Seaweed-Based Products and Mushroom β-Glucan as Tomato Plant Immunological Inducers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of the Experiment
2.2. Preparation of Inoculum and Inoculation
2.3. Inducers
2.4. Enzymatic Assays
2.4.1. β-1,3-Glucanase
2.4.2. Peroxidase
2.4.3. Phenylalanine Ammonia Lyase (PAL)
2.5. Statistical Analysis
3. Results
3.1. Tomato Plant Hypersensitivity Reaction to Fusarium oxysporum f. sp. Lycopersici
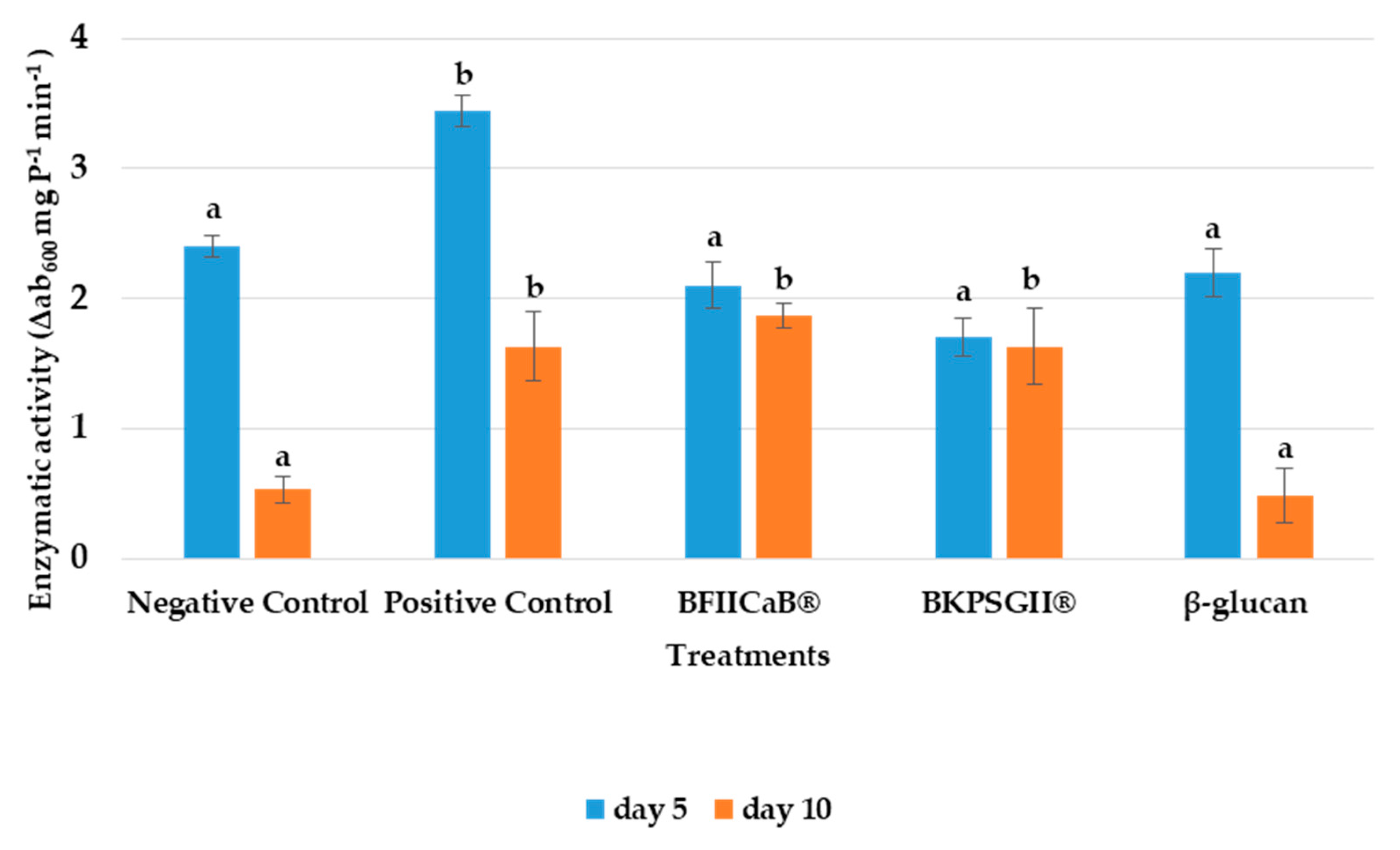
3.2. β-1,3-Glucanase Activity
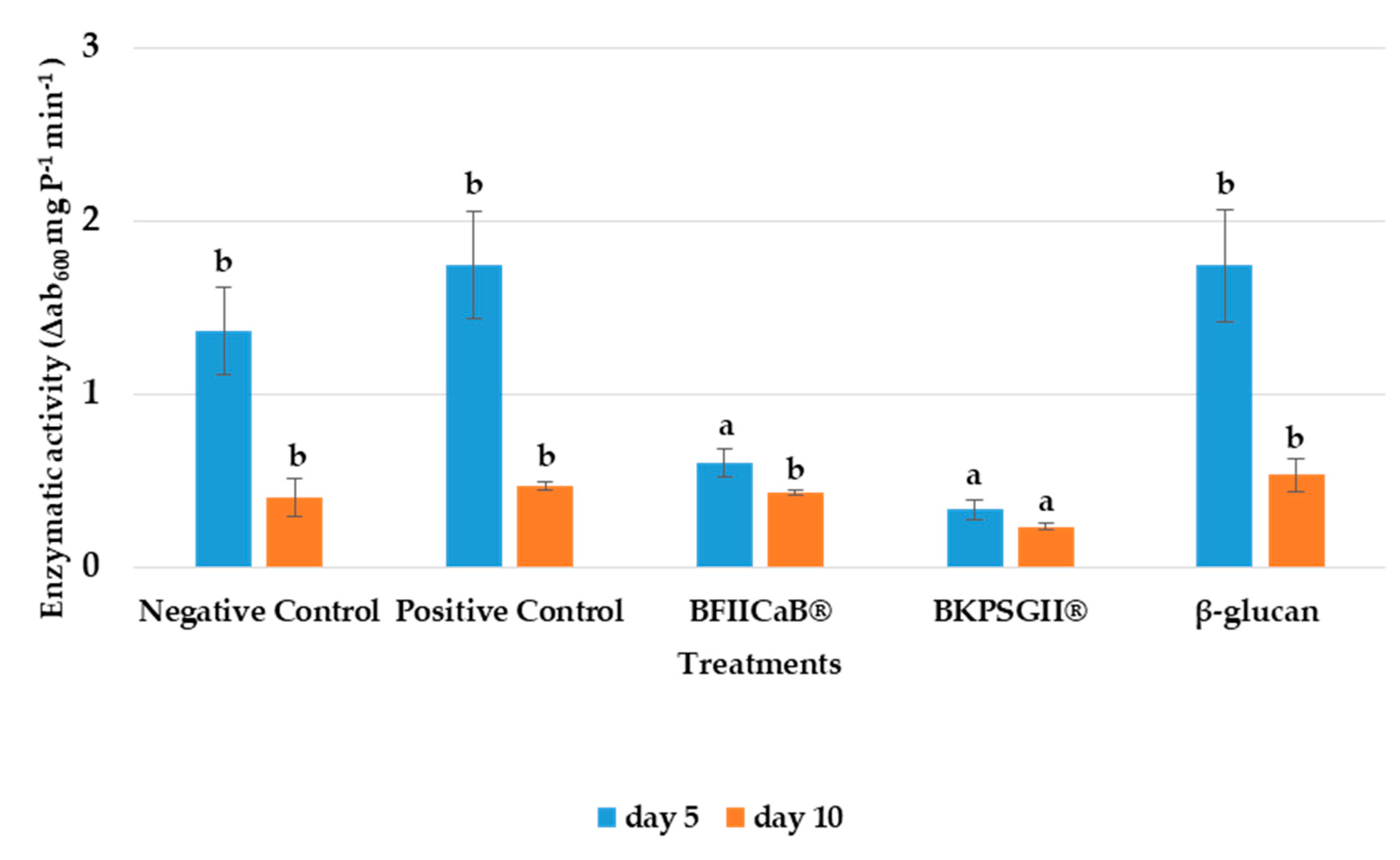
3.3. Peroxidase Activity
3.4. Phenylalanine Ammonia Lyase (PAL) Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- FAO (Food and Agriculture Organization of the United Nations). The Food Insecurity Experience Scale—Frequently Asked Questions—FAQs. Available online: http://www.fao.org/3/a-bl354e.pdf (accessed on 23 July 2020).
