Perfluorosulfonic Acid Membranes with Short and Long Side Chains and Their Use in Sensors for the Determination of Markers of Viral Diseases in Saliva
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Conditioning and Pretreatment of Membranes
2.3. Formation of Polymer Dispersions
2.4. Membrane Formation by Casting Procedure
2.5. Membrane Characterization
2.6. Preparation of Artificial Saliva Solution and Measurements with DP-Sensors
2.7. Regeneration of Membranes
3. Results and Discussion
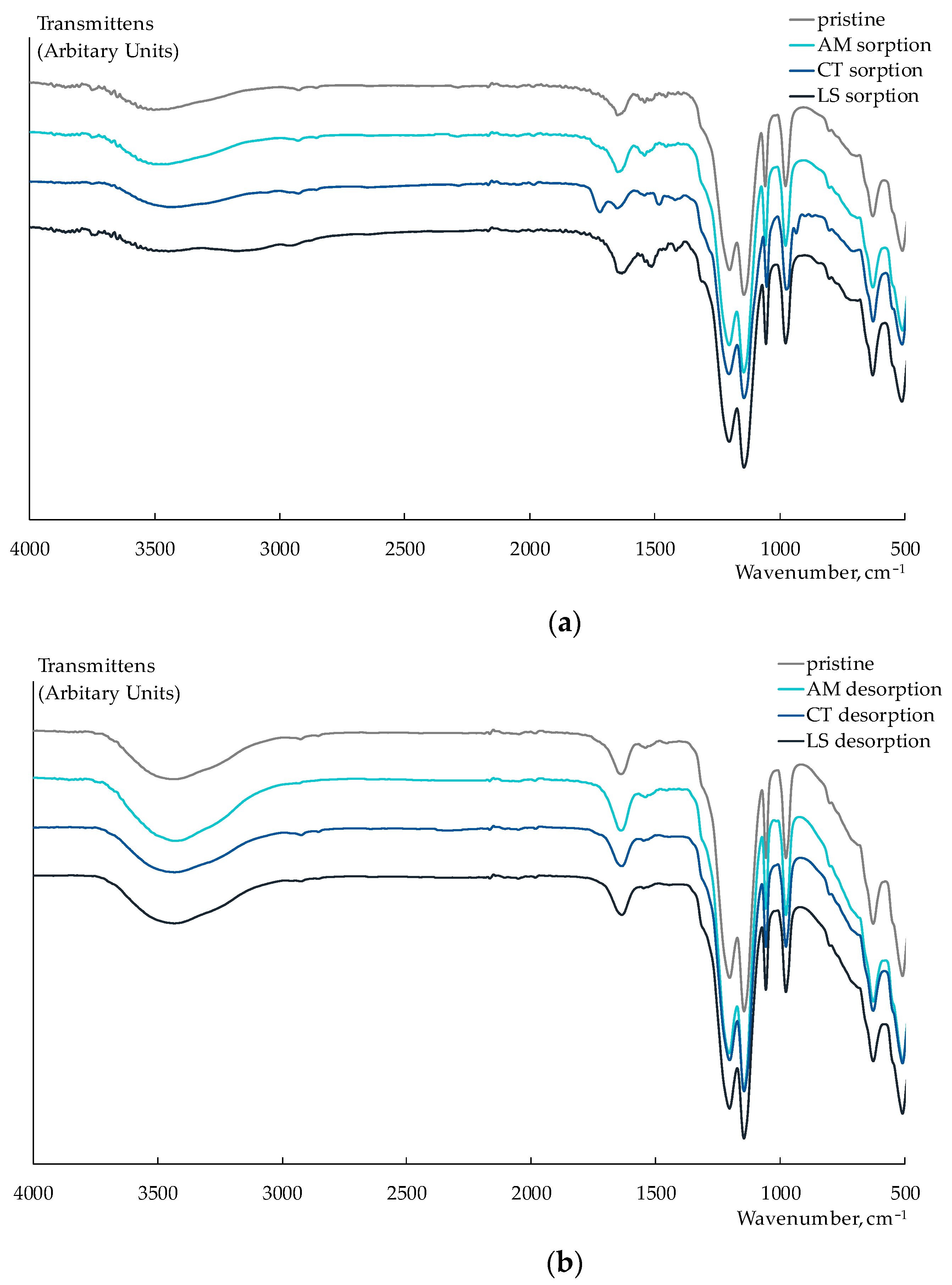
3.1. Membrane Characteristics
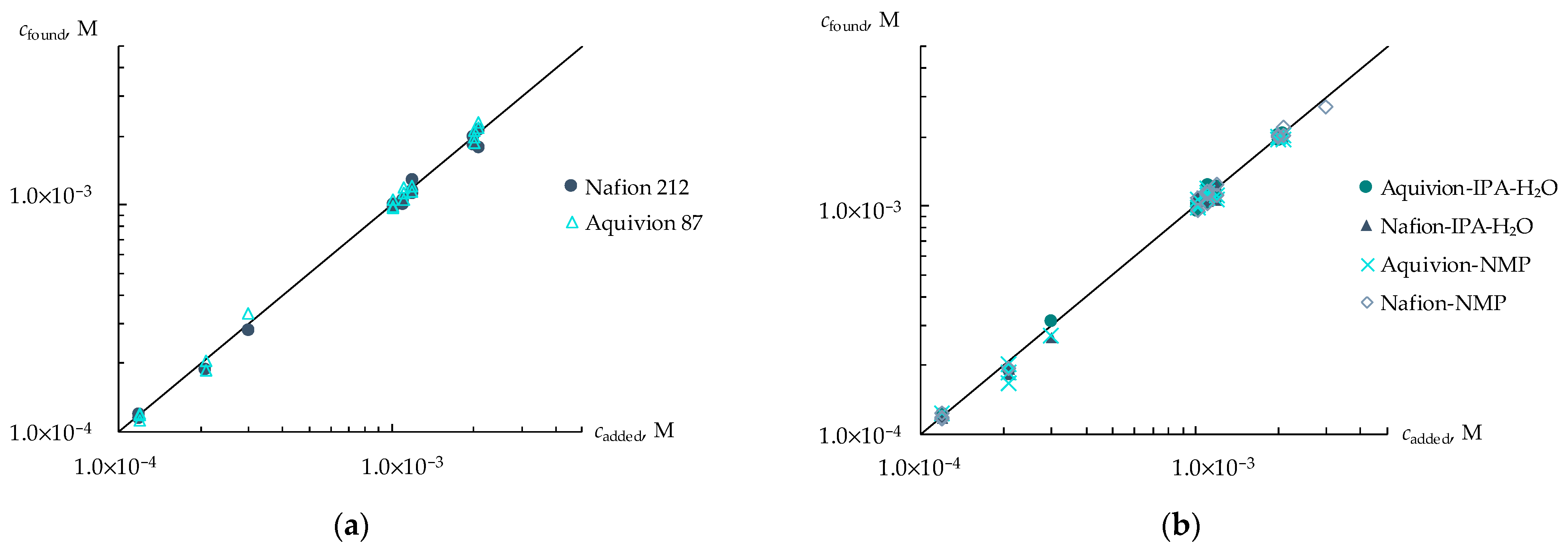
3.2. Determination of Viral Markers in Artificial Saliva Solutions
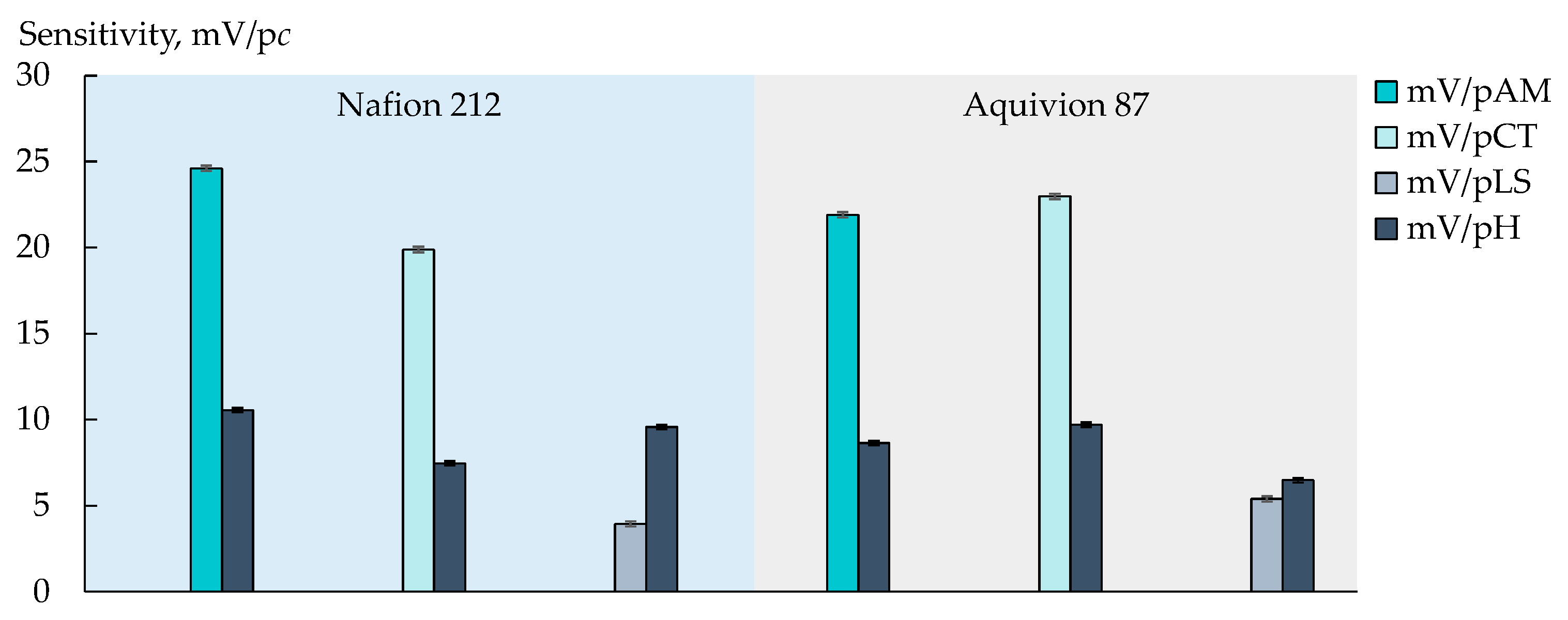
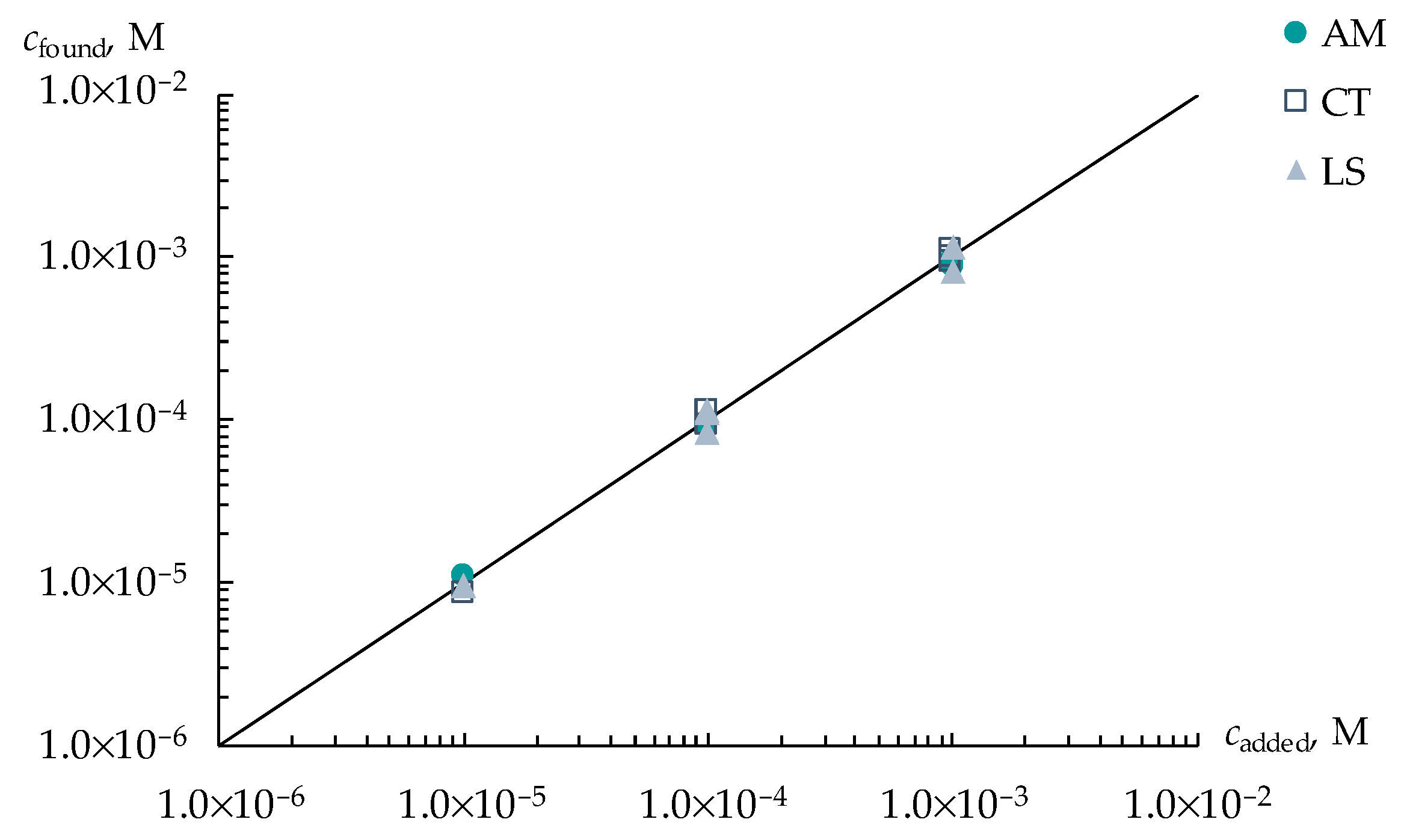
3.3. Group Analysis of Viral Markers in Artificial Saliva Solutions
3.4. Regeneration of Membranes and Stability of Calibrations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Issaoui, M.; Jellali, S.; Zorpas, A.A.; Dutournie, P. Membrane Technology for Sustainable Water Resources Management: Challenges and Future Projections. Sustain. Chem. Pharm. 2022, 25, 100590. [Google Scholar] [CrossRef]
- Goh, P.S.; Wong, K.C.; Ismail, A.F. Membrane Technology: A Versatile Tool for Saline Wastewater Treatment and Resource Recovery. Desalination 2022, 521, 115377. [Google Scholar] [CrossRef]
- Bu, Y.; Hu, Q.; Bao, T.; Xie, X.; Wang, S. Recent Advances in Cell Membrane-Coated Technology for Drug Discovery from Natural Products. TrAC Trends Anal. Chem. 2022, 151, 116601. [Google Scholar] [CrossRef]
- Gkotsis, P.; Kougias, P.; Mitrakas, M.; Zouboulis, A. Biogas Upgrading Technologies—Recent Advances in Membrane-Based Processes. Int. J. Hydrogen Energy 2023, 48, 3965–3993. [Google Scholar] [CrossRef]
- Morker, H.; Saini, B.; Dey, A. Role of Membrane Technology in Food Industry Effluent Treatment. Mater. Today Proc. 2023, 77, 314–321. [Google Scholar] [CrossRef]
- Zhao, Z.; Muylaert, K.; Vankelecom, I.F.J. Applying Membrane Technology in Microalgae Industry: A Comprehensive Review. Renew. Sustain. Energy Rev. 2023, 172, 113041. [Google Scholar] [CrossRef]
- Filippov, S.P.; Yaroslavtsev, A.B. Hydrogen Energy: Development Prospects and Materials. Russ. Chem. Rev. 2021, 90, 627–643. [Google Scholar] [CrossRef]
- Alent’ev, A.Y.; Volkov, A.V.; Vorotyntsev, I.V.; Maksimov, A.L.; Yaroslavtsev, A.B. Membrane Technologies for Decarbonization. Membr. Membr. Technol. 2021, 3, 255–273. [Google Scholar] [CrossRef]
- Belov, N.A.; Pashkevich, D.S.; Alentiev, A.Y.; Tressaud, A. Effect of Direct Fluorination on the Transport Properties and Swelling of Polymeric Materials: A Review. Membranes 2021, 11, 713. [Google Scholar] [CrossRef]
- Robeson, L.M. Polymer Membranes. Polym. Sci. A Compr. Ref. 2012, 1–10, 325–347. [Google Scholar]
- Quast, M.J.; Mueller, A. Membrane Preparation from Hyperbranched Perfluorinated Polymers. J. Polym. Sci. Part A Polym. Chem. 2019, 57, 961–972. [Google Scholar] [CrossRef]
- Doyle, M.; Rajendran, G. Perfluorinated Membranes. In Handbook of Fuel Cells; Vielstich, W., Lamm, A., Gasteiger, H.A., Yokokawa, H., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2010; p. f303034. [Google Scholar] [CrossRef]
