Cytokine Profiling of End Stage Cancer Patients Treated with Immunotherapy
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. Treatment
2.3. Sample Collection and Handling
2.4. Cytokines Analyses
2.5. Statistical Analysis
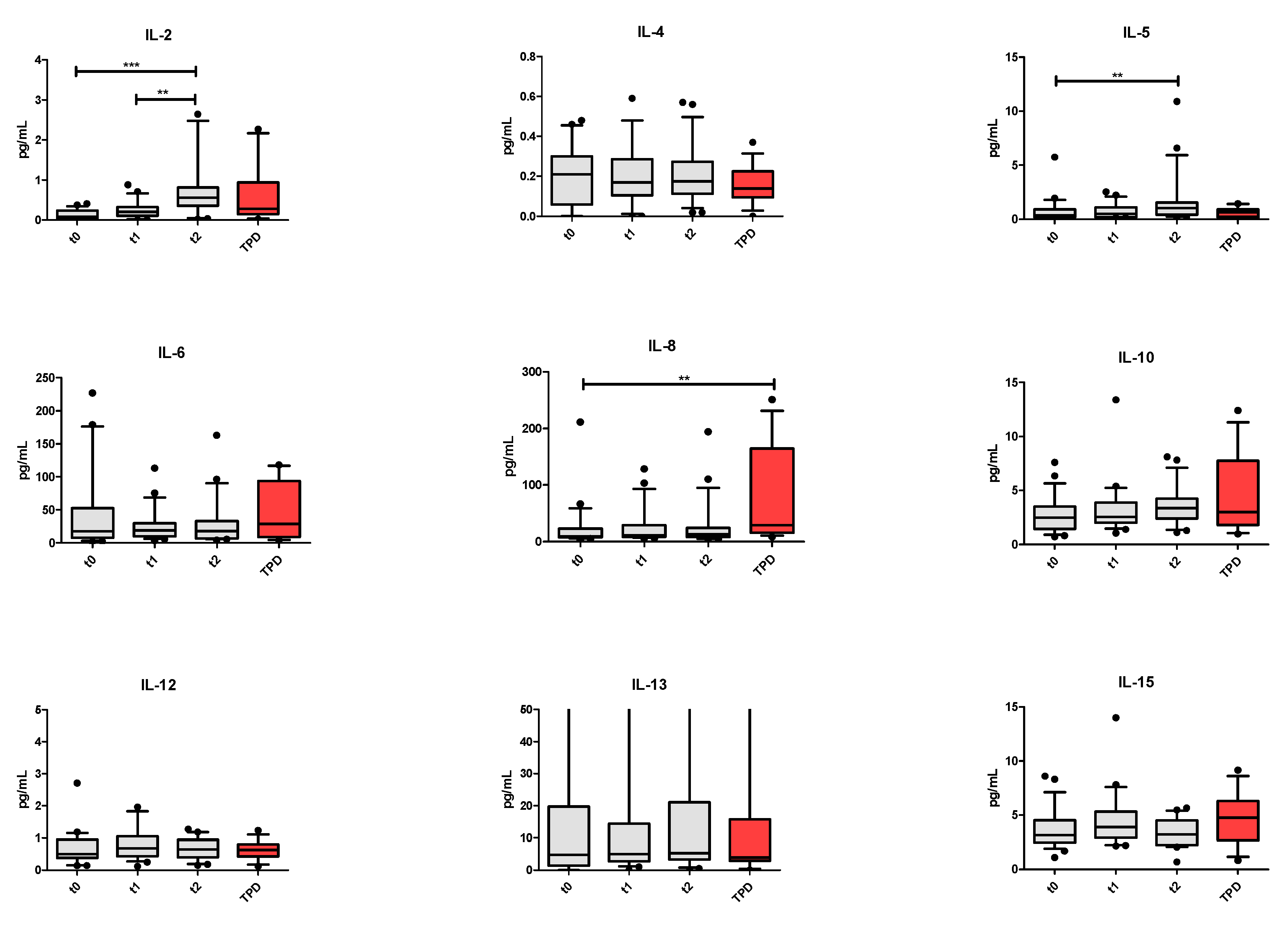
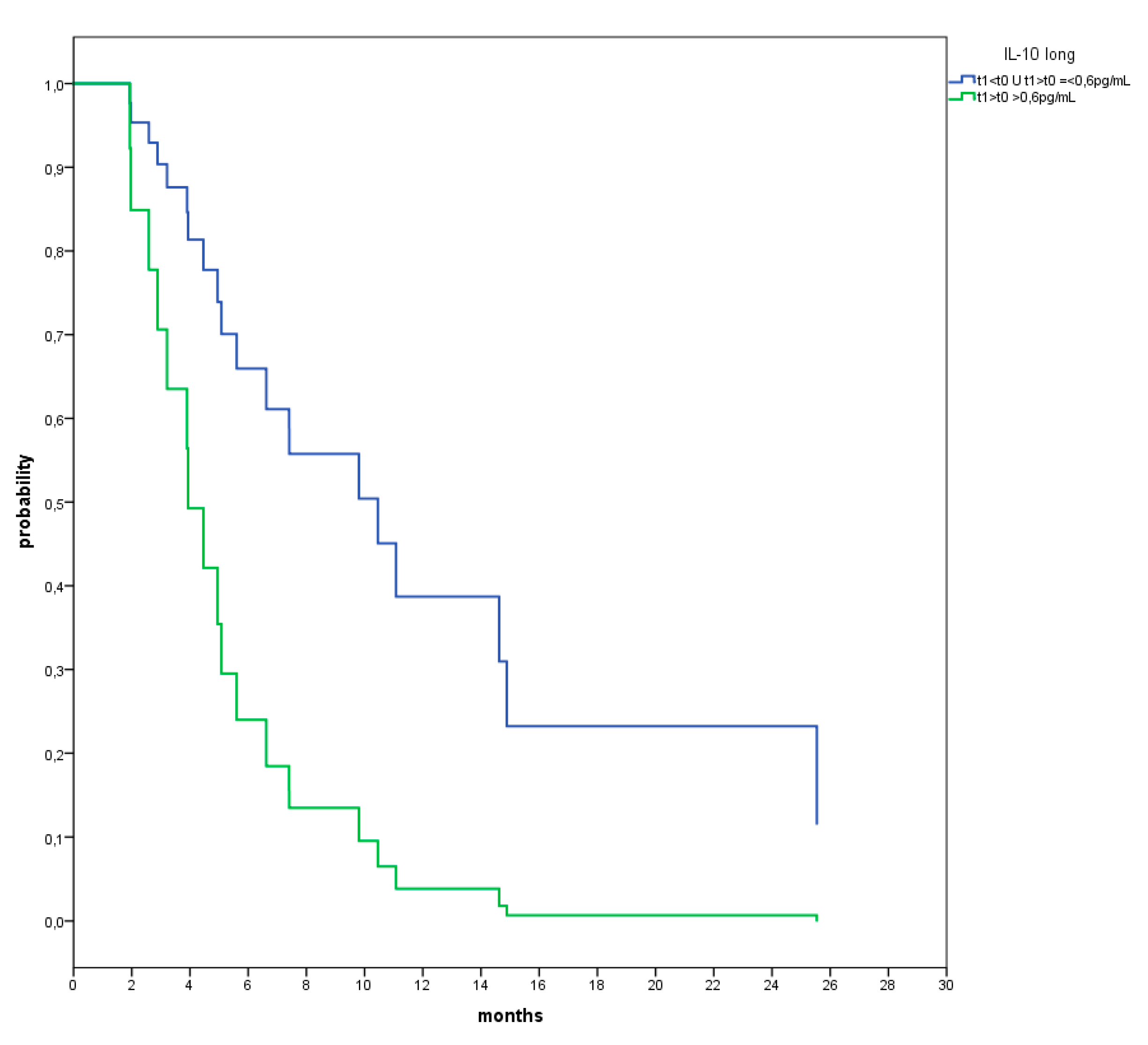
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Qin, S.; Xu, L.; Yi, M.; Yu, S.; Wu, K.; Luo, S. Novel immune checkpoint targets: Moving beyond PD-1 and CTLA-4. Mol. Cancer 2019, 18, 155. [Google Scholar] [CrossRef]
- Barbari, C.; Fontaine, T.; Parajuli, P.; Lamichhane, N.; Jackubski, S.; Lamichhane, P.; Deshmukh, R.R. Immunotherapies and combination strategies for immune-oncology. Int. J. Mol. Sci. 2020, 21, 5009. [Google Scholar] [CrossRef]
- Heinhuis, K.M.; Ros, W.; Kok, M.; Steeghs, N.; Beijnen, J.H.; Schellens, J.H.M. Enhancing antitumor response by combining immune checkpoint inhibitors with chemotherapy. Ann. Oncol. 2019, 30, 219–235. [Google Scholar] [CrossRef] [PubMed]
- Merlano, M.; Denaro, N.; Garrone, O. Immune escape mechanisms in head and neck squamous cell carcinoma and implication for new immunotherapy approach. Curr. Opin. Oncol. 2020, 32, 203–209. [Google Scholar] [CrossRef]
- Kim, R.; Emi, M.; Tanabe, K. Cancer immunoediting from immune surveillance to immune escape. Immunology 2007, 121, 1–14. [Google Scholar] [CrossRef]
