32, 168–207. [Google Scholar] [CrossRef]
- Amemiya, K.; Dankmeyer, J.L.; Bernhards, R.C.; Fetterer, D.P.; Waag, D.M.; Worsham, P.L.; DeShazer, D. Activation of Toll-Like Receptors by Live Gram-Negative Bacterial Pathogens Reveals Mitigation of TLR4 Responses and Activation of TLR5 by Flagella. Front. Cell. Infect. Microbiol. 2021, 11, 745325. [Google Scholar] [CrossRef]
- Lavelle, E.C.; Murphy, C.; O’Neill, L.A.; Creagh, E.M. The role of TLRs, NLRs, and RLRs in mucosal innate immunity and homeostasis. Mucosal Immunol. 2010, 3, 17–28. [Google Scholar] [CrossRef]
- Zhang, G.; Ghosh, S. Toll-like receptor-mediated NF-kappaB activation: A phylogenetically conserved paradigm in innate immunity. J. Clin. Invest. 2001, 107, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Kaiko, G.E.; Horvat, J.C.; Beagley, K.W.; Hansbro, P.M. Immunological decision-making: How does the immune system decide to mount a helper T-cell response? Immunology 2008, 123, 326–338. [Google Scholar] [CrossRef] [PubMed]
- Muraille, E.; Leo, O.; Moser, M. Th1/Th2 Paradigm Extended: Macrophage Polarization as an Unappreciated Pathogen-Driven Escape Mechanism? Front. Immunol. 2014, 5, 603. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef]
- Guo, M.; Tang, X.; Sheng, X.; Xing, J.; Zhan, W. The Immune Adjuvant Effects of Flounder (Paralichthys olivaceus) Interleukin-6 on E. tarda Subunit Vaccine OmpV. Int. J. Mol. Sci. 2017, 18, 1445. [Google Scholar] [CrossRef]
- Baggiolini, M.; Loetscher, P.; Moser, B. Interleukin-8 and the chemokine family. Int. J. Immunopharmacol. 1995, 17, 103–108. [Google Scholar] [CrossRef]
- Stillie, R.; Farooq, S.M.; Gordon, J.R.; Stadnyk, A.W. The functional significance behind expressing two IL–8 receptor types on PMN. J. Leukoc. Biol. 2009, 86, 529–543. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, M.; Abualrous, E.T.; Sticht, J.; Álvaro-Benito, M.; Stolzenberg, S.; Noé, F.; Freund, C. Major Histocompatibility Complex (MHC) Class I and MHC Class II Proteins: Conformational Plasticity in Antigen Presentation. Front Immunol 2017, 8, 292. [Google Scholar] [CrossRef]
- Bengte, E.; Quiniou, S.M.-A.; Stuge, T.B.; Katagiri, T.; Miller, N.W.; Clem, L.W.; Warr, G.W.; Wilson, M. The IgH Locus of the Channel Catfish, Ictalurus punctatus, Contains Multiple Constant Region Gene Sequences: Different Genes Encode Heavy Chains of Membrane and Secreted IgD. J. Immunol. 2002, 169, 2488–2497. [Google Scholar]
- Xu, J.; Zhang, X.; Luo, Y.; Wan, X.; Yao, Y.; Zhang, L.; Yu, Y.; Ai, T.; Wang, Q.; Xu, Z. IgM and IgD heavy chains of yellow catfish (Pelteobagrus fulvidraco): Molecular cloning, characterization and expression analysis in response to bacterial infection. Fish Shellfish Immunol. 2019, 84, 233–243. [Google Scholar] [CrossRef]
- Lund, V.; Børdal, S.; Kjellsen, O.; Mikkelsen, H.; Schrøder, M.B. Comparison of antibody responses in Atlantic cod (Gadus morhua L.) to Aeromonas salmonicida and Vibrio anguillarum. Dev. Comp. Immunol. 2006, 30, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- Santander, J.; Golden, G.; Wanda, S.-Y.; Curtiss, R. Fur-Regulated Iron Uptake System of Edwardsiella ictaluri and Its Influence on Pathogenesis and Immunogenicity in the Catfish Host. Infect. Immun. 2012, 80, 2689–2703. [Google Scholar] [CrossRef] [PubMed]
| Strain or Plasmid | Genotype and Relevant Characteristics | Source or Reference(s) |
|---|---|---|
| Strains | ||
| Escherichia coli | ||
| χ7213 | thi-1 thr-1 leuB6 glnV44 fhuA21 lacY1 recA1 RP4-2-Tc::Mu λpir ∆asdA4 ∆zhf-2::Tn10 | [18] |
| E. piscicida | ||
