Opsonic Activity of Conservative Versus Variable Regions of the Group A Streptococcus M Protein
Abstract
1. Introduction
2. Materials and Methods
2.1. General Methods
2.2. Vaccine Synthesis
2.2.1. VC-1
2.2.2. VC-2
2.2.3. VC-3
2.3. Dynamic Light Scattering
2.4. Transmisssion Electron Microsocpy
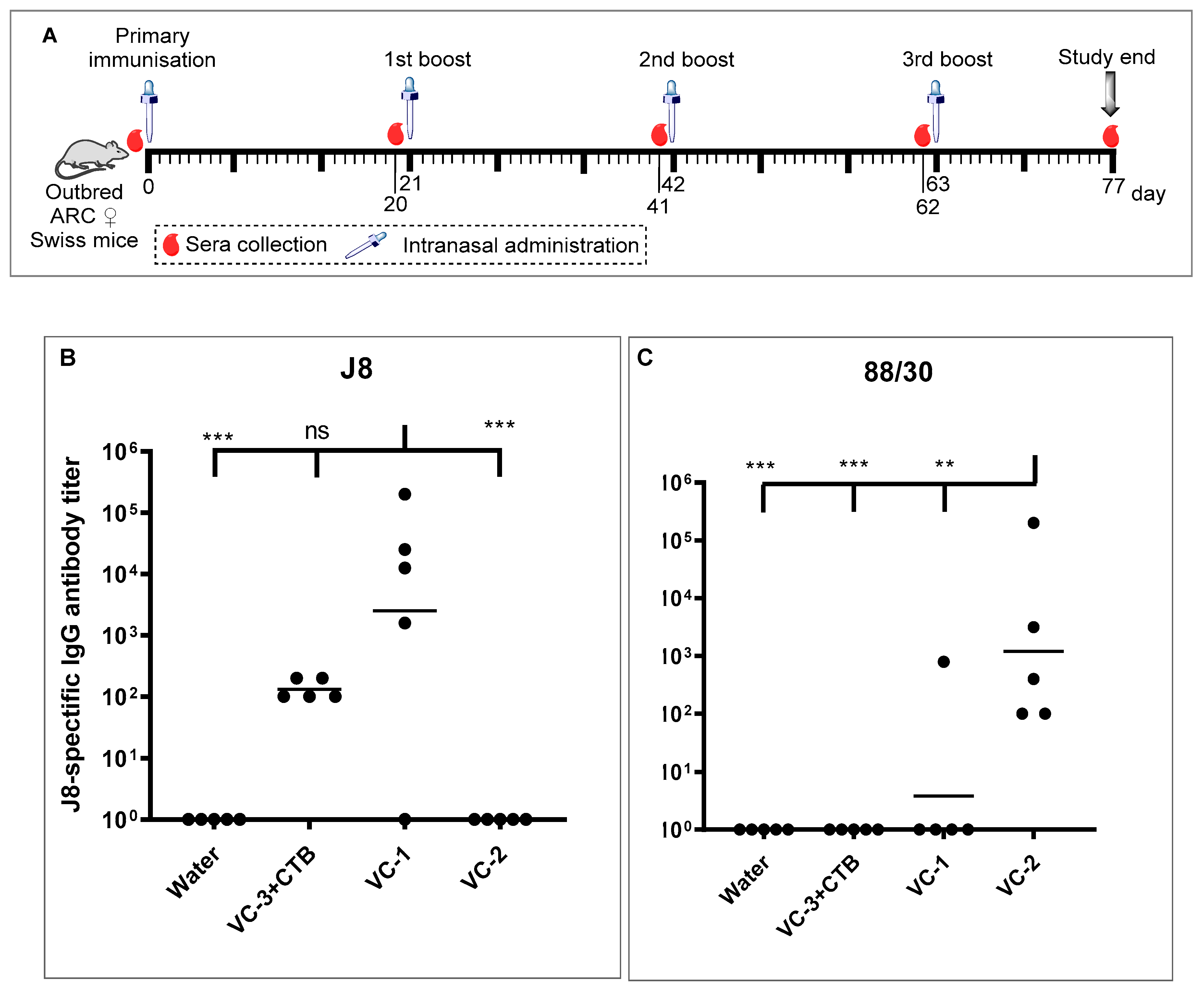
2.5. In Vivo Immunogenicity Study
2.6. ELISA Study
2.7. In Vitro Opsonisation Study
3. Results and Discussion
3.1. Vaccine Design and Synthesis
3.2. Physicochemical Evaluation
3.3. In Vivo Immunological Study
3.4. In Vitro Opsonisation Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Walker, M.J.; Barnett, T.C.; McArthur, J.D.; Cole, J.N.; Gillen, C.M.; Henningham, A.; Sriprakash, K.S.; Sanderson-Smith, M.L.; Nizet, V. Disease Manifestations and Pathogenic Mechanisms of Group A Streptococcus. Clin. Microbiol. Rev. 2014, 27, 264–301. [Google Scholar] [CrossRef] [PubMed]
- Carapetis, J.R.; Steer, A.C.; Mulholland, E.K.; Weber, M. The global burden of group A streptococcal diseases. Lancet Infect. Dis. 2005, 5, 685–694. [Google Scholar] [CrossRef]
- Salehi, S.; Hohn, C.M.; Penfound, T.A.; Dale, J.B. Development of an Opsonophagocytic Killing Assay Using HL-60 Cells for Detection of Functional Antibodies against Streptococcus pyogenes. mSphere 2018, 3, e00617-18. [Google Scholar] [CrossRef] [PubMed]
- Bisno, A.L. Group A Streptococcal Infections and Acute Rheumatic Fever. N. Engl. J. Med. 1991, 325, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Rivera-Hernandez, T.; Carnathan, D.G.; Jones, S.; Cork, A.J.; Davies, M.R.; Moyle, P.M.; Toth, I.; Batzloff, M.R.; McCarthy, J.S.; Nizet, V.; et al. An Experimental Group A Streptococcus Vaccine That Reduces Pharyngitis and Tonsillitis in a Nonhuman Primate Model. mBio 2019, 10, e00693-19. [Google Scholar] [CrossRef] [PubMed]
- Azuar, A.; Jin, W.; Mukaida, S.; Hussein, W.M.; Toth, I.; Skwarczynski, M. Recent Advances in the Development of Peptide Vaccines and Their Delivery Systems Against Group A Streptococcus. Vaccines 2019, 7, 58. [Google Scholar] [CrossRef]
- Jones, S.; Moreland, N.J.; Zancolli, M.; Raynes, J.; Loh, J.M.S.; Smeesters, P.R.; Sriskandan, S.; Carapetis, J.R.; Fraser, J.D.; Goldblatt, D. Development of an opsonophagocytic killing assay for group a streptococcus. Vaccine 2018, 36, 3756–3763. [Google Scholar] [CrossRef]
- Good, M.F.; Batzloff, M.R.; Pandey, M. Strategies in the development of vaccines to prevent infections with group A streptococcus. Hum. Vaccines Immunother. 2013, 9, 2393–2397. [Google Scholar] [CrossRef]