- Bettiol, W. Conversão de Sistemas de Produção. In Métodos Alternativos de Controle de 3 Insetos-Praga, Doenças e Plantas Daninhas: Panorama Atual e Perspectivas; Poltronieri, L.S., Ishida, A.K.N., Eds.; Embrapa Amazônia Oriental: Belém, Brazil, 2008. [Google Scholar]
- ODS2|ONU Brasil. Available online: https://nacoesunidas.org/pos2015/ods2/ (accessed on 23 July 2020).
- Bird, G.W.; Edens, T.; Drummond, F.; Groden, E. Design of pest management systems for sustainable agriculture. In Sustainable Agriculture in Temperate Zones; Francis, C.A., Flora, C.B., King, L.D., Eds.; John Willey: New York, NY, USA, 1990; pp. 55–110. [Google Scholar]
- Morandi, M.A.B.; Bettiol, W. Controle biológico de doenças de plantas no brasil marcos históricos do controle biológico de doenças de plantas no brasil. In Biocontrole de Doenças de Plantas: Uso e Perspectivas; Embrapa, Ed.; Ministério da Agricultura, Pecuária e Abastecimento: Esplanada dos Ministérios Bloco D, Brasília, Brasil, 2008; pp. 7–15. ISBN 9788585771478. [Google Scholar]
- Michalak, I.; Chojnacka, K. Algae as production systems of bioactive compounds. Eng. Life Sci. 2015, 15, 160–176. [Google Scholar] [CrossRef]
- Khan, W.; Rayirath, U.P.; Subramanian, S.; Jithesh, M.N.; Rayorath, P.; Hodges, D.M.; Critchley, A.T.; Craigie, J.S.; Norrie, J.; Prithiviraj, B. Seaweed Extracts as Biostimulants of Plant Growth and Development. J. Plant Growth Regul. 2009, 28, 386–399. [Google Scholar] [CrossRef]
- Boukhari, M.E.M.E.; Barakate, M.; Bouhia, Y.; Lyamlouli, K. Trends in Seaweed Extract Based Biostimulants: Manufacturing Process and Beneficial Effect on Soil-Plant Systems. Plants 2020, 9, 359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heil, M.; Bostock, R.M. Induced Systemic Resistance (ISR) Against Pathogens in the Context of Induced Plant Defences. Ann. Bot. 2002, 89, 503–512. [Google Scholar] [CrossRef] [Green Version]
- Van Loon, L.C.; Bakker, P.A.H.M.; Pieterse, C.M.J. Systemic resistance induced by rhizosphere bacteria. Annu. Rev. Phytopathol. 1998, 36, 453–483. [Google Scholar] [CrossRef] [Green Version]
- Sticher, L.; Mauch-Mani, B.; Métraux, A.J. SYSTEMIC ACQUIRED RESISTANCE. Annu. Rev. Phytopathol. 1997, 35, 235–270. [Google Scholar] [CrossRef]
- Aziz, A.; Poinssot, B.; Daire, X.; Adrian, M.; Bézier, A.; Lambert, B.; Joubert, J.-M.; Pugin, A. Laminarin Elicits Defense Responses in Grapevine and Induces Protection against Botrytis cinerea and Plasmopara viticola. Mol. Plant Microbe Interact. 2003, 16, 1118–1128. [Google Scholar] [CrossRef] [Green Version]
- Huffaker, A.; Pearce, G.; Veyrat, N.; Erb, M.; Turlings, T.C.J.; Sartor, R.; Shen, Z.; Briggs, S.P.; Vaughan, M.M.; Alborn, H.T.; et al. Plant elicitor peptides are conserved signals regulating direct and indirect antiherbivore defense. Proc. Natl. Acad. Sci. USA 2013, 110, 5707–5712. [Google Scholar] [CrossRef] [Green Version]
- Cole, D.L. The efficacy of acibenzolar-S-methyl, an inducer of systemic acquired resistance, against bacterial and fungal diseases of tobacco. Crop. Prot. 1999, 18, 267–273. [Google Scholar] [CrossRef]
