CO2 Desorption Performance from Imidazolium Ionic Liquids by Membrane Vacuum Regeneration Technology
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Method
3. Model Development of MVR System in Absorption–Desorption Process
3.1. Transport Equations
3.2. Physical Properties and Some Concerns of Model Parameters
3.3. Numerical Analysis
4. Results and Discussion
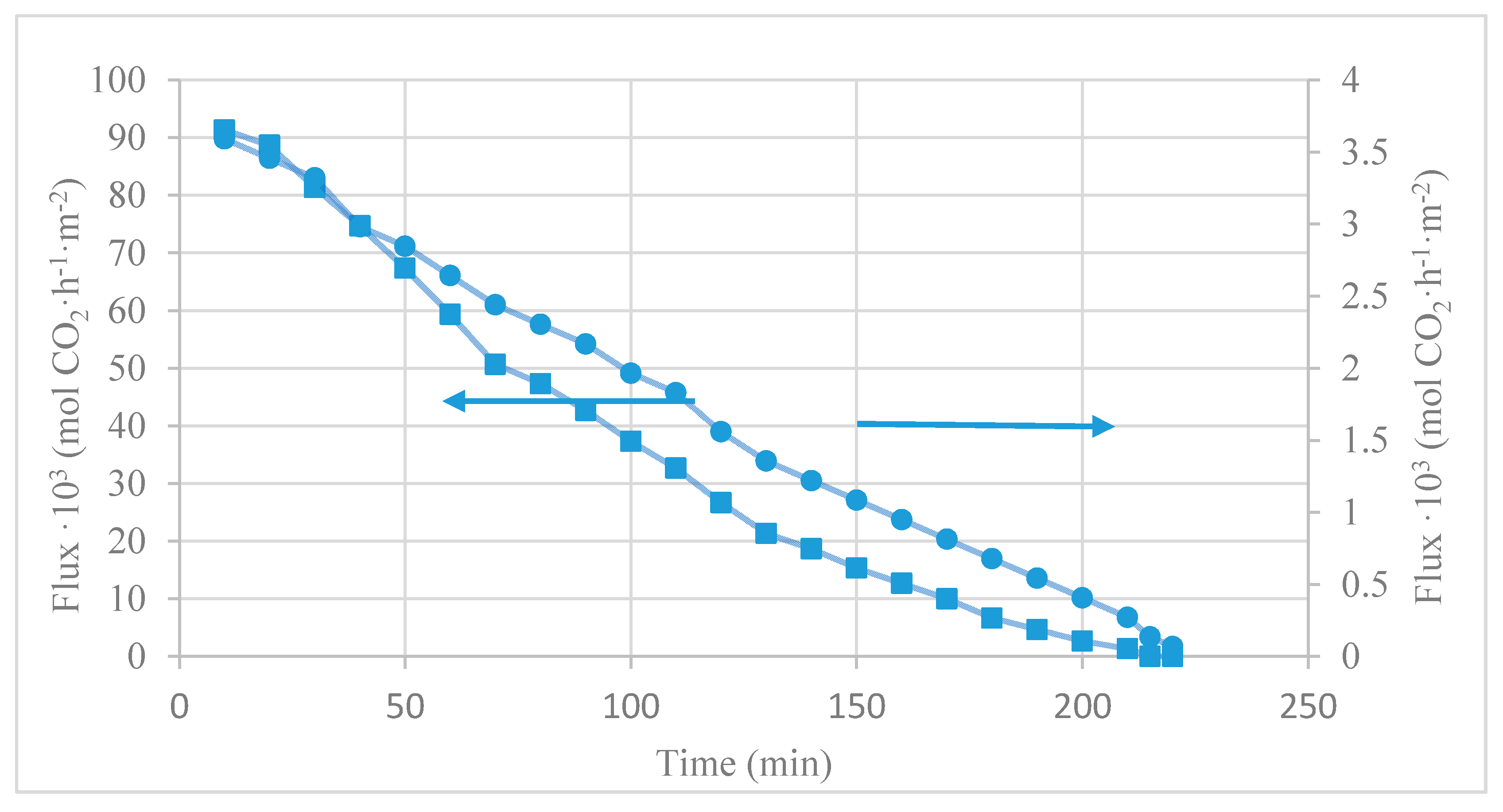
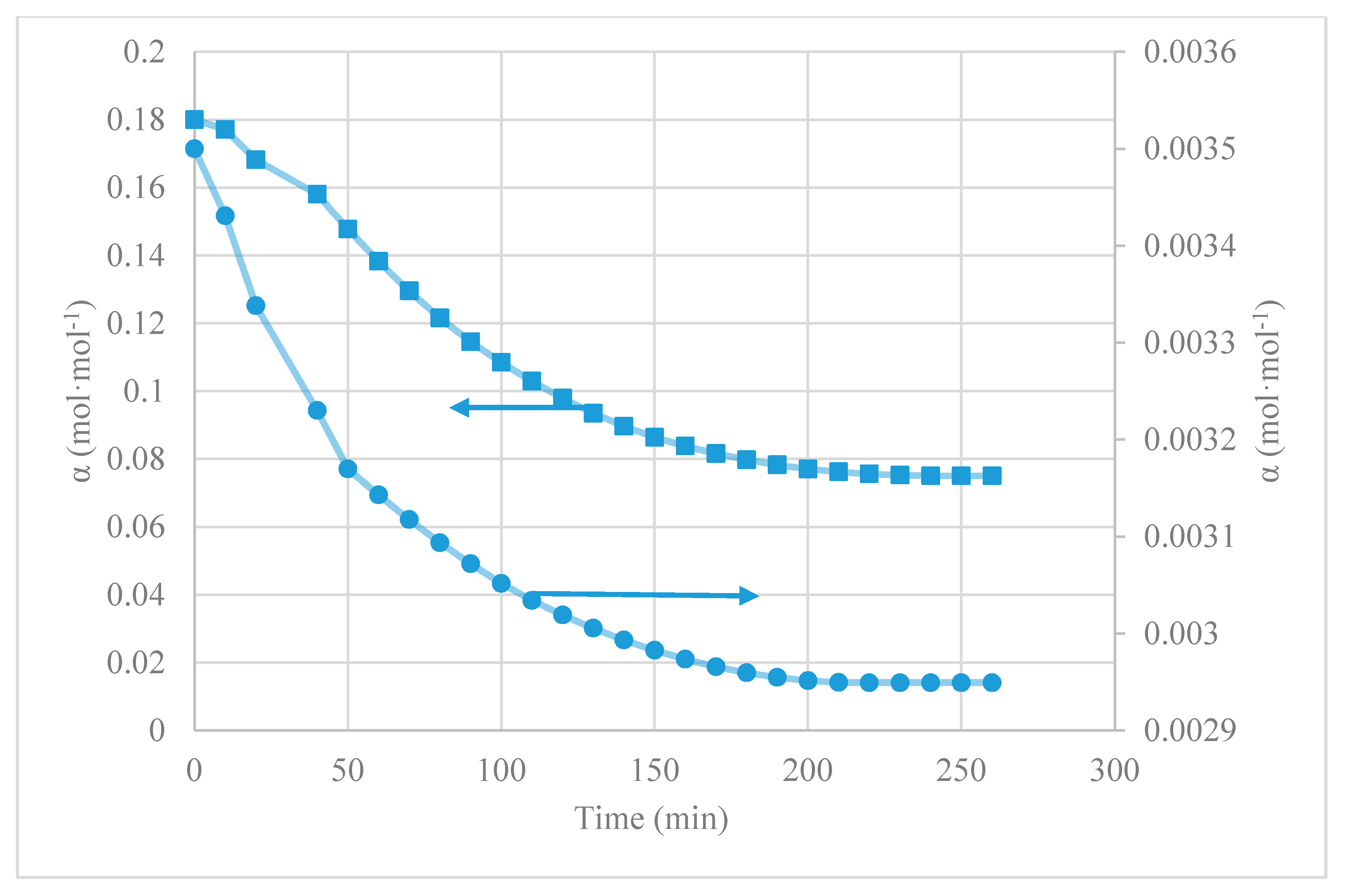
4.1. CO2 Desorption Test
4.2. Absorption–Desorption Process
4.3. Model Validation
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| molar concentration, mol CO2·mL−1 | |
| diffusion coefficient, m2·s−1 | |
| diameter of the contactor, m | |
| hydraulic diameter of the shell side, m | |
| inside diameter of the fiber, m | |
| outside diameter of the fiber, m | |
| enhancement factor | |
| activation energy, KJ·mol−1 | |
| Henry’s constant, MPa | |
| enthalpy, KJ·mol−1 | |
| first order reaction rate constant, s−1 | |
| equilibrium constant | |
| mass transfer coefficient, m·s−1 | |
| distribution coefficient between the liquid and gas | |
| molecular weight, g·mol−1 | |
| desorption molar flux, mol·m−2·h−1 | |
| pressure, bar | |
| vacuum pressure applied in MVR process, bar | |
| volumetric flow rate, mL min−1 | |
| MVR efficiency, % | |
| inner radius of membrane fiber, m | |
| radial coordinate | |
| CO2 reaction rate, mol·m−3·s−1 | |
| temperature, K | |
| volume, mL | |
| velocity, m·s−1 | |
| liquid mean velocity, m·s−1 | |
| molar volume of CO2, cm3·mol−1 | |
| axial coordinate | |
| Reynolds number | |
| Schmidt number | |
| Sherwood number | |
| Subscripts | |
| carbon dioxide | |
| cyclic | |
| external | |
| initial | |
| gas | |
| ionic liquid | |
| in | inlet |
| liquid | |
| membrane | |
| net | |
| outlet | |
| vacuum | |
| Greek Letters | |
| CO2 loading, mol_CO2·mol_IL−1 | |
| viscosity, cP | |
| density, Kg·m−3 | |
| molar volume of CO2, cm3·mol−1 | |
Appendix A
Appendix A.1. Liquid Diffusivity
Appendix A.2. Gas and Membrane Diffusivities
Appendix A.3. Henry Constant
References
- Cuéllar-Franca, R.M.; Azapagic, A. Carbon Capture, Storage and Utilisation Technologies: A Critical Analysis and Comparison of Their Life Cycle Environmental Impacts. J. CO2 Util. 2015, 9, 82–102. [Google Scholar] [CrossRef]
- He, X.; Hägg, M.B. Membranes for Environmentally Friendly Energy Processes. Membranes 2012, 2, 706–726. [Google Scholar] [CrossRef] [PubMed]
- Rúa, J.; Bui, M.; Nord, L.O.; Mac Dowell, N. Does CCS Reduce Power Generation Flexibility? A Dynamic Study of Combined Cycles with Post-Combustion CO2 Capture. Int. J. Greenh. Gas Control. 2020, 95, 102984. [Google Scholar] [CrossRef]
- Raksajati, A.; Ho, M.T.; Wiley, D.E. Comparison of Solvent Development Options for Capture of CO2 from Flue Gases. Ind. Eng. Chem. Res. 2018, 57, 6746–6758. [Google Scholar] [CrossRef]
- Nieminen, H.; Järvinen, L.; Ruuskanen, V.; Laari, A.; Koiranen, T.; Ahola, J. Insights into a Membrane Contactor Based Demonstration Unit for CO2 Capture. Sep. Purif. Technol. 2020, 231, 115951. [Google Scholar] [CrossRef]
- Xu, Y.; Goh, K.; Wang, R.; Bae, T. A Review on Polymer-Based Membranes for Gas-Liquid Membrane Contacting Processes: Current Challenges and Future Direction. Sep. Purif. Technol. 2019, 229, 115791. [Google Scholar] [CrossRef]
- Zhao, S.; Feron, P.H.M.; Deng, L.; Favre, E.; Chabanon, E.; Yan, S.; Hou, J.; Chen, V.; Qi, H. Status and Progress of Membrane Contactors in Post-Combustion Carbon Capture: A State-of-the-Art Review of New Developments. J. Membr. Sci. 2016, 511, 180–206. [Google Scholar] [CrossRef]
- Luis, P.; Van Gerven, T.; Van Der Bruggen, B. Recent Developments in Membrane-Based Technologies for CO2 Capture. Prog. Energy Combust. Sci. 2012, 38, 419–448. [Google Scholar] [CrossRef]
- Luis, P. Fundamental Modelling on Membrane Systems; Elsevier Inc.: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Chuah, C.Y.; Kim, K.; Lee, J.; Koh, D.Y.; Bae, T.H. CO2 Absorption Using Membrane Contactors: Recent Progress and Future Perspective. Ind. Eng. Chem. Res. 2020, 59, 6773–6794. [Google Scholar] [CrossRef]
- Hamzehie, M.E.; Najibi, H. Experimental and Theoretical Study of Carbon Dioxide Solubility in Aqueous Solution of Potassium Glycinate Blended with Piperazine as New Absorbents. J. CO2 Util. 2016, 16, 64–77. [Google Scholar] [CrossRef]
- Zeng, S.; Zhang, X.; Bai, L.; Zhang, X.; Wang, H.; Wang, J.; Bao, D.; Li, M.; Liu, X.; Zhang, S. Ionic-Liquid-Based CO2 Capture Systems: Structure, Interaction and Process. Chem. Rev. 2017, 117, 9625–9673. [Google Scholar] [CrossRef] [PubMed]
- Ferro, V.R.; Moya, C.; Moreno, D.; Santiago, R.; De Riva, J.; Pedrosa, G.; Larriba, M.; Diaz, I.; Palomar, J. Enterprise Ionic Liquids Database (ILUAM) for Use in Aspen ONE Programs Suite with COSMO-Based Property Methods. Ind. Eng. Chem. Res. 2018, 57, 980–989. [Google Scholar] [CrossRef]
- Venkatraman, V.; Alsberg, B.K. Predicting CO2 Capture of Ionic Liquids Using Machine Learning. J. CO2 Util. 2017, 21, 162–168. [Google Scholar] [CrossRef]
- Mei, K.; He, X.; Chen, K.; Zhou, X.; Li, H.; Wang, C. Highly Efficient CO2 Capture by Imidazolium Ionic Liquids through a Reduction in the Formation of the Carbene-CO2 Complex. Ind. Eng. Chem. Res. 2017, 56, 8066–8072. [Google Scholar] [CrossRef]
- Simons, T.J.; Hield, P.; Pas, S.J. A Novel Experimental Apparatus for the Study of Low Temperature Regeneration CO2 Capture Solvents Using Hollow Fibre Membrane Contactors. Int. J. Greenh. Gas Control 2018, 78, 228–235. [Google Scholar] [CrossRef]
- Bai, L.; Zeng, S.; Han, J.; Yang, B.; Deng, L.; Gao, H.; Zhang, X.; Zhang, X.; Zhang, S. Ionic Liquid–Based Membranes for CO2 Separation; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 235–260. [Google Scholar]
- Dai, Z.; Noble, R.D.; Gin, D.L.; Zhang, X.; Deng, L. Combination of Ionic Liquids with Membrane Technology: A New Approach for CO2 Separation. J. Membr. Sci. 2016, 497, 1–20. [Google Scholar] [CrossRef]
- Gómez-Coma, L.; Garea, A.; Irabien, A. Non-Dispersive Absorption of CO2 in [Emim][EtSO4] and [Emim][Ac]: Temperature Influence. Sep. Purif. Technol. 2014, 132, 120–125. [Google Scholar] [CrossRef]
- Albo, J.; Irabien, A. Non-Dispersive Absorption of CO2 in Parallel and Cross-Flow Membrane Modules Using EMISE. J. Chem. Technol. Biotechnol. 2012, 87, 1502–1507. [Google Scholar] [CrossRef]
- Sohaib, Q.; Vadillo, J.M.; Gómez-Coma, L.; Albo, J.; Druon-Bocquet, S.; Irabien, A.; Sanchez-Marcano, J. Post-Combustion CO2 Capture by Coupling [Emim] Cation Based Ionic Liquids with a Membrane Contactor; Pseudo-Steady-State Approach. Int. J. Greenh. Gas Control 2020, 99, 103076. [Google Scholar] [CrossRef]
- Ghasem, N. Modeling and Simulation of the Simultaneous Absorption/Stripping of CO2 with Potassium Glycinate Solution in Membrane Contactor. Membranes 2020, 10, 72. [Google Scholar] [CrossRef]
- Zaidiza, D.A.; Belaissaoui, B.; Rode, S.; Favre, E. Intensification Potential of Hollow Fiber Membrane Contactors for CO2 Chemical Absorption and Stripping Using Monoethanolamine Solutions. Sep. Purif. Technol. 2017, 188, 38–51. [Google Scholar] [CrossRef]
- Bazhenov, S.D.; Lyubimova, E.S. Gas—Liquid Membrane Contactors for Carbon Dioxide Capture from Gaseous Streams. Pet. Chem. 2016, 56, 889–914. [Google Scholar] [CrossRef]
- Klingberg, P.; Wilkner, K.; Schlüter, M.; Grünauer, J.; Shishatskiy, S. Separation of Carbon Dioxide from Real Power Plant Flue Gases by Gas Permeation Using a Supported Ionic Liquid Membrane: An Investigation of Membrane Stability. Membranes 2019, 9, 35. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Fang, M.; Pan, Y.; Yan, S.; Luo, Z. Comparison and Selection of Amine-Based Absorbents in Membrane Vacuum Regeneration Process for CO2 Capture with Low Energy Cost. Energy Procedia 2013, 37, 1085–1092. [Google Scholar] [CrossRef][Green Version]
- Fang, M.; Wang, Z.; Yan, S.; Cen, Q.; Luo, Z. CO2 Desorption from Rich Alkanolamine Solution by Using Membrane Vacuum Regeneration Technology. Int. J. Greenh. Gas Control 2012, 9, 507–521. [Google Scholar] [CrossRef]
- Nii, S.; Iwata, Y.; Takahashi, K.; Takeuchi, H. Regeneration of CO2-Loaded Carbonate Solution by Reducing Pressure. J. Chem. Eng. Jpn. 1995, 28, 148–153. [Google Scholar] [CrossRef]
- Li, J.L.; Chen, B.H. Review of CO2 Absorption Using Chemical Solvents in Hollow Fiber Membrane Contactors. Sep. Purif. Technol. 2005, 41, 109–122. [Google Scholar] [CrossRef]
- Shukla, S.K.; Khokarale, S.G.; Bui, T.Q.; Mikkola, J.P.T. Ionic Liquids: Potential Materials for Carbon Dioxide Capture and Utilization. Front. Mater. 2019, 6, 42. [Google Scholar] [CrossRef]
- Rea, R.; De Angelis, M.G.; Baschetti, M.G. Models for Facilitated Transport Membranes: A Review. Membranes 2019, 9, 26. [Google Scholar] [CrossRef]
- Pinto, A.M.; Rodríguez, H.; Arce, A.; Soto, A. Combined Physical and Chemical Absorption of Carbon Dioxide in a Mixture of Ionic Liquids. J. Chem. Thermodyn. 2014, 77, 197–205. [Google Scholar] [CrossRef]
- Elhajj, J.; Al-Hindi, M.; Azizi, F. A Review of the Absorption and Desorption Processes of Carbon Dioxide in Water Systems. Ind. Eng. Chem. Res. 2014, 53, 2–22. [Google Scholar] [CrossRef]
- Gabelman, A.; Hwang, S.T. Hollow Fiber Membrane Contactors. J. Membr. Sci. 1999, 159, 61–106. [Google Scholar] [CrossRef]
- Zareiekordshouli, F.; Lashanizadehgan, A.; Darvishi, P. Study on the Use of an Imidazolium-Based Acetate Ionic Liquid for CO2 Capture from Fl Ue Gas in Absorber/Stripper Packed Columns: Experimental and Modeling. Int. J. Greenh. Gas Control 2018, 70, 178–192. [Google Scholar] [CrossRef]
- Ghasem, N. Chemical Absorption of CO2 Enhanced by Nanoparticles Using a Membrane Contactor: Modeling and Simulation. Membranes 2019, 9, 150. [Google Scholar] [CrossRef]
- Sander, R. Modeling atmospheric chemistry: Interactions between gas-phase species and liquid cloud/aerosol particles. Surv. Geophys. 1999, 20, 1–31. [Google Scholar] [CrossRef]
- Gomez-Coma, L.; Garea, A.; Irabien, A. Carbon Dioxide Capture by [Emim][Ac] Ionic Liquid in a Polysulfone Hollow Fiber Membrane Contactor. Int. J. Greenh. Gas Control 2016, 52, 401–409. [Google Scholar] [CrossRef]
- Yokozeki, A.; Shiflett, M.B.; Junk, C.P.; Grieco, L.M.; Foo, T. Physical and Chemical Absorptions of Carbon Dioxide in Room-Temperature Ionic Liquids. J. Phys. Chem. B 2008, 112, 16654–16663. [Google Scholar] [CrossRef]
- Qazi, S.; Gómez-coma, L.; Albo, J.; Druon-bocquet, S.; Irabien, A. Mathematical Modelling of CO2 Absorption with Ionic Liquids in a Membrane Contactor, Study of Absorption Kinetics and Influence of Temperature. J. Chem Technol. Biotechnol. 2019, 95, 1844–1857. [Google Scholar] [CrossRef]
- Baghban, A.; Mohammadi, A.H.; Taleghani, M.S. Rigorous Modeling of CO2 Equilibrium Absorption in Ionic Liquids. Int. J. Greenh. Gas Control 2017, 58, 19–41. [Google Scholar] [CrossRef]
- Shiflett, M.B.; Yokozeki, A. Phase Behavior of Carbon Dioxide in Ionic Liquids: [Emim][Acetate], [Emim][Trifluoroacetate], and [Emim][Acetate] + [Emim][Trifluoroacetate] Mixtures. J. Chem. Eng. Data 2009, 54, 108–114. [Google Scholar] [CrossRef]
- Ramdin, M.; De Loos, T.W.; Vlugt, T.J.H. State-of-the-Art of CO2 Capture with Ionic Liquids. Ind. Eng. Chem. Res. 2012, 51, 8149–8177. [Google Scholar] [CrossRef]
- Hospital-Benito, D.; Lemus, J.; Moya, C.; Santiago, R.; Palomar, J. Process Analysis Overview of Ionic Liquids on CO2 Chemical Capture. Chem. Eng. J. 2020, 390, 124509. [Google Scholar] [CrossRef]
- Hospital-Benito, D.; Lemus, J.; Santiago, R.; Palomar, J. Thermodynamic and Kinetic Evaluation of Ionic Liquids + Tetraglyme Mixtures on CO2 Capture. J. CO2 Util. 2020, 35, 185–193. [Google Scholar] [CrossRef]
- Qazi, S.; Gómez-Coma, L.; Albo, J.; Druon-Bocquet, S.; Irabien, A.; Sanchez-Marcano, J. CO2 Capture in a Hollow Fiber Membrane Contactor Coupled with Ionic Liquid: Influence of Membrane Wetting and Process Parameters. Sep. Purif. Technol. 2020, 233, 115986. [Google Scholar] [CrossRef]
- Tijl, P.; Steinbuch, M.M.; Backx, A.; Perkins, G. Assessment of the Parameter Estimation Capabilities of GPROMS and Aspen Custom Modeler, Using the Sec-Butyl-Alcohol Stripper Kinetics Case Study. In Graduation Report; Eindhoven Technical University: Eindhoven, The Netherlands, 2005. [Google Scholar]
- Ma, T.; Wang, J.; Du, Z.; Abdeltawab, A.A.; Al-Enizi, A.M.; Chen, X.; Yu, G. A Process Simulation Study of CO2 Capture by Ionic Liquids. Int. J. Greenh. Gas Control 2017, 58, 223–231. [Google Scholar] [CrossRef]
- Lu, J.G.; Lu, C.T.; Chen, Y.; Gao, L.; Zhao, X.; Zhang, H.; Xu, Z.W. CO2 Capture by Membrane Absorption Coupling Process: Application of Ionic Liquids. Appl. Energy 2014, 115, 573–581. [Google Scholar] [CrossRef]
- Lu, J.G.; Hua, A.C.; Xu, Z.W.; Li, J.T.; Liu, S.Y.; Wang, Z.L.; Zhao, Y.L.; Pan, C. CO2 Capture by Membrane Absorption Coupling Process: Experiments and Coupling Process Evaluation. J. Membr. Sci. 2013, 431, 9–18. [Google Scholar] [CrossRef]
- Yan, S.; Fang, M.; Wang, Z.; Luo, Z. Regeneration Performance of CO2-Rich Solvents by Using Membrane Vacuum Regeneration Technology: Relationships between Absorbent Structure and Regeneration Efficiency. Appl. Energy 2012, 98, 357–367. [Google Scholar] [CrossRef]
- Koonaphapdeelert, S.; Wu, Z.; Li, K. Carbon Dioxide Stripping in Ceramic Hollow Fibre Membrane Contactors. Chem. Eng. Sci. 2009, 64, 1–8. [Google Scholar] [CrossRef]
- Gebremariam, S.K. Modelling a Membrane Contactor for CO2 Capture. In Graduation Report; Norwegian University of Science and Technology: Trondheim, Norway, 2017. [Google Scholar]
- Mulukutla, T.; Obuskovic, G.; Sirkar, K.K. Novel Scrubbing System for Post-Combustion CO2 Capture and Recovery: Experimental Studies. J. Membr. Sci. 2014, 471, 16–26. [Google Scholar] [CrossRef]
- Wang, Z.; Fang, M.; Ma, Q.; Zhao, Z.; Wang, T.; Luo, Z. Membrane Stripping Technology for CO2 Desorption from CO2-Rich Absorbents with Low Energy Consumption. Energy Procedia 2014, 63, 765–772. [Google Scholar] [CrossRef]
- Morgan, D.; Ferguson, L.; Scovazzo, P. Diffusivities of Gases in Room-Temperature Ionic Liquids: Data and Correlations Obtained Using a Lag-Time Technique. Ind. Eng. Chem. Res. 2005, 44, 4815–4823. [Google Scholar] [CrossRef]
- Ziobrowski, Z.; Krupiczka, R.; Rotkegel, A. Carbon Dioxide Absorption in a Packed Column Using Imidazolium Based Ionic Liquids and MEA Solution. Int. J. Greenh. Gas Control 2016, 47, 8–16. [Google Scholar] [CrossRef]
- Gilliland, E.R. Diffusion Coefficients in Gaseous Systems. Ind. Eng. Chem. 1934, 26, 681–685. [Google Scholar] [CrossRef]
- Moya, C.; Gonzalez-Miquel, M.; Rodriguez, F.; Soto, A.; Rodriguez, H.; Palomar, J. Non-Ideal Behavior of Ionic Liquid Mixtures to Enhance CO2 Capture. Fluid Phase Equilibria 2017, 450, 175–183. [Google Scholar] [CrossRef]












| Membrane Material | Polypropylene |
|---|---|
| Module i.d., dcont (m) | 25 × 10−3 |
| Fiber outside diameter, do (m) | 3 × 10−4 |
| Fiber inside diameter, di (m) | 2.2 × 10−4 |
| Fiber length, L (m) | 0.115 |
| Number of fibers, n | 2300 |
| Effective inner membrane area, A (m2) | 0.18 |
| Membrane thickness, δ (m) | 4 × 10−5 |
| Membrane pore diameter, dp (m) | 4 × 10−6 |
| Porosity, (%) | 40 |
| Packing factor, φ | 0.39 |
| Tortuosity, τ | 2.50 |
| Parameter/Property | Value | Unit |
|---|---|---|
| Ionic Liquid | [emim][Ac] | - |
| Volume, V | 100 | mL |
| Temperature, T | 289–310 | K |
| Feed Gas flow rate, Qg | 60 | mL·min−1 |
| Liquid flow rate, Ql | 60 | mL·min−1 |
| Feed gas pressure, Pg,in | 1.03 | bar |
| Liquid pressure, Pl,in | 1.31 | bar |
| Vacuum pressure, Pv | 0.04–0.5 | bar |
| Parameter | Unit | Value | Reference |
|---|---|---|---|
| Diffusion coefficient of CO2 in the liquid phase, | m2·s−1 | 5.58·10−10 | Appendix A |
| Diffusion coefficient of CO2 in the gas phase, | m2·s−1 | 6.62·10−6 | Appendix A |
| Diffusion coefficient of CO2 through the membrane, | m2·s−1 | 1.86·10−6 | Appendix A |
| Henry’s constant, | MPa | 8.8 | Appendix A |
| Liquid viscosity, | cP | 0.17 | [38] |
| Liquid density, | g·cm−3 | 1.1 | [19] |
| Equilibrium constant, | - | 136 | [39] |
| Enthalpy, | KJ·mol−1 | −30.18 | [40] |
| Activation energy, | KJ·mol−1 | 9.2 | [40] |
| Solvent | Reference | Operational Conditions | ||||
|---|---|---|---|---|---|---|
| [emim][Ac] | 0.180 | 0.075 | 0.105 | 58 | our work | Room T; 40 mbar |
| [emim][EtSO4] | 0.0035 | 0.003 | 0.001 | 15 | our work | Room T; 40 mbar |
| Aqueous [bmim][BF4] | 0.018 | 0.000 | 0.018 | 100 | [49] | Room T; 500 mbar |
| Aqueous [apmim][BF4] | 0.380 | 0.150 | 0.230 | 61 | [49] | Room T; 500 mbar |
| Aqueous PG + AMP | 0.900 | 0.550 | 0.350 | 39 | [50] | Room T; 600 mbar |
| Aqueous MEA | 0.69 | 0.454 | 0.236 | 34 | [51] | 70 °C; 100 mbar |
| Experimental | Model | Error. | |||
|---|---|---|---|---|---|
| SET | Pv (mbar) | T (K) | (%) | (%) | (%) |
| 1 | 500 | 289 | 2.3 | 2.5 | 7.3 |
| 2 | 200 | 289 | 60.7 | 60.7 | 0.1 |
| 3 | 40 | 289 | 91.4 | 86.6 | 5.3 |
| 4 | 500 | 310 | 10.6 | 11.1 | 4.5 |
| 5 | 200 | 310 | 77.8 | 76.4 | 1.8 |
| 6 | 40 | 310 | 93.8 | 87.5 | 6.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vadillo, J.M.; Gómez-Coma, L.; Garea, A.; Irabien, A. CO2 Desorption Performance from Imidazolium Ionic Liquids by Membrane Vacuum Regeneration Technology. Membranes 2020, 10, 234. https://doi.org/10.3390/membranes10090234
Vadillo JM, Gómez-Coma L, Garea A, Irabien A. CO2 Desorption Performance from Imidazolium Ionic Liquids by Membrane Vacuum Regeneration Technology. Membranes. 2020; 10(9):234. https://doi.org/10.3390/membranes10090234
Chicago/Turabian StyleVadillo, Jose Manuel, Lucia Gómez-Coma, Aurora Garea, and Angel Irabien. 2020. "CO2 Desorption Performance from Imidazolium Ionic Liquids by Membrane Vacuum Regeneration Technology" Membranes 10, no. 9: 234. https://doi.org/10.3390/membranes10090234
APA StyleVadillo, J. M., Gómez-Coma, L., Garea, A., & Irabien, A. (2020). CO2 Desorption Performance from Imidazolium Ionic Liquids by Membrane Vacuum Regeneration Technology. Membranes, 10(9), 234. https://doi.org/10.3390/membranes10090234