- Ascierto, P.A.; Simeone, E.; Sileni, V.C.; Pigozzo, J.; Maio, M.; Altomonte, M.; Del Vecchio, M.; Di Guardo, L.; Marchetti, P.; Ridolfi, R.; et al. Clinical experience with ipilimumab 3 mg/kg: Real-world efficacy and safety data from an expanded access programme cohort. J. Transl. Med. 2014, 12, 116. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, L.; Zitvogel, L.; Eggermont, A.; Marabelle, A. PD-Loma: A cancer entity with a shared sensitivity to the PD-1/PD-L1 pathway blockade. Br. J. Cancer 2019, 120, 3–5. [Google Scholar] [CrossRef]
- Pluvy, J.; Brosseau, S.; Naltet, C.; Opsomer, M.-A.; Cazes, A.; Danel, C.; Khalil, A.; Zalcman, G.; Gounant, V. Lazarus syndrome in nonsmall cell lung cancer patients with poor performance status and major leukocytosis following nivolumab treatment. Eur. Respir. J. 2017, 50, 17. [Google Scholar] [CrossRef]
- Glish, C.; Hagiwara, Y.; Gibelrtson-White, S.; Gao, Y.; Lyckholm, L. Immune checkpoint inhibitor use near the end of life is associated with poor performance status, lower hospice enrolment, and dying in the hospital. Am. J. Hosp. Palliat. Care 2020, 37, 179–184. [Google Scholar] [CrossRef]
- Associazione Italiana Oncologia Medica (AIOM) 2016 Guidelines. Available online: http://media.aiom.it/userfiles/files/doc/LG/metodologia_aggiornamento/2016_LG_AIOM_manuale_operativo.pdf (accessed on 15 July 2016).
