Influence of the Human Development Index on the Maternal–Perinatal Morbidity and Mortality of Pregnant Women with SARS-CoV-2 Infection: Importance for Personalized Medical Care
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
- Mild—clinical symptoms mild and no sign of pneumonia on imaging.
- Moderate—fever and respiratory symptoms plus radiological findings of pneumonia.
- Severe—any of the following conditions:
- Respiratory distress (respiratory rate of ≥30 per min).
- Oxygen saturation in room air at rest ≤93%.
- Partial pressure of oxygen in arterial blood/fraction of inspired oxygen ≤300 mmHg.
- Critical cases—any of the following conditions:
- Respiratory failure and need for mechanical ventilation.
- Shock.
- Patients with failure of an organ requiring ICU care.
2.2. Data Analysis
2.3. Ethical Approval
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease (COVID-19) Dashboard. 2021. Available online: https://covid19.who.int (accessed on 22 February 2021).
- Jamieson, D.J.; Theiler, R.; Rasmussen, S. Emerging Infections and Pregnancy. Emerg. Infect. Dis. 2006, 12, 1638–1643. [Google Scholar] [CrossRef] [PubMed]
- Dashraath, P.; Wong, J.L.J.; Lim, M.X.K.; Lim, L.M.; Li, S.; Biswas, A.; Choolani, M.; Mattar, C.; Su, L.L. Coronavirus disease 2019 (COVID-19) pandemic and preg-nancy. Am. J. Obstet. Gynecol. 2020, 222, 521–531. [Google Scholar] [CrossRef]
- Wang, C.L.; Liu, Y.Y.; Wu, C.H.; Wang, C.Y.; Wang, C.H.; Long, C.Y. Impact of COVID-19 on Pregnancy. Int. J. Med. Sci. 2021, 18, 763–767. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Lemini, M.; Ferriols Perez, E.; de la Cruz Conty, M.L.; Caño Aguilar, A.; Encinas Pardilla, M.B.; Prats Rodríguez, P.; Muner Hernando, M.; Forcen Acebal, L.; Pintado Recarte, P.; Medina Mallen, M.D.C.; et al. Obstetric Outcomes of SARS-CoV-2 Infection in Asymptomatic Pregnant Women. Viruses 2021, 13, 112. [Google Scholar] [CrossRef]
- Cuñarro-López, Y.; Cano-Valderrama, Ó.; Pintado-Recarte, P.; Cueto-Hernández, I.; González-Garzón, B.; García-Tizón, S.; Bujan, J.; Asúnsolo, Á.; Ortega, M.A.; De León-Luis, J.A. Maternal and Perinatal Outcomes in Patients with Suspected COVID-19 and Their Relationship with a Negative RT-PCR Result. J. Clin. Med. 2020, 9, 3552. [Google Scholar] [CrossRef]
- Di Mascio, D. Maternal and Perinatal Outcomes of Pregnant Women with SARS-COV-2 infection. Ultrasound. Obstet. Gynecol. 2021, 57, 232–241. [Google Scholar] [CrossRef]
- Di Mascio, D.; Sen, C.; Saccone, G.; Galindo, A.; Grünebaum, A.; Yoshimatsu, J.; Stanojevic, M.; Kurjak, A.; Chervenak, F.; Suárez, M.J.R.; et al. Risk factors associated with adverse fetal outcomes in pregnancies affected by Coronavirus disease 2019 (COVID-19): A secondary analysis of the WAPM study on COVID-19. J. Perinat. Med. 2020, 48, 950–958. [Google Scholar] [CrossRef]
