Abstract
In this novel conceptual fuel cell vehicle (FCV), an on-board CH4 steam reforming (MSR) membrane reformer (MR) is considered to generate pure H2 for supplying a Fuel Cell (FC) system, as an alternative to the conventional automobile engines. Two on-board tanks are forecast to store CH4 and water, useful for feeding both a combustion chamber (designed to provide the heat required by the system) and a multi tubes Pd-Ag MR useful to generate pure H2 via methane steam reforming (MSR) reaction. The pure H2 stream is hence supplied to the FC. The flue gas stream coming out from the combustion chamber is used to preheat the MR feed stream by two heat exchangers and one evaporator. Then, this theoretical work demonstrates by a 1-D model the feasibility of the MR based system in order to generate 5 kg/day of pure H2 required by the FC system for cruising a vehicle for around 500 km. The calculated CH4 and water consumptions were 50 and 70 kg, respectively, per 1 kg of pure H2. The on-board MR based FCV presents lower CO2 emission rates than a conventional gasoline-powered vehicle, also resulting in a more environmentally friendly solution.
1. Introduction
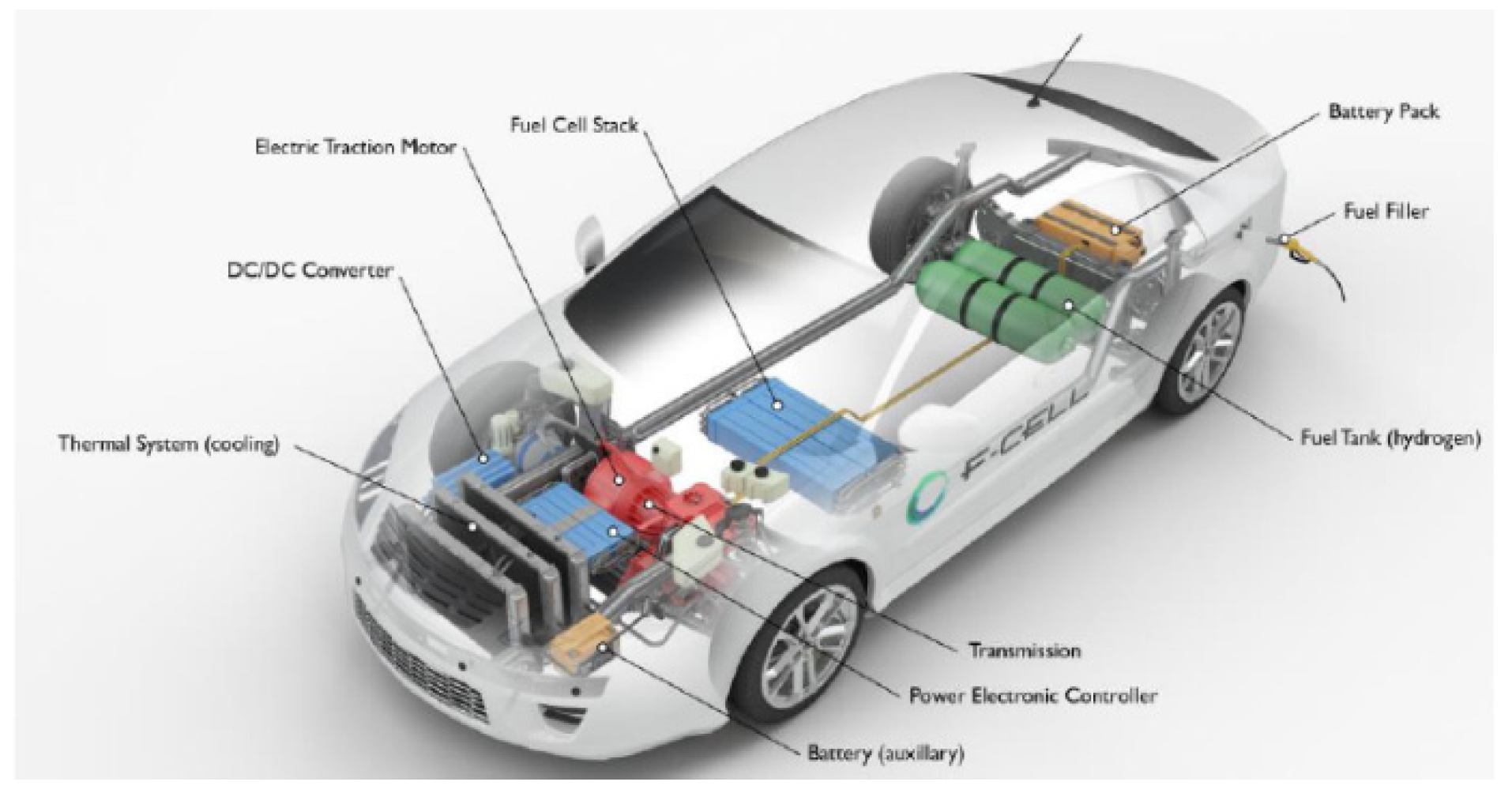
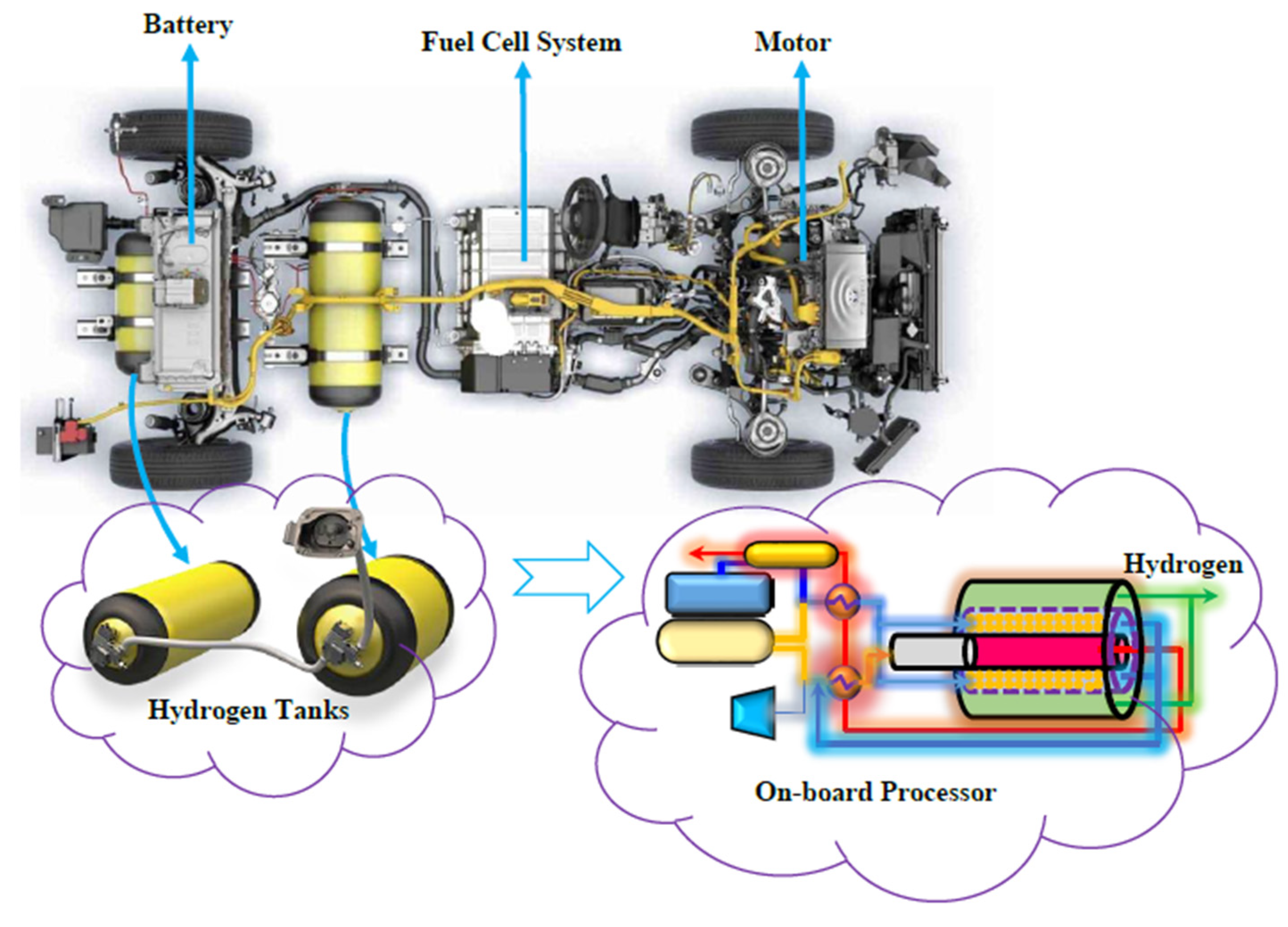
Nowadays, fuel cell vehicles (FCVs) supplied by pure H2 are considered as a viable alternative to the internal combustion engine (ICE) powered vehicles [1]. In a H2 powered FCV, H2 reacts with O2 to generate power that is further transformed into mechanical energy, Figure 1.
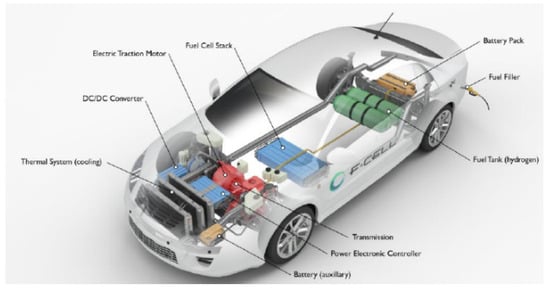
Figure 1.