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours; revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Ghiringhelli, F.; Menard, C.; Puig, P.E.; Ladoire, S.; Roux, S.; Martin, F.; Solary, E.; Le Cesne, A.; Zitvogel, L.; Chauffert, B. Metronomic cyclophosphamide regimen selectively depletes CD4+CD25+ regulatory T cells and restores T and NK effector functions in end stage cancer patients. Cancer Immunol. Immunother. 2007, 56, 641–648. [Google Scholar] [CrossRef]
- Hong, C.C.; Yao, S.; McCann, S.E.; Dolnick, R.Y.; Wallace, P.K.; Gong, Z.; Quan, L.; Lee, K.P.; Evans, S.S.; Repasky, E.A.; et al. Pretreatment levels of circulating Th1 and Th2 cytokines, and their ratios, are associated with ER-negative and triple negative breast cancers. Breast Cancer Res. Treat. 2013, 139, 477–488. [Google Scholar] [CrossRef]
- Dubitzky, W.; Wolkenhauer, O.; Cho, K.-H.; Yokota, H. Encyclopedia of Systems Biology; Springer: New York, NY, USA, 2013; pp. 154–155. [Google Scholar]
- Amin, A.; White, R.L. Interleukin-2 in renal cell carcinoma: A has-been or a still-viable option? J. Kidney Cancer VHL 2014, 1, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Korbecki, J.; Kojder, K.; Siminska, D.; Bohatyrewicz, R.; Gutowska, I.; Chlubek, D.; Baranowska-Bosiacka, I. CC Chemokine in a tumor: A review of Pro-cancer and anti-cancer properties of the ligands of receptos CCR1, CCR2, CCR3 and CCR4. Int. J. Mol. Sci. 2020, 21, 8412. [Google Scholar] [CrossRef]
- Bakouny, Z.; Choueiri, T.K. IL-8 and cancer prognosis on immunotherapy. Nat. Med. 2020, 26, 650–651. [Google Scholar] [CrossRef] [PubMed]
- Garrone, O.; Michelotti, A.; Paccagnella, M.; Montemurro, F.; Vandone, A.M.; Abbona, A.; Geuna, E.; Vanella, P.; De Angelis, C.; Lo Nigro, C.; et al. Exploratory analysis of circulating cytokines in patients with metastatic breast cancer treated with eribulin: The TRANSERI-GONO (Gruppo Oncologico del Nord-Ovest) study. ESMO Open 2020, 5, e000876. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Wu, D.; Wu, P.; Wang, Z.; Huang, J. Serum IL-10 predicts worse outcome in cancer patients: A meta-analysis. PLoS ONE 2015, 10, e0139598. [Google Scholar] [CrossRef]
