Supervised Resistance Training on Functional Capacity, Muscle Strength and Vascular Function in Peripheral Artery Disease: An Updated Systematic Review and Meta-Analysis
Abstract
:1. Introduction
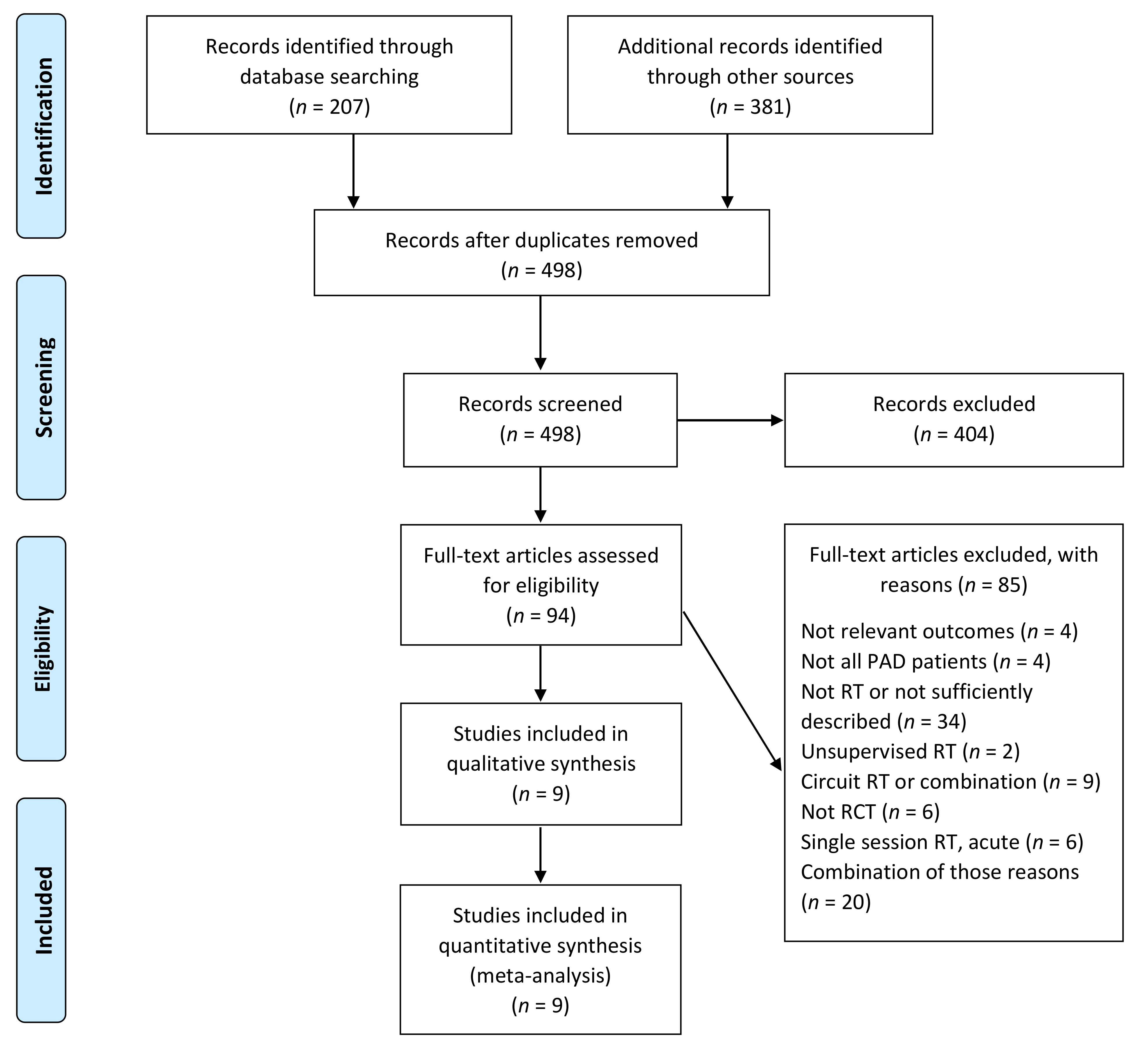
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Exclusion Criteria
2.4. Risk of Bias
2.5. Outcome Measures
2.6. Data Extraction
2.7. Data Synthesis and Analyses
3. Results
3.1. Description of Studies and Exercise Interventions
3.2. Risk of Bias of Included Studies
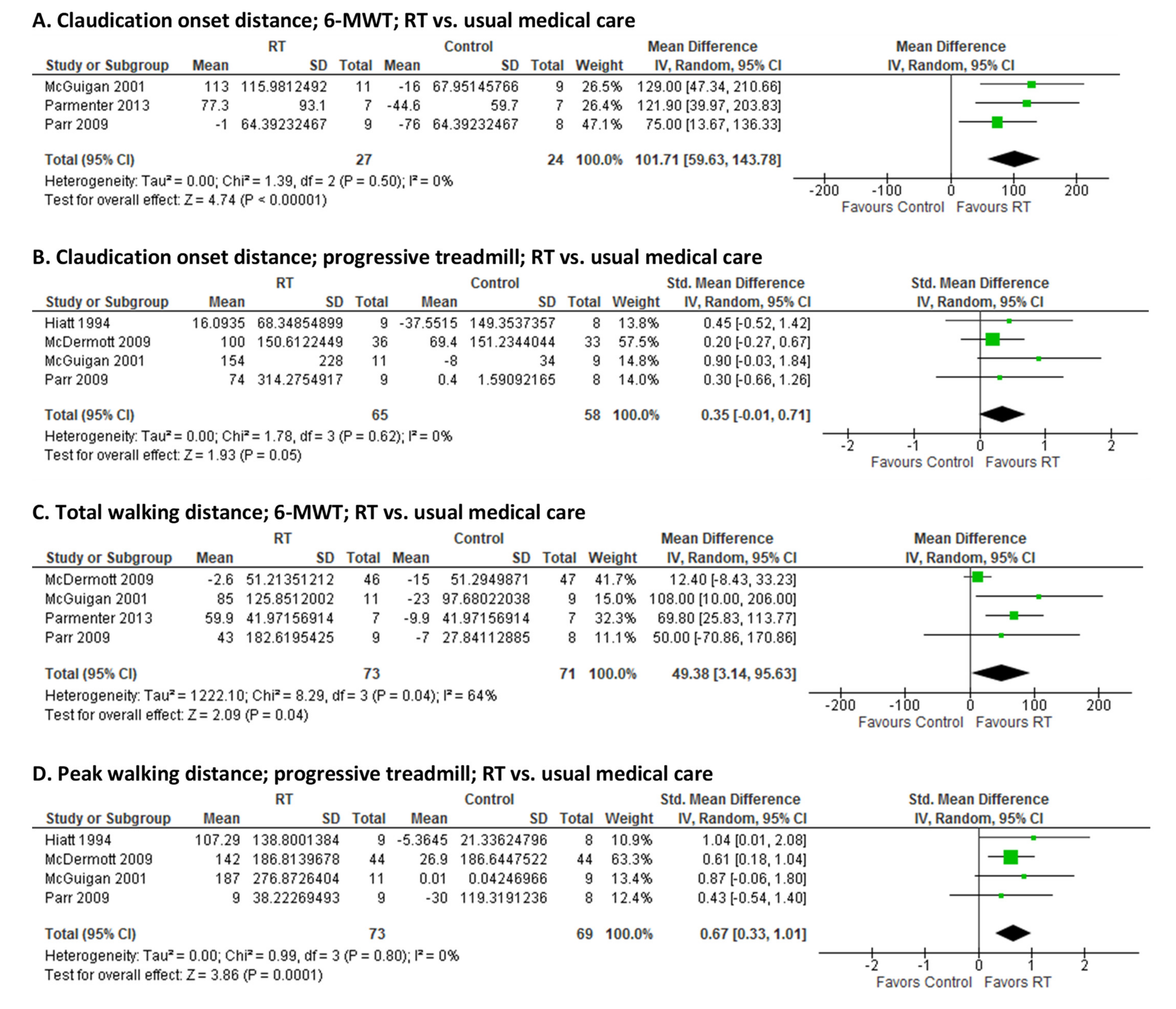
3.3. Effect of RT on Walking Capacity
3.4. Effect of RT on Muscle Strength, Blood Pressure, ABI and Vascular Function
4. Discussion
Limitations and Future Research
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fowkes, F.G.; Rudan, D.; Rudan, I.; Aboyans, V.; Denenberg, J.O.; McDermott, M.M.; Norman, P.E.; Sampson, U.K.; Williams, L.J.; Mensah, G.A.; et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: A systematic review and analysis. Lancet 2013, 382, 1329–1340. [Google Scholar] [CrossRef]