- Chuan, Y.P.; Rivera-Hernandez, T.; Wibowo, N.; Connors, N.K.; Wu, Y.; Hughes, F.K.; Lua, L.H.; Middelberg, A.P. Effects of pre-existing anti-carrier immunity and antigenic element multiplicity on efficacy of a modular virus-like particle vaccine. Biotechnol. Bioeng. 2013, 110, 2343–2351. [Google Scholar] [CrossRef]
- Batzloff, M.R.; Hayman, W.A.; Davies, M.R.; Zeng, M.; Pruksakorn, S.; Brandt, E.R.; Good, M.F. Protection against Group A Streptococcus by Immunization with J8–Diphtheria Toxoid: Contribution of J8- and Diphtheria Toxoid–Specific Antibodies to Protection. J. Infect. Dis. 2003, 187, 1598–1608. [Google Scholar] [CrossRef]
- Pandey, M.; Mortensen, R.; Calcutt, A.; Powell, J.; Batzloff, M.R.; Dietrich, J.; Good, M.F. Combinatorial Synthetic Peptide Vaccine Strategy Protects against Hypervirulent CovR/S Mutant Streptococci. J. Immunol. 2016, 196, 3364–3374. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.R.; McIntyre, L.; Mutreja, A.; Lacey, J.A.; Lees, J.A.; Towers, R.J.; Duchene, S.; Smeesters, P.R.; Frost, H.R.; Price, D.J.; et al. Atlas of group A streptococcal vaccine candidates compiled using large-scale comparative genomics. Nat. Genet. 2019, 51, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Sekuloski, S.; Batzloff, M.R.; Griffin, P.; Parsonage, W.; Elliott, S.; Hartas, J.; O’Rourke, P.; Marquart, L.; Pandey, M.; Rubin, F.A.; et al. Evaluation of safety and immunogenicity of a group A streptococcus vaccine candidate (MJ8VAX) in a randomized clinical trial. PLoS ONE 2018, 13, e0198658. [Google Scholar] [CrossRef] [PubMed]
- Hayman, W.A.; Brandt, E.R.; Relf, W.A.; Cooper, J.; Saul, A.; Good, M.F. Mapping the minimal murine T cell and B cell epitopes within a peptide vaccine candidate from the conserved region of the M protein of group A streptococcus. Int. Immunol. 1997, 9, 1723–1733. [Google Scholar] [CrossRef]
- Raynes, J.M.; Young, P.G.; Proft, T.; Williamson, D.A.; Baker, E.N.; Moreland, N.J. Protein adhesins as vaccine antigens for Group A Streptococcus. Pathog. Dis. 2018, 76. [Google Scholar] [CrossRef] [PubMed]
- Martin, W.J.; Steer, A.C.; Smeesters, P.R.; Keeble, J.; Inouye, M.; Carapetis, J.; Wicks, I.P. Post-infectious group A streptococcal autoimmune syndromes and the heart. Autoimmun. Rev. 2015, 14, 710–725. [Google Scholar] [CrossRef]
- Brandt, E.R.; Sriprakash, K.S.; Hobb, R.I.; Hayman, W.A.; Zeng, W.; Batzloff, M.R.; Jackson, D.C.; Good, M.F. New multi-determinant strategy for a group A streptococcal vaccine designed for the Australian Aboriginal population. Nat. Med. 2000, 6, 455–459. [Google Scholar] [CrossRef]