Conceptual scheme of a fuel cell vehicle (FCV) provided by a H2 pressurized tank [2].
Proton exchange membrane fuel cells (PEMFCs) are the most used fuel cells typology in transportation applications [1,2]. They are commonly supplied by pure H2, provided from an external source and stored in a pressurized tank [2]. With respect to the common ICE powered vehicles, a FCV presents several advantages including silent operations, lower temperature, rapid start-up, lower leakage and corrosion concerns, and lower greenhouse gases (GHGs) emission [3]. Typically, about 4–7 kg of pure H2 are stored in a FCV by means of pressurized tanks [1,2]. In particular, 4 kg of H2 enables a FCV driving-range autonomy without refueling for around 320 km [1].
Nevertheless, some open issues still remain regarding the adoption of FCVs implementing an on-board H2 production or a H2 storage system, both presenting drawbacks needing further development prior to be proposed at larger scale [4]. In this regard, the U.S. Department of Energy (DOE) first established, and it is still under development, the on-board H2 storage program [5], also to introduce new methods to meet the needs of the customers. Another important aspect of research is related to low energy-density of H2, which is responsible for its difficult storage in a car. Indeed, too large or too heavy H2 pressurized tanks would be required to ensure an adequate driving-range as currently guaranteed by the conventional ICEs vehicles [4]. Consequently, the on-board H2 generation via fuel reforming processors seems to be a quite attractive option, particularly if MSR reformers are used to generate H2, because this solution could exploit the CH4 pipeline infrastructure still existing for CH4 fueled ICE vehicles, even though further complexity would be added to the system and an increase of the costs as well. The former issues were analyzed in deep by Ma and Spataru [4]. Different studies in literature address the development of on-board H2 generation systems using different kind of fuels such as methanol, ethanol, and other hydrocarbons. Boettner and Moran [6] and Wu et al. [7] worked on the on-board FCVs development, implementing methanol fuel processors for H2 generation, and they demonstrated the economic advantage of this solution in terms of capital cost with respect to FCVs adopting a direct H2 supplying by tank. Purnima and Jayanti [8] simulated the on-board reforming of ethanol for a FC powered vehicle, also coupling a methane reformer to provide the heat of ethanol reforming reaction. They demonstrated that an overall efficiency of a bit less than 50% could be reached by the proposed system. Zhang et al. [9] analyzed the use of dimethyl ether (DME) as a fuel for on-board reformer in a PEM fuel cell system. They proved that the DME reformer could supply acceptable hydrogen for a FCV [9]. An on-board n-heptane fuel processor system was simulated for driving a 2–3 kW FCV by Karakaya and Avci [10], coupling a methane combustor also for supplying the required heat for n-heptane steam reforming. Darwish at al. [11] investigated the feasibility of hydrogen generation in a 50 kW FCV by on-board naphtha reforming process. They founded that a 70 L fuel tank should be required to guarantee 5 h of continuous driving time due to a consume of 14 L/h of naphtha in the reformer. Myers et al. [12] pointed out how MSR represents an efficient process for H2 generation for FCVs supplying, highlighting the following advantages: (1) N2 dilution effect could be ignored and, consequently, the highest H2 generation may be attained by MSR reaction among the other H2 generation methods (autothermal reforming and partial oxidation); (2) the high temperature of the output gas stream could provide the heat inputs required, (3) no need to compress air to the system because the reaction pressure could be achieved by pumping the reactants; (4) the CH4 price is consistently lower than that of other fuels.
To the aforementioned examples, a consistent number of research studies was dedicated in the last decade to find alternative solutions to the conventional reformers under the purpose of pursuing the principles of Process Intensification Strategy (PIS) [13]. In this regard, the role of membrane engineering in the application of PIS in the fuel reforming was largely studied, demonstrating several advantages over the conventional technologies [14,15,16,17,18,19,20,21]. In the field of H2 generation, many studies were dedicated to the conversion of CH4 into pure H2 via reforming reactions in membrane reformers (MRs), highlighting the operational and economic benefits over the conventional reformers [16,17,18,22,23,24,25]. In particular, metallic membranes were largely studied in MR applications, with palladium and its alloys resulting the dominant materials for preparing inorganic membranes due to the high solubility and permeability of H2 through them [19,20,26,27,28,29,30,31].
The H2 produced in a fully H2 perm-selective Pd-based MR is hence directly useful for a PEMFC supply without needing any additional H2 purification stage. This constitutes the superiority of an on-board Pd-based MR adoption over an on-board conventional reformer, which would require further H2 purification stage processes to purify the reformed H2-rich stream in order to meet the strict purity requirements of a PEMFC (CO content below 10 ppm).
The H2 transport through a dense palladium or palladium-alloy film occurs in six stages under a driving force (from a high to a low pressure gas region): (a) diffusion of molecular H2 at the Pd membrane surface, (b) reversible dissociative adsorption on the Pd surface, (c) dissolution of atomic H into the bulk metal, (d) diffusion of atomic H through the bulk metal, (e) association of H atom on the Pd surface, (f) desorption of molecular H2 from the surface, (g) diffusion of molecular H2 away from the surface [20].
Hence, the H2 permeation through Pd-based membranes is generally described by the Sieverts–Fick law (1):
where, JH2 represents the H2 permeating flux, Q the H2 permeability, δ the thickness of the palladium/palladium alloy film, phps and plps are the H2 partial pressures on the high pressure (feed) and low pressure (permeate) sides, respectively, while “0.5” is the Sieverts pressure exponent, representing the bulk diffusion controlling step of the H2 permeation mechanism [21].
Several applications are noticed in literature regarding the application of Pd-based MRs to carry out MSR reaction for generating pure H2 to be directly supplied to PEMFCs (with standard requirements of highly H2 concentrated streams showing CO concentration below 10 ppm) [5,8,16,18,19], reaching high CH4 conversions generally at lower temperatures than the equivalent conventional reformers [17,32] due to the selective permeation of H2 through the membrane that is responsible for the shift of MSR reaction towards the products, enhancing both the conversion and H2 yield [16,22,23,24,25]. Although a large body of literature on MRs for H2 for generation may be noticed, to our best knowledge there are no applications of FCVs adopting MRs on-board. This choice would result more convenient also than a solid oxide electrolysis cell (SOEC) utilization for generating pure hydrogen. Indeed, the former shows as main issue (still unsolved) the high degradation under a longer operation, which would result as a limit for ensuring stable and long vehicle cruising. The SOEC degradation may occur in cell components such as hydrogen/air electrodes and electrolytes due to structural, electrochemical, and thermal modifications in the components, which in turn would affect the SOEC performance.
The novelty of this work consists of the modeling of an on-board Pd-Ag based multi-tubes MR used for generating 5 kg/day of pure H2 from MSR reaction to be supplied to a FCV (in this case, a pure CH4 feed stream was assumed without sulphur based odorants as-on the contrary-normally present in the already existing CH4 refueling stations). As the diffusion could be neglected in angular direction at low pressures [26,27], a 1-D model was adopted to evaluate macroscopically the size, the efficiency, and the hydrogen generation performance of the proposed on-board membrane-based fuel processor as a viable option for the FCVs development.
2. On-Board Processor Description
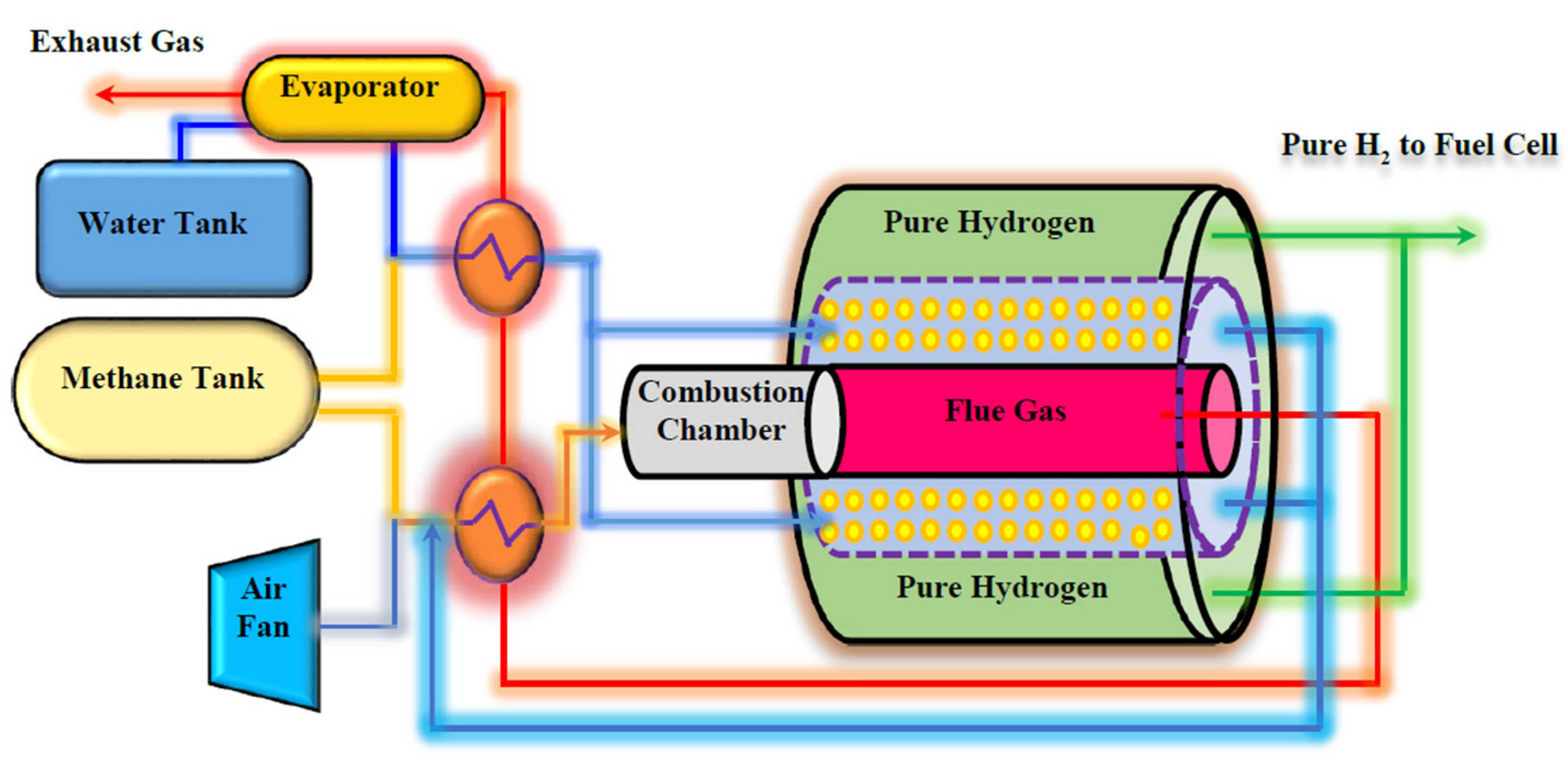
Figure 2 shows the schematic diagram of the proposed Pd-Ag MR on-board to produce pure H2 from MSR reaction for a FCV application.