- Ziegler-Graham, K.; MacKenzie, E.J.; Ephraim, P.L.; Travison, T.G.; Brookmeyer, R. Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch. Phys. Med. Rehabil. 2008, 89, 422–429. [Google Scholar] [CrossRef]
- McDermott, M.M.; Liu, K.; Greenland, P.; Guralnik, J.M.; Criqui, M.H.; Chan, C.; Pearce, W.H.; Schneider, J.R.; Ferrucci, L.; Celic, L.; et al. Functional decline in peripheral arterial disease: Associations with the ankle brachial index and leg symptoms. JAMA 2004, 292, 453–461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morris, D.R.; Rodriguez, A.J.; Moxon, J.V.; Cunningham, M.A.; McDermott, M.M.; Myers, J.; Leeper, N.J.; Jones, R.E.; Golledge, J. Association of lower extremity performance with cardiovascular and all-cause mortality in patients with peripheral artery disease: A systematic review and meta-analysis. J. Am. Heart Assoc. 2014, 3. [Google Scholar] [CrossRef] [Green Version]
- Gerhard-Herman, M.D.; Gornik, H.L.; Barrett, C.; Barshes, N.R.; Corriere, M.A.; Drachman, D.E.; Fleisher, L.A.; Fowkes, F.G.R.; Hamburg, N.M.; Kinlay, S.; et al. 2016 AHA/ACC Guideline on the Management of Patients With Lower Extremity Peripheral Artery Disease: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 2017, 135, e726–e779. [Google Scholar] [CrossRef] [PubMed]
- Treat-Jacobson, D.; McDermott Mary, M.; Bronas Ulf, G.; Campia, U.; Collins Tracie, C.; Criqui Michael, H.; Gardner Andrew, W.; Hiatt William, R.; Regensteiner Judith, G.; Rich, K.; et al. Optimal Exercise Programs for Patients With Peripheral Artery Disease: A Scientific Statement From the American Heart Association. Circulation 2019, 139, e10–e33. [Google Scholar] [CrossRef]
- Center for Medicare & Medicaid Services. Decision Memo for Supervised Exercise Therapy (SET) for Symptomatic Peripheral Artery Disease (PAD) (CAG-00449N). Available online: https://www.cms.gov/medicare-coverage-database/details/nca-tracking-sheet.aspx?NCAId=287 (accessed on 1 September 2020).