- Pandey, M.; Ozberk, V.; Calcutt, A.; Langshaw, E.; Powell, J.; Rivera-Hernandez, T.; Ho, M.F.; Phillips, Z.; Batzloff, M.R.; Good, M.F. Streptococcal Immunity Is Constrained by Lack of Immunological Memory following a Single Episode of Pyoderma. PLoS Pathog. 2016, 12, e1006122. [Google Scholar] [CrossRef]
- Olive, C.; Ho, M.-F.; Dyer, J.; Lincoln, D.; Barozzi, N.; Toth, I.; Good, M.F. Immunization with a Tetraepitopic Lipid Core Peptide Vaccine Construct Induces Broadly Protective Immune Responses against Group A Streptococcus. J. Infect. Dis. 2006, 193, 1666–1676. [Google Scholar] [CrossRef][Green Version]
- Brandt, E.R.; Teh, T.; Relf, W.A.; Hobb, R.I.; Good, M.F. Protective and Nonprotective Epitopes from Amino Termini of M Proteins from Australian Aboriginal Isolates and Reference Strains of Group A Streptococci. Infect. Immun. 2000, 68, 6587–6594. [Google Scholar] [CrossRef][Green Version]
- McNeil, S.A.; Halperin, S.A.; Langley, J.M.; Smith, B.; Warren, A.; Sharratt, G.P.; Baxendale, D.M.; Reddish, M.A.; Hu, M.C.; Stroop, S.D.; et al. Safety and Immunogenicity of 26-Valent Group A Streptococcus Vaccine in Healthy Adult Volunteers. Clin. Infect. Dis. 2005, 41, 1114–1122. [Google Scholar] [CrossRef] [PubMed]
- Kotloff, K.L.; Corretti, M.; Palmer, K.; Campbell, J.D.; Reddish, M.A.; Hu, M.C.; Wasserman, S.S.; Dale, J.B. Safety and Immunogenicity of a Recombinant Multivalent Group A Streptococcal Vaccine in Healthy Adults: Phase 1 trial. JAMA 2004, 292, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Moyle, P.M.; Olive, C.; Ho, M.-F.; Good, M.F.; Toth, I. Synthesis of a Highly Pure Lipid Core Peptide Based Self-Adjuvanting Triepitopic Group A Streptococcal Vaccine, and Subsequent Immunological Evaluation. J. Med. Chem. 2006, 49, 6364–6370. [Google Scholar] [CrossRef] [PubMed]
- Dale, J.B.; Penfound, T.A.; Chiang, E.Y.; Walton, W.J. New 30-valent M protein-based vaccine evokes cross-opsonic antibodies against non-vaccine serotypes of group a streptococci. Vaccine 2011, 29, 8175–8178. [Google Scholar] [CrossRef] [PubMed]
- Pastural, E.; McNeil, S.A.; MacKinnon-Cameron, D.; Ye, L.; Langley, J.M.; Stewart, R.; Martin, L.H.; Hurley, G.J.; Salehi, S.; Penfound, T.A.; et al. Safety and immunogenicity of a 30-valent M protein-based group a streptococcal vaccine in healthy adult volunteers: A randomized, controlled phase I study. Vaccine 2020, 38, 1384–1392. [Google Scholar] [CrossRef]