- Romero, R.S.; García, F.A.O. Indução de resistência em plantas a patógenos por eliciadores de natureza bacteriana. In Biocontrole de Doenças de Plantas: Uso e Perspectivas; Embrapa Meio Ambiente: Jaguariúna-SP, Brazil, 2009; pp. 85–99. [Google Scholar]
- Choudhary, D.K.; Kasotia, A.; Jain, S.; Vaishnav, A.; Kumari, S.; Sharma, K.P.; Varma, A. Bacterial-Mediated Tolerance and Resistance to Plants under Abiotic and Biotic Stresses. J. Plant Growth Regul. 2015, 35, 276–300. [Google Scholar] [CrossRef]
- Singh, P.; Singh, J.; Ray, S.; Rajput, R.S.; Vaishnav, A.; Singh, R.K.; Singh, H.B. Seed biopriming with antagonistic microbes and ascorbic acid induce resistance in tomato against Fusarium wilt. Microbiol. Res. 2020, 237, 126482. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, C.M.J.; Van Loon, L.C. Salicylic acid-independent plant defence pathways. Trends Plant Sci. 1999, 4, 52–58. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, A.A.C.; Neto, E.B.; Coêlho, R.S. Indução de resistência a Fusarium oxysporum f. sp. Tracheiphilum em Caupi: Eficiência de indutores abióticos e atividade enzimática elicitada. Fitopatol. Bras. 2006, 31, 492–499. [Google Scholar] [CrossRef] [Green Version]
- Food and Agriculture Organization of the United Nations. Tomato|Land & Water. Available online: http://www.fao.org/land-water/databases-and-software/crop-information/tomato/en/ (accessed on 23 July 2020).
- Food and Agriculture Organization Corporate Statistical Database (FAOSTAT). New Food Balances. Available online: http://www.fao.org/faostat/en/#data/FBS (accessed on 2 December 2019).
- Reis, A.; Urben, A.; Costa, H.; Boiteux, L.S. Estabelecimento e Caracterização a Nível de Raça de Uma Coleção de Isolados de Fusarium oxysporum f. sp. Lycopersici; Embrapa: Brasilia, Brazil, 2006; pp. 3–6. [Google Scholar]
- Srinivas, C.; Devi, D.N.; Murthy, K.N.; Mohan, C.D.; Lakshmeesha, T.; Singh, B.P.; Kalagatur, N.K.; Niranjana, S.; Hashem, A.; Alqarawi, A.A.; et al. Fusarium oxysporum f. sp. lycopersici causal agent of vascular wilt disease of tomato: Biology to diversity–A review. Saudi J. Boil. Sci. 2019, 26, 1315–1324. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Singh, H.B.; Upadhyay, R.S. Role of fusaric acid in the development of ‘Fusarium wilt’ symptoms in tomato: Physiological, biochemical and proteomic perspectives. Plant Physiol. Biochem. 2017, 118, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Maymon, M.; Hirsch, A.M. Combating Fusarium Infection Using Bacillus-Based Antimicrobials. Microorganisms 2017, 5, 75. [Google Scholar] [CrossRef] [Green Version]
- Soares, C.; Švarc-Gajić, J.; Oliva-Teles, M.T.; Pinto, E.; Nastić, N.; Savić, S.; Almeida, A.; Delerue-Matos, C. Mineral Composition of Subcritical Water Extracts of Saccorhiza Polyschides, a Brown Seaweed Used as Fertilizer in the North of Portugal. J. Mar. Sci. Eng. 2020, 8, 244. [Google Scholar] [CrossRef] [Green Version]
- Chatzissavvidis, C.; Therios, I. Role of algae in agriculture. In Seaweeds: Agricultural Uses, Biological and Antioxidant Agents; Pomin, V.H., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2014; pp. 1–37. ISBN 9781631175756. [Google Scholar]