- Yampolskii, Y.P.; Belov, N.A.; Alentiev, A.Y. Fluorine in the Structure of Polymers: Influence on the Gas Separation Properties. Russ. Chem. Rev. 2019, 88, 387–405. [Google Scholar] [CrossRef]
- Yampolskii, Y.; Belov, N.; Alentiev, A. Perfluorinated Polymers as Materials of Membranes for Gas and Vapor Separation. J. Membr. Sci. 2020, 598, 117779. [Google Scholar] [CrossRef]
- Javed, A.; Palafox Gonzalez, P.; Thangadurai, V. A Critical Review of Electrolytes for Advanced Low- and High-Temperature Polymer Electrolyte Membrane Fuel Cells. ACS Appl. Mater. Interfaces 2023, 15, 29674–29699. [Google Scholar] [CrossRef] [PubMed]
- Pourcelly, G.; Nikonenko, V.V.; Pismenskaya, N.D.; Yaroslavtsev, A.B. Applications of Charged Membranes in Separation, Fuel Cells, and Emerging Processes. In Ionic Interactions in Natural and Synthetic Macromolecules; Ciferri, A., Perico, A., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 761–815. [Google Scholar] [CrossRef]
- Olabi, A.G.; Wilberforce, T.; Alanazi, A.; Vichare, P.; Sayed, E.T.; Maghrabie, H.M.; Elsaid, K.; Abdelkareem, M.A. Novel Trends in Proton Exchange Membrane Fuel Cells. Energies 2022, 15, 4949. [Google Scholar] [CrossRef]
- Shi, X.; Esan, O.C.; Huo, X.; Ma, Y.; Pan, Z.; An, L.; Zhao, T.S. Polymer Electrolyte Membranes for Vanadium Redox Flow Batteries: Fundamentals and Applications. Prog. Energy Combust. Sci. 2021, 85, 100926. [Google Scholar] [CrossRef]
- Voropaeva, D.Y.; Novikova, S.A.; Yaroslavtsev, A.B. Polymer Electrolytes for Metal-Ion Batteries. Russ. Chem. Rev. 2020, 89, 1132–1155. [Google Scholar] [CrossRef]
- Xu, G.; Dong, X.; Xue, B.; Huang, J.; Wu, J.; Cai, W. Recent Approaches to Achieve High Temperature Operation of Nafion Membranes. Energies 2023, 16, 1565. [Google Scholar] [CrossRef]
- Kusoglu, A.; Weber, A.Z. New Insights into Perfluorinated Sulfonic-Acid Ionomers. Chem. Rev. 2017, 117, 987–1104. [Google Scholar] [CrossRef]
- Primachenko, O.N.; Marinenko, E.A.; Odinokov, A.S.; Kononova, S.V.; Kulvelis, Y.V.; Lebedev, V.T. State of the Art and Prospects in the Development of Proton-conducting Perfluorinated Membranes with Short Side Chains: A Review. Polym. Adv. Technol. 2021, 32, 1386–1408. [Google Scholar] [CrossRef]
- Zhao, N.; Edwards, D.; Lei, C.; Wang, K.; Li, J.; Zhang, Y.; Holdcroft, S.; Shi, Z. The Importance of Water Transport on Short-Side Chain Perfluorosulfonic Acid Membrane Fuel Cells Operating under Low Relative Humidity. J. Power Source 2013, 242, 877–883. [Google Scholar] [CrossRef]
- Prikhno, I.A.; Safronova, E.Y.; Stenina, I.A.; Yurova, P.A.; Yaroslavtsev, A.B. Dependence of the Transport Properties of Perfluorinated Sulfonated Cation-Exchange Membranes on Ion-Exchange Capacity. Membr. Membr. Technol. 2020, 2, 265–271. [Google Scholar] [CrossRef]
- Shi, S.; Weber, A.Z.; Kusoglu, A. Structure-Transport Relationship of Perfluorosulfonic-Acid Membranes in Different Cationic Forms. Electrochim. Acta 2016, 220, 517–528. [Google Scholar] [CrossRef]