- Treat-Jacobson, D.; McDermott, M.M.; Beckman, J.A.; Burt, M.A.; Creager, M.A.; Ehrman, J.K.; Gardner, A.W.; Mays, R.J.; Regensteiner, J.G.; Salisbury, D.L.; et al. Implementation of Supervised Exercise Therapy for Patients With Symptomatic Peripheral Artery Disease: A Science Advisory From the American Heart Association. Circulation 2019, 140, e700–e710. [Google Scholar] [CrossRef]
- Bolton, C.E.; Bevan-Smith, E.F.; Blakey, J.D.; Crowe, P.; Elkin, S.L.; Garrod, R.; Greening, N.J.; Heslop, K.; Hull, J.H.; Man, W.D.C.; et al. British Thoracic Society guideline on pulmonary rehabilitation in adults: Accredited by NICE. Thorax 2013, 68, ii1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iepsen, U.W.; Jørgensen, K.J.; Ringbaek, T.; Hansen, H.; Skrubbeltrang, C.; Lange, P. A Systematic Review of Resistance Training Versus Endurance Training in COPD. J. Cardiopulm. Rehabil. Prev. 2015, 35, 163–172. [Google Scholar] [CrossRef]
- Abaraogu, U.; Ezenwankwo, E.; Dall, P.; Tew, G.; Stuart, W.; Brittenden, J.; Seenan, C. Barriers and enablers to walking in individuals with intermittent claudication: A systematic review to conceptualize a relevant and patient-centered program. PLoS ONE 2018, 13, e0201095. [Google Scholar] [CrossRef]
- McDermott, M.M.; Greenland, P.; Tian, L.; Kibbe, M.R.; Green, D.; Zhao, L.; Criqui, M.H.; Guralnik, J.M.; Ferrucci, L.; Liu, K.; et al. Association of 6-Minute Walk Performance and Physical Activity With Incident Ischemic Heart Disease Events and Stroke in Peripheral Artery Disease. J. Am. Heart Assoc. 2015, 4, e001846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parmenter, B.J.; Mavros, Y.; Ritti Dias, R.; King, S.; Fiatarone Singh, M. Resistance training as a treatment for older persons with peripheral artery disease: A systematic review and meta-analysis. Br. J. Sports Med. 2020, 54, 452. [Google Scholar] [CrossRef] [PubMed]
- Aboyans, V.; Criqui, M.H.; Abraham, P.; Allison, M.A.; Creager, M.A.; Diehm, C.; Fowkes, F.G.; Hiatt, W.R.; Jonsson, B.; Lacroix, P.; et al. Measurement and interpretation of the ankle-brachial index: A scientific statement from the American Heart Association. Circulation 2012, 126, 2890–2909. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Higgins, J.P.T.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [Green Version]
- Anderson, T.J.; Phillips, S.A. Assessment and Prognosis of Peripheral Artery Measures of Vascular Function. Prog. Cardiovasc. Dis. 2015, 57, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions version 6.2 (updated February 2021). 2021. Available online: https://training.cochrane.org/handbook (accessed on 20 February 2021).
- Jelicic Kadic, A.; Vucic, K.; Dosenovic, S.; Sapunar, D.; Puljak, L. Extracting data from figures with software was faster, with higher interrater reliability than manual extraction. J. Clin. Epidemiol. 2016, 74, 119–123. [Google Scholar] [CrossRef]
- Fu, R.; Vandermeer, B.W.; Shamliyan, T.A.; O’Neil, M.E.; Yazdi, F.; Fox, S.H.; Morton, S.C. Handling Continuous Outcomes in Quantitative Synthesis. In Methods Guide for Effectiveness and Comparative Effectiveness Reviews [Internet]; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2013. [Google Scholar]
- Gardner, A.W.; Parker, D.E.; Montgomery, P.S.; Blevins, S.M. Step-monitored home exercise improves ambulation, vascular function, and inflammation in symptomatic patients with peripheral artery disease: A randomized controlled trial. J. Am. Heart Assoc. 2014, 3, e001107. [Google Scholar] [CrossRef] [Green Version]