- Kawaguchi, K.; Sakurai, M.; Yamamoto, Y.; Suzuki, E.; Tsuda, M.; Kataoka, T.R.; Hirata, M.; Nishie, M.; Nojiri, T.; Kumazoe, M.; et al. Alteration of specific cytokine expression patterns in patients with breast cancer. Sci. Rep. 2019, 9, 24. [Google Scholar] [CrossRef]
- Manetti, R.; Gerosa, F.; Giudizi, M.G.; Biagiotti, R.; Parronchi, P.; Piccinni, M.P.; Sampognaro, S.; Maggi, E.; Romagnani, S.; Trinchieri, G. Interleukin 12 induces stable priming for interferon gamma (IFN-gamma) production during differentiation of human T helper (Th) cells and transient IFN-gamma production in established Th2 cell clones. J. Exp. Med. 1994, 179, 1273–1283. [Google Scholar] [CrossRef]
- Wigginton, J.M.; Park, J.W.; Gruys, M.E.; Young, H.A.; Jorcyk, C.L.; Back, T.C.; Brunda, M.J.; Strieter, R.M.; Ward, J.; Green, J.E.; et al. Complete regression of established spontaneous mammary carcinoma and the therapeutic prevention of genetically programmed neoplastic transition by IL-12/pulse IL-2: Induction of local T cell infiltration, Fas/Fas ligand gene expression, and mammary epithelial apoptosis. J. Immunol. 2001, 166, 1156–1168. [Google Scholar]
- Wigginton, J.M.; Komschlies, K.L.; Back, T.C.; Franco, J.L.; Brunda, M.J.; Wiltrout, R.H. Administration of interleukin 12 with pulse interleukin 2 and the rapid and complete eradication of murine renal carcinoma. J. Natl. Cancer Inst. 1996, 88, 38–43. [Google Scholar] [CrossRef]
- Gollob, J.A.; Schnipper, C.P.; Murphy, E.A.; Ritz, J.; Frank, D.A. The functional synergy between IL-12 and IL-2 involves p38 mitogen-activated protein kinase and is associated with the augmentation of STAT serine phosphorylation. J. Immunol. 1999, 162, 4472–4481. [Google Scholar]
- Konrad, M.W.; Hemstreet, G.; Hersh, E.M.; Mansell, P.W.L.; Mertelsmann, R.; Kolitz, J.E.; Bradley, E.C. Pharmacokinetics of recombinant interleukin-2 in humans. Cancer Res. 1990, 50, 2009–2017. [Google Scholar]
- Tomova, R.; Pomakov, J.; Jacobs, J.J.L.; Adjarov, D.; Popova, S.; Altankova, I.; Den Otter, W.; Krastev, Z. Changes il cytokines profile during local IL-2 therapy in cancer patients. Anticancer Res. 2006, 26, 2037–2048. [Google Scholar] [PubMed]