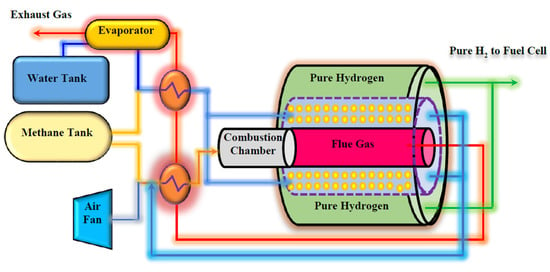
Figure 2.
The proposed system for pure hydrogen producing in an on-board engine.
Two separate tanks are embedded to provide water and CH4 for the system. MSR reaction is carried out in a multi-tubes MR packed with a Ni-based catalyst, adopting tubular unsupported Pd-Ag membranes, 50 µm thick. The heat required for carrying out the MSR reaction is supplied by burning a portion of the CH4 in a combustion chamber, placed before the MR. The combustion chamber produces high temperature flue gas, giving the possibility of generating steam and hot combustion air by flue gas heat exchanging with water and air in heat exchangers and evaporators. The produced steam and hot air are used in the MR.
3. Mathematical Modeling
This theoretical study evaluates the feasibility of the proposed on-board MR and the entire FCV system, which is modeled at steady state and non-isothermal conditions.
3.1. Combustion Chamber Mass and Energy Balances
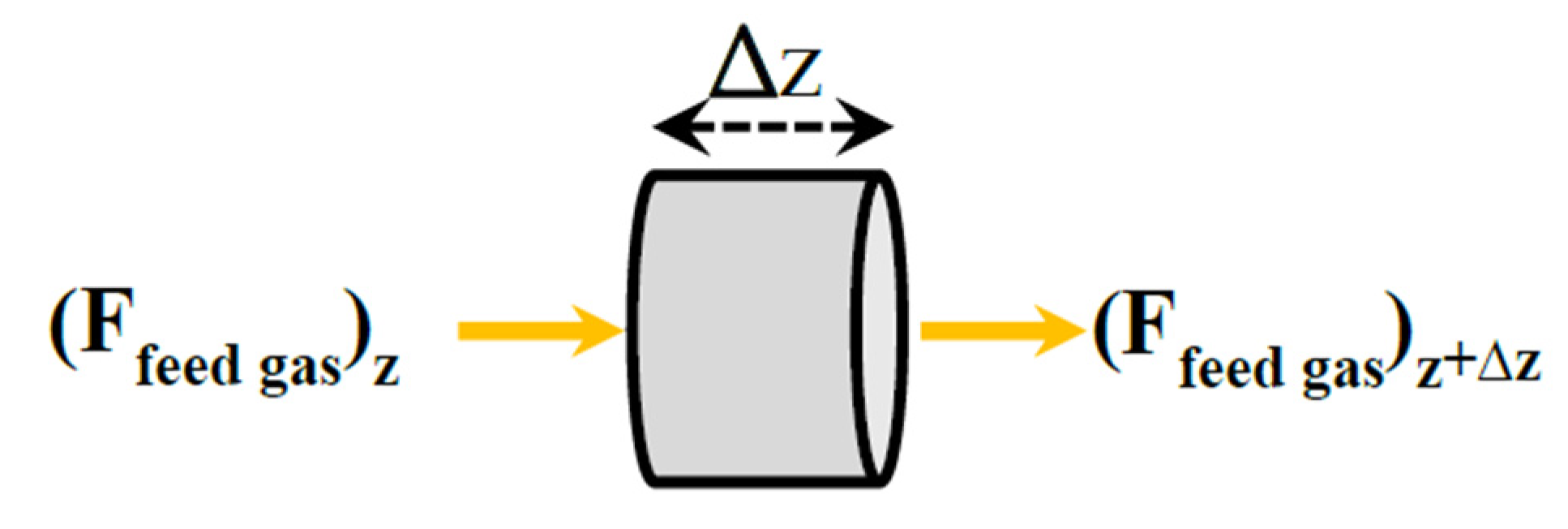
The differential volume element of thickness Δz for the combustion chamber is shown in Figure 3.
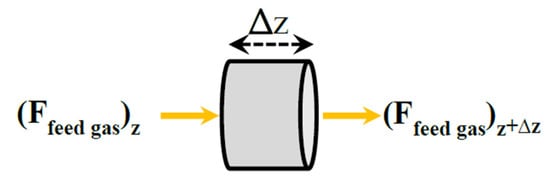
Figure 3.
An elemental volume of the combustion chamber.
The combustion process is assumed complete. The mass and energy balances are written for an adiabatic reactor.
where Fi is the molar flow rate of component i, η is catalyst effectiveness factor, rj is rate of reaction of component j, ρB is the bed density, Tcombustion is the combustion chamber temperature, ∆Hf, is the enthalpy change of combustion reaction and AC is the cross area of the chamber. The boundary conditions of combustion chamber are as following:
where Fi feed1 and Tfeed1 are the molar flow rate of component i and the temperature of the combustion chamber feed.
3.2. Reformer Mass and Energy Balances
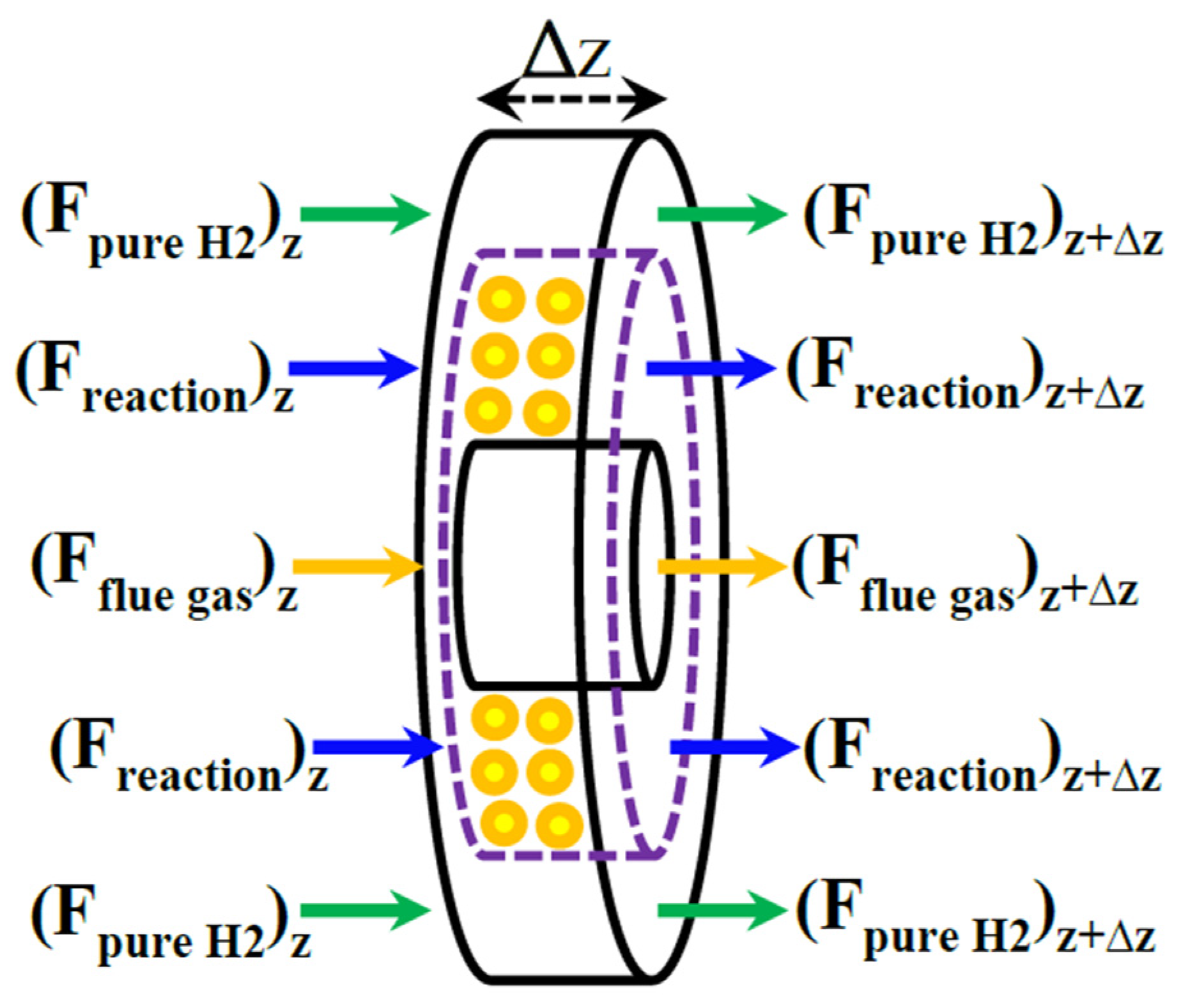
The Pd-Ag MR packed with a Ni-based catalyst is modeled on the basis of an elemental volume of the MR, as shown in Figure 4.