- Sentilhes, L.; De Marcillac, F.; Jouffrieau, C.; Kuhn, P.; Thuet, V.; Hansmann, Y.; Ruch, Y.; Fafi-Kremer, S.; Deruelle, P. Coronavirus disease 2019 in pregnancy was associated with maternal morbidity and preterm birth. Am. J. Obstet. Gynecol. 2020, 223, 914.e1–914.e15. [Google Scholar] [CrossRef] [PubMed]
- Antoun, L.; El Taweel, N.; Ahmed, I.; Patni, S.; Honest, H. Maternal COVID-19 infection, clinical characteristics, pregnancy, and neonatal outcome: A prospective cohort study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 252, 559–562. [Google Scholar] [CrossRef]
- Coronado-Arroyo, J.C.; Concepción-Zavaleta, M.J.; Zavaleta-Gutiérrez, F.E.; Concepción-Urteaga, L.A. Is COVID-19 a risk factor for severe preeclampsia? Hospital experience in a developing country. Eur. J. Obstet. Gynecol. Reprod. Biol. 2021, 256, 502–503. [Google Scholar] [CrossRef] [PubMed]
- Cuñarro-López, Y.; Pintado-Recarte, P.; Cueto-Hernández, I.; Hernández-Martín, C.; Payá-Martínez, M.P.; Muñóz-Chápuli, M.D.M.; Cano-Valderrama, Ó.; Bravo, C.; Bujan, J.; Álvarez-Mon, M.; et al. The Profile of the Obstetric Patients with SARS-CoV-2 Infection According to Country of Origin of the Publication: A Systematic Review of the Literature. J. Clin. Med. 2021, 10, 360. [Google Scholar] [CrossRef] [PubMed]
- United Nations Development Programme. Human Development Report. 2019. Available online: http://hdr.undp.org/sites/default/files/hdr2019.pdf (accessed on 3 July 2020).
- Shahbazi, F.; Khazaei, S. Socio-economic inequality in global incidence and mortality rates from coronavirus disease 2019: An ecological study. New Microbes New Infect. 2020, 38, 100762. [Google Scholar] [CrossRef]
- Liu, K.; He, M.; Zhuang, Z.; He, D.; Li, H. Unexpected positive correlation between human development index and risk of infections and deaths of COVID-19 in Italy. One Health 2020, 10, 100174. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Int. J. Surg. 2014, 12, 1495–1499. [Google Scholar] [CrossRef] [Green Version]
- Encinas Pardilla, M.B.; Cano Aguilar, A.; Marcos Puig, B.; Sanz Lorenzana, A.; de la Torre, I.; de la Manzanara, P.; Fernandez Bernardo, A.; Martinez Perez, O. Spanish registry of Covid-19 screening in asymptomatic pregnant women. Rev. Esp. Salud Publica 2020, 94. [Google Scholar]
- Larroca, S.G.T.; Valera, F.A.; Herrera, E.A.; Hernandez, I.C.; Lopez, Y.C.; De Leon-Luis, J. Human Development Index of the maternal country of origin and its relationship with maternal near miss: A systematic review of the literature. BMC Pregnancy Childbirth 2020, 20, 224. [Google Scholar]
- United Nations Development Programme (UNDP). Human Developmet Report 2020. The Next Frontier. Human Development and the Anthropocene. New York: United Nations Development Programme (UNDP). 2020. Available online: http://hdr.undp.org/sites/default/files/hdr2020.pdf (accessed on 22 February 2021).