- Herrera, R.M.H.; Virgen-Calleros, G.; Ruiz-López, M.; Zañudo-Hernandez, J.; Délano-Frier, J.P.; Sánchez-Hernández, C.V. Extracts from green and brown seaweeds protect tomato (Solanum lycopersicum) against the necrotrophic fungus Alternaria solani. Environ. Boil. Fishes 2013, 26, 1607–1614. [Google Scholar] [CrossRef]
- Cluzet, S.; Torregrosa, C.; Jacquet, C.; Lafitte, C.; Fournier, J.; Mercier, L.; Salamagne, S.; Briand, X.; Esquerre-Tugaye, M.-T.; Dumas, B. Gene expression profiling and protection of Medicago truncatula against a fungal infection in response to an elicitor from green algae Ulva spp. Plant Cell Environ. 2004, 27, 917–928. [Google Scholar] [CrossRef]
- Esserti, S.; Smaili, A.; Rifai, L.A.; Koussa, T.; Makroum, K.; Belfaiza, M.; Kabil, E.M.; Faize, L.; Burgos, L.; Alburquerque, N.; et al. Protective effect of three brown seaweed extracts against fungal and bacterial diseases of tomato. Environ. Boil. Fishes 2016, 29, 1081–1093. [Google Scholar] [CrossRef]
- Paulert, R.; Talamini, V.; Cassolato, J.E.F.; Duarte, M.E.R.; Noseda, M.D.; Smania, A., Jr.; Stadnik, M.J. Effects of sulfated polysaccharide and alcoholic extracts from green seaweed Ulva fasciata on anthracnose severity and growth of common bean (Phaseolus vulgaris L.). J. Plant Dis. Prot. 2009, 116, 263–270. [Google Scholar] [CrossRef]
- Paulert, R.; Ebbinghaus, D.; Urlass, C.; Moerschbacher, B.M. Priming of the oxidative burst in rice and wheat cell cultures by ulvan, a polysaccharide from green macroalgae, and enhanced resistance against powdery mildew in wheat and barley plants. Plant Pathol. 2010, 59, 634–642. [Google Scholar] [CrossRef]
- Vera, J.; Castro, J.; Gonzalez, A.; Moenne, A. Seaweed Polysaccharides and Derived Oligosaccharides Stimulate Defense Responses and Protection against Pathogens in Plants. Mar. Drugs 2011, 9, 2514–2525. [Google Scholar] [CrossRef] [PubMed]
- Se-Kwon Kim, K.C. Marine Algae Extracts: Processes, Products, and Applications, 1st ed.; Wiley-VCH: Weinheim, Germany, 2015; ISBN 9783527333271. [Google Scholar]
- Patil, S.; Sriram, S.; Savitha, M.J. 2011—Evaluation of non-pathogenic Fusarium for antagonistic activity against Fusarium wilts of tomato. J. Biol. Control. 2011, 25, 118–123. [Google Scholar]
- Mudgil, D. The interaction between insoluble and soluble fiber. In Dietary Fiber for the Prevention of Cardiovascular Disease; Samaan, R.A., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 35–59. [Google Scholar]
- Park, Y.K.; Ikegaki, M.; Alencar, S.M.; Aguiar, C.L. Determinação da concentração de beta-glucano em cogumelo Agaricus blazei Murill por método enzimático. Food Sci. Technol. 2003, 23, 312–316. [Google Scholar] [CrossRef] [Green Version]
- Bula Serenade—Bayer. Available online: https://www.agrolink.com.br/agrolinkfito/produto/serenade_8518.html (accessed on 2 September 2020).
- Bayer. Serenade (Serenade Opti Fungicide); Bayer S.A.: São Paulo, Brazil, 2019. [Google Scholar]
- Hibar, K.; Daami-Remadi, M.; Hamada, W.; El-Mahjoub, M. Bio-fungicides as an alternative for tomato Fusarium crown and root rot control. Tunis. J. Plant Prot. 2006, 1, 19. [Google Scholar]
- Serenade ASO Fungicide|Crop Science, USA. Available online: https://www.cropscience.bayer.us/products/fungicides/serenade-aso?fbclid=IwAR3hHa6rryVliDXUaYYl6pwxVZDXKwmV96FYWo9mfmEq5V4y2QTXmwiRVcI (accessed on 2 September 2020).