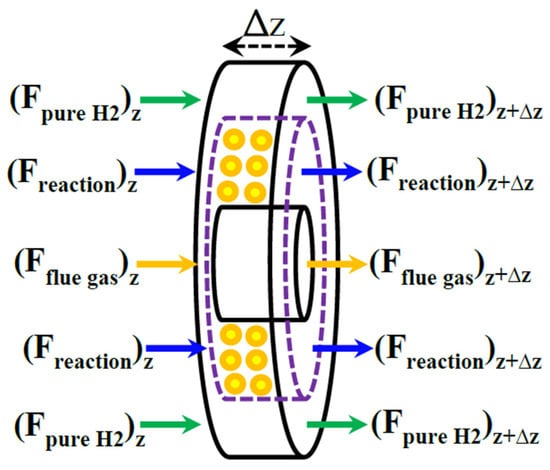
Figure 4.
An elemental volume of the membrane reformer (MR) during MSR reaction process.
It may be schematized in three zones: reaction side, membrane side, and heating media (flue gas zone).
3.2.1. Reaction Side
The reaction side mass balance is written according to Equation (2).
Equation (5) shows the expression to calculate the H2 permeation flux ():
In this equation, and are H2 partial pressures in the reaction and membrane sides, respectively. Furthermore, and are the Pd-Ag membrane thickness and H2 permeability, respectively.
For the dense Pd-Ag membrane is determined by [28], assuming also its full H2 perm-selectivity:
where p0 is the pre-exponential factor (6.82 × 10−5 ) and E0 is the apparent activation energy (13,412 j/mol). R is the universal gas constant, and T is the bulk temperature.
By considering the poisoning effect of possible byproducts formed during MSR reaction such as CO or CO2 on the H2 permeability of the dense Pd-Ag membrane, the following equations developed by Perez et al. [28] were considered:
In Equations (7) and (8), and are the H2 permeation flux in the presence of CO or CO2 and the normalized flux of H2, respectively. is a dimensionless parameter depending only on the temperature (it accounts for additional effects of the adsorbed gas), the adsorption equilibrium constant (Pa−1), and average partial pressure (Pa) of species i. Table 1 shows the quantities of α and Ki at different temperatures. At T > 400 °C, the CO effect on H2 permeability is not comparable with the CO2 one and, at T > 450 °C, also the CO2 effect results to be negligible [28].

Table 1.
The α and Ki parameters of Equation (9) taken from Perez et al. [28].
By considering the Equations from (5) to (8), the H2 flow rate in the reaction side could be calculated by the following equation:
The energy balance of the reaction side could be written as follows:
where Treact is the reaction side temperature, ∆Hf,j is the enthalpy change of reaction i, As is the cross area of reaction side, and Dso is the external diameter of the reaction side.
The following boundary conditions should be applied:
3.2.2. Membrane Side
The membrane side mass balance is:
The boundary conditions for the membrane side are as follows:
3.2.3. Flue Gas Side
The energy balance of the flue gas could be written as follows:
where Tflue gas is the temperature of heating media. The following boundary condition should be applied:
The overall heat transfer coefficient is given by the following correlation:
where hi and ho are the heat transfer coefficients obtained by the following correlation [33]:
where, in the above equation, V is velocity of gas and the other parameters are those of bulk gas phase.
3.3. Reaction Kinetics
Considering the Westbrook and Dryer (WD) global one step mechanism, the following reaction could be considered for the combustion chamber [34]:
Equation (19) addresses the concerning reaction rate for CH4 combustion [34]:
CH4 conversion takes place in the MR via the steam reforming reaction over a Ni based catalyst.
The corresponding reaction rate equations are as follows [31,32]:
The catalyst effectiveness factors for R1, R2, and R3 are 0.01, 0.01, and 0.3, respectively, (taken from [32]). They were considered constant due to the short length of the reaction tubes (50 cm). Thermodynamic and rate constants for Equations (23)–(25) over Ni-based catalysts are given in Table 2, where reactions rates are in mol.kg−1.s−1, and the various partial pressures in bar.

Table 2.
Thermodynamic and rate constants for Equations (23)–(25) are taken from Xu and Froment [32].
3.4. Heat Exchangers and Evaporator Modeling
As shown in Figure 2, two heat exchangers and an evaporator are designed for preheating the CH4, water, and air on their way to the membrane processor. They are placed before the combustion chamber and MR. The general energy balance equations for these two exchangers are:
where CPC and CPH are heat capacities of cold and hot streams in heat exchangers. T, F, and QHEX are the temperature, flow, and heat transfer rate, respectively. The general energy balance equations for evaporators are:
where Qevap. is the heat transfer rate in the evaporator.
4. Numerical Solution
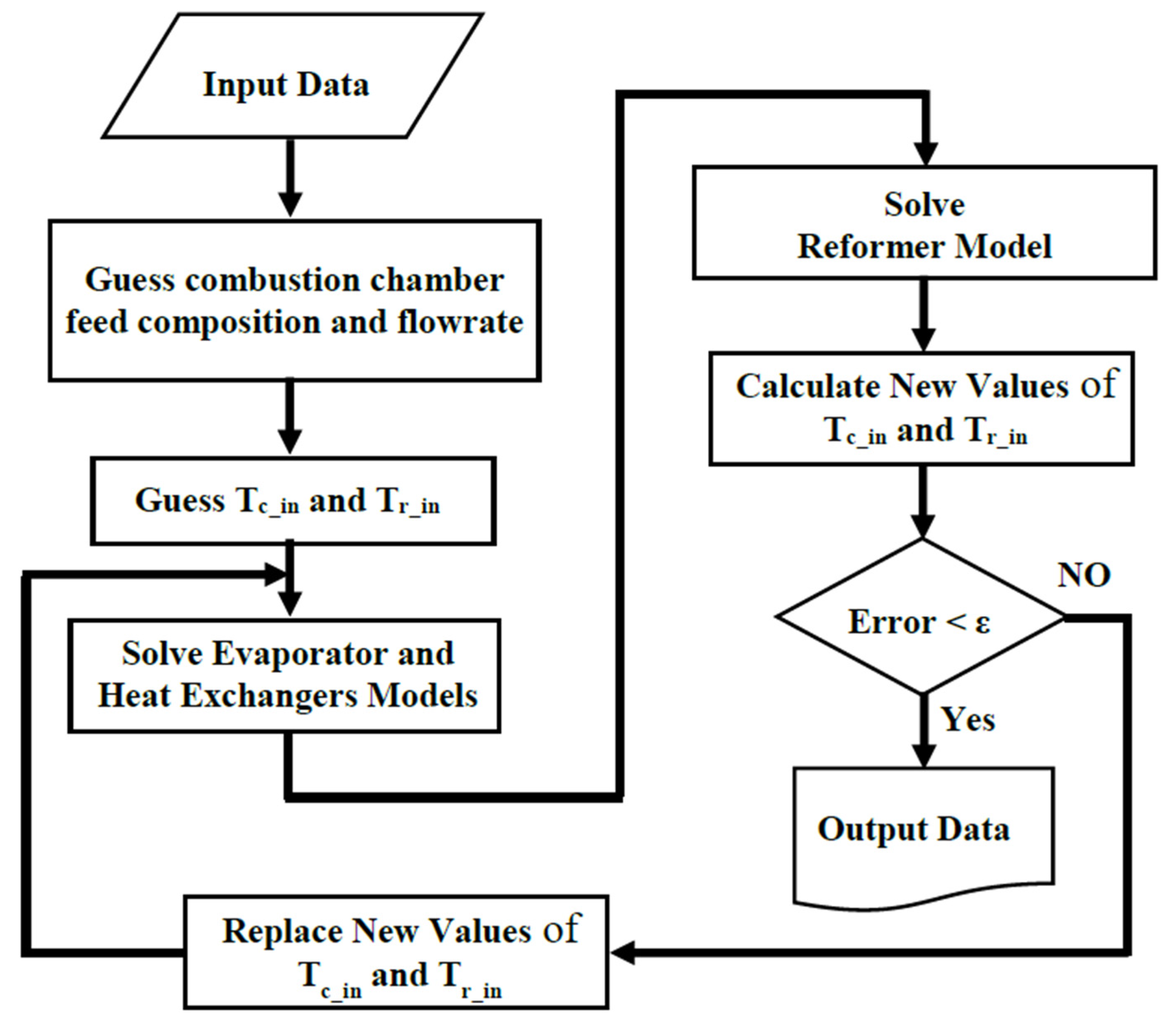
Figure 5 shows the flowchart of the program developed for modeling the proposed on-board processor.