- Say, L.; Souza, J.P.; Pattinson, R.C. Maternal near miss–towards a standard tool for monitoring quality of maternal health care. Best Pr. Res. Clin. Obstet. Gynaecol. 2009, 23, 287–296. [Google Scholar] [CrossRef]
- Pileggi-Castro, C.; Camelo, J.C., Jr.; Perdoná, G.; Mussi-Pinhata, M.; Cecatti, J.G.; Mori, R.; Morisaki, N.; Yunis, K.; Vogel, J.; Tunçalp, Ö.; et al. Development of criteria for identifying neonatal near-miss cases: Analysis of two WHO multicountry cross-sectional studies. Int. J. Obstet. Gynaecol. 2014, 121, 110–118. [Google Scholar] [CrossRef]
- Peng, F.; Tu, L.; Yang, Y.; Hu, P.; Wang, R.; Hu, Q.; Cao, F.; Jiang, T.; Sun, J.; Xu, G.; et al. Management and Treatment of COVID-19: The Chinese Experience. Can. J. Cardiol. 2020, 36, 915–930. [Google Scholar] [CrossRef]
- Hussain, A.; Mahawar, K.; Xia, Z.; Yang, W.; El-Hasani, S. RETRACTED: Obesity and mortality of COVID-19. Meta-analysis. Obes. Res. Clin. Pract. 2020, 14, 295–300. [Google Scholar] [CrossRef]
- Hessami, K.; Homayoon, N.; Hashemi, A.; Vafaei, H.; Kasraeian, M.; Asadi, N. COVID-19 and maternal, fetal and neonatal mortality: A systematic review. J. Matern. Neonatal Med. 2020, 1–6, 1–6. [Google Scholar] [CrossRef]
- Kayem, G.; Lecarpentier, E.; Deruelle, P.; Bretelle, F.; Azria, E.; Blanc, J.; Bohec, C.; Bornes, M.; Ceccaldi, P.-F.; Chalet, Y.; et al. A snapshot of the Covid-19 pandemic among pregnant women in France. J. Gynecol. Obstet. Hum. Reprod. 2020, 49, 101826. [Google Scholar] [CrossRef]
- Knight, M.; Bunch, K.; Vousden, N.; Morris, E.; Simpson, N.; Gale, C.; O’Brien, P.; Quigley, M.; Brocklehurst, P.; Kurinczuk, J.J. Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: National population based cohort study. BMJ 2020, 369, m2107. [Google Scholar] [CrossRef]
- Narang, K.; Enninga, E.A.L.; Gunaratne, M.D.; Ibirogba, E.R.; Trad, A.T.A.; Elrefaei, A.; Theiler, R.N.; Ruano, R.; Szymanski, L.M.; Chakraborty, R.; et al. SARS-CoV-2 Infection and COVID-19 During Pregnancy: A Multidisciplinary Review. Mayo Clin. Proc. 2020, 95, 1750–1765. [Google Scholar] [CrossRef]
- Khalil, A.; Kalafat, E.; Benlioglu, C.; O’Brien, P.; Morris, E.; Draycott, T.; Thangaratinam, S.; Le Doare, K.; Heath, P.; Ladhani, S. SARS-CoV-2 infection in pregnancy: A systematic review and meta-analysis of clinical features and pregnancy outcomes. EClinicalMedicine 2020, 25, 100446. [Google Scholar] [CrossRef]
- Ministry of Health. Federal Government. 2021. Available online: https://www.saude.gov.br/images/pdf/2020/May/29/2020-05-25—BEE17—Boletim-do-COE.pdf (accessed on 22 February 2021). (In Portuguese)
- Atanasova, V.B.; Arevalo-Serrano, J.; Alvarado, E.A.; Larroca, S.G.T. Maternal mortality in Spain and its association with country of origin: Cross-sectional study during the period 1999–2015. BMC Public Health 2018, 18, 1171. [Google Scholar]
- National Statistics Institute. Perinatal Mortality Rate by Sex. 2019. Available online: https://www.ine.es/jaxiT3/Tabla.htm?t=1698&L=1 (accessed on 22 February 2021).