- Canfield, S.M.; Morrison, S.L. The binding affinity of human IgG for its high affinity Fc receptor is determined by multiple amino acids in the CH2 domain and is modulated by the hinge region. J. Exp. Med. 1991, 173, 1483–1491. [Google Scholar] [CrossRef] [PubMed]
- Van De Bovenkamp, F.S.; Hafkenscheid, L.; Rispens, T.; Rombouts, Y. The Emerging Importance of IgG Fab Glycosylation in Immunity. J. Immunol. 2016, 196, 1435–1441. [Google Scholar] [CrossRef] [PubMed]
- Chlewicki, L.K.; Holler, P.D.; Monti, B.C.; Clutter, M.R.; Kranz, D.M. High-affinity, Peptide-specific T Cell Receptors can be Generated by Mutations in CDR1, CDR2 or CDR3. J. Mol. Boil. 2005, 346, 223–239. [Google Scholar] [CrossRef]
- Zha, H.; Matsunami, E.; Blazon-Brown, N.; Koutsogiannaki, S.; Hou, L.; Bu, W.; Babazada, H.; Odegard, K.C.; Liu, R.; Eckenhoff, R.G.; et al. Volatile anesthetics affect macrophage phagocytosis. PLoS ONE 2019, 14, e0216163. [Google Scholar] [CrossRef]
- Ghaffar, K.A.; Marasini, N.; Giddam, A.K.; Batzloff, M.R.; Good, M.F.; Skwarczynski, M.; Toth, I. Liposome-based intranasal delivery of lipopeptide vaccine candidates against group A streptococcus. Acta Biomater. 2016, 41, 161–168. [Google Scholar] [CrossRef]
- Dai, C.; Stephenson, R.J.; Skwarczynski, M.; Toth, I. Application of Fmoc-SPPS, Thiol-Maleimide Conjugation, and Copper(I)-Catalyzed Alkyne-Azide Cycloaddition “Click” Reaction in the Synthesis of a Complex Peptide-Based Vaccine Candidate Against Group A Streptococcus. Methods Mol. Biol. 2020, 2103, 13–27. [Google Scholar] [CrossRef] [PubMed]
- Azuar, A.; Zhao, L.; Hei, T.T.; Nevagi, R.J.; Bartlett, S.; Hussein, W.M.; Khalil, Z.G.; Capon, R.J.; Toth, I.; Skwarczynski, M. Cholic Acid-based Delivery System for Vaccine Candidates against Group A Streptococcus. ACS Med. Chem. Lett. 2019, 10, 1253–1259. [Google Scholar] [CrossRef] [PubMed]
- Nevagi, R.J.; Khalil, Z.G.; Hussein, W.M.; Powell, J.; Batzloff, M.R.; Capon, R.J.; Good, M.F.; Skwarczynski, M.; Toth, I. Polyglutamic acid-trimethyl chitosan-based intranasal peptide nano-vaccine induces potent immune responses against group A streptococcus. Acta Biomater. 2018, 80, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Giudice, E.L.; Campbell, J.D. Needle-free vaccine delivery. Adv. Drug Deliv. Rev. 2006, 58, 68–89. [Google Scholar] [CrossRef] [PubMed]