- Nakamura, Y.; Ozaki, T.; Yanagawa, H.; Yasuoka, S.; Ogura, T. Eosinophil colony-stimulating factor induced by administration of IL-2 into the pleural cavity of patients with malignant pleurisy. Am. J. Respir. Cell Mol. Biol. 1990, 3, 291–300. [Google Scholar] [CrossRef]
- Tomaki, M.; Zhao, L.L.; Lundhal, J.; Sjostrand, M.; Jordana, M.; Linden, A.; O’Byrne, P.; Lotvall, J. Eosinophilopoiesis in a murine model of allergic airway eosinophilia: Involvement of bone marrow IL-5 and IL-5 receptor alpha. J. Immunol. 2000, 165, 4040–4050. [Google Scholar] [CrossRef] [PubMed]
- Schaafsma, M.R.; Falkenburg, J.H.; Landergent, J.E.; Duinkerken, N.; Osantom, S.; Ralph, P.; Kaushansky, K.; Wagenmaker, G.; Van Damme, J.; Willemze, R. In vivo production of interleukin-5, Granulocute-macrophage colony stimulatimg factor, macrophage colony stimulatimg factor, and interleukin-6 during intravenous administration of high dose IL-2 in cancer patients. Blood 1991, 78, 1981–1987. [Google Scholar] [CrossRef]
- Spoormakers, T.J.; Klein, W.R.; Jacobs, J.J.L.; Van Den Ingh, T. Comparison of the efficacy of local treatment of equine sarcoids with IL-2 or cisplatin/IL-2. Cancer Immunol. Immunother. 2003, 52, 179–184. [Google Scholar] [CrossRef]
- Varicchi, G.; Galdiero, M.R.; Loffredo, S.; Lucarini, V.; Marone, G.; Mattei, F.; Marone, G.; Schiavone, G. Eosinophils: The unsung heroes in cancer? Oncoimmunology 2018, 7, e1393134. [Google Scholar] [CrossRef]
- Waugh, D.D.J.; Wilson, C. The Interleukin-8 Pathway in Cancer. Clin. Cancer Res. 2008, 14, 6735–6741. [Google Scholar] [CrossRef]
- Dvorak, H.F. Vascular permeability factor/vascular endothelial growth factor: A critical cytokine in tumor angiogenesis and a potential target for diagnosis and therapy. J. Clin. Oncol. 2002, 20, 4368–4380. [Google Scholar] [CrossRef] [PubMed]
- Roviello, G.; Zanotti, L.; Correale, P.; Gobbi, A.; Wigfield, S.; Guglielmi, A.; Pacifico, C.; Generali, D. Is still there a role for IL-2 for solid tumors other than melanoma or renal cancer? Immunotherapy 2017, 9, 25–32. [Google Scholar] [CrossRef]