- Cucu, M.A.; Gilardi, G.; Pugliese, M.; Gullino, M.L.; Garibaldi, A. An assessment of the modulation of the population dynamics of pathogenic Fusarium oxysporum f. sp. lycopersici in the tomato rhizosphere by means of the application of Bacillus subtilis QST 713, Trichoderma sp. TW2 and two composts. Boil. Control. 2020, 142, 104158. [Google Scholar] [CrossRef]
- Tian, X.; Zheng, Y. Evaluation of biological control agents for Fusarium wilt in Hiemalis begonia. Can. J. Plant Pathol. 2013, 35, 363–370. [Google Scholar] [CrossRef]
- Dann, E.; Deverall, B.J. Activation of systemic disease resistance in pea by an avirulent bacterium or a benzothiadiazole, but not by a fungal leaf spot pathogen. Plant Pathol. 2000, 49, 324–332. [Google Scholar] [CrossRef]
- Medeiros, F.; Resende, M.; Zhang, H.; Paré, P.; Medeiros, F. Defense gene expression induced by a coffee-leaf extract formulation in tomato. Physiol. Mol. Plant Pathol. 2009, 74, 175–183. [Google Scholar] [CrossRef]
- Ferreira, D.F. SISVAR: A program for statistical analysis and teaching. Rev. Científica Symp. 2008, 6, 36–41. [Google Scholar]
- Swarupa, V.; Ravishankar, K.V.; Rekha, A. Plant defense response against Fusarium oxysporum and strategies to develop tolerant genotypes in banana. Planta 2014, 239, 735–751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiménez, E.; Dorta, F.; Medina, C.; Ramírez, A.; Ramírez, I.; Peña-Cortés, H. Anti-Phytopathogenic Activities of Macro-Algae Extracts. Mar. Drugs 2011, 9, 739–756. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lahaye, M.; Robic, A. Structure and Functional Properties of Ulvan, a Polysaccharide from Green Seaweeds. Biomacromolecules 2007, 8, 1765–1774. [Google Scholar] [CrossRef]
- Shanmugam, V.; Kanoujia, N. Biological management of vascular wilt of tomato caused by Fusarium oxysporum f.sp. lycospersici by plant growth-promoting rhizobacterial mixture. Boil. Control. 2011, 57, 85–93. [Google Scholar] [CrossRef]
- Mohamed, M.A.; Shousha, W.G.; Mahdy, E.M.; Ghazy, A.E.M.; Mohamed, M.M. Biochemical alterations induced in tomato in response to Fusarium oxysporum infection: Purification and characterization of an acidic β-1,3-glucanase. Res. J. Agric. Biol. Sci. 2007, 3, 939–949. [Google Scholar]
- Krebs, S.L.; Grumet, R. Affinity purification and characterization of a β-1,3-glucanase from celery. Plant Sci. 1993, 93, 31–39. [Google Scholar] [CrossRef]
- Kang, L.; Zhou, J.; Wang, R.; Zhang, X.; Liu, C.; Liu, Z.; Yuan, S. Glucanase-Induced Stipe Wall Extension Shows Distinct Differences from Chitinase-Induced Stipe Wall Extension of Coprinopsis cinerea. Appl. Environ. Microbiol. 2019, 85, 85. [Google Scholar] [CrossRef]
- Trouvelot, S.; Varnier, A.-L.; Allegre, M.; Mercier, L.; Baillieul, F.; Arnould, C.; Gianinazzi-Pearson, V.; Klarzynski, O.; Joubert, J.-M.; Pugin, A.; et al. A β-1,3 Glucan Sulfate Induces Resistance in Grapevine against Plasmopara viticola through Priming of Defense Responses, Including HR-like Cell Death. Mol. Plant-Microbe Interact. 2008, 21, 232–243. [Google Scholar] [CrossRef] [Green Version]
- De Aguiar, T.; Schimidt, E.C.; Neto, A.C.D.R.; Di Piero, R.M. Physiological and histological aspects of innate and shiitake-induced resistance against bacterial spot on tomatoes. Eur. J. Plant Pathol. 2020, 157, 1–11. [Google Scholar] [CrossRef]
- Morales, D.; Smiderle, F.R.; Villalva, M.; Abreu, H.; Rico, C.; Santoyo, S.; Iacomini, M.; Soler-Rivas, C. Testing the effect of combining innovative extraction technologies on the biological activities of obtained β-glucan-enriched fractions from Lentinula edodes. J. Funct. Foods 2019, 60, 103446. [Google Scholar] [CrossRef]
- Bindschedler, L.V.; Dewdney, J.; Blee, K.A.; Stone, J.M.; Asai, T.; Plotnikov, J.; Denoux, C.; Hayes, T.; Gerrish, C.; Davies, D.R.; et al. Peroxidase-dependent apoplastic oxidative burst in Arabidopsis required for pathogen resistance. Plant J. 2006, 47, 851–863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chandrashekhara; Raj, S.N.; Manjunath, G.; Deepak, S.; Shetty, H.S. Seed treatment with aqueous extract of Viscum album induces resistance to pearl millet downy mildew pathogen. J. Plant Interact. 2010, 5, 283–291. [Google Scholar] [CrossRef]
- Fernandes, C.F. Estudo da Atividade Peroxidásica em Folhas Primárias de Feijão-de-Corda [Vigna unguiculata (L.) Walp.] cv. Vita 3. Master’s Thesis, Universidade Federal do Ceará, Fortaleza, CE, Brazil, 1998. [Google Scholar]
- Nakazawa, A.; Nozue, M.; Yasuda, H.; Takeba, G.; Kubo, H. Expression Pattern and Gene Structure of Phenylalanine Ammonia-Lyase in Pharbitis nil. J. Plant Res. 2001, 114, 323–328. [Google Scholar] [CrossRef]
- Campos, Â.D.; Ferreira, A.G.; Hampe, M.M.V.; Antunes, I.F.; Brancão, N.; Silveira, E.P.; Da Silva, J.B.; Osório, V.A. Induction of chalcone synthase and phenylalanine ammonia-lyase by salicylic acid and Colletotrichum lindemuthianum in common bean. Braz. J. Plant Physiol. 2003, 15, 129–134. [Google Scholar] [CrossRef]
- Mercier, L.; Lafitte, C.; Borderies, G.; Briand, X.; Esquerre-Tugaye, M.T.; Fournier, J. The algal polysaccharide carrageenans can act as an elicitor of plant defence. New Phytol. 2001, 149, 43–51. [Google Scholar] [CrossRef]
- Saravanan, T.; Bhaskaran, R.; Muthusamy, M. Pseudomonas fluorescens Induced Enzymological Changes in Banana Roots (Cv. Rasthali) against Fusarium Wilt Disease. Plant Pathol. J. 2004, 3, 72–80. [Google Scholar] [CrossRef]
- Saltveit, M.E. Wound induced changes in phenolic metabolism and tissue browning are altered by heat shock. Postharvest Boil. Technol. 2000, 21, 61–69. [Google Scholar] [CrossRef]
- Chen, Y.-A.; Shin, J.-W.; Liu, Z.-H. Effect of light on peroxidase and lignin synthesis in mungbean hypocotyls. Plant Physiol. Biochem. 2002, 40, 33–39. [Google Scholar] [CrossRef]
- Jeyanthi Rebecca, L.; Sharmila, S.; Kowsalya, E. Partial purification of peroxidase from marine algae. Pharm. Lett. 2016, 8, 352–354. [Google Scholar]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melo, P.C.d.; Collela, C.F.; Sousa, T.; Pacheco, D.; Cotas, J.; Gonçalves, A.M.M.; Bahcevandziev, K.; Pereira, L. Seaweed-Based Products and Mushroom β-Glucan as Tomato Plant Immunological Inducers. Vaccines 2020, 8, 524. https://doi.org/10.3390/vaccines8030524
Melo PCd, Collela CF, Sousa T, Pacheco D, Cotas J, Gonçalves AMM, Bahcevandziev K, Pereira L. Seaweed-Based Products and Mushroom β-Glucan as Tomato Plant Immunological Inducers. Vaccines. 2020; 8(3):524. https://doi.org/10.3390/vaccines8030524
Chicago/Turabian StyleMelo, Paulo César de, Carolina Figueiredo Collela, Tiago Sousa, Diana Pacheco, João Cotas, Ana M. M. Gonçalves, Kiril Bahcevandziev, and Leonel Pereira. 2020. "Seaweed-Based Products and Mushroom β-Glucan as Tomato Plant Immunological Inducers" Vaccines 8, no. 3: 524. https://doi.org/10.3390/vaccines8030524
APA StyleMelo, P. C. d., Collela, C. F., Sousa, T., Pacheco, D., Cotas, J., Gonçalves, A. M. M., Bahcevandziev, K., & Pereira, L. (2020). Seaweed-Based Products and Mushroom β-Glucan as Tomato Plant Immunological Inducers. Vaccines, 8(3), 524. https://doi.org/10.3390/vaccines8030524