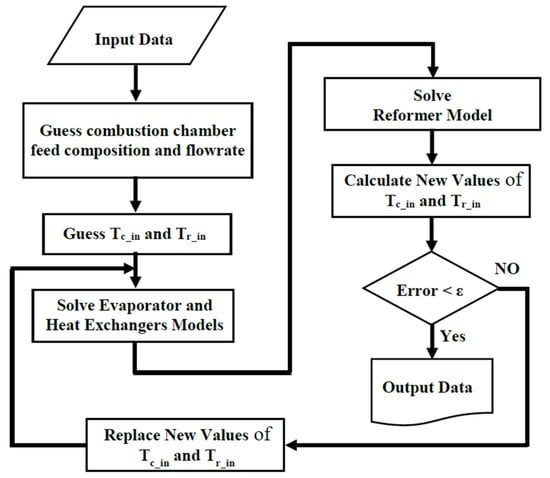
Figure 5.
The flowchart of the program for solving the on-board MSR processor model.
As shown, at first the initial guesses are declared. The guessing parameters are:
- The combustion chamber inlet temperature (Tc,in),
- The reformer reaction side inlet temperature (Tr,in),
- The combustion chamber feed composition and flowrate.
The modeling codes were written by MATLAB 2016a software. The modified Rosenbrock method (ode23s) was used to solve the set of stiff ordinary differential equations (ODEs). A total of 100 nodes were considered for numerical solution of ODEs [35].
5. Results and Discussion
In this section, the developed model is used to investigate the loop’s performance and the impact of various parameters on it. Table 3 shows the specifications of the Toyota Mirai FCV, in which it is reported a H2 tank capacity of 5 kg, a H2 consumption in a combined cycle cruising equal to 0.76 kgH2/100 km, and the total cruising range for this car equal to 500 km [36].

Table 3.
The specifications for Toyota Mirai FCV [36].
The proposed system could be hence replaced with the proposed innovative on-board H2 production system (Figure 6). It should be mentioned that the methane consumption is proportional to FCV speed. Therefore, a fuel injection system should be applied to set the amount of methane injected to the on-board processor.
Figure 6.
The replacement of the on-board MR combined to a H2 storage tank on a FCV [36].
The operating and geometrical parameters of the on-board processor model are listed in Table 4. The parameters were chosen to give a reasonable fuel processor geometry for a five-passenger, mid-size sedan (Toyota Mirai). The membrane tubes thickness of 50 µm was chosen due to the higher permeability and lower manufacturing cost than ticker dense and unsupported membranes present in the market [30,37].

Table 4.
The operating and geometric parameters of the on-board processor.
5.1. The Processor Performance
The methane conversion and H2 recovery were calculated with the following equations:
The developed MR model was validated in our previous work [37]. The required feed flow rates (CH4, water, and air) for the system are summarized in Table 5.

Table 5.
Feed flow rates for the proposed on-board MR.
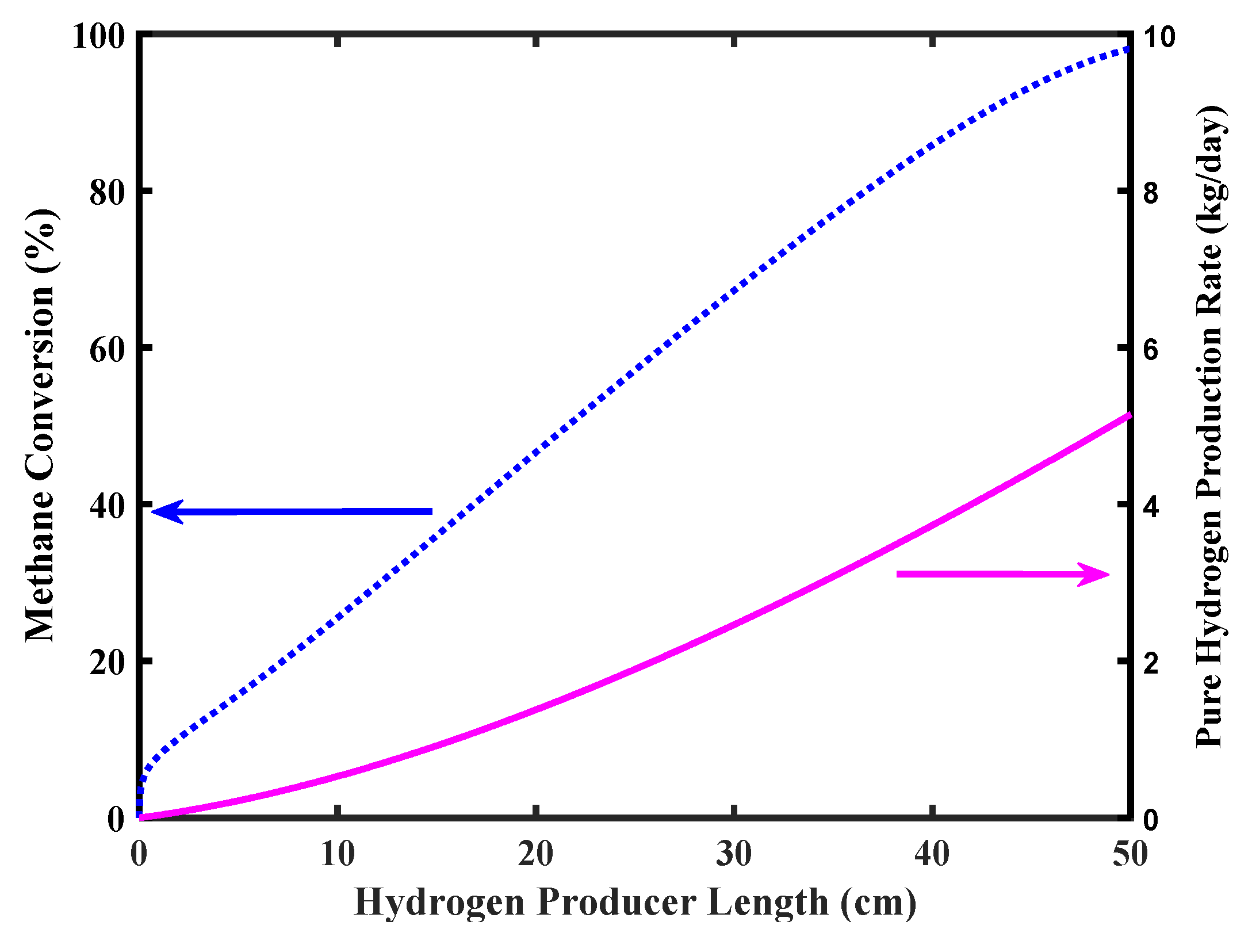
Figure 7 shows CH4 conversion and pure H2 production rate versus the reformer length. Complete CH4 conversion is achieved in the MR outlet stream with a pure H2 production a bit higher than 5 kg/day as in the target of the FCV requirements.
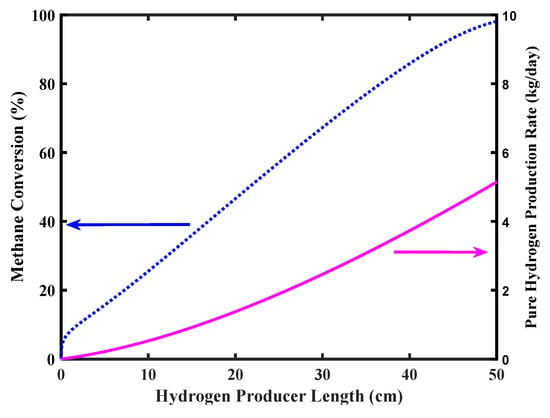
Figure 7.
Methane conversion and pure hydrogen production rate of the MR, Pd-Ag membrane thickness = 50 µm, water to methane ratio = 2.5, inlet temperature = 500 °C, and inlet pressure = 10 bar.