- Allen, F.I.; Comolli, L.R.; Kusoglu, A.; Modestino, M.A.; Minor, A.M.; Weber, A.Z. Morphology of Hydrated As-Cast Nafion Revealed through Cryo Electron Tomography. ACS Macro Lett. 2015, 4, 1–5. [Google Scholar] [CrossRef]
- Wu, W.; Li, Y.; Liu, J.; Wang, J.; He, Y.; Davey, K.; Qiao, S. Molecular-Level Hybridization of Nafion with Quantum Dots for Highly Enhanced Proton Conduction. Adv. Mater. 2018, 30, 1707516. [Google Scholar] [CrossRef]
- Guan, P.; Zou, Y.; Zhang, M.; Zhong, W.; Xu, J.; Lei, J.; Ding, H.; Feng, W.; Liu, F.; Zhang, Y. High-Temperature Low-Humidity Proton Exchange Membrane with “Stream-Reservoir” Ionic Channels for High-Power-Density Fuel Cells. Sci. Adv. 2023, 9, eadh1386. [Google Scholar] [CrossRef] [PubMed]
- Guan, P.; Lei, J.; Liu, X.; Xu, K.; Pai, S.; Ding, H.; Zou, Y.; Feng, W.; Liu, F.; Zhang, Y. Origins of water state and ionic cluster morphology for high proton conductivity of short side-chain perfluorinated sulfonic acid membranes. Chem. Mater. 2022, 34, 7845–7857. [Google Scholar] [CrossRef]
- Crothers, A.R.; Radke, C.J.; Weber, A.Z. Impact of Nano- and Mesoscales on Macroscopic Cation Conductivity in Perfluorinated-Sulfonic-Acid Membranes. J. Phys. Chem. C 2017, 121, 28262–28274. [Google Scholar] [CrossRef]
- Clothers, A.R.; Kusoglu, A.; Radke, C.J.; Weber, A.Z. Influence of mesoscale interactions on proton, water, and electrokinetic transport in solvent-filled membranes: Theory and simulation. Langmuir 2022, 38, 10362–10374. [Google Scholar] [CrossRef]
- Haubold, H.-G.; Vad, T.; Jungbluth, H.; Hiller, P. Nano Structure of NAFION: A SAXS Study. Electrochim. Acta 2001, 46, 1559–1563. [Google Scholar] [CrossRef]
- Hsu, W.Y.; Gierke, T.D. Ion Transport and Clustering in Nafion Perfluorinated Membranes. J. Membr. Sci. 1983, 13, 307–326. [Google Scholar] [CrossRef]
- Safronova, E.Y.; Voropaeva, D.Y.; Novikova, S.A.; Yaroslavtsev, A.B. On the Influence of Solvent and Ultrasonic Treatment on Properties of Cast Nafion® Membranes. Membr. Membr. Technol. 2022, 4, 39–47. [Google Scholar] [CrossRef]
- Kim, Y.S.; Welch, C.F.; Hjelm, R.P.; Mack, N.H.; Labouriau, A.; Orler, E.B. Origin of Toughness in Dispersion-Cast Nafion Membranes. Macromolecules 2015, 48, 2161–2172. [Google Scholar] [CrossRef]
- Berlinger, S.A.; Dudenas, P.J.; Bird, A.; Chen, X.; Freychet, G.; McCloskey, B.D.; Kusoglu, A.; Weber, A.Z. Impact of Dispersion Solvent on Ionomer Thin Films and Membranes. ACS Appl. Polym. Mater. 2020, 2, 5824–5834. [Google Scholar] [CrossRef]
- Collette, F.M.; Thominette, F.; Mendil-Jakani, H.; Gebel, G. Structure and Transport Properties of Solution-Cast Nafion® Membranes Subjected to Hygrothermal Aging. J. Membr. Sci. 2013, 435, 242–252. [Google Scholar] [CrossRef]
- Safronova, E.Y.; Voropaeva, D.Y.; Safronov, D.V.; Stretton, N.; Parshina, A.V.; Yaroslavtsev, A.B. Correlation between Nafion Morphology in Various Dispersion Liquids and Properties of the Cast Membranes. Membranes 2022, 13, 13. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Chen, X.; Yu, X.; Chen, X.; Ding, X.; Zhao, X.; Tang, K. Ultrahighly Sensitive QCM Humidity Sensor Based on Nafion/MoS2 Hybrid Thin Film. IEEE Trans. Electron Devices 2022, 69, 1321–1326. [Google Scholar] [CrossRef]