- Gardner Andrew, W.; Parker Donald, E.; Montgomery Polly, S.; Scott Kristy, J.; Blevins Steve, M. Efficacy of Quantified Home-Based Exercise and Supervised Exercise in Patients With Intermittent Claudication. Circulation 2011, 123, 491–498. [Google Scholar] [CrossRef] [Green Version]
- Gardner, A.W.; Montgomery, P.S.; Wang, M. Minimal clinically important differences in treadmill, 6-minute walk, and patient-based outcomes following supervised and home-based exercise in peripheral artery disease. Vasc. Med. 2018, 23, 349–357. [Google Scholar] [CrossRef] [Green Version]
- Gomes, A.P.F.; Correia, M.A.; Soares, A.H.G.; Cucato, G.G.; Lima, A.; Cavalcante, B.R.; Sobral-Filho, D.C.; Ritti-Dias, R.M. Effects of Resistance Training on Cardiovascular Function in Patients With Peripheral Artery Disease: A Randomized Controlled Trial. J. Strength Cond. Res. Natl. Strength Cond. Assoc. 2018, 32, 1072–1080. [Google Scholar] [CrossRef]
- Hiatt, W.R.; Wolfel, E.E.; Meier, R.H.; Regensteiner, J.G. Superiority of treadmill walking exercise versus strength training for patients with peripheral arterial disease. Implications for the mechanism of the training response. Circulation 1994, 90, 1866–1874. [Google Scholar] [CrossRef] [Green Version]
- Hiatt, W.R.; Regensteiner, J.G.; Wolfel, E.E.; Carry, M.R.; Brass, E.P. Effect of exercise training on skeletal muscle histology and metabolism in peripheral arterial disease. J. Appl. Physiol. 1996, 81, 780–788. [Google Scholar] [CrossRef]
- Regensteiner, J.G.; Steiner, J.F.; Hiatt, W.R. Exercise training improves functional status in patients with peripheral arterial disease. J. Vasc. Surg. 1996, 23, 104–115. [Google Scholar] [CrossRef] [Green Version]
- McDermott, M.M.; Ades, P.; Guralnik, J.M.; Dyer, A.; Ferrucci, L.; Liu, K.; Nelson, M.; Lloyd-Jones, D.; Van Horn, L.; Garside, D.; et al. Treadmill exercise and resistance training in patients with peripheral arterial disease with and without intermittent claudication: A randomized controlled trial. JAMA 2009, 301, 165–174. [Google Scholar] [CrossRef] [PubMed]
- McGuigan, M.R.; Bronks, R.; Newton, R.U.; Sharman, M.J.; Graham, J.C.; Cody, D.V.; Kraemer, W.J. Resistance training in patients with peripheral arterial disease: Effects on myosin isoforms, fiber type distribution, and capillary supply to skeletal muscle. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2001, 56, B302–B310. [Google Scholar] [CrossRef] [PubMed]
- Parmenter, B.J.; Raymond, J.; Dinnen, P.; Lusby, R.J.; Fiatarone Singh, M.A. High-Intensity Progressive Resistance Training Improves Flat-Ground Walking in Older Adults with Symptomatic Peripheral Arterial Disease. J. Am. Geriatr. Soc. 2013, 61, 1964–1970. [Google Scholar] [CrossRef]
- Parr, B.M.; Noakes, T.D.; Derman, E.W. Peripheral arterial disease and intermittent claudication: Efficacy of short-term upper body strength training, dynamic exercise training, and advice to exercise at home. South Afr. Med J. 2009, 99, 800–804. [Google Scholar]