- Azegami, T.; Yuki, Y.; Kiyono, H. Challenges in mucosal vaccines for the control of infectious diseases. Int. Immunol. 2014, 26, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Miquel-Clopés, A.; Bentley, E.G.; Stewart, J.; Carding, S.R. Mucosal vaccines and technology. Clin. Exp. Immunol. 2019, 196, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Basto, A.P.; Leitao, A. Targeting TLR2 for Vaccine Development. J. Immunol. Res. 2014, 2014, 1–22. [Google Scholar] [CrossRef]
- Zaman, M.; Toth, I. Immunostimulation by Synthetic Lipopeptide-Based Vaccine Candidates: Structure-Activity Relationships. Front. Immunol. 2013, 4, 318. [Google Scholar] [CrossRef]
- Zaman, M.; Abdel-Aal, A.B.; Fujita, Y.; Phillipps, K.S.; Batzloff, M.R.; Good, M.F.; Toth, I. Immunological Evaluation of Lipopeptide Group A Streptococcus (GAS) Vaccine: Structure-Activity Relationship. PLoS ONE 2012, 7, e30146. [Google Scholar] [CrossRef]
- Fuaad, A.A.; Pearson, M.S.; Pickering, D.A.; Becker, L.; Zhao, G.; Loukas, A.C.; Skwarczynski, M.; Toth, I. Lipopeptide Nanoparticles: Development of Vaccines against Hookworm Parasite. Chem. Med. Chem. 2015, 10, 1647–1654. [Google Scholar] [CrossRef]
- Zhong, W.; Skwarczynski, M.; Toth, I. Lipid Core Peptide System for Gene, Drug, and Vaccine Delivery. Aust. J. Chem. 2009, 62, 956–967. [Google Scholar] [CrossRef]
- Skwarczynski, M.; Toth, I. Lipid-Core-Peptide System for Self-Adjuvanting Synthetic Vaccine Delivery. Methods Mol. Biol. 2011, 751, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, T.; Flies, A.; Efebera, Y.; Sherr, D.H. CD40 Ligand-activated, antigen-specific B cells are comparable to mature dendritic cells in presenting protein antigens and major histocompatibility complex class I- and class II-binding peptides. Immunology 2008, 124, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Walker, J.; Jackson, D.C. Identification of canine helper T-cell epitopes from the fusion protein of canine distemper virus. Immunology 2001, 104, 58–66. [Google Scholar] [CrossRef]
- Torresi, J.; Fischer, A.; Grollo, L.; Zeng, W.; Drummer, H.; Jackson, D.C. Induction of neutralizing antibody responses to hepatitis C virus with synthetic peptide constructs incorporating both antibody and T-helper epitopes. Immunol. Cell Biol. 2007, 85, 169–173. [Google Scholar] [CrossRef]
- Chandrudu, S.; Skwarczynski, M.; Pattinson, D.; Apte, S.H.; Doolan, D.L.; Toth, I. Synthesis and immunological evaluation of peptide-based vaccine candidates against malaria. Biochem. Compd. 2016, 4, 1. [Google Scholar] [CrossRef][Green Version]
- Bhattacharjee, S. DLS and zeta potential – What they are and what they are not? J. Control. Release 2016, 235, 337–351. [Google Scholar] [CrossRef]