- Maciak, E. Low-Coherence Interferometric Fiber Optic Sensor for Humidity Monitoring Based on Nafion® Thin Film. Sensors 2019, 19, 629. [Google Scholar] [CrossRef]
- Liu, S.; Ji, Y.; Yang, J.; Sun, W.; Li, H. Nafion Film Temperature/Humidity Sensing Based on Optical Fiber Fabry-Perot Interference. Sens. Actuators A Phys. 2018, 269, 313–321. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Hsiao, Y.-S.; Lu, H.-C.; Chen, P.; Whang, W.-T. Humidity-Switch Chromism of Aniline-Pentamer in Nafion. J. Polym. Res. 2016, 23, 196. [Google Scholar] [CrossRef]
- Zhi, Z.; Gao, W.; Yang, J.; Geng, C.; Yang, B.; Tian, C.; Fan, S.; Li, H.; Li, J.; Hua, Z. Amperometric Hydrogen Gas Sensor Based on Pt/C/Nafion Electrode and Ionic Electrolyte. Sens. Actuators B Chem. 2022, 367, 132137. [Google Scholar] [CrossRef]
- Kumaravel, A.; Chandrasekaran, M. A Biocompatible Nano TiO2/Nafion Composite Modified Glassy Carbon Electrode for the Detection of Fenitrothion. J. Electroanal. Chem. 2011, 650, 163–170. [Google Scholar] [CrossRef]
- Liu, J.; Cao, Y. An Electrochemical Sensor Based on an Anti-Fouling Membrane for the Determination of Histamine in Fish Samples. Anal. Methods 2021, 13, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Xu, Q.; Li, Y.; Long, N.; Li, P.; Wang, J.; Zhou, L.; Sheng, P.; Kong, W. Ultrasensitive Electrochemical Aptasensor with Nafion-Stabilized f-MWCNTs as Signal Enhancers for OTA Detection. Bioelectrochemistry 2023, 151, 108399. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, W.; Wajrak, M.; Alameh, K. Manufacture and Application of RuO2 Solid-State Metal-Oxide PH Sensor to Common Beverages. Talanta 2018, 180, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Finnerty, N.J.; O’Riordan, S.L.; Brown, F.O.; Serra, P.A.; O’Neill, R.D.; Lowry, J.P. In Vivo Characterisation of a Nafion®-Modified Pt Electrode for Real-Time Nitric Oxide Monitoring in Brain Extracellular Fluid. Anal. Methods 2012, 4, 550. [Google Scholar] [CrossRef]
- Pasqualotto, E.; Cretaio, E.; Scaramuzza, M.; De Toni, A.; Franchin, L.; Paccagnella, A.; Bonaldo, S. Optical System Based on Nafion Membrane for the Detection of Ammonia in Blood Serum Samples. Biosensors 2022, 12, 1079. [Google Scholar] [CrossRef] [PubMed]
- Stozhko, N.; Bukharinova, M.; Galperin, L.; Brainina, K. A Nanostructured Sensor Based on Gold Nanoparticles and Nafion for Determination of Uric Acid. Biosensors 2018, 8, 21. [Google Scholar] [CrossRef] [PubMed]
- Karthika, A.; Suganthi, A.; Rajarajan, M. An Ultrahigh Selective Uric Acid Sensor Based on SrWO4 Nanocomposite Using Pomelo Leaf Extract Solubilized Nafion Modified Glassy Carbon Electrode. J. Sci. Adv. Mater. Devices 2021, 6, 186–196. [Google Scholar] [CrossRef]
- Karthika, A.; Rosaline, D.R.; Inbanathan, S.S.R.; Suganthi, A.; Rajarajan, M. Fabrication of Cupric Oxide Decorated β-Cyclodextrin Nanocomposite Solubilized Nafion as a High Performance Electrochemical Sensor for l-Tyrosine Detection. J. Phys. Chem. Solids 2020, 136, 109145. [Google Scholar] [CrossRef]
- Alam, M.M.; Asiri, A.M.; Rahman, M.M. Wet-Chemically Synthesis of SnO2-Doped Ag2O Nanostructured Materials for Sensitive Detection of Choline by an Alternative Electrochemical Approach. Microchem. J. 2021, 165, 106092. [Google Scholar] [CrossRef]