- Ritti-Dias, R.M.; Wolosker, N.; de Moraes Forjaz, C.L.; Carvalho, C.R.F.; Cucato, G.G.; Leão, P.P.; de Fátima Nunes Marucci, M. Strength training increases walking tolerance in intermittent claudication patients: Randomized trial. J. Vasc. Surg. 2010, 51, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Menêses, A.L.; de Lima, G.H.; Forjaz, C.L.; Lima, A.H.; Silva, G.Q.; Cucato, G.G.; Rodrigues, S.L.; Wolosker, N.; Marucci Mde, F.; Dias, R.M. Impact of a supervised strength training or walking training over a subsequent unsupervised therapy period on walking capacity in patients with claudication. J. Vasc. Nurs. Off. Publ. Soc. Peripher. Vasc. Nurs. 2011, 29, 81–86. [Google Scholar] [CrossRef]
- Grizzo Cucato, G.; de Moraes Forjaz, C.L.; Kanegusuku, H.; da Rocha Chehuen, M.; Riani Costa, L.A.; Wolosker, N.; Kalil Filho, R.; de Fátima Nunes Marucci, M.; Mendes Ritti-Dias, R. Effects of walking and strength training on resting and exercise cardiovascular responses in patients with intermittent claudication. Vasa 2011, 40, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Szymczak, M.; Oszkinis, G.; Majchrzycki, M. The Impact of Walking Exercises and Resistance Training upon the Walking Distance in Patients with Chronic Lower Limb Ischaemia. BioMed Res. Int. 2016, 2016, 7515238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, P.; Rudan, D.; Zhu, Y.; Fowkes, F.J.I.; Rahimi, K.; Fowkes, F.G.R.; Rudan, I. Global, regional, and national prevalence and risk factors for peripheral artery disease in 2015: An updated systematic review and analysis. Lancet Glob. Health 2019, 7, e1020–e1030. [Google Scholar] [CrossRef] [Green Version]
- Fakhry, F.; van de Luijtgaarden, K.M.; Bax, L.; den Hoed, P.T.; Hunink, M.G.; Rouwet, E.V.; Spronk, S. Supervised walking therapy in patients with intermittent claudication. J. Vasc. Surg. 2012, 56, 1132–1142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Parmenter, B.J.; Dieberg, G.; Smart, N.A. Exercise training for management of peripheral arterial disease: A systematic review and meta-analysis. Sports Med. 2015, 45, 231–244. [Google Scholar] [CrossRef]
- Jansen, S.C.P.; Abaraogu, U.O.; Lauret, G.J.; Fakhry, F.; Fokkenrood, H.J.P.; Teijink, J.A.W. Modes of exercise training for intermittent claudication. Cochrane Database Syst. Rev. 2020. [Google Scholar] [CrossRef]
- Golledge, J.; Singh, T.P.; Alahakoon, C.; Pinchbeck, J.; Yip, L.; Moxon, J.V.; Morris, D.R. Meta-analysis of clinical trials examining the benefit of structured home exercise in patients with peripheral artery disease. Br. J. Surg. 2019, 106, 319–331. [Google Scholar] [CrossRef]
- Fokkenrood, H.J.; Bendermacher, B.L.; Lauret, G.J.; Willigendael, E.M.; Prins, M.H.; Teijink, J.A. Supervised exercise therapy versus non-supervised exercise therapy for intermittent claudication. Cochrane Database Syst. Rev. 2013, CD005263. [Google Scholar] [CrossRef]
- McDermott, M.M.; Guralnik, J.M.; Criqui, M.H.; Liu, K.; Kibbe, M.R.; Ferrucci, L. Six-minute walk is a better outcome measure than treadmill walking tests in therapeutic trials of patients with peripheral artery disease. Circulation 2014, 130, 61–68. [Google Scholar] [CrossRef] [Green Version]
- Hiatt William, R.; Rogers, R.K.; Brass Eric, P. The Treadmill Is a Better Functional Test Than the 6-Minute Walk Test in Therapeutic Trials of Patients With Peripheral Artery Disease. Circulation 2014, 130, 69–78. [Google Scholar] [CrossRef] [Green Version]
- Egger, M.; Smith, G.D.; Altman, D.G. Systematic Reviews in Health Care Meta-Analysis in Context, 2nd ed.; BMJ: London, UK, 2001. [Google Scholar]