| J118 | Wild-type E. piscicida EIB202, highly virulent, fish isolated, Colr | [19] |
| χ16016 | J118 derivative; ΔPmurA180::TT araC ParaBAD murA | This study |
| Plasmids | ||
| pRE112 | Suicide vector; sacB mobRP4 R6K ori; Cmr | [20] |
| pYA3700 | Cloning vector containing TT araC ParaBAD cassette | [21] |
| pG8R8025 | ΔPmurA180::TT araC ParaBAD murA, pRE112 | This study |
| Primer | Sequence (5′-3′) |
|---|---|
| MurA1-SphI | F-CATGCATGCCGGGCATCACGTGTGTGGATATC |
| MurA2-BglII | R-CATAGATCTTCACAGACCGCTCAGCTTGCG |
| MurA3-KpnI | F-CATGGTACC AGGAGGAATTAAATGGATAAATTTCGT |
| MurA4-EcoRI | R-CCGGAATTCCGCTGACCTTGTCCATCACGATA |
| MurA5-XmaI | R-CATCCCGGGCGCTGACCTTGTCCATCACGATA |
| pYA3700 | F-ATAACCTTTCATTCCCAGCGG |
| (A) | |||||
| J118 (Wild-Type E. piscicida) | |||||
| Dose CFU/Fish | Mortality | Deaths | Survivals | Mortality % | LD50 |
| 9 × 102 | 1/10 | 1 | 9 | 5 | 1.7 × 104 |
| 9 × 103 | 4/10 | 4 | 6 | 38 | |
| 9 × 104 | 8/10 | 8 | 2 | 87 | |
| 9 × 105 | 10/10 | 10 | 0 | 100 | |
| 9 × 106 | 10/10 | 10 | 0 | 100 | |
| χ16016 (ΔPmurA180::TT araC ParaBAD murA) | |||||
| 4 × 104 | 0/10 | 0 | 10 | 0 | 5 × 106 |
| 4 × 105 | 0/10 | 0 | 10 | 0 | |
| 4 × 106 | 4/10 | 4 | 6 | 40 | |
| 4 × 107 | 10/10 | 10 | 0 | 100 | |
| 4 × 108 | 10/10 | 10 | 0 | 100 | |
| (B) | |||||
| J118 (Wild-Type E. piscicida) CFU/mL | |||||
| 9 × 105 | 0/10 | 0 | 10 | 0 | 4.8 × 107 |
| 9 × 106 | 2/10 | 2 | 8 | 20 | |
| 9 × 107 | 10/10 | 10 | 0 | 100 | |
| χ16016 (ΔPmurA180::TT araC ParaBAD murA) | |||||
| 4 × 106 | 0/10 | 0 | 10 | 0 | - |
| 4 × 107 | 0/10 | 0 | 10 | 0 | |
| 4 × 108 | 0/10 | 0 | 10 | 0 | |
| Gene | Sequence 5′-3′ | PCR Product Size in bp | GenBank # |
|---|---|---|---|
| 18S rRNA | F-GAGAAACGGCTACCACATCC | 128 | AF021880.1 |
| R-GATACGCTCATTCCGATTACAG | |||
| CD4-1 | F-GGGGATTTTTGCACAGTTCGG | 243 | DQ435305.1 |
| R-TCCACAAGGTGATGCTGTTCA | |||
| CD4-2 | F-TGAACATCGGTGTTGTTGGTC | 238 | DQ435304.1 |
| R-TCCTGTTCAGTGAAGGTGACTT | |||
| CD8-α | F-TCGTACAGCAAGACTAAGAAATCA | 198 | HQ446239.1 |
| R-TTTGATTGTTGTTGGGACTGTCT | |||
| CD8-β | F-CTTAAGAACAGAGACACACCGGA | 299 | HQ446240.1 |
| R-ACACGCTTTGGATTTTTCACTG | |||
| IL-1β | F-TGCTGGAAAGGTACTTTGACAG | 296 | DQ157743.1 |
| R-GCACTTCAAATTGATCACCATGC | |||
| IL-8 | F-GCTGCCCTTGATCATCTTTGC | 219 | AY145142.1 |
| R-GGATCCAGGCAGACCTTCATTC | |||
| IFN-γ | F-TACTGGACATGAGATTCACAACCT | 257 | NM_001200217.1 |
| R-CTTTCACACTTTCCTGGAGTTC | |||
| MHC-II | F-CACTGAAGCACAGATCAAACACC | 236 | AF103002.2 |
| R-CTGTTCTGTACACTCCTAGGTTGG | |||
| TNF-α | F-TCTTCAGGAGTTTGGAGAACCTG | 255 | AJ417565.2 |
| R-CAGTTTCAAGCCTGAAGAGAAGG | |||
| IL-6 | F-GAAACATCTGGAGTCGAGCTGC | 214 | XM_017455306.1 |
| R-GACATTTTTGCGGGCTGAAGTC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Swain, B.; Campodonico, V.A.; Curtiss, R., III. Recombinant Attenuated Edwardsiella piscicida Vaccine Displaying Regulated Lysis to Confer Biological Containment and Protect Catfish against Edwardsiellosis. Vaccines 2023, 11, 1470. https://doi.org/10.3390/vaccines11091470
Swain B, Campodonico VA, Curtiss R III. Recombinant Attenuated Edwardsiella piscicida Vaccine Displaying Regulated Lysis to Confer Biological Containment and Protect Catfish against Edwardsiellosis. Vaccines. 2023; 11(9):1470. https://doi.org/10.3390/vaccines11091470
Chicago/Turabian StyleSwain, Banikalyan, Vanessa A. Campodonico, and Roy Curtiss, III. 2023. "Recombinant Attenuated Edwardsiella piscicida Vaccine Displaying Regulated Lysis to Confer Biological Containment and Protect Catfish against Edwardsiellosis" Vaccines 11, no. 9: 1470. https://doi.org/10.3390/vaccines11091470