- Mahmoudpour, S.H.; Jankowski, M.; Valerio, L.; Becker, C.; Espinola-klein, C.; Konstantinides, S.; Quitzau, K.; Barco, S. Safety of low-dose subcutaneous recombinant interleukin-2: Systematic review and meta-analysis of randomized controlled trials. Sci. Rep. 2019, 9, 7145. [Google Scholar] [CrossRef]
- Merlano, M.; Merlotti, A.; Licitra, L.; Denaro, N.; Fea, E.; Galizia, D.; Di Maio, M.; Fruttero, C.; Curcio, P.; Vecchio, S.; et al. Activation of immune responses in patients with relapsed-metastatic head and neck cancer (CONFRONT phase I-II trial): Multimodality immunotherapy with avelumab, short-course radiotherapy, and cyclophosphamide. Clin. Transl. Radiat. Oncol. 2018, 12, 47–52. [Google Scholar] [CrossRef]


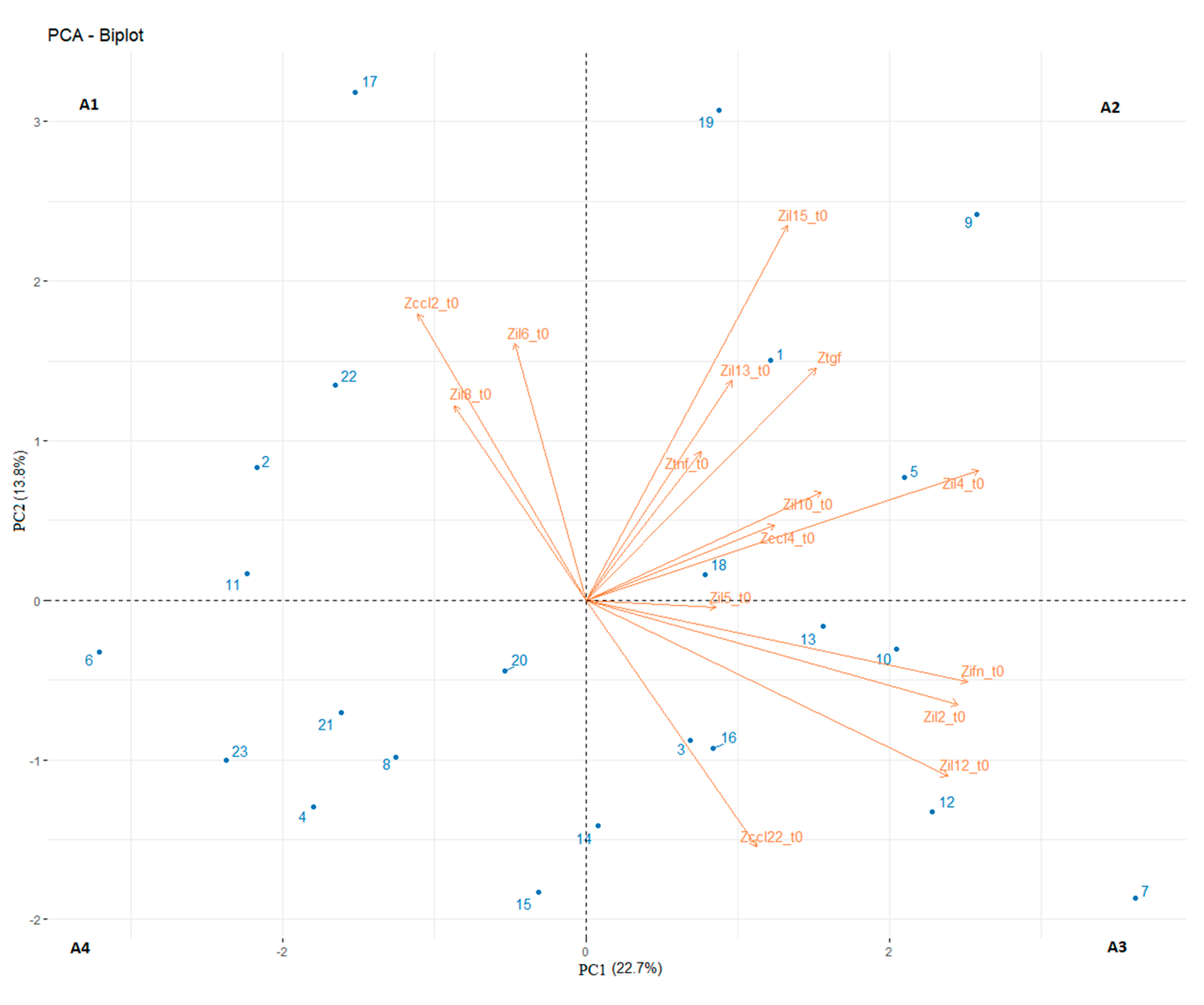


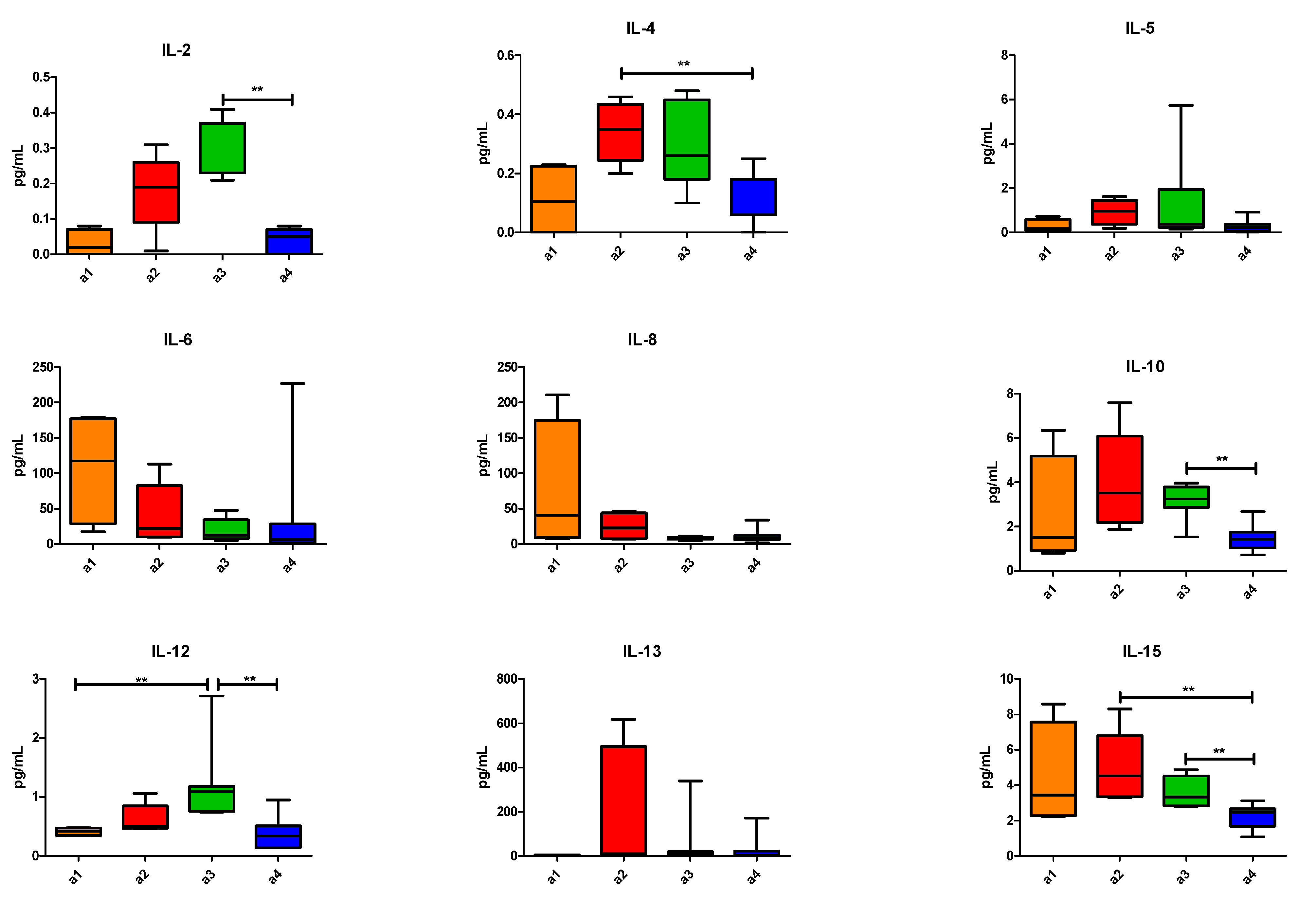
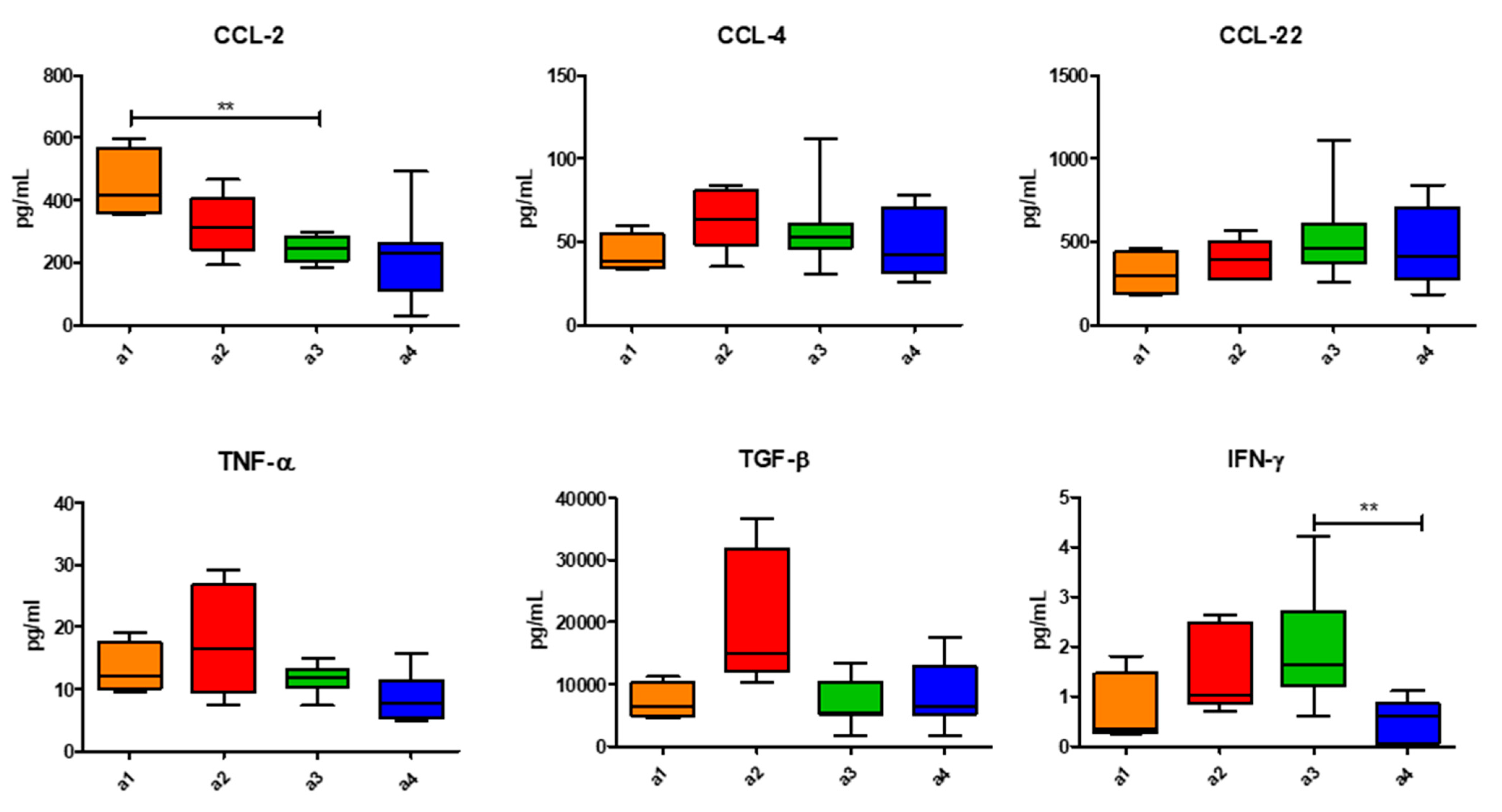



| Patients | Age (Y.O.) | Sex | PS | N° of Prev. Th. | Types of Prev. Th. | Last But One Th-Best Response | Last Th-Best Response | TTF (Weeks) | NLR | LDH | OS (Weeks) | PCA |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| COLON | ||||||||||||
| TC1 | 50 | F | 1 | 3 | Fol + Beva, Fil, Myt + Cape | PD | PD | 19.6 | 5.6 | 511 | 111.1 | 15 |
| TC2 | 72 | F | 1 | 5 | Fol, Fil + Beva, Xil, Fil + 5FU, EDX | PD | PD | 28.8 | 4.3 | 444 | 28.8 | 16 |
| TC3 | 71 | M | 0 | 3 | Xel + Beva, Fil + Beva, Pani | SD | PD | 8.4 | 7.9 | 5942 | 8.4 | 17 |
| TC4 | 69 | F | 0 | 5 | Xel + Beva, Cape, Fil + Beva, Fol, Rego | SD | PD | 12 | 5.6 | 707 | 45.6 | 18 |
| TC5 | 48 | F | 1 | 3 | Fil + Cetu, Fol + Beva, Cetu + CPT11 | SD | PD | 11.7 | 6.9 | 2142 | 21.6 | 19 |
| TC6 | 64 | M | 0 | 3 | Xel, Fil + Beva, Pani | PD | PD | 13.6 | 7.9 | 566 | 63.7 | 20 |
| TC7 | 76 | M | 0 | 6 | Fol + Beva, Cape + Beva, Xil + Beva, Cape, Rego, Fil | PD | PD | 9.4 | 5.1 | 842 | 17 | 21 |
| TC8 | 73 | M | 1 | 2 | Fil + Pani, Xol | SD | PD | 9.3 | 9.3 | 4806 | 22.1 | 22 |
| TC11 | 55 | M | 0 | 3 | Fol, Fil + Beva, Xol | SD | PD | 13.4 | 3.5 | 448 | 49.8 | 23 |
| KIDNEY | ||||||||||||
| TK3 | 75 | M | 1 | 4 | Sun, Eve, Sor, Cabo | PD | SD | 39 | 7.0 | 338 | 73.3 | 13 |
| TK2 | 68 | M | 0 | 4 | Sun, Eve, Sor, NVB | PD | SD | 177.6 | 2.4 | 485 | 177.6 | 12 |