- Takeshima, N.; Sozu, T.; Tajika, A.; Ogawa, Y.; Hayasaka, Y.; Furukawa, T.A. Which is more generalizable, powerful and interpretable in meta-analyses, mean difference or standardized mean difference? BMC Med Res. Methodol. 2014, 14, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hiatt, W.R.; Armstrong, E.J.; Larson, C.J.; Brass, E.P. Pathogenesis of the limb manifestations and exercise limitations in peripheral artery disease. Circ. Res. 2015, 116, 1527–1539. [Google Scholar] [CrossRef] [Green Version]
- Rontoyanni, V.G.; Nunez Lopez, O.; Fankhauser, G.T.; Cheema, Z.F.; Rasmussen, B.B.; Porter, C. Mitochondrial Bioenergetics in the Metabolic Myopathy Accompanying Peripheral Artery Disease. Front. Physiol. 2017, 8, 141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baker, W.B.; Li, Z.; Schenkel, S.S.; Chandra, M.; Busch, D.R.; Englund, E.K.; Schmitz, K.H.; Yodh, A.G.; Floyd, T.F.; Mohler, E.R., 3rd. Effects of exercise training on calf muscle oxygen extraction and blood flow in patients with peripheral artery disease. J. Appl. Physiol. 2017, 123, 1599–1609. [Google Scholar] [CrossRef]
- Maruhashi, T.; Kajikawa, M.; Kishimoto, S.; Hashimoto, H.; Takaeko, Y.; Yamaji, T.; Harada, T.; Han, Y.; Aibara, Y.; Mohamad Yusoff, F.; et al. Diagnostic Criteria of Flow-Mediated Vasodilation for Normal Endothelial Function and Nitroglycerin-Induced Vasodilation for Normal Vascular Smooth Muscle Function of the Brachial Artery. J. Am. Heart Assoc. 2020, 9, e013915. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; L. Erlbaum Associates: Hillsdale, MI, USA, 1988. [Google Scholar]
- Jones, P.P.; Skinner, J.S.; Smith, L.K.; John, F.M.; Bryant, C.X. Functional Improvements Following StairMaster vs. Treadmill Exercise Training for Patients With Intermittent Claudication. J. Cardiopulm. Rehabil. Prev. 1996, 16, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Kent, D.M.; Paulus, J.K.; van Klaveren, D.; D’Agostino, R.; Goodman, S.; Hayward, R.; Ioannidis, J.P.A.; Patrick-Lake, B.; Morton, S.; Pencina, M.; et al. The Predictive Approaches to Treatment effect Heterogeneity (PATH) Statement. Ann. Intern. Med. 2020, 172, 35–45. [Google Scholar] [CrossRef] [Green Version]
- Manfredini, F.; Malagoni, A.M.; Mandini, S.; Felisatti, M.; Mascoli, F.; Basaglia, N.; Manfredini, R.; Mikhailidis, D.P.; Zamboni, P. Near-Infrared Spectroscopy Assessment Following Exercise Training in Patients With Intermittent Claudication and in Untrained Healthy Participants. Vasc. Endovasc. Surg. 2012, 46, 315–324. [Google Scholar] [CrossRef]
- Manfredini, F.; Lamberti, N.; Rossi, T.; Mascoli, F.; Basaglia, N.; Zamboni, P. A Toe Flexion NIRS assisted Test for Rapid Assessment of Foot Perfusion in Peripheral Arterial Disease: Feasibility, Validity, and Diagnostic Accuracy. Eur. J. Vasc. Endovasc. Surg. 2017, 54, 187–194. [Google Scholar] [CrossRef]
- Yost, M.L. Cost-benefit analysis of critical limb ischemia in the era of the Affordable Care Act. Endovasc. Today 2014, 5, 29–36. [Google Scholar]
- Mahoney, E.M.; Wang, K.; Cohen, D.J.; Hirsch, A.T.; Alberts, M.J.; Eagle, K.; Mosse, F.; Jackson, J.D.; Steg, P.G.; Bhatt, D.L.; et al. One-year costs in patients with a history of or at risk for atherothrombosis in the United States. Circ. Cardiovasc. Qual. Outcomes 2008, 1, 38–45. [Google Scholar] [CrossRef] [PubMed] [Green Version]


| RCT | Group (n) | Mean Age (Years) | MeanABI | Supervised RT Group | Comparison Group | Session (Min) | RT Intensity | Set × Rep × Ex (n) | Sessions per Wk | Program Duration (wk) | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Supervised RT vs. Usual Medical Care Control | |||||||||||
| Gomes 2018 (Brazil) [23] | RT (15) Control (15) | 61 66 | 0.73 0.70 | Whole body machine-based RT | Stretching and relaxation exercises | 40 | Moderate | 3 × 10 × 8 | 2 | 12 | |
| Hiatt 1994 (US) [24,25,26] | RT (9) Control (10) | 67 67 | 0.52 0.61 | Lower limb isotonic free-weight RT | Usual medical care | 60 | Moderate-High | 3 × 6 × 5/leg | 3 | 12 | |
| McDermott 2009 (US) [27] | RT (52) Control (51) | 72 69 | 0.62 0.60 | Lower limb machine-based/BW RT | Attention control | 40 | Moderate-High | 3 × 8 × 5 | 3 | 24 | |
| McGuigan 2001 (US) [28] | RT (11) Control (9) | 70 69 | 0.61 0.67 | Whole body machine-based/free-weights/BW RT | Usual medical care | NR | Moderate-High | 2 × 8–15 × 8 | 3 | 24 | |
| Parmenter 2013 (AU) [29] | RT (8) Control (7) | 79 71 | 0.53 0.55 | Whole body machine-based RT | Usual medical care | NR | High | 3 × 8 × 8 | 3 | 24 | |
| Parr 2009 (South Africa) [30] | RT (9) Control (8) | 66 62 | NR NR | Upper body machine-based/ free-weights RT | Usual medical care | 45 | Moderate | 1 × 15–30 × 14 | 3 | 6 | |
| Supervised RT vs. Supervised Aerobic Training | |||||||||||
| Gardner 2014 (US) [20,21,22] | RT (60) SupAer (60) | 65 65 | 0.74 0.68 | Whole body machine-based RT | Treadmill walking | NR 15–45 min | Light | 1 × 15 × 9 | 3 | 12 | |
| Hiatt 1994 (US) [24,25,26] | RT (9) SupAer (10) | 67 67 | 0.52 0.55 | Lower limb isotonic free-weights RT | Treadmill walking | 60 | Moderate-High | 3 × 6 × 5/leg | 3 | 12 | |
| McDermott 2009 (US) [27] | RT (52) SupAer (53) | 72 72 | 0.62 0.60 | Lower limb machine-based RT | Treadmill walking | 40 | Moderate-High | 3 × 8 × 5 | 3 | 24 | |
| Ritti-Dias 2010 (Brazil) [31,32,33] | RT (15) SupAer (15) | 66 65 | 0.63 0.66 | Whole body machine-based RT | Treadmill walking | 68 | Moderate | 3 × 10 × 8 | 2 | 12 | |
| Szymczak 2016 (Poland) [34] | RT (26) SupAer (24) | NR NR | 0.70 0.67 | Lower limb machine-based RT | Treadmill walking | 50 | Light-Moderate | 3 × 15 × 6 | 2 | 12 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blears, E.E.; Elias, J.K.; Tapking, C.; Porter, C.; Rontoyanni, V.G. Supervised Resistance Training on Functional Capacity, Muscle Strength and Vascular Function in Peripheral Artery Disease: An Updated Systematic Review and Meta-Analysis. J. Clin. Med. 2021, 10, 2193. https://doi.org/10.3390/jcm10102193
Blears EE, Elias JK, Tapking C, Porter C, Rontoyanni VG. Supervised Resistance Training on Functional Capacity, Muscle Strength and Vascular Function in Peripheral Artery Disease: An Updated Systematic Review and Meta-Analysis. Journal of Clinical Medicine. 2021; 10(10):2193. https://doi.org/10.3390/jcm10102193
Chicago/Turabian StyleBlears, Elizabeth E., Jessica K. Elias, Christian Tapking, Craig Porter, and Victoria G. Rontoyanni. 2021. "Supervised Resistance Training on Functional Capacity, Muscle Strength and Vascular Function in Peripheral Artery Disease: An Updated Systematic Review and Meta-Analysis" Journal of Clinical Medicine 10, no. 10: 2193. https://doi.org/10.3390/jcm10102193
APA StyleBlears, E. E., Elias, J. K., Tapking, C., Porter, C., & Rontoyanni, V. G. (2021). Supervised Resistance Training on Functional Capacity, Muscle Strength and Vascular Function in Peripheral Artery Disease: An Updated Systematic Review and Meta-Analysis. Journal of Clinical Medicine, 10(10), 2193. https://doi.org/10.3390/jcm10102193