- Bartlett, S.; Eichenberger, R.M.; Nevagi, R.J.; Ghaffar, K.A.; Marasini, N.; Dai, Y.; Loukas, A.; Toth, I.; Skwarczynski, M. Lipopeptide-Based Oral Vaccine Against Hookworm Infection. J. Infect. Dis. 2019, 221, 934–942. [Google Scholar] [CrossRef]
- Marasini, N.; Giddam, A.K.; Khalil, Z.G.; Hussein, W.M.; Capon, R.J.; Batzloff, M.R.; Good, M.F.; Toth, I.; Skwarczynski, M. Double adjuvanting strategy for peptide-based vaccines: Trimethyl chitosan nanoparticles for lipopeptide delivery. Nanomedicine 2016, 11, 3223–3235. [Google Scholar] [CrossRef]
- Faruck, M.O.; Zhao, L.; Hussein, W.M.; Khalil, Z.G.; Capon, R.J.; Skwarczynski, M.; Toth, I. Polyacrylate-Peptide Antigen Conjugate as a Single-Dose Oral Vaccine against Group A Streptococcus. Vaccines 2020, 8, 23. [Google Scholar] [CrossRef]
- Lortan, J.E.; Kaniuk, A.S.; Monteil, M.A. Relationship of in vitro phagocytosis of serotype 14 Streptococcus pneumoniae to specific class and IgG subclass antibody levels in healthy adults. Clin. Exp. Immunol. 1993, 91, 54–57. [Google Scholar] [CrossRef] [PubMed]





| Vaccine Constructs | Particle Size (nm) ± STD/Ratio (%) | PDI ± STD |
|---|---|---|
| VC-1 | 113 ± 26/(88.1) 7 ± 188.1/(11.7) 5560 ± 0/(0.2) | 0.42 ± 0.071 |
| VC-2 | 134 ± 42/(91.5) 17 ± 3/(5.8) 4574 ± 847/(2.6) | 0.33 ± 0.073 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, C.; Khalil, Z.G.; Hussein, W.M.; Yang, J.; Wang, X.; Zhao, L.; Capon, R.J.; Toth, I.; Stephenson, R.J. Opsonic Activity of Conservative Versus Variable Regions of the Group A Streptococcus M Protein. Vaccines 2020, 8, 210. https://doi.org/10.3390/vaccines8020210
Dai C, Khalil ZG, Hussein WM, Yang J, Wang X, Zhao L, Capon RJ, Toth I, Stephenson RJ. Opsonic Activity of Conservative Versus Variable Regions of the Group A Streptococcus M Protein. Vaccines. 2020; 8(2):210. https://doi.org/10.3390/vaccines8020210
Chicago/Turabian StyleDai, Chuankai, Zeinab G. Khalil, Waleed M. Hussein, Jieru Yang, Xiumin Wang, Lili Zhao, Robert J. Capon, Istvan Toth, and Rachel J. Stephenson. 2020. "Opsonic Activity of Conservative Versus Variable Regions of the Group A Streptococcus M Protein" Vaccines 8, no. 2: 210. https://doi.org/10.3390/vaccines8020210
APA StyleDai, C., Khalil, Z. G., Hussein, W. M., Yang, J., Wang, X., Zhao, L., Capon, R. J., Toth, I., & Stephenson, R. J. (2020). Opsonic Activity of Conservative Versus Variable Regions of the Group A Streptococcus M Protein. Vaccines, 8(2), 210. https://doi.org/10.3390/vaccines8020210