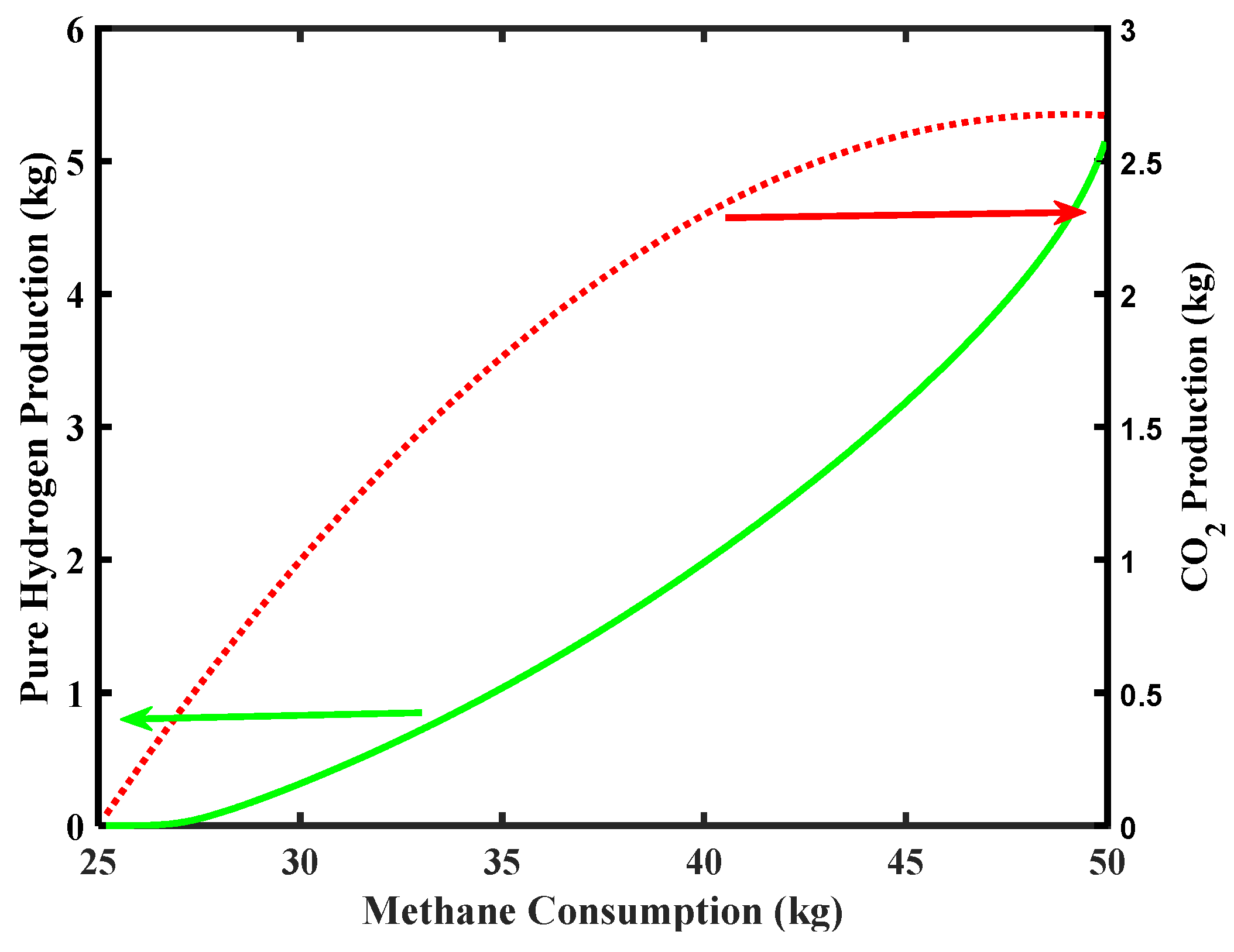
Figure 8 reports the simulation related to CO2 and pure H2 productions along the CH4 consumption. Considering that the Environmental Protection Agency (EPA) report (2018) [38] sets the average CO2 emission for a five-passenger car equal to around 253 g/km (126.5 kg for a cruising range of 500 km), the simulation of the MR of Figure 8 shows that the maximum CO2 production is below this requirement in the outlet stream coming out from the MR, making the MR process theoretically feasible under the aforementioned limitations.
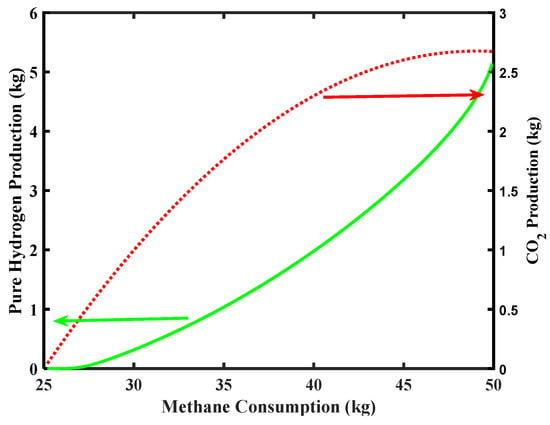
Figure 8.
CO2 and pure H2 productions versus CH4 consumption for Pd-Ag membrane thickness = 50 µm and water to methane ratio = 2.5, inlet temperature = 500 °C, and inlet pressure = 10 bar.
In addition, taking into account that the feed of CH4 depends on the vehicle’s H2 consumption, CH4 consumption is highly depleted for a FCV presenting lower engine power. Steam to CH4 mole ratio is a significant parameter affecting MSR process efficiency. This ratio ranges from 2 to 3 for industrial steam methane reformers [22].
5.2. The Processor Flexibility
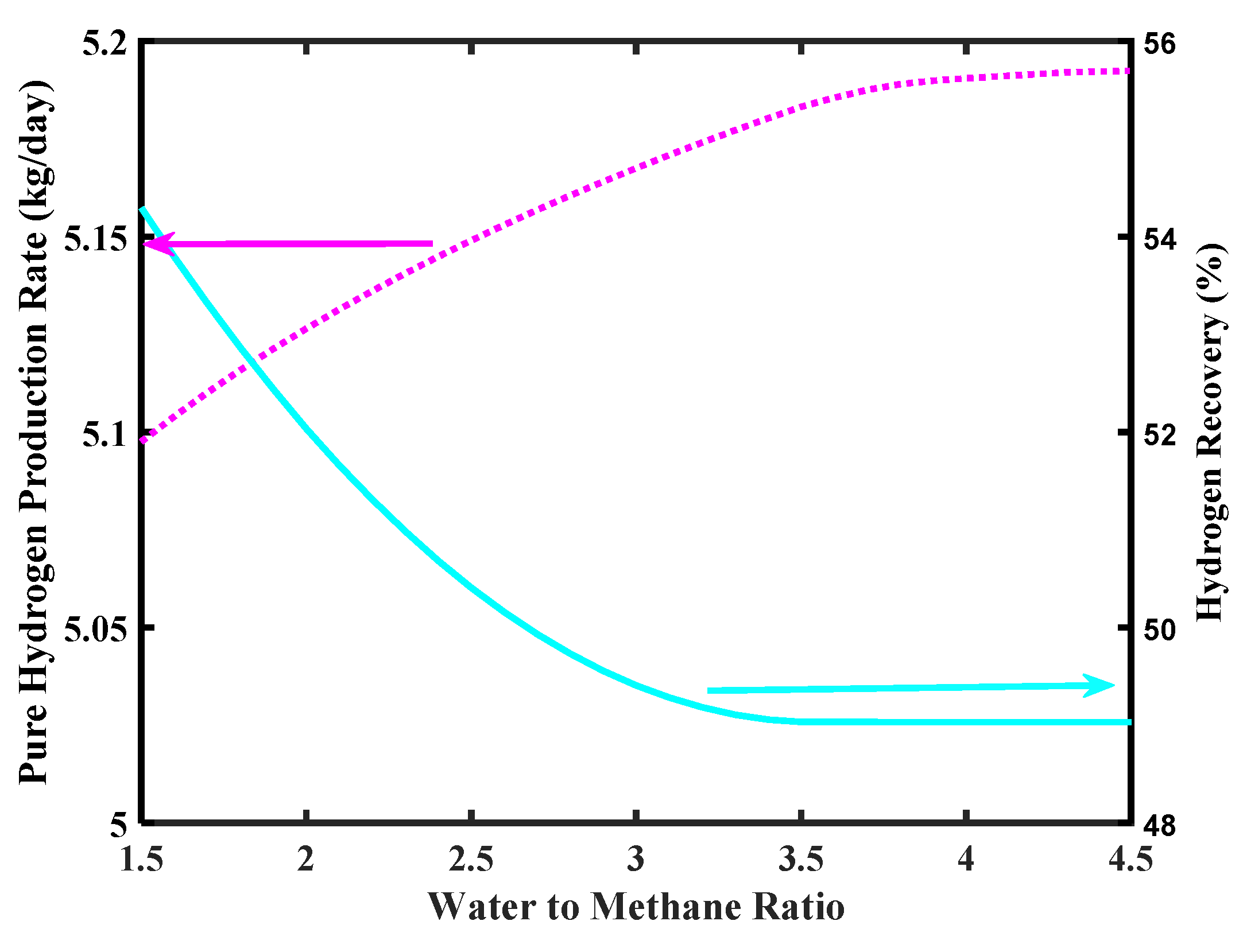
Figure 9 shows the effect of feed ratio on pure H2 production rate. As shown in the simulation, the H2 production increases as the steam to CH4 mole ratio increases, even though values higher than 4 do not affect significantly the H2 production rate, meanwhile overcoming 5 kg/day as requested by the FCV target. On the contrary, the H2 recovery decreases as a consequence of a steam/CH4 ratio increase. Indeed, at higher steam/CH4 ratio the amount of steam in excess lowers the H2 partial pressure in the reaction side, globally determining a lower H2 permeation driving force, which involves a lower amount of H2 collected in the permeate side.
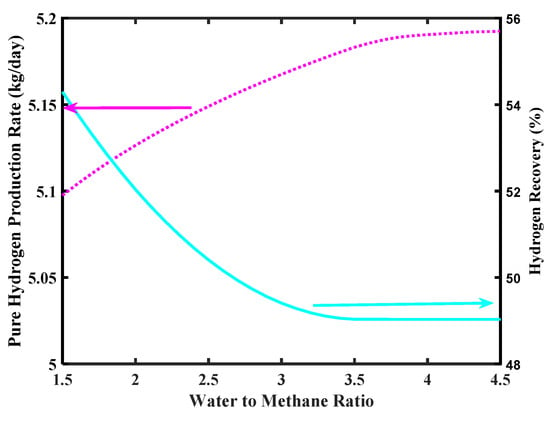
Figure 9.
Pure H2 production rate and hydrogen recovery ratio versus water to methane ratio, Pd-Ag membrane thickness = 50 µm, inlet temperature = 500 °C, and inlet pressure = 10 bar.
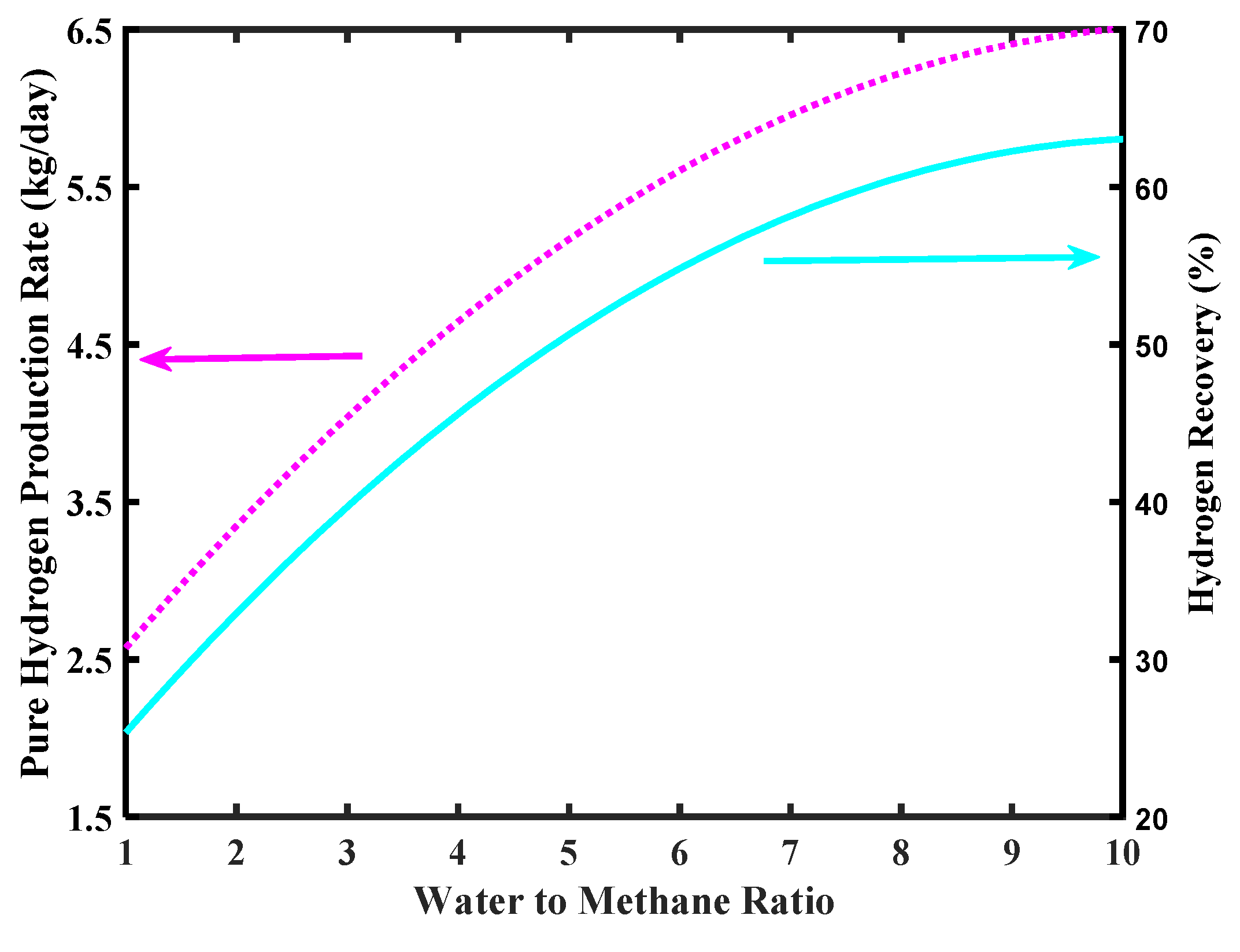
The simulations of Figure 10 show the effect of an increase of pressure on the H2 production rate and H2 recovery. In both cases, a higher pressure favors an increase of production rate and the recovery. This because a higher pressure determines a larger H2 permeation driving force, which enhances CH4 conversion due to an improved “shift effect” on the reaction system, with consequent larger H2 production. For the same reason, more H2 is collected in the permeate stream and the recovery is consequently improved. Nevertheless, unsupported Pd-based membranes suffer relatively high pressure due to structural limits. Therefore, realistically, an operating pressure with unsupported Pd-based membranes should not overcome 5–6 bar.
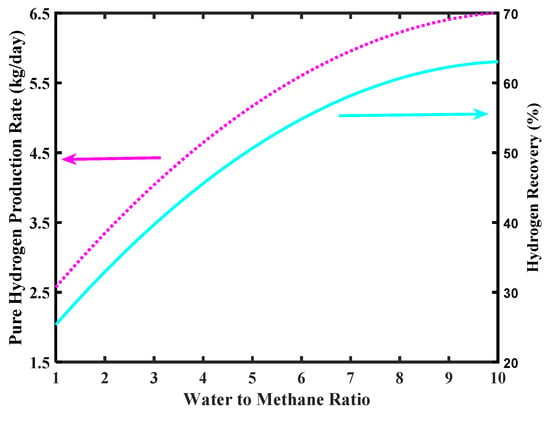
Figure 10.
Pure H2 production rate and hydrogen recovery versus MR pressure, Pd-Ag membrane thickness = 50 µm, water to methane ratio = 2.5, and inlet temperature = 500 °C.
A technical solution for this issue was found by Basile et al. [39], who increased the permeate pressure to respect a total pressure difference across the membrane not higher than 5 bar. Nevertheless, the H2 permeation driving force depends on the H2 partial pressure difference and not on the total pressure difference; high H2 recovery was, then, achieved without structural problems for the dense unsupported Pd-Ag membrane.
However, fuel cell systems for vehicle applications are much more complex than stationary systems because of the need to accommodate load-following transients, identifying this requirement as a fundamental challenge of on-board fuel reforming [40]. In this regard, transient performance for the MRs used in on-board FCVs applications result to be still an open issue, particularly at the shout-down and restart phases. Larger experimental tests will be required for solving the aforementioned problems, which were out of the scopes of this work.
6. Conclusions
H2 storage in FCVs represents one of the most challenging issues in this field. This theoretical work studied the feasibility of implementing a novel on-board MR able to produce pure H2 on demand from MSR reaction. CH4 and water were preheated by waste flue gas and reacted within a synthesis loop equipped with a Pd-Ag MR. The results showed that the system was able to produce 5 kg/day of pure H2, which is suitable for a FCV cruising range of 500 km. A total of 50 kg of CH4 and 70 kg of water were needed as feeding reactants to generate 1 kg of pure H2.
The effects of various parameters such as CH4 consumption rate, water to methane ratio, and operating pressure on the processor performance were investigated. The model showed that large pure H2 production rates were reached by the Pd-Ag MR at both higher steam to CH4 ratio and operating pressure.
Author Contributions
Conceptualization and formal analysis, P.P. and S.M.J.; writing—original draft preparation, P.P., S.M.J. and A.B.; writing—review and editing, A.I. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The authors would like to appreciate the Shiraz University of Technology for protecting this work.
Conflicts of Interest
The authors declare no conflict of interest.
Nomenclature
| Ac | Cross area of reaction side (m) |
| Cpi | Specific heat of component i (J kmol−1K−1) |
| DSo | External diameter of reaction side (m) |
| E0 | Activation energy of membrane |
| Fi | Molar flow rate of component i |
| Standard Gibbs free energy change of reaction i | |
| ∆Hf,i | Enthalpy change of reaction i |
| ki | Reaction rate constant |
| Ki | Adsorption equilibrium constant |
| Pi | Partial pressure of component i (bar) |
| r0 | Membrane tube radius (m) |
| QPd | Hydrogen permeability |
| rj | Rate of reaction of component j (kmol m−3 s−1) |
| R | Universal gas constant (J kmol−1 K−1) |
| Tcombustion | Reaction side temperature (K) |
| Treact | Reaction side temperature (K) |
| Tshell | Temperature of heating stream (K) |
| Ushell | Overall heat transfer coefficient (W m−1 s−1) |
| Methane conversion | |
| Carbon dioxide conversion | |
| z | Axial direction |
| Membrane thickness | |
| Catalyst effectiveness factor | |
| Bed density (kg m−3) | |
| DME | Dimethyl ether |
| DOE | Department of energy |
| FCV | Fuel cell vehicle |
| GHG | Greenhouse gases |
| ICE | Internal combustion engine |
| MR | Membrane reformer |
| MSR | Methane steam reforming |
| ODE | Ordinary differential equations |
| PEMFC | Proton exchange membrane fuel cell |
| PIS | Process intensification strategy |
References
- Wilberforce, T.; El-Hassan, Z.; Khatib, F.N.; Makky, A.A.; Baroutaji, A.; Carton, J.G.; Olabi, A.G. Developments of electric cars and fuel cell hydrogen electric cars. Int. J. Hydrog. Energ. 2017, 42, 25695–25734. [Google Scholar] [CrossRef]
- Manoharan, Y.; Hosseini, S.E.; Butler, B.; Alzhahrani, H.; Senior, B.T.F.; Ashuri, T.; Krohn, J. Hydrogen Fuel Cell Vehicles; Current Status and Future Prospect. Appl. Sci. 2019, 9, 2296. [Google Scholar] [CrossRef]
- Achour, H.; Carton, J.G.; Olabi, A.G. Estimating vehicle emissions from road transport, case study: Dublin City. Appl. Energy 2011, 88, 1957–1964. [Google Scholar] [CrossRef]
- Ma, L.; Spataru, C. The use of natural gas pipeline network with different energy carriers. Ener. Strategy Rev. 2015, 8, 72–81. [Google Scholar] [CrossRef]
- Qi, A.; Peppley, B.; Karan, K. Integrated fuel processors for fuel cell application: A review. Fuel Process. Technol. 2007, 88, 3–22. [Google Scholar] [CrossRef]
- Boettner, D.D.; Moran, M.J. Proton exchange membrane (PEM) fuel cell-powered vehicle performance using direct-hydrogen fueling and on-board methanol reforming. J. Energy 2004, 29, 2317–2330. [Google Scholar] [CrossRef]
- Wu, W.; Chuang, B.N.; Hwang, J.J.; Lin, G.K.; Yang, S.B. Techno-economic evaluation of a hybrid fuel cell vehicle with on-board MeOH-to-H2 processor. Appl. Energy 2019, 238, 401–412. [Google Scholar] [CrossRef]
- Purnima, P.; Jayanti, S. A high-efficiency, auto-thermal system for onboard hydrogen production for low temperature PEM fuel cells using dual reforming of ethanol. Int. J. Hydrog. Energ. 2016, 41, 13800–13810. [Google Scholar] [CrossRef]
- Zhang, T.; Ou, K.; Jung, S.; Choi, B.; Kim, Y.B. Dynamic analysis of a PEM fuel cell hybrid system with an on-board dimethyl ether (DME) steam reformer (SR). Int. J. Hydrog. Energ. 2018, 43, 13521–13531. [Google Scholar] [CrossRef]
- Karakaya, M.; Avci, A.K. Comparison of compact reformer configurations for on-board fuel processing. Int. J. Hydrog. Energ. 2010, 35, 2305–2316. [Google Scholar] [CrossRef]
- Darwish, N.A.; Hilal, N.; Versteeg, G.; Heesink, B. Feasibility of the direct generation of hydrogen for fuel-cell-powered vehicles by on-board steam reforming of naphtha. Fuel 2004, 83, 409–417. [Google Scholar] [CrossRef]
- Myers, D.B.; Ariff, G.D.; James, B.D. Cost and Performance Comparison of Stationary Hydrogen Fueling Appliances. Task 2 Report 2002. Available online: https://www.nrel.gov/docs/fy02osti/32405b2.pdf (accessed on 13 February 2020).
- Boodhoo, K.; Harvey, A. Process intensification for green chemistry. Chem. Listy. 2013, 107, 665–669. [Google Scholar]
- Drioli, E.; Barbieri, G.; Brunetti, A. Membrane Engineering for the Treatment of Gases: Vol. 2, Gas-Separation Issues Combined with Membrane Reactors; RSC: Cambridge, UK, 2017; pp. 1–366. ISBN ISBN 978-1-78262-875-0. [Google Scholar] [CrossRef]
- Bagnato, G.; Iulianelli, A.; Sanna, A.; Basile, A. Glycerol production and transformation: A critical review with particular emphasis on glycerol reforming reaction for producing hydrogen in conventional and membrane reactors. Membranes 2017, 7, 17. [Google Scholar] [CrossRef]
- Iulianelli, A.; Liguori, S.; Wilcox, J.; Basile, A. Advances on methane steam reforming to produce hydrogen through membrane reactors technology: A review. Catal. Rev. Sci. Eng. 2016, 58, 1–35. [Google Scholar] [CrossRef]
- Wang, H.; Liu, M.; Kong, H.; Hao, Y. Thermodynamic analysis on mid/low temperature solar methane steam reforming with hydrogen permeation membrane reactors. Appl. Therm. Eng. 2019, 152, 925–936. [Google Scholar] [CrossRef]
- Yan, Y.-F.; Zhang, L.; Li, L.-X.; Tang, Q. Progress in catalytic membrane reactors for high purity hydrogen production. J. Inorg. Mat. 2011, 26, 1233–1243. [Google Scholar] [CrossRef]
- Al-Mufachi, N.A.; Rees, N.V.; Steinberger-Wilkens, R. Hydrogen selective membranes: A review of palladium-based dense metal membranes. Ren. Sust. En. Rev. 2015, 47, 540–551. [Google Scholar] [CrossRef]
- Yun, S.; Oyama, S.T. Correlations in palladium membranes for hydrogen separation: A review. J. Membr. Sci. 2011, 375, 28. [Google Scholar] [CrossRef]
- Caravella, A.; Scura, F.; Barbieri, G.; Drioli, E. Sieverts law empirical exponent for Pd-based membranes: Critical analysis in pure H2 permeation. J. Phys. Chem. B 2010, 114, 6033–6047. [Google Scholar] [CrossRef]
- Iaquaniello, G.; Giacobbe, F.; Morico, B.; Cosenza, S.; Farace, A. Membrane reforming in converting natural gas to hydrogen: Production costs, Part II. Int. J. Hydrog. Energ. 2008, 33, 6559–6601. [Google Scholar] [CrossRef]
- Lu, N.; Xie, D. Novel membrane reactor concepts for hydrogen production from hydrocarbons: A review. Int. J. Chem. Reactor Eng. 2016, 14, 1–31. [Google Scholar] [CrossRef]
- Yücel, O.; Alaittin, H.M. Comprehensive study of steam reforming of methane in membrane reactors. J. Energy. Resour. Technol. 2016, 138, 052204. [Google Scholar] [CrossRef]
- Jørgensen, S.L.; Nielsen, P.E.H.; Lehrmann, P. Steam reforming of methane in a membrane reactor. Catal. Today 1995, 25, 303–307. [Google Scholar] [CrossRef]
- Oyama, S.T.; Hacarlioglu, P. The boundary between simple and complex descriptions of membrane reactors: The transition between 1-D and 2-D analysis. Int. J. Hydrog. Energ. 2009, 337, 188–199. [Google Scholar] [CrossRef]
- Rahimpour, M.R.; Samimia, F.; Babapoor, A.; Tohidian, T.; Mohebi, S. Palladium membranes applications in reaction systems for hydrogen separation and purification: A review. Chem. Eng. Proc. Proc. Intens. 2017, 121, 24–49. [Google Scholar] [CrossRef]
- Perez, P.; Cornaglia, C.A.; Mendes, A.; Madeira, L.M.; Tosti, S. Surface effects and CO/CO2 influence in the H2 permeation through a Pd-Ag membrane: A comprehensive model. Int. J. Hydrog. Energ. 2015, 40, 6566–6572. [Google Scholar] [CrossRef]
- Hamilton, H. Palladium-based membranes for hydrogen separation. Platin. Met. Rev. 2012, 56, 117–123. [Google Scholar]
- Plazaola, A.A.; Pacheco, T.A.D.; Van, S.A.M.; Gallucci, F. Recent advances in Pd-based membranes for membrane reactors. Membranes 2017, 22, 51. [Google Scholar]
- Iulianelli, A.; Liguori, S.; Vita, A.; Italiano, C.; Fabiano, C.; Huang, Y.; Basile, A. The oncoming energy vector: Hydrogen produced in Pd-composite membrane reactor via bioethanol reforming over Ni/CeO2 catalyst. Catal. Today 2016, 259, 368–375. [Google Scholar] [CrossRef]
- Xu, J.; Froment, G.F. Methane steam reforming, methanation and water-gas shift: I. Intrinsic kinetics. Amer. Inst. Chem. Eng. J. 1989, 35, 88–103. [Google Scholar] [CrossRef]
- Smith, J.M. Chemical Engineering Kinetics; McGraw-Hill: New York, NY, USA, 1980. [Google Scholar]
- Acampora, L.; Marra, F.S. Investigation by thermodynamic properties of methane combustion mechanisms under harmonic oscillations in perfectly stirred reactor. Chem. Eng. Trans. 2017, 57, 1459–1464. [Google Scholar]
- Shampine, L.F.; Reichelt, M.W. The Matlab ODE suite. SIAM. J. Sci. Comput. 1977, 18, 1–12. [Google Scholar]
- Toyota Mirai Hydrogen Fuel Cell Electric Vehicle. Available online: https://h2.live/en/wasserstoffautos/toyota-mirai (accessed on 9 April 2020).
- Jokar, S.M.; Parvasi, P.; Basile, A. The evaluation of methane mixed reforming reaction in an industrial membrane reformer for hydrogen production. Int. J. Hydrog. Energ. 2018, 43, 15321–15329. [Google Scholar] [CrossRef]
- Greenhouse Gas Emissions from a Typical Passenger Vehicle, U.S. Environmental Protection Agency (EPA) Report. 2018. Available online: https://nepis.epa.gov/Exe/ZyPDF.cgi?Dockey=P100U8YT.pdf (accessed on 9 April 2020).
- Basile, A.; Campanari, S.; Manzolini, G.; Iulianelli, A.; Longo, T.; Liguori, S.; De, F.M.; Piemonte, V. Methane steam reforming in a Pd-Ag membrane reformer: An experimental study on reaction pressure influence at middle temperature. Int. J. Hydrog. Energ. 2011, 36, 1531–1539. [Google Scholar] [CrossRef]
- Brown, L.F. A comparative study of fuels for on-board hydrogen production for fuel-cell-powered automobiles. Int. J. Hydrog. Energ. 2001, 26, 381–397. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).