Evaluating Fertilizer-Drawn Forward Osmosis Performance in Treating Anaerobic Palm Oil Mill Effluent
Abstract
:1. Introduction
2. Materials and Methods
2.1. Membrane
2.2. Feed and Draw Solutions
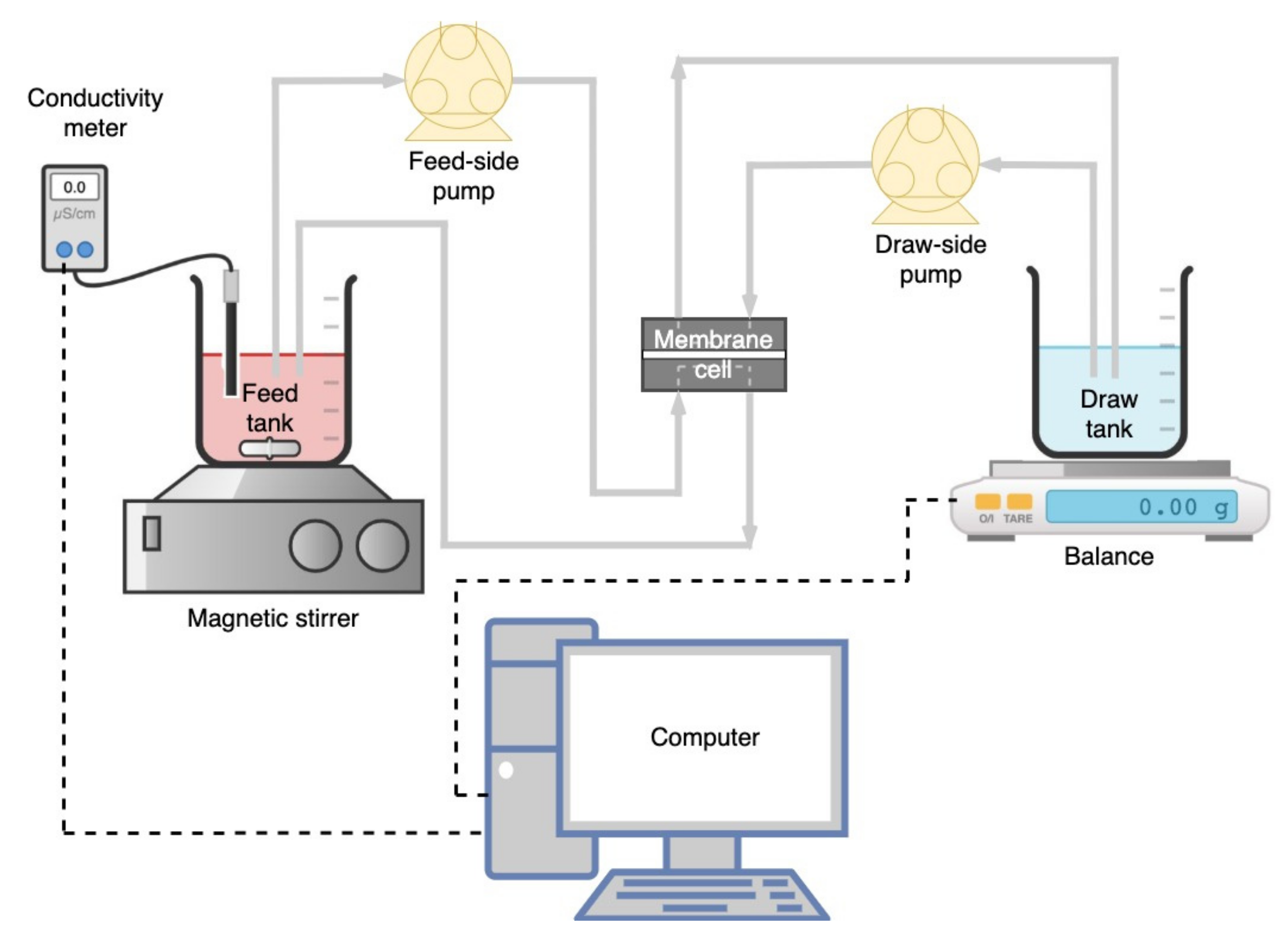
2.3. Lab-Scale FO System
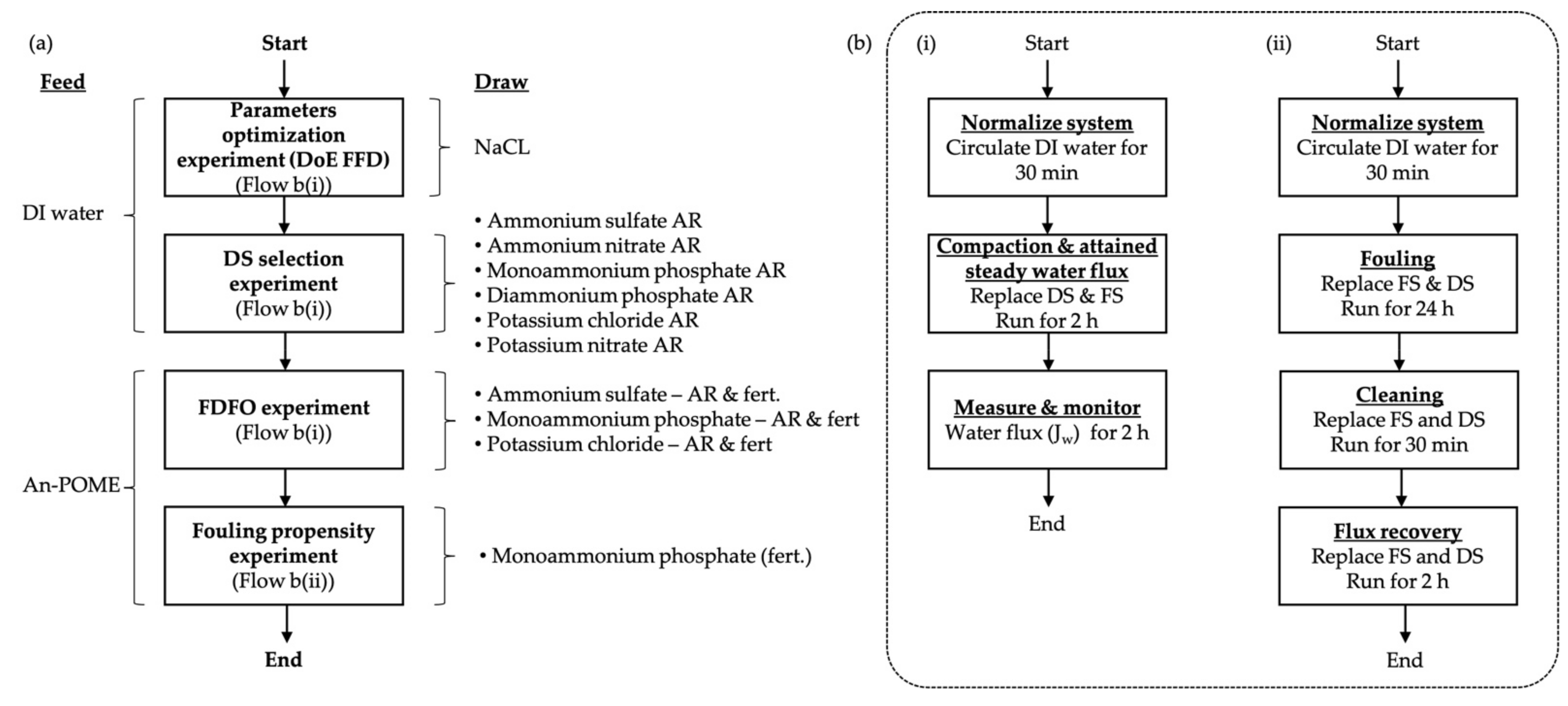
2.4. FO Performance Experiments
2.5. Measurement and Analysis
2.5.1. Analytical Methods
2.5.2. Water Flux and Reverse Salt Flux
2.5.3. Specific Reverse Salt Flux and Performance Ratio
2.5.4. Water Recovery and Water Flux Recovery
2.5.5. Factorial Design of Experiment (DoE) for Parameter Optimization
3. Results and Discussion
3.1. Effect of Process Parameters on FO Performance
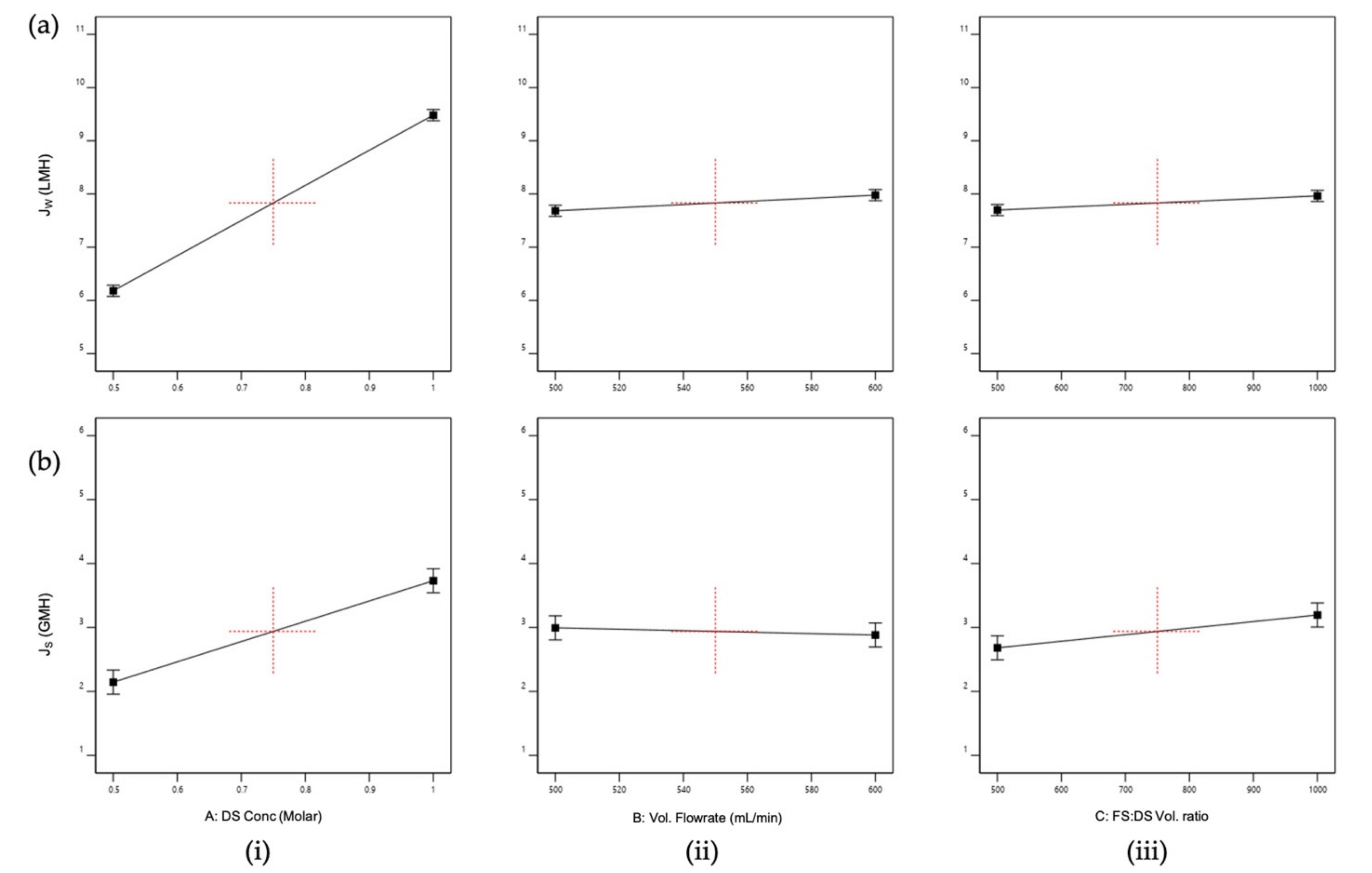
3.1.1. Analysis of Variance (ANOVA) and Normal Distribution
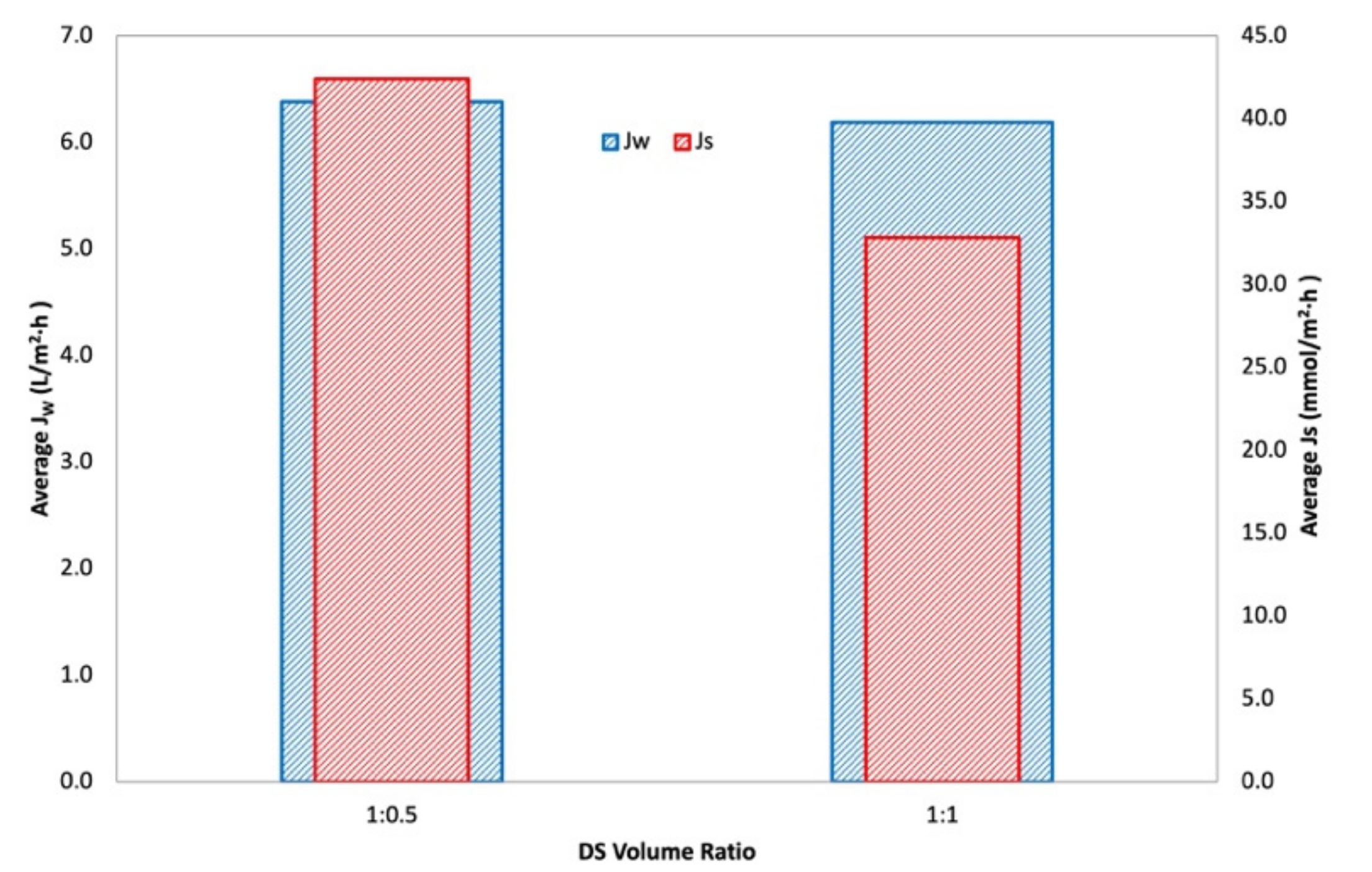
3.1.2. Main and Interactions Effects
3.1.3. Optimization
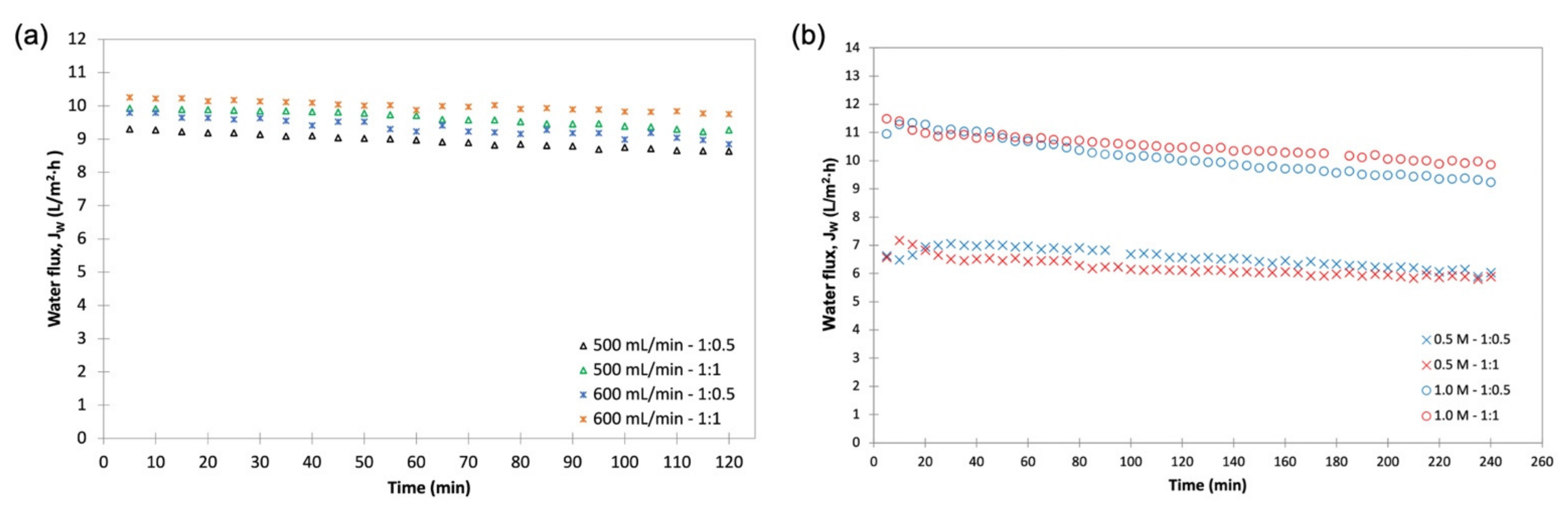
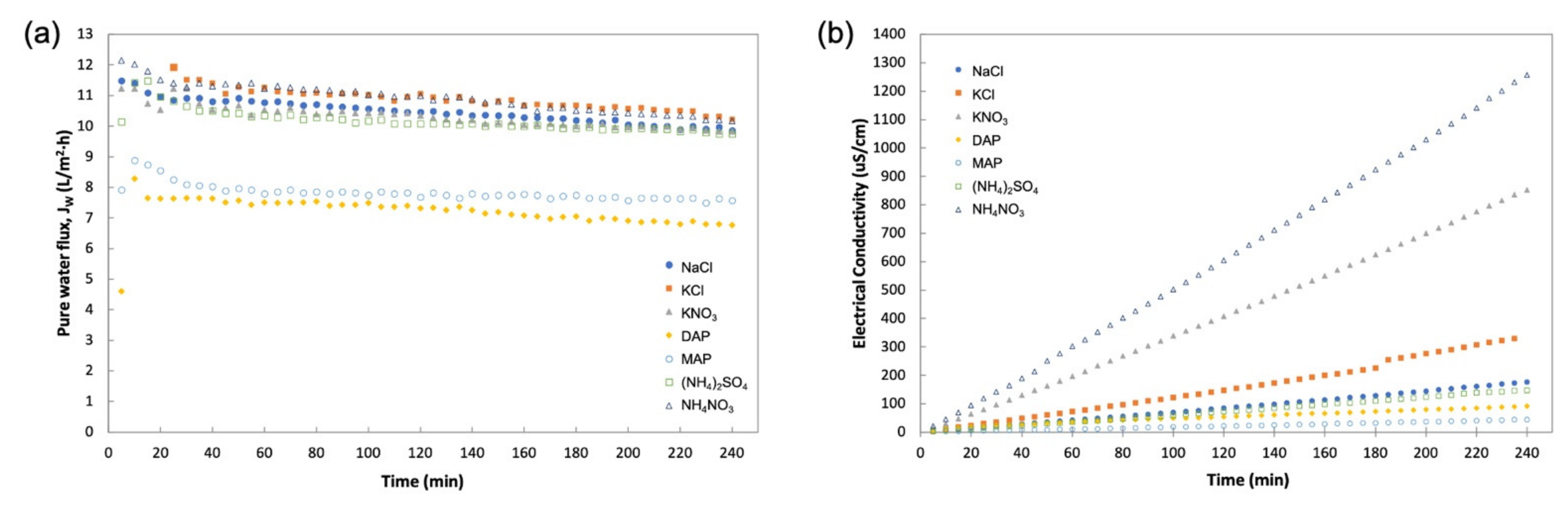
3.2. FO Performance of Draw Solutions—Initial Screening
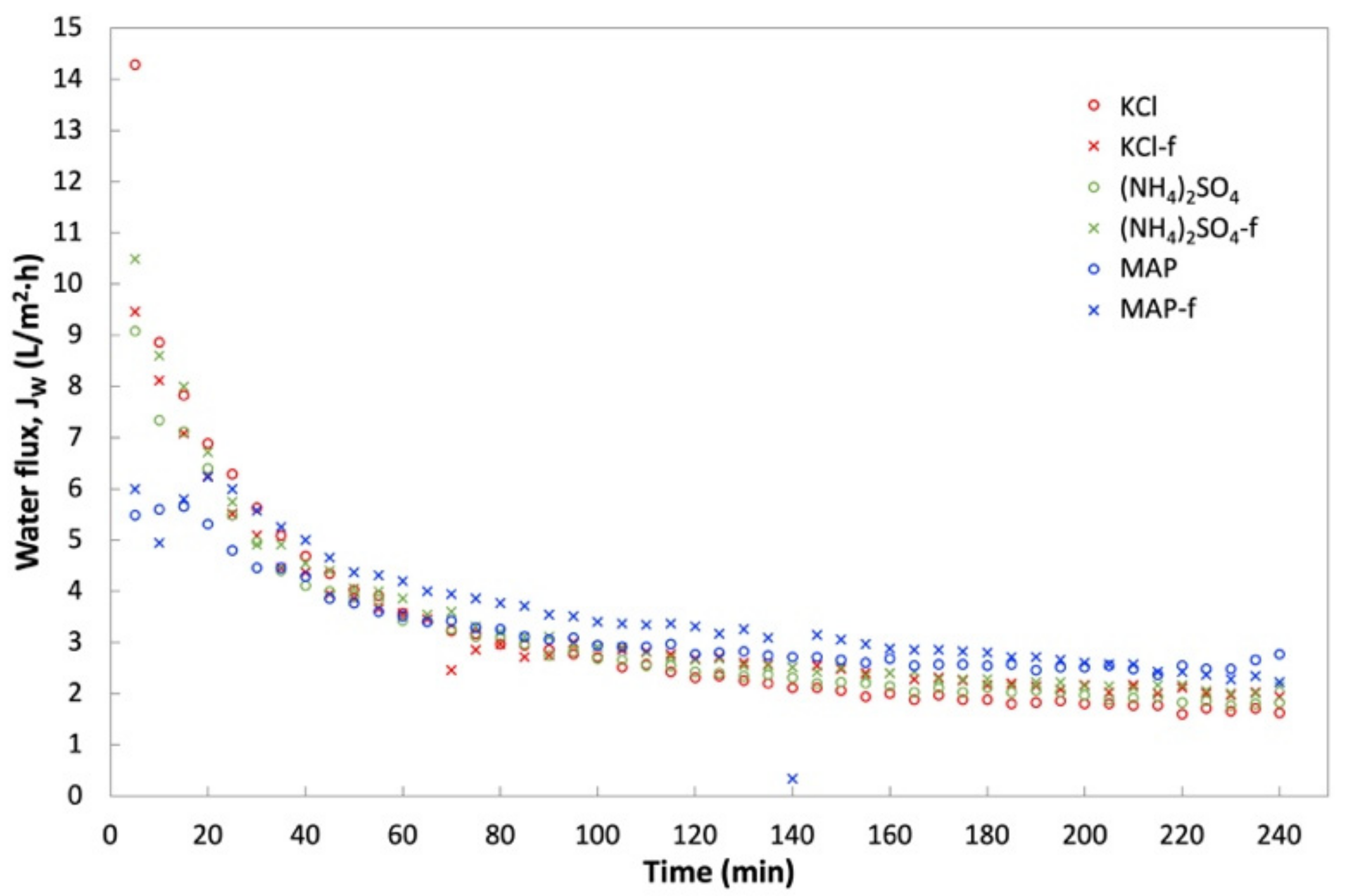
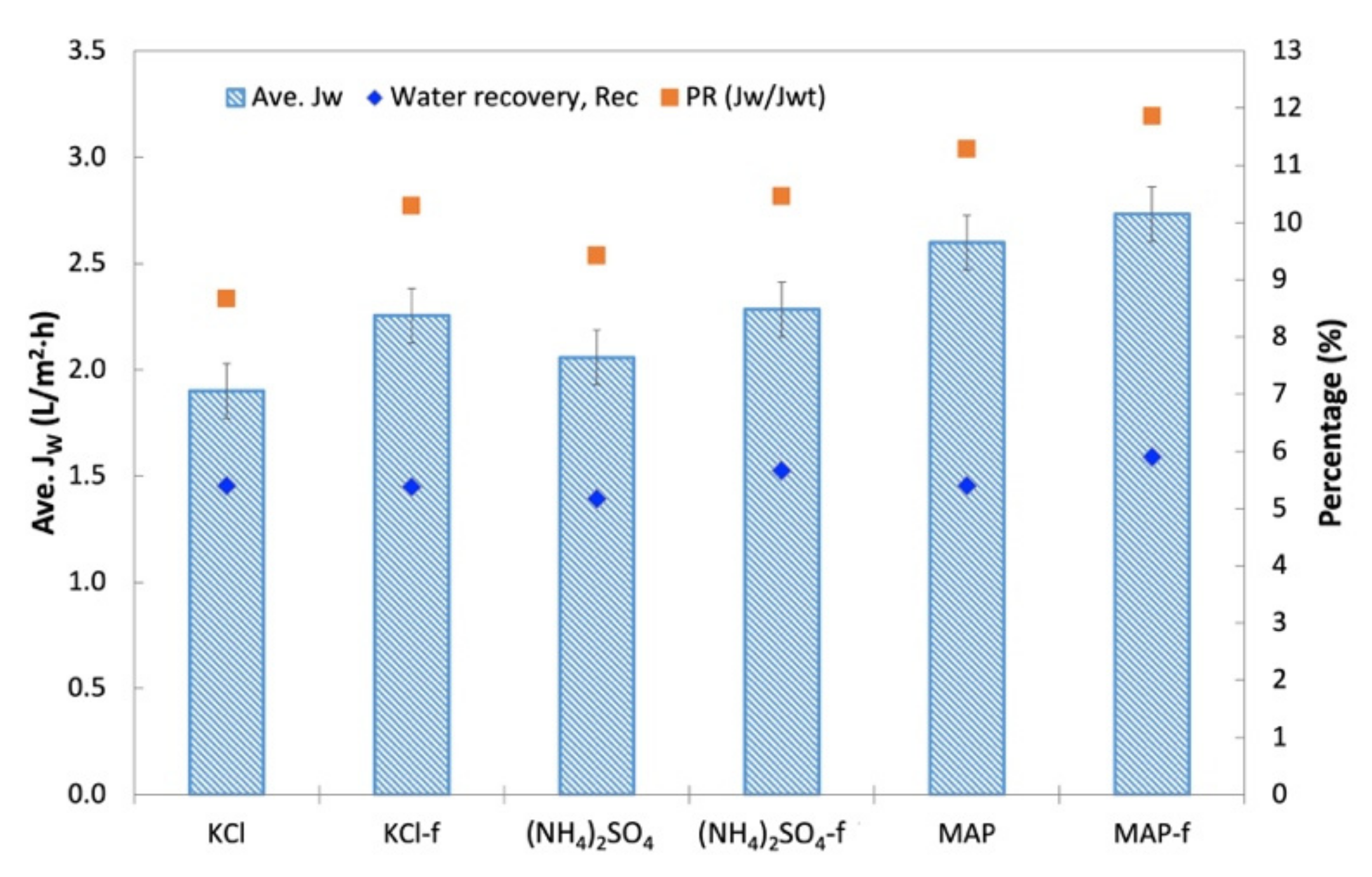
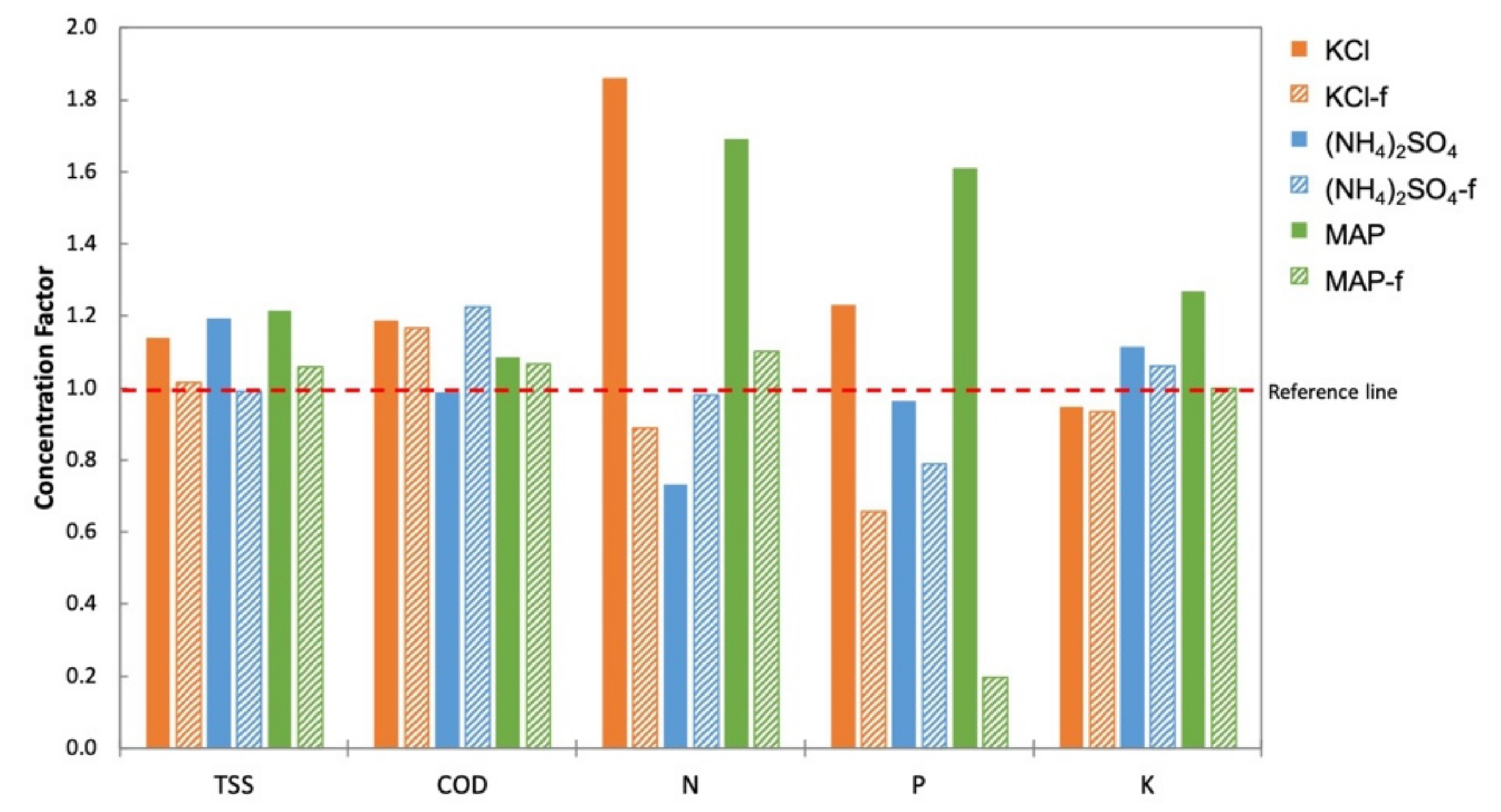
3.3. FO Performance of Fertilizers as DS for the Treatment of Anaerobic POME
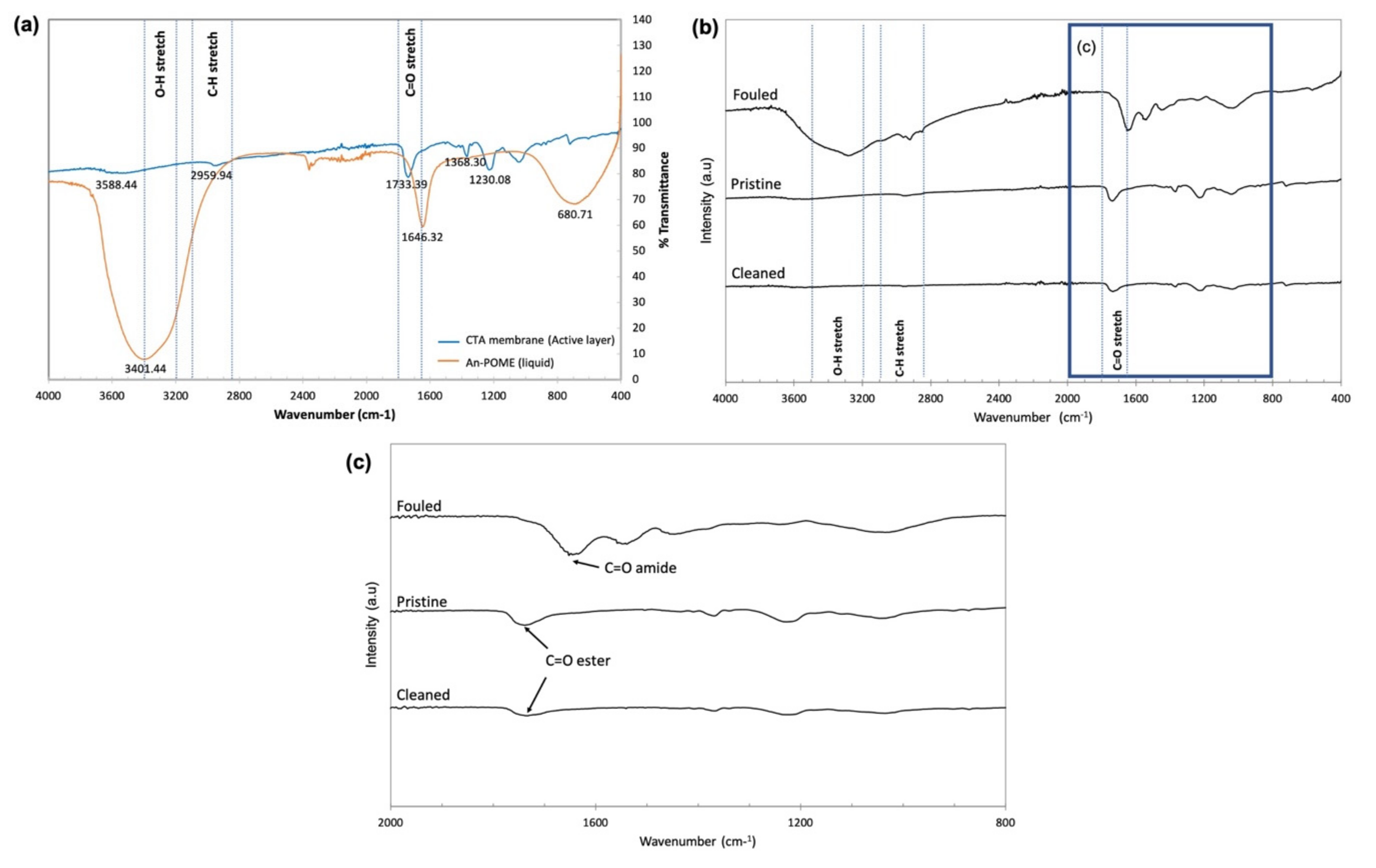
3.4. Fouling Behavior and Water Flux Recovery
4. Conclusions
- With DI water feed, KCl and NH4NO3 showed highest average JW; however, (NH4)2SO4, MAP, and DAP demonstrated the lowest JS and SRSF. The optimum DSs with sufficiently good average JW, low JS, and low SRSF were (NH4)2SO4, MAP, and DAP.
- With An-POME feed, (NH4)2SO4, MAP, and KCl were chosen as the N-source, P-source, and K-source fertilizer, respectively, for DS. Although it started out with a lower initial JW than KCl, both reagent and technical grades of MAP showed high average JW and low flux decline due to the ability to sustain a relatively high water flux throughout the process. MAP also had higher PR compared to other DSs that indicated a sufficient osmotic gradient for driving water removal from An-POME.
- Generally, no significant changes in TSS and COD in the An-POME feed were observed, as the FDFO process only involves water migration. Significant variation was observed in nutrient (NPK) concentrations in the feed that are associated with the type of fertilizer DS (salinity build up due to RSF from the draw solution) and the characteristics of the initial feed.
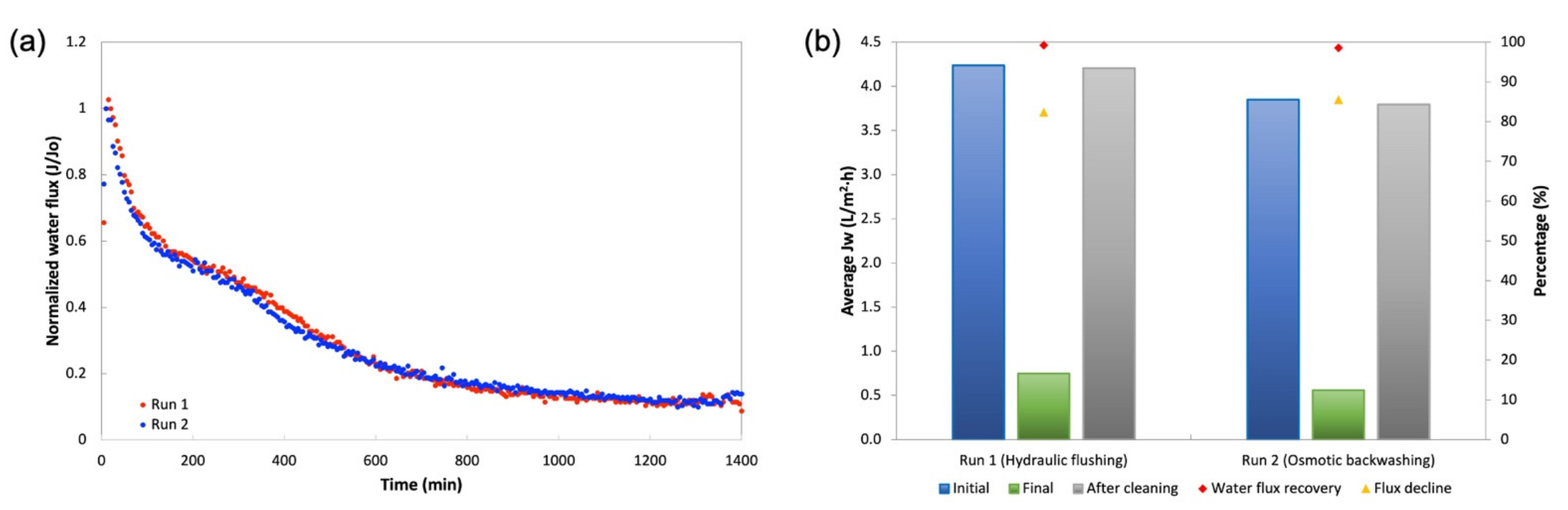
- The long-term fouling using An-POME feed and MAP as DSs yielded a flux decline of around 84% and an average 15% water recovery for a 24-h operation. Sufficient cleaning using hydraulic flushing and osmotic backwashing was able to effectively restore the water flux, although complete flux recovery was not achieved, with 99.2% and 98.6% water flux recovery, respectively.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Agriculture, Forestry, and Fishing, Value Added (% of GDP)|Data. Available online: https://data.worldbank.org/indicator/NV.AGR.TOTL.ZS?end=2019&start=1996&view=chart (accessed on 20 April 2021).
- World Bank. Health, Nutrition and Population Data and Statistics, World Bank-Population Dashboard. Available online: http://datatopics.worldbank.org/health/population (accessed on 20 March 2019).
- Arias, A.; Behera, C.R.; Feijoo, G.; Sin, G.; Moreira, M.T. Unravelling the environmental and economic impacts of innovative technologies for the enhancement of biogas production and sludge management in wastewater systems. J. Environ. Manag. 2020, 270, 110965. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, E.; Nellemann, C.; Baker, E.; Bos, R.; Osborn Savelli, H. (Eds.) Sick Water? The Central Role of Wastewater Management in Sustainable Development; United Nations Environment Programme (UNEP): Nairobi, Kenya, 2010. [Google Scholar]
- Chin, M.J.; Poh, P.E.; Tey, B.T.; Chan, E.S.; Chin, K.L. Biogas from Palm Oil Mill Effluent (POME): Opportunities and Challenges from Malaysia’s Perspective. Renew. Sustain. Energy Rev. 2013, 26, 717–726. [Google Scholar] [CrossRef]
- Subramaniam, V.; Ma, A.N.; Choo, Y.M.; Nik Sulaiman, N.M. Life Cycle Inventory of the Production of Crude Palm Oil-A Gate to Gate Case Study of 12 Palm Oil Mills. J. Oil Palm Res. 2008, 20, 484–494. [Google Scholar]
- Nasution, M.A.; Wibawa, D.S.; Ahamed, T.; Noguchi, R. Comparative Environmental Impact Evaluation of Palm Oil Mill Effluent Treatment Using a Life Cycle Assessment Approach: A Case Study Based on Composting and a Combination for Biogas Technologies in North Sumatera of Indonesia. J. Clean. Prod. 2018, 184, 1028–1040. [Google Scholar] [CrossRef]
- Sharvini, S.R.; Noor, Z.Z.; Chong, C.S.; Lindsay, C.; Glew, D. Energy Generation from Palm Oil Mill Effluent: A Life Cycle Assessment of Two Biogas Technologies. Energy 2019, 191, 116513. [Google Scholar] [CrossRef]
- Kim, Y.; Woo, Y.C.; Phuntsho, S.; Nghiem, L.D.; Shon, H.K.; Hong, S. Evaluation of Fertilizer-Drawn Forward Osmosis for Coal Seam Gas Reverse Osmosis Brine Treatment and Sustainable Agricultural Reuse. J. Memb. Sci. 2017, 537, 22–31. [Google Scholar] [CrossRef] [Green Version]
- Schneider, C.; Rajmohan, R.S.; Zarebska, A.; Tsapekos, P.; Hélix-Nielsen, C. Treating Anaerobic Effluents Using Forward Osmosis for Combined Water Purification and Biogas Production. Sci. Total Environ. 2019, 647, 1021–1030. [Google Scholar] [CrossRef]
- Lambrechts, R.; Sheldon, M.S. Performance and Energy Consumption Evaluation of a Fertiliser Drawn Forward Osmosis (FDFO) System for Water Recovery from Brackish Water. Desalination 2019, 456, 64–73. [Google Scholar] [CrossRef]
- Go, G.-W.; Im, S.-J.; Lee, S.-H.; Jang, A. Evaluation of Forward Osmosis Membrane Performance by Using Wastewater Treatment Plant Effluents as Feed Solution. Desalin. Water Treat. 2016, 57, 26657–26669. [Google Scholar] [CrossRef]
- Xie, M.; Shon, H.K.; Gray, S.R.; Elimelech, M. Membrane-Based Processes for Wastewater Nutrient Recovery: Technology, Challenges, and Future Direction. Water Res. 2016, 89, 210–221. [Google Scholar] [CrossRef] [Green Version]
- Phuntsho, S.; Shon, H.K.; Hong, S.; Lee, S.; Vigneswaran, S. A Novel Low Energy Fertilizer Driven Forward Osmosis Desalination for Direct Fertigation: Evaluating the Performance of Fertilizer Draw Solutions. J. Memb. Sci. 2011, 375, 172–181. [Google Scholar] [CrossRef]
- Zou, S.; He, Z. Enhancing Wastewater Reuse by Forward Osmosis with Self-Diluted Commercial Fertilizers as Draw Solutes. Water Res. 2016, 99, 235–243. [Google Scholar] [CrossRef] [Green Version]
- Chekli, L.; Kim, J.E.; El Saliby, I.; Kim, Y.; Phuntsho, S.; Li, S.; Ghaffour, N.; Leiknes, T.; Kyong Shon, H. Fertilizer Drawn Forward Osmosis Process for Sustainable Water Reuse to Grow Hydroponic Lettuce Using Commercial Nutrient Solution. Sep. Purif. Technol. 2017, 181, 18–28. [Google Scholar] [CrossRef] [Green Version]
- Gulied, M.; Al Momani, F.; Khraisheh, M.; Bhosale, R.; AlNouss, A. Influence of Draw Solution Type and Properties on the Performance of Forward Osmosis Process: Energy Consumption and Sustainable Water Reuse. Chemosphere 2019, 233, 234–244. [Google Scholar] [CrossRef]
- Achilli, A.; Cath, T.Y.; Childress, A.E. Selection of Inorganic-Based Draw Solutions for Forward Osmosis Applications. J. Memb. Sci. 2010, 364, 233–241. [Google Scholar] [CrossRef]
- Jang, Y.; Cho, H.; Shin, Y.; Choi, Y.; Lee, S.; Koo, J. Comparison of Fouling Propensity and Physical Cleaning Effect in Forward Osmosis, Reverse Osmosis, and Membrane Distillation. Desalin. Water Treat. 2016, 57, 24532–24541. [Google Scholar] [CrossRef]
- Lotfi, F.; Samali, B.; Hagare, D. Cleaning Efficiency of the Fouled Forward Osmosis Membranes under Different Experimental Conditions. J. Environ. Chem. Eng. 2018, 6, 4555–4563. [Google Scholar] [CrossRef]
- Tiraferri, A.; Yip, N.Y.; Straub, A.P.; Romero-Vargas Castrillon, S.; Elimelech, M. A Method for the Simultaneous Determination of Transport and Structural Parameters of Forward Osmosis Membranes. J. Memb. Sci. 2013, 444, 523–538. [Google Scholar] [CrossRef]
- Kim, B.; Gwak, G.; Hong, S. Review on Methodology for Determining Forward Osmosis (FO) Membrane Characteristics: Water Permeability (A), Solute Permeability (B), and Structural Parameter (S). Desalination 2017, 422, 5–16. [Google Scholar] [CrossRef]
- Emadzadeh, D.; Lau, W.J.; Rahbari-Sisakht, M.; Ilbeygi, H.; Rana, D.; Matsuura, T.; Ismail, A.F. Synthesis, Modification and Optimization of Titanate Nanotubes-Polyamide Thin Film Nanocomposite (TFN) Membrane for Forward Osmosis (FO) Application. Chem. Eng. J. 2015, 281, 243–251. [Google Scholar] [CrossRef]
- Chekli, L.; Kim, Y.; Phuntsho, S.; Li, S.; Ghaffour, N.; Leiknes, T.O.; Shon, H.K. Evaluation of Fertilizer-Drawn Forward Osmosis for Sustainable Agriculture and Water Reuse in Arid Regions. J. Environ. Manag. 2017, 187, 137–145. [Google Scholar] [CrossRef] [Green Version]
- Phuntsho, S.; Shon, H.K.; Majeed, T.; El Saliby, I.; Vigneswaran, S.; Kandasamy, J.; Hong, S.; Lee, S. Blended Fertilizers as Draw Solutions for Fertilizer-Drawn Forward Osmosis Desalination. Environ. Sci. Technol. 2012, 46, 4567–4575. [Google Scholar] [CrossRef] [PubMed]
- Phuntsho, S.; Lotfi, F.; Hong, S.; Shaffer, D.L.; Elimelech, M.; Shon, H.K. Membrane Scaling and Flux Decline during Fertiliser-Drawn Forward Osmosis Desalination of Brackish Groundwater. Water Res. 2014, 57, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Phuntsho, S.; Sahebi, S.; Majeed, T.; Lotfi, F.; Kim, J.E.; Shon, H.K. Assessing the Major Factors Affecting the Performances of Forward Osmosis and Its Implications on the Desalination Process. Chem. Eng. J. 2013, 231, 484–496. [Google Scholar] [CrossRef]
- Suwaileh, W.A.; Johnson, D.J.; Sarp, S.; Hilal, N. Advances in Forward Osmosis Membranes: Altering the Sub-Layer Structure via Recent Fabrication and Chemical Modification Approaches. Desalination 2018, 436, 176–201. [Google Scholar] [CrossRef] [Green Version]
- Johnson, D.J.; Suwaileh, W.A.; Mohammed, A.W.; Hilal, N. Osmotic’s Potential: An Overview of Draw Solutes for Forward Osmosis. Desalination 2018, 434, 100–120. [Google Scholar] [CrossRef] [Green Version]
- McCutcheon, J.R.; Elimelech, M. Influence of Concentrative and Dilutive Internal Concentration Polarization on Flux Behavior in Forward Osmosis. J. Memb. Sci. 2006, 284, 237–247. [Google Scholar] [CrossRef]
- Tansel, B.; Sager, J.; Rector, T.; Garland, J.; Strayer, R.F.; Levine, L.; Roberts, M.; Hummerick, M.; Bauer, J. Significance of Hydrated Radius and Hydration Shells on Ionic Permeability during Nanofiltration in Dead End and Cross Flow Modes. Sep. Purif. Technol. 2006, 51, 40–47. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, Y.; Yuan, B.; Wang, Z.; Li, X.; Ren, Y. Comparison of Biofouling Mechanisms between Cellulose Triacetate (CTA) and Thin-Film Composite (TFC) Polyamide Forward Osmosis Membranes in Osmotic Membrane Bioreactors. Bioresour. Technol. 2016, 202, 50–58. [Google Scholar] [CrossRef]
- Lay, W.C.L.; Chong, T.H.; Tang, C.Y.; Fane, A.G.; Zhang, J.; Liu, Y. Fouling Propensity of Forward Osmosis: Investigation of the Slower Flux Decline Phenomenon. Water Sci. Technol. 2010, 61, 927–936. [Google Scholar] [CrossRef]
- Li, S.; Kim, Y.; Chekli, L.; Phuntsho, S.; Shon, H.K.; Leiknes, T.O.; Ghaffour, N. Impact of Reverse Nutrient Diffusion on Membrane Biofouling in Fertilizer-Drawn Forward Osmosis. J. Memb. Sci. 2017, 539, 108–115. [Google Scholar] [CrossRef] [Green Version]
- Johnson, D.; Lun, A.W.; Mohammed, A.W.; Hilal, N. Dewatering of POME Digestate Using Lignosulfonate Driven Forward Osmosis. Sep. Purif. Technol. 2020, 235, 116151. [Google Scholar] [CrossRef]
- Adnan, M.; Khan, S.J.; Manzoor, K.; Hankins, N.P. Performance Evaluation of Fertilizer Draw Solutions for Forward Osmosis Membrane Bioreactor Treating Domestic Wastewater. Process Saf. Environ. Prot. 2019, 127, 133–140. [Google Scholar] [CrossRef]
- Ansari, A.J.; Hai, F.I.; He, T.; Price, W.E.; Nghiem, L.D. Physical Cleaning Techniques to Control Fouling during the Pre-Concentration of High Suspended-Solid Content Solutions for Resource Recovery by Forward Osmosis. Desalination 2018, 429, 134–141. [Google Scholar] [CrossRef]
- Kim, Y.; Li, S.; Ghaffour, N. Evaluation of Different Cleaning Strategies for Different Types of Forward Osmosis Membrane Fouling and Scaling. J. Memb. Sci. 2020, 596, 117731. [Google Scholar] [CrossRef]
- Kim, C.; Lee, S.; Hong, S. Application of Osmotic Backwashing in Forward Osmosis: Mechanisms and Factors Involved. Desalin. Water Treat. 2012, 43, 314–322. [Google Scholar] [CrossRef]
- Baharuddin, A.S.; Rahman, N.A.A.; Shah, U.K.M.; Hassan, M.A.; Wakisaka, M.; Shirai, Y. Evaluation of Pressed Shredded Empty Fruit Bunch (EFB)-Palm Oil Mill Effluent (POME) Anaerobic Sludge Based Compost Using Fourier Transform Infrared (FTIR) and Nuclear Magnetic Resonance (NMR) Analysis. Afr. J. Biotechnol. 2011, 10, 8082–8089. [Google Scholar]
- Ganapathy, B.; Yahya, A.; Ibrahim, N. Bioremediation of Palm Oil Mill Effluent (POME) Using Indigenous Meyerozyma Guilliermondii. Environ. Sci. Pollut. Res. 2019, 26, 11113–11125. [Google Scholar] [CrossRef]
- Yoochatchaval, W.; Kumakura, S.; Tanikawa, D.; Yamaguchi, T.; Yunus, M.F.M.; Chen, S.S.; Kubota, K.; Harada, H.; Syutsubo, K. Anaerobic Degradation of Palm Oil Mill Effluent (POME). Water Sci. Technol. 2011, 64, 2001–2008. [Google Scholar] [CrossRef]
- IR Absorption Table. Available online: https://webspectra.chem.ucla.edu/irtable.html (accessed on 23 May 2021).
- Li, F.; Sun, M.; Cheng, Q.; Yang, B. Preparation and Characterization of Graphene Oxide/Cellulose Triacetate Forward Osmosis Membranes. In Proceedings of the MATEC Web of Conferences, Chiang Mai, Thailand, 20–21 August 2016; Volume 67. [Google Scholar]
- Westphalen, H.; Saadati, S.; Eduok, U.; Abdelrasoul, A.; Shoker, A.; Choi, P.; Doan, H.; Ein-Mozaffari, F. Case Studies of Clinical Hemodialysis Membranes: Influences of Membrane Morphology and Biocompatibility on Uremic Blood-Membrane Interactions and Inflammatory Biomarkers. Sci. Rep. 2020, 10, 14808. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Cho, H.; Shin, Y.; Jang, Y.; Lee, S. Economic Evaluation of a Hybrid Desalination System Combining Forward and Reverse Osmosis. Membranes 2015, 6, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El Zayat, H.; Nasr, P.; Sewilam, H. Investigating sustainable management of desalination brine through concentration using forward osmosis. Environ. Sci. Pollut. Res. 2021, 1–14. [Google Scholar] [CrossRef]
- Lok, X.; Chan, Y.J.; Foo, D.C.Y. Simulation and Optimisation of Full-Scale Palm Oil Mill Effluent (POME) Treatment Plant with Biogas Production. J. Water Process Eng. 2020, 38, 101558. [Google Scholar] [CrossRef]




| Water Permeability Coefficient, A (LMH/bar) | Salt Permeability Coefficient, B (LMH) | Structural Parameter, S μm |
|---|---|---|
| 0.51 | 0.156 | 480 |
| Parameters | Concentration (mg/L) |
|---|---|
| pH | 7.76 |
| Chemical oxygen demand (COD) | 13.646 |
| Total suspended solid (TSS) | 7.701 |
| Total Nitrogen (TN) | 617 |
| Phosphorus (P) | 1.264 |
| Potassium (K) | 5.450 |
| Chemicals | Chemical Formula | Molecular Weight, MW (g/mol) | Supplier | Osmotic Pressure (bar) a | Diffusivity, D (10−9 m2/s) a |
|---|---|---|---|---|---|
| Reagent grade: | |||||
| Sodium chloride | NaCl | 58.44 | Chemiz, Malaysia | 47.39 | |
| Ammonium sulfate | (NH4)2SO4 | 132.14 | Systerm | 46.75 | 1.14 |
| Ammonium nitrate | NH4NO3 | 80.04 | Hamburg Chemical | 34.13 | 1.65 |
| Monoammonium phosphate | NH4H2PO4 | 115.03 | Chemiz, Malaysia | 44.40 | 1.06 |
| Diammonium phosphate | (NH4)2HPO4 | 132.06 | Chemiz, Malaysia | 51.23 | 0.912 |
| Potassium chloride | KCl | 74.56 | Systerm | 44.55 | 1.79 |
| Potassium nitrate | KNO3 | 101.10 | Chemiz, Malaysia | 37.68 | 1.78 |
| Commercial grade: | |||||
| Ammonium sulfate | (NH4)2SO4 | 132.14 b | n/a | - | - |
| Monoammonium phosphate | NH4H2PO4 | 115.03 b | n/a | - | - |
| Muriate of potash (MOP) | KCl | 74.56 b | Behn-Meyer Agricare | - | - |
| Factors | Coded Symbol | Range and Level | |
|---|---|---|---|
| Low (−) | High (+) | ||
| DS concentration (Molar) | A | 0.5 | 1.0 |
| Flowrate (mL/min) | B | 500 | 600 |
| FS:DS volume ratio | C | 1:0.5 | 1:1 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 67.40 | 7 | 9.63 | 162.80 | <0.0001 | significant |
| A-DS Conc. | 65.42 | 1 | 65.42 | 1106.01 | <0.0001 | |
| B-Vol. Flowrate | 0.5207 | 1 | 0.5207 | 8.80 | 0.0091 | |
| C-FS:DS Vol. ratio | 0.4248 | 1 | 0.4248 | 7.18 | 0.0164 | |
| AB | 0.0452 | 1 | 0.0452 | 0.7634 | 0.3952 | |
| AC | 0.9886 | 1 | 0.9886 | 16.71 | 0.0009 | |
| BC | 0.0071 | 1 | 0.0071 | 0.1202 | 0.7334 | |
| ABC | 0.0018 | 1 | 0.0018 | 0.0308 | 0.8630 | |
| Pure Error | 0.9463 | 16 | 0.0591 | |||
| Cor Total | 68.35 | 23 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | |
|---|---|---|---|---|---|---|
| Model | 21.26 | 7 | 3.04 | 16.07 | <0.0001 | significant |
| A-DS Conc. | 15.09 | 1 | 15.09 | 79.90 | <0.0001 | |
| B-Vol. Flowrate | 0.0745 | 1 | 0.0745 | 0.3943 | 0.5389 | |
| C-FS:DS Vol. ratio | 1.58 | 1 | 1.58 | 8.38 | 0.0106 | |
| AB | 0.0556 | 1 | 0.0556 | 0.2942 | 0.5950 | |
| AC | 4.04 | 1 | 4.04 | 21.38 | 0.0003 | |
| BC | 0.4121 | 1 | 0.4121 | 2.18 | 0.1591 | |
| ABC | 0.0004 | 1 | 0.0004 | 0.0021 | 0.9642 | |
| Pure Error | 3.02 | 16 | 0.1889 | |||
| Cor Total | 24.28 | 23 |
| Ion | Hydrated Radius (nm) |
|---|---|
| Cation: | |
| K+ | 0.201 |
| Na+ | 0.178 |
| NH4+ | 0.104 |
| Anion: | |
| Cl− | 0.195 |
| NO3− | 0.340 |
| SO42− | 0.300 |
| PO42− | 0.339 |
| Run | Average Water Flux, JW (L/m2 h) | Water Flux Recovery (%) | Flux Decline (%) | Water Recovery (%) | ||
|---|---|---|---|---|---|---|
| Initial | Final | After Cleaning | ||||
| Run 1 (Hydraulic flushing) | 4.24 | 0.75 | 4.20 | 99.2 | 82.4 | 16.4 |
| Run 2 (Osmotic backwashing) | 3.85 | 0.56 | 3.80 | 98.6 | 85.5 | 12.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdul Wahid, R.; Ang, W.L.; Mohammad, A.W.; Johnson, D.J.; Hilal, N. Evaluating Fertilizer-Drawn Forward Osmosis Performance in Treating Anaerobic Palm Oil Mill Effluent. Membranes 2021, 11, 566. https://doi.org/10.3390/membranes11080566
Abdul Wahid R, Ang WL, Mohammad AW, Johnson DJ, Hilal N. Evaluating Fertilizer-Drawn Forward Osmosis Performance in Treating Anaerobic Palm Oil Mill Effluent. Membranes. 2021; 11(8):566. https://doi.org/10.3390/membranes11080566
Chicago/Turabian StyleAbdul Wahid, Ruwaida, Wei Lun Ang, Abdul Wahab Mohammad, Daniel James Johnson, and Nidal Hilal. 2021. "Evaluating Fertilizer-Drawn Forward Osmosis Performance in Treating Anaerobic Palm Oil Mill Effluent" Membranes 11, no. 8: 566. https://doi.org/10.3390/membranes11080566
APA StyleAbdul Wahid, R., Ang, W. L., Mohammad, A. W., Johnson, D. J., & Hilal, N. (2021). Evaluating Fertilizer-Drawn Forward Osmosis Performance in Treating Anaerobic Palm Oil Mill Effluent. Membranes, 11(8), 566. https://doi.org/10.3390/membranes11080566