| TK1 | 49 | M | 1 | 5 | Sun, Axi, Eve, Sor, Pazo | SD | PD | 12.6 | 6.2 | 283 | 12.6 | 11 |
| TK4 | 81 | F | 1 | 4 | Sun, Pazo, NVB, Cabo | SD | SD | 29.7 | 5.3 | 486 | 48.3 | 14 |
| PROSTATE | ||||||||||||
| TP1 | 64 | M | 0 | 3 | Doce, Caba, CTX | SD | SD | 16.7 | 4.8 | 917 | 60.1 | 7 |
| TP2 | 69 | M | 0 | 5 | Doce, Caba, NVB, Enza, CTX | PR | PR | 13.9 | 1.6 | 478 | 24.4 | 8 |
| TP3 | 67 | M | 1 | 4 | BAT, Doce, Caba, Enza | PR | PR | 20 | 3.2 | 450 | 42.7 | 9 |
| TP4 | 77 | M | 0 | 5 | LHRHa, Doce, Abi, Caba, Enza | SD | PR | 18.1 | 3.6 | 571 | 32.3 | 10 |
| BREAST | ||||||||||||
| TB1 | 73 | F | 0 | 8 | Exe, Cape + NVB + CTX, Ful, Doxo, NabPa, Cape, Eri, NVB | SD | PD | 11.3 | 2.4 | 872 | 14 | 1 |
| TB2 | 46 | F | 2 | 9 | Tam, NVB, Cape, Meg, Ana, EDX + MTX, PegDoxo, Eri, NabPa | PD | PD | 19.4 | 6.6 | 1431 | 19.4 | 2 |
| TB3 | 62 | F | 1 | 4 | Letro, Ful, Cape, CTX | PD | SD | 9.9 | 4.2 | 821 | 454.1 | 3 |
| TB4 | 64 | F | 0 | 8 | FEC + Pacli, Letro, ExeFul, Cape, Eri, NVB, NabPa | PD | SD | 13.3 | 3.4 | 621 | 17.1 | 4 |
| TB5 | 79 | F | 1 | 8 | Pacli, Ana, Exe, Ful, Cape, PegDoxo, Cape + NVB, Eri | PD | PD | 8.6 | 2.2 | 787 | 8.6 | 5 |
| TB8 | 66 | F | 2 | 8 | FEC + Pacli, Letro, Eve + Exe, Ful, Cape + NVB, Eri, Doxo, NVB | SD | PD | 11.3 | 5.0 | 378 | 11.3 | 6 |
| Group | mPFS (CI) (Months) | mOS (CI) (Months) |
|---|---|---|
| TB | 2.7 (2.1–3.2) | 3.2 (1.6–4.8) |
| TP | 2.8 (2.6–3.0) | 7.4 (3.3–11.5) |
| TK | 6.8 (0.7–12.8) | 11.1 (0.0–24.7) |
| TC | 2.8 (2.6–3.0) | 6.6 (2.1–11.1) |
| Univariate Cox Model for PFS | ||||||
|---|---|---|---|---|---|---|
| Variables | Group | N | HR | S.E. | 95% CI | p Value |
| Cancer site | Kidney | 4 | 0.093 | 1.049 | 0.012–0.723 | 0.023 |
| Other sites | 19 | 1 | ||||
| N° prev. Th. | ≤4 prev. Th. | 12 | 0.549 | 0.456 | 0.225–1.343 | 0.189 |
| >4 prev. Th. | 11 | 1 | ||||
| Areas | Area 3 | 7 | 0.158 | 0.650 | 0.044–0.563 | 0.004 |
| Others | 16 | 1 | ||||
| Multivariate Cox Model for PFS | ||||||
| Cancer site | Kidney | 4 | 0.178 | 1.115 | 0.020–1.587 | 0.122 |
| Other sites | 19 | 1 | ||||
| N° prev. Th. | ≤4 prev. Th. | 12 | 0.796 | 0.467 | 0.318–1.988 | 0.625 |