- Allotey, J.; Stallings, E.; Bonet, M.; Yap, M.; Chatterjee, S.; Kew, T.; Debenham, L.; Llavall, A.C.; Dixit, A.; Zhou, D.; et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: Living systematic review and meta-analysis. BMJ 2020, 370, m3320. [Google Scholar] [CrossRef]
- Cuñarro-López, Y.; Pintado-Recarte, P.; Hernández-Martín, C.; Paya-Martínez, P.; López-Pérez, R.; Cueto-Hernández, I.; Ruiz-Labarta, J.; Cano-Valderrama, Ó.; Martínez-Pérez, Ó.; Bravo-Arribas, C.; et al. Comparing Infection Profiles of Expectant Mothers with COVID-19 and Impacts on Maternal and Perinatal Outcomes between the First Two Waves of the Pandemic. J. Pers. Med. 2021, 11, 599. [Google Scholar] [CrossRef]
- Crozier, T.M. General Care of the Pregnant Patient in the Intensive Care Unit. Semin. Respir. Crit. Care Med. 2017, 38, 208–217. [Google Scholar] [CrossRef]
- Larroca, S.G.T.; Arevalo-Serrano, J.; Vila, A.D.; Recarte, M.P.; Hernandez, I.C.; Pierna, A.S.; Bonelli, S.L.; De Leon-Luis, J. Human Development Index (HDI) of the maternal country of origin as a predictor of perinatal outcomes—A longitudinal study conducted in Spain. BMC Pregnancy Childbirth 2017, 17, 314. [Google Scholar]
- World Health Organization. Comisión Sobre Determinantes Sociales de la Salud: Informe de la Secretaría. 2009. Available online: https://apps.who.int/iris/bitstream/handle/10665/4148/A62_9-sp.pdf (accessed on 30 July 2021).
- World Health Organization. Trends in Maternal Mortality: 1990 to 2008. 2010. Available online: https://apps.who.int/iris/handle/10665/44423 (accessed on 30 July 2021).
- Urquia, M.L.; Glazier, R.H.; Mortensen, L.; Andersen, A.-M.N.; Small, R.; Davey, M.-A.; Rööst, M.; Essén, B.; for the ROAM (Reproductive Outcomes and Migration. An International Collaboration). Severe maternal morbidity associated with maternal birthplace in three high-immigration settings. Eur. J. Public Health 2015, 25, 620–625. [Google Scholar] [CrossRef] [Green Version]