- De Cássia Mendonça, J.; Da Rocha, L.R.; Capelari, T.B.; Prete, M.C.; Angelis, P.N.; Segatelli, M.G.; Tarley, C.R.T. Design and Performance of Novel Molecularly Imprinted Biomimetic Adsorbent for Preconcentration of Prostate Cancer Biomarker Coupled to Electrochemical Determination by Using Multi-Walled Carbon Nanotubes/Nafion®/Ni(OH)2-Modified Screen-Printed Electrode. J. Electroanal. Chem. 2020, 878, 114582. [Google Scholar] [CrossRef]
- Pedrozo-Peñafiel, M.J.; Lópes, T.; Gutiérrez-Beleño, L.M.; Da Costa, M.E.H.M.; Larrudé, D.G.; Aucelio, R.Q. Voltammetric Determination of Creatinine Using a Gold Electrode Modified with Nafion Mixed with Graphene Quantum Dots-Copper. J. Electroanal. Chem. 2020, 878, 114561. [Google Scholar] [CrossRef]
- Parrilla, M.; Vanhooydonck, A.; Watts, R.; De Wael, K. Wearable Wristband-Based Electrochemical Sensor for the Detection of Phenylalanine in Biofluids. Biosens. Bioelectron. 2022, 197, 113764. [Google Scholar] [CrossRef]
- Kalasin, S.; Sangnuang, P.; Khownarumit, P.; Tang, I.M.; Surareungchai, W. Salivary Creatinine Detection Using a Cu(I)/Cu(II) Catalyst Layer of a Supercapacitive Hybrid Sensor: A Wireless IoT Device To Monitor Kidney Diseases for Remote Medical Mobility. ACS Biomater. Sci. Eng. 2020, 6, 5895–5910. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Liu, M.; Shi, Y.; Yang, X.; Li, L.; Lu, Q.; Zheng, H.; Feng, S.; Bai, Y.; Zhang, T. Vertically Aligned Conductive Metal-Organic Framework Nanowires Array Composite Fiber as Efficient Solid-Contact for Wearable Potentiometric Sweat Sensing. Sens. Actuators B Chem. 2022, 369, 132290. [Google Scholar] [CrossRef]
- Hoekstra, R.; Blondeau, P.; Andrade, F.J. IonSens: A Wearable Potentiometric Sensor Patch for Monitoring Total Ion Content in Sweat. Electroanalysis 2018, 30, 1536–1544. [Google Scholar] [CrossRef]
- Kang, B.-C.; Park, B.-S.; Ha, T.-J. Highly Sensitive Wearable Glucose Sensor Systems Based on Functionalized Single-Wall Carbon Nanotubes with Glucose Oxidase-Nafion Composites. Appl. Surf. Sci. 2019, 470, 13–18. [Google Scholar] [CrossRef]
- Cánovas, R.; Blondeau, P.; Andrade, F.J. Modulating the Mixed Potential for Developing Biosensors: Direct Potentiometric Determination of Glucose in Whole, Undiluted Blood. Biosens. Bioelectron. 2020, 163, 112302. [Google Scholar] [CrossRef]
- Liu, C.-T.; Liu, C.-H.; Lai, Y.-T.; Lee, C.-Y.; Gupta, S.; Tai, N.-H. A Salivary Glucose Biosensor Based on Immobilization of Glucose Oxidase in Nafion-Carbon Nanotubes Nanocomposites Modified on Screen Printed Electrode. Microchem. J. 2023, 191, 108872. [Google Scholar] [CrossRef]
- Parrilla, M.; Detamornrat, U.; Domínguez-Robles, J.; Donnelly, R.F.; De Wael, K. Wearable Hollow Microneedle Sensing Patches for the Transdermal Electrochemical Monitoring of Glucose. Talanta 2022, 249, 123695. [Google Scholar] [CrossRef] [PubMed]