| >4 prev. Th. | 11 | 1 | ||||
| Areas | Area 3 | 7 | 0.261 | 0.666 | 0.071–0.962 | 0.044 |
| Others | 16 | 1 | ||||
| Univariate Cox Model for OS | ||||||
| Cancer site | Kidney | 4 | 0.273 | 0.754 | 0.062–1.194 | 0.085 |
| Other sites | 19 | 1 | ||||
| N° prev. Th. | ≤4 prev. Th. | 12 | 0.148 | 0.613 | 0.044–0.491 | 0.002 |
| >4 prev. Th. | 11 | 1 | ||||
| Areas | Area 3 | 7 | 0.304 | 0.524 | 0.109–0.849 | 0.023 |
| Others | 16 | 1 | ||||
| Multivariate Cox Model for OS | ||||||
| Cancer site | Kidney | 4 | 0.712 | 0.875 | 0.128–3.960 | 0.698 |
| Other sites | 19 | 1 | ||||
| N° prev. Th. | ≤4 prev. Th. | 12 | 0.177 | 0.612 | 0.053–0.588 | 0.005 |
| >4 prev. Th. | 11 | 1 | ||||
| Areas | Area 3 | 7 | 0.396 | 0.599 | 0.122–1.282 | 0.122 |
| Others | 16 | 1 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Merlano, M.C.; Abbona, A.; Paccagnella, M.; Falletta, A.; Granetto, C.; Ricci, V.; Fea, E.; Denaro, N.; Ruatta, F.; Merlotti, A.; et al. Cytokine Profiling of End Stage Cancer Patients Treated with Immunotherapy. Vaccines 2021, 9, 235. https://doi.org/10.3390/vaccines9030235
Merlano MC, Abbona A, Paccagnella M, Falletta A, Granetto C, Ricci V, Fea E, Denaro N, Ruatta F, Merlotti A, et al. Cytokine Profiling of End Stage Cancer Patients Treated with Immunotherapy. Vaccines. 2021; 9(3):235. https://doi.org/10.3390/vaccines9030235
Chicago/Turabian StyleMerlano, Marco Carlo, Andrea Abbona, Matteo Paccagnella, Antonella Falletta, Cristina Granetto, Vincenzo Ricci, Elena Fea, Nerina Denaro, Fiorella Ruatta, Anna Merlotti, and et al. 2021. "Cytokine Profiling of End Stage Cancer Patients Treated with Immunotherapy" Vaccines 9, no. 3: 235. https://doi.org/10.3390/vaccines9030235
APA StyleMerlano, M. C., Abbona, A., Paccagnella, M., Falletta, A., Granetto, C., Ricci, V., Fea, E., Denaro, N., Ruatta, F., Merlotti, A., Bertetto, O., Crosetto, N., Galizia, D., Basiricò, M., Gammaitoni, L., Sangiolo, D., Aglietta, M., & Garrone, O. (2021). Cytokine Profiling of End Stage Cancer Patients Treated with Immunotherapy. Vaccines, 9(3), 235. https://doi.org/10.3390/vaccines9030235