- Ikram, U.Z.; MacKenbach, J.P.; Harding, S.; Rey, G.; Bhopal, R.S.; Regidor, E.; Rosato, M.; Juel, K.; Stronks, K.; Kunst, A.E. All-cause and cause-specific mortality of different migrant populations in Europe. Eur. J. Epidemiol. 2015, 31, 655–665. [Google Scholar] [CrossRef] [Green Version]
- Hantoushzadeh, S.; Shamshirsaz, A.A.; Aleyasin, A.; Seferovic, M.D.; Aski, S.K.; Arian, S.E.; Pooransari, P.; Ghotbizadeh, F.; Aalipour, S.; Soleimani, Z.; et al. Maternal death due to COVID-19. Am. J. Obstet. Gynecol. 2020, 223, 109.e1–109.e16. [Google Scholar] [CrossRef] [PubMed]

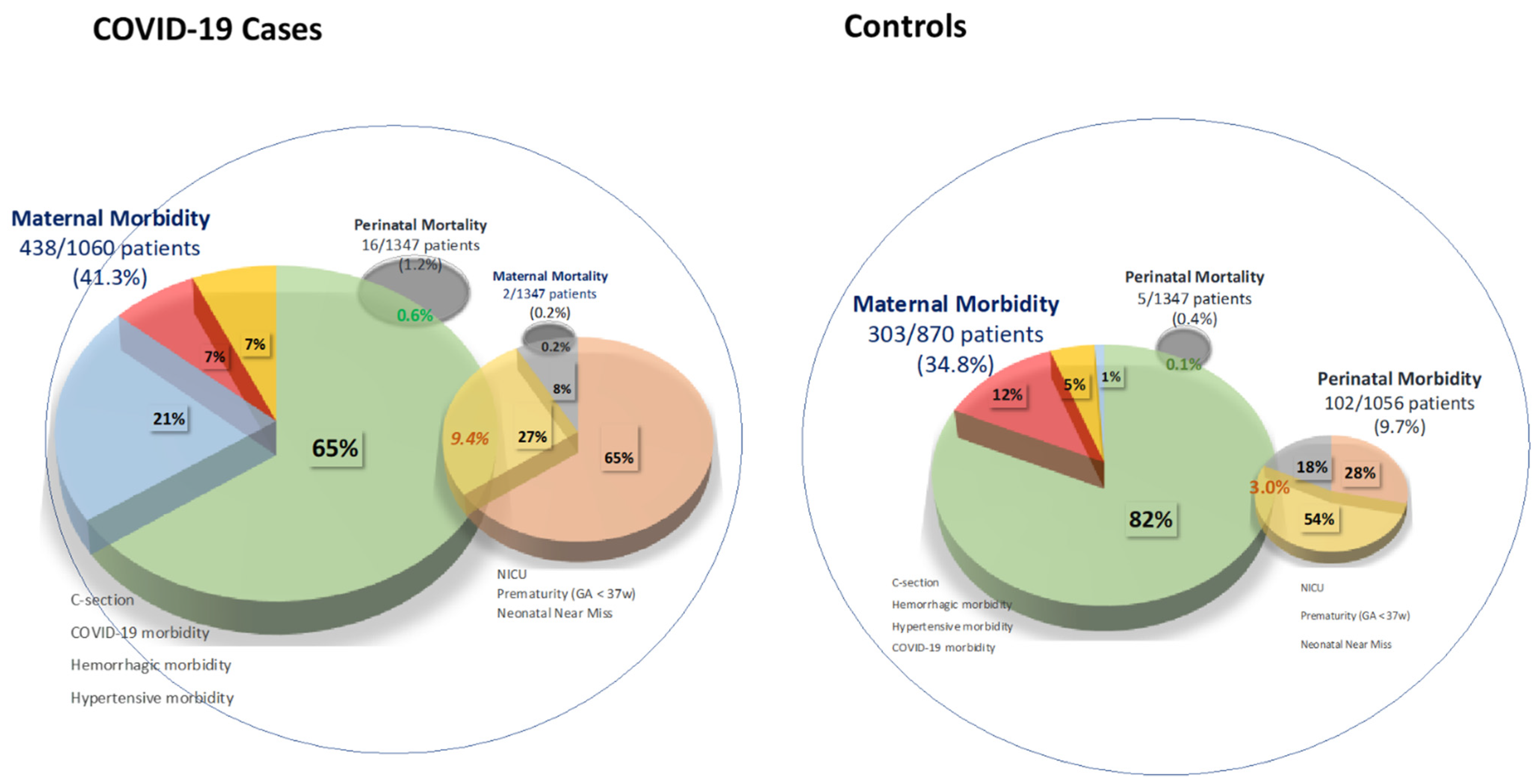

| Number (%) of Patients Reporting Results | Overall | Cases | Controls | p Value | |
|---|---|---|---|---|---|
| n, % | 2694 (100) | 2694 (100) | 1347 (50) | 1347 (50) | |
| Maternal characteristics | |||||
| Maternal age, mean, CI 95% | 2664 (98.9) | 32.6 (32.4–32.9) | 32.7 (32.3–33.0) | 32.6 (32.3–32.9) | 0.734 |
| Tobacco, n, % | 2554 (94.8) | 293 (11.5) | 131 (10.2) | 162 (12.8) | 0.035 |
| BMI, mean, CI 95% | 2314 (85.9) | 26.5 (26.3–26.8) | 26.6 (26.3–26.9) | 26.5 (26.2–26.8) | 0.532 |
| Maternal morbidities, n, % | 2432 (90.3) | 893 (36.7) | 483 (39.2) | 410 (34.1) | 0.009 |
| Nuliparous, n, % | 2670 (99.1) | 1059 (39.7) | 516 (48.7) | 543 (51.3) | 0.315 |
| Symptomps at triage, n, % | 1457 (54.1) | 644 (44.2) | 632 (55.0) | 12 (3.9) | <0.001 |
| Pneumonia, n, % | 1346 (50.0) | 124 (9.2) | 124 (12.2) | 0 (0.0) | <0.001 |
| Obstetric and perinatal characteristics | |||||
| Obstetric morbidities, n, % | 2246 (83.4) | 879 (39.1) | 468 (42.5) | 411 (35.9) | 0.001 |
| GA at triage, mean, CI 95% | 2691 (99.9) | 36.9 (36.7–37.1) | 34.9 (34.5–35.3) | 38.9 (38.8–39.0) | <0.001 |
| GA at delivery, mean, CI 95 % | 2694 (100) | 38.8 (38.7–38.9) | 38.5 (38.4–38.7) | 39.0 (38.9–39.1) | <0.001 |
| Birthweight, mean, CI 95% | 2659 (98.7) | 3219.5 (3198.2–3240.8) | 3181.2 (3148.6–3213.7) | 3257.7 (3230.3–3285.1) | <0.001 |
| Maternal and perinatal mortality | |||||
| Maternal mortality, n, % | 2694 (100) | 2 (0.1) | 2 (0.1) | 0 (0) | 0.096 |
| Perinatal mortality, n, % | 2694 (100) | 21 (0.8) | 16 (1.2) | 5 (0.4) | <0.001 |