- Xuan, X.; Yoon, H.S.; Park, J.Y. A Wearable Electrochemical Glucose Sensor Based on Simple and Low-Cost Fabrication Supported Micro-Patterned Reduced Graphene Oxide Nanocomposite Electrode on Flexible Substrate. Biosens. Bioelectron. 2018, 109, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Myndrul, V.; Iatsunskyi, I.; Babayevska, N.; Jarek, M.; Jesionowski, T. Effect of Electrode Modification with Chitosan and Nafion® on the Efficiency of Real-Time Enzyme Glucose Biosensors Based on ZnO Tetrapods. Materials 2022, 15, 4672. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Zhao, F.; Zeng, B. A Molecularly Imprinted Copolymer Based Electrochemical Sensor for the Highly Sensitive Detection of L-Tryptophan. Talanta 2020, 206, 120245. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Liu, L.; Ou, G.; Huang, W.; Wu, K. Development of Nafion/Single-Walled Carbon Nanotube Integrated Arrays for the Rapid Detection of Salbutamol Doping. Anal. Chim. Acta 2023, 1249, 340907. [Google Scholar] [CrossRef]
- Hao, Y.; Zhang, H.; Yue, X.; Zhao, Z.; Zhao, S. A Novel Electrochemical Sensor Based on Cu(II) Metal Organic Framework for the Determination of Metformin. Microchem. J. 2023, 191, 108849. [Google Scholar] [CrossRef]
- Senel, M. Electrochemistry Test Strip as Platform for In Situ Detection of Blood Levels of Antipsychotic Clozapine in Finger-Pricked Sample Volume. Biosensors 2023, 13, 346. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.; Ansari, M.S.; Satsangee, S.P.; Alam, M.G.; Jain, R. Electrochemical Sensing Platform Based on ZrO2/BiVO4 Nanocomposite for Gastro-Prokinetic Drug in Human Blood Serum. J. Nanostruct. Chem. 2023, 13, 361–375. [Google Scholar] [CrossRef]
- Madhuvilakku, R.; Yen, Y.-K.; Yan, W.-M.; Huang, G.-W. Laser-Scribed Graphene Electrodes Functionalized with Nafion/Fe3O4 Nanohybrids for the Ultrasensitive Detection of Neurotoxin Drug Clioquinol. ACS Omega 2022, 7, 15936–15950. [Google Scholar] [CrossRef]
- Safronova, E.; Safronov, D.; Lysova, A.; Parshina, A.; Bobreshova, O.; Pourcelly, G.; Yaroslavtsev, A. Sensitivity of Potentiometric Sensors Based on Nafion®-Type Membranes and Effect of the Membranes Mechanical, Thermal, and Hydrothermal Treatments on the on Their Properties. Sens. Actuators B Chem. 2017, 240, 1016–1023. [Google Scholar] [CrossRef]
- Parshina, A.; Kolganova, T.; Safronova, E.; Osipov, A.; Lapshina, E.; Yelnikova, A.; Bobreshova, O.; Yaroslavtsev, A. Perfluorosulfonic Acid Membranes Thermally Treated and Modified by Dopants with Proton-Acceptor Properties for Asparaginate and Potassium Ions Determination in Pharmaceuticals. Membranes 2019, 9, 142. [Google Scholar] [CrossRef]
- Parshina, A.; Yelnikova, A.; Safronova, E.; Kolganova, T.; Bobreshova, O.; Yaroslavtsev, A. Potentiometric Sensor Arrays Based on Hybrid PFSA/CNTs Membranes for the Analysis of UV-Degraded Drugs. Polymers 2023, 15, 2682. [Google Scholar] [CrossRef] [PubMed]
- Parshina, A.; Yelnikova, A.; Kolganova, T.; Titova, T.; Yurova, P.; Stenina, I.; Bobreshova, O.; Yaroslavtsev, A. Perfluorosulfonic Acid Membranes Modified with Polyaniline and Hydrothermally Treated for Potentiometric Sensor Arrays for the Analysis of Combination Drugs. Membranes 2023, 13, 311. [Google Scholar] [CrossRef]
- Skulimowska, A.; Dupont, M.; Zaton, M.; Sunde, S.; Merlo, L.; Jones, D.J.; Rozière, J. Proton Exchange Membrane Water Electrolysis with Short-Side-Chain Aquivion® Membrane and IrO2 Anode Catalyst. Int. J. Hydrogen Energy 2014, 39, 6307–6316. [Google Scholar] [CrossRef]
- Mohammed, A.; Alghetaa, H.F.K.; Miranda, K.; Wilson, K.; Singh, N.P.; Cai, G.; Putluri, N.; Nagarkatti, P.; Nagarkatti, M. Δ9-Tetrahydrocannabinol Prevents Mortality from Acute Respiratory Distress Syndrome through the Induction of Apoptosis in Immune Cells, Leading to Cytokine Storm Suppression. Int. J. Mol. Sci. 2020, 21, 6244. [Google Scholar] [CrossRef]
- Welch, C.; Labouriau, A.; Hjelm, R.; Orler, B.; Johnston, C.; Kim, Y.S. Nafion in Dilute Solvent Systems: Dispersion or Solution? ACS Macro Lett. 2012, 1, 1403–1407. [Google Scholar] [CrossRef]
- Tarokh, A.; Karan, K.; Ponnurangam, S. Atomistic MD Study of Nafion Dispersions: Role of Solvent and Counterion in the Aggregate Structure, Ionic Clustering, and Acid Dissociation. Macromolecules 2020, 53, 288–301. [Google Scholar] [CrossRef]
- Shimoaka, T.; Wakai, C.; Sakabe, T.; Yamazaki, S.; Hasegawa, T. Hydration Structure of Strongly Bound Water on the Sulfonic Acid Group in a Nafion Membrane Studied by Infrared Spectroscopy and Quantum Chemical Calculation. Phys. Chem. Chem. Phys. 2015, 17, 8843–8849. [Google Scholar] [CrossRef]
- Kreuer, K.-D.; Paddison, S.J.; Spohr, E.; Schuster, M. Transport in Proton Conductors for Fuel-Cell Applications: Simulations, Elementary Reactions, and Phenomenology. Chem. Rev. 2004, 104, 4637–4678. [Google Scholar] [CrossRef]
- Gebel, G.; Moore, R.B. Small-Angle Scattering Study of Short Pendant Chain Perfuorosulfonated Ionomer Membranes. Macromolecules 2000, 33, 4850–4855. [Google Scholar] [CrossRef]
- Atanassova, S.; Neykov, K.; Gutzow, I. Solubility and Dissolution Kinetics of Calcium Oxalate Renal Calculi in Solutions, Containing Dl-Lysine: In Vitro Experiments. J. Cryst. Growth 2000, 212, 233–238. [Google Scholar] [CrossRef]


| Name | Side Chain Composition | Form | EW, g/mol |
|---|---|---|---|
| Nafion 212 | -O-CF2-CF(CF3)-O-CF2-CF2-SO3H | Film | 1100 |
| Aquivion E87-05S | -O-CF2-CF2-SO3H | Film | 870 |
| Aquivion PW79S | -O-CF2-CF2-SO3H | Powder | 790 |
| Name | Polymer | Counter Ion | Dispersing Liquid | Mass Fraction of Polymer, wt% | Dispersing Conditions | ηdisp., mPa·s |
|---|---|---|---|---|---|---|
| Nafion-IPA-H2O | Nafion 212 | Li+ | IPA-H2O 80-20 | 5.0 | 80 °C, 2 h | 15.1 |
| Nafion-NMP | Nafion 212 | Li+ | NMP | 5.0 | 100 °C, 2 h | 11.6 |
| Aquivion-IPA-H2O | Aquivion PW79S | H+ | IPA-H2O 80-20 | 2.5 | 95 °C, 3 h | 37.7 |
| Aquivion-NMP | Aquivion PW79S | H+ | NMP | 2.5 | 110 °C, 3 h | 13.7 |
| Membrane | IEC, mg-eqv/g | Water Uptake, H+ Form | Water Uptake, Na+ Form | ||
|---|---|---|---|---|---|
| W, % | λ (H2O/−SO3−) | W, % | λ (H2O/−SO3−) | ||
| Nafion 212 | 0.98 | 29.2 | 16.5 | 20.7 | 11.7 |
| Aquivion 87 | 1.13 | 44.0 | 21.6 | 32.1 | 15.8 |
| Nafion-IPA-H2O | 0.97 | 42.2 | 24.2 | 32.3 | 18.5 |
| Nafion-NMP | 0.97 | 38.9 | 22.9 | 26.8 | 15.3 |
| Aquivion-IPA-H2O | 1.23 | 58.2 | 26.3 | 53.5 | 24.2 |
| Aquivion-NMP | 1.23 | 67.3 | 30.4 | 52.5 | 23.7 |
| Membrane | H+ Form | Na+ Form | ||
|---|---|---|---|---|
| σ, mS/cm | S, mV/pc | σ, mS/cm | S, mV/pc | |
| Nafion 212 | 4.9 | 51.4 | 3.5 | 50.1 |
| Aquivion 87 | 6.3 | 51.6 | 2.8 | 52.0 |
| Nafion-IPA-H2O | 6.8 | 51.2 | 5.1 | 47.5 |
| Nafion-NMP | 5.2 | 52.4 | 3.4 | 48.0 |
| Aquivion-IPA-H2O | 15.6 | 55.7 | 7.5 | 51.1 |
| Aquivion-NMP | 17.3 | 54.9 | 8.3 | 52.3 |
| Membrane | b1, mV/pY | b2, mV/pH | ε, % |
|---|---|---|---|
| Nafion 212 | 13.07 ± 0.09 | 5.13 ± 0.06 | 2 |
| Aquivion 87 | 10.67 ± 0.08 | 3.99 ± 0.05 | 1.6 |
| Nafion-IPA-H2O | 11.74 ± 0.05 | 4.83 ± 0.04 | 1.9 |
| Nafion-NMP | 11.05 ± 0.06 | 3.98 ± 0.04 | 2 |
| Aquivion-IPA-H2O | 11.11 ±0.06 | 4.94 ± 0.04 | 1.9 |
| Aquivion-NMP | 11.48 ± 0.04 | 5.25 ± 0.03 | 1.9 |
| Membrane | t-Test | t-Test, f = 5, p = 0.95 | F-Test | F-Test, f1 = 2, f2 = 3, p = 0.95 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| b0, mV | b1, mV/pAM | b2, mV/pCT | b3, mV/pLS | b4, mV/pH | b0, mV | b1, mV/pAM | b2, mV/pCT | b3, mV/pLS | b4, mV/pH | |||
| Nafion 212 | 2.07 | 0.75 | 0.72 | 0.81 | 0.64 | 2.57 | 2.36 | 1.90 | 2.37 | 2.09 | 2.11 | 9.95 |
| Aquivion 87 | 0.48 | 1.18 | 0.28 | 0.92 | 0.07 | 1.07 | 1.13 | 1.19 | 0.87 | 1.18 | ||
| Nafion-NMP | 1.24 | 1.39 | 0.18 | 0.28 | 0.52 | 1.57 | 1.46 | 1.72 | 1.56 | 1.56 | ||
| Membrane | t-Test | t-Test, f = 5, p = 0.95 | F-Test | F-Test, f1 = 2, f2 = 3, p = 0.95 | ||||
|---|---|---|---|---|---|---|---|---|
| b0, mV | b1, mV/pY | b2, mV/pH | b0, mV | b1, mV/pY | b2, mV/pH | |||
| Aquivion 87 | 0.50 | 1.46 | 1.69 | 2.57 | 1.53 | 1.46 | 1.58 | 9.95 |
| Nafion-NMP | 0.43 | 0.11 | 0.55 | 1.57 | 1.55 | 1.56 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parshina, A.V.; Safronova, E.Y.; Novikova, S.A.; Stretton, N.; Yelnikova, A.S.; Zhuchkov, T.R.; Bobreshova, O.V.; Yaroslavtsev, A.B. Perfluorosulfonic Acid Membranes with Short and Long Side Chains and Their Use in Sensors for the Determination of Markers of Viral Diseases in Saliva. Membranes 2023, 13, 701. https://doi.org/10.3390/membranes13080701
Parshina AV, Safronova EY, Novikova SA, Stretton N, Yelnikova AS, Zhuchkov TR, Bobreshova OV, Yaroslavtsev AB. Perfluorosulfonic Acid Membranes with Short and Long Side Chains and Their Use in Sensors for the Determination of Markers of Viral Diseases in Saliva. Membranes. 2023; 13(8):701. https://doi.org/10.3390/membranes13080701
Chicago/Turabian StyleParshina, Anna V., Ekaterina Yu. Safronova, Svetlana A. Novikova, Nastasia Stretton, Anastasia S. Yelnikova, Timur R. Zhuchkov, Olga V. Bobreshova, and Andrey B. Yaroslavtsev. 2023. "Perfluorosulfonic Acid Membranes with Short and Long Side Chains and Their Use in Sensors for the Determination of Markers of Viral Diseases in Saliva" Membranes 13, no. 8: 701. https://doi.org/10.3390/membranes13080701
APA StyleParshina, A. V., Safronova, E. Y., Novikova, S. A., Stretton, N., Yelnikova, A. S., Zhuchkov, T. R., Bobreshova, O. V., & Yaroslavtsev, A. B. (2023). Perfluorosulfonic Acid Membranes with Short and Long Side Chains and Their Use in Sensors for the Determination of Markers of Viral Diseases in Saliva. Membranes, 13(8), 701. https://doi.org/10.3390/membranes13080701