| Overall Maternal Morbidity | 1930 (71.6) | 741 (38.4) | 438 (41.3) | 303 (34.8) | 0.003 |
| COVID-19 Maternal Morbidity | 2694 (100) | 96 (3.6) | 93 (6.9) | 3 (0.2) | <0.001 |
| Oxygen therapy, n, % | 2694 (100) | 74 (2.8) | 73 (5.4) | 1 (0.1) | <0.001 |
| Mechanical ventilation, n, % | 2694 (100) | 17 (0.6) | 17 (1.3) | 0 (0) | <0.001 |
| Admission to ICU, n, % | 2694 (100) | 38 (1.4) | 36 (2.7) | 2 (0.2) | <0.001 |
| Not COVID-19 Maternal Morbidity | 1925 (71.5) | 707 (36.7) | 405 (38.4) | 302 (34.7) | 0.096 |
| C-section, n, % | 2687 (99.7) | 640 (23.8) | 373 (27.8) | 267 (19.9) | <0.001 |
| Haemorrhagic maternal disorders, n, % | 1741 (64.6) | 81 (4.7) | 43 (4.4) | 38 (5.0) | 0.582 |
| Hypertensive maternal disorders, n, % | 2157 (80.1) | 58 (2.7) | 40 (3.6) | 18 (1.7) | 0.006 |
| Perinatal morbidity | 2165 (80.4) | 313 (14.5) | 211 (19.0) | 102 (9.7) | <0.001 |
| GA< 37 weeks, n, % | 2694 (100) | 230 (8.5) | 149 (11.1) | 81 (6.0) | <0.001 |
| Neonatal Near miss, n, % | 2642 (98.1) | 90 (3.4) | 65 (4.9) | 25 (1.9) | <0.001 |
| GA < 33 weeks, n, % | 2694 (100) | 68 (2.5) | 53 (3.9) | 15 (1.1) | <0.001 |
| Birthweight < 1750 grs, n, % | 2659 (98.7) | 47 (1.8) | 34 (2.6) | 13 (1.0) | 0.002 |
| Apgar 5 minutes < 7, n, % | 2664 (98.9) | 24 (0.9) | 12 (0.9) | 12 (0.9) | 0.988 |
| Admission to NICU, n, % | 2694 (100) | 166 (6.2) | 137 (10.2) | 29 (2.2) | <0.001 |
| Number (%) of Patients Reporting Results | High/Medium/Low HDI | Very High HDI | p Value | |
|---|---|---|---|---|
| n, % | 2263 (84.0) | 696 (30.8) | 1567 (69.2) | |
| Cases, n, % | 2263 (84.0) | 456 (65.5) | 678 (43.3) | <0.001 |
| Control cases, n, % | 2263 (84.0) | 240 (34.5) | 889 (56.7) | <0.001 |
| Maternal and perinatal mortality | ||||
| Maternal mortality, n, % | 1549 (57.5) | 1 (0.2) | 1 (0.1) | 0.565 |
| Perinatal mortality, n, % | 2263 (84.0) | 7 (1.0) | 10 (0.6) | 0.362 |
| Overall Maternal Morbidity | 1659 (61.6) | 211 (39.4) | 423 (37.6) | 0.480 |
| COVID-19 Maternal Morbidity | 2263 (84.0) | 42 (6.0) | 38 (2.4) | <0.001 |
| Oxygen therapy, n, % | 2263 (84.0) | 33 (4.7) | 28 (1.8) | <0.001 |
| Mechanical ventilation, n, % | 2263 (84.0) | 8 (1.2) | 9 (0.6) | 0.158 |
| Admission to ICU, n, % | 2263 (84.0) | 18 (2.6) | 16 (1.0) | 0.007 |
| Not COVID-19 Maternal Morbidity | 1656 (61.5) | 196 (36.7) | 411 (36.6) | 0.977 |
| C-section, n, % | 2258 (83.8) | 172 (24.8) | 377 (24.1) | 0.729 |
| Haemorrhagic maternal disorders, n, % | 1499 (55.6) | 27 (5.5) | 45 (4.5) | 0.364 |
| Hypertensive maternal disorders, n, % | 1841 (68.3) | 22 (3.8) | 30 (2.4) | 0.106 |
| Perinatal morbidity | 1801 (66.9) | 84 (14.6) | 179 (14.6) | 0.996 |
| GA< 37 weeks, n, % | 2263 (84.0) | 56 (8.1) | 143 (9.1) | 0.399 |
| Neonatal Near miss, n, % | 1768 (65.6) | 28 (4.9) | 45 (3.8) | 0.251 |
| GA < 33 weeks, n, % | 2263 (84.0) | 23 (3.3) | 38 (2.4) | 0.242 |
| Birthweight < 1750 grs, n, % | 2237 (83.0) | 16 (2.3) | 27 (1.8) | 0.369 |
| Apgar 5 minutes < 7, n, % | 2239 (83.1) | 5 (0.7) | 13 (0.8) | 0.786 |
| Admission to NICU, n, % | 2263 (84.0) | 52 (7.5) | 84 (5.4) | 0.056 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuñarro-López, Y.; Larroca, S.G.-T.; Pintado-Recarte, P.; Hernández-Martín, C.; Prats-Rodríguez, P.; Cano-Valderrama, Ó.; Cueto-Hernández, I.; Ruiz-Labarta, J.; Muñoz-Chápuli, M.d.M.; Martínez-Pérez, Ó.; et al. Influence of the Human Development Index on the Maternal–Perinatal Morbidity and Mortality of Pregnant Women with SARS-CoV-2 Infection: Importance for Personalized Medical Care. J. Clin. Med. 2021, 10, 3631. https://doi.org/10.3390/jcm10163631
Cuñarro-López Y, Larroca SG-T, Pintado-Recarte P, Hernández-Martín C, Prats-Rodríguez P, Cano-Valderrama Ó, Cueto-Hernández I, Ruiz-Labarta J, Muñoz-Chápuli MdM, Martínez-Pérez Ó, et al. Influence of the Human Development Index on the Maternal–Perinatal Morbidity and Mortality of Pregnant Women with SARS-CoV-2 Infection: Importance for Personalized Medical Care. Journal of Clinical Medicine. 2021; 10(16):3631. https://doi.org/10.3390/jcm10163631
Chicago/Turabian StyleCuñarro-López, Yolanda, Santiago García-Tizón Larroca, Pilar Pintado-Recarte, Concepción Hernández-Martín, Pilar Prats-Rodríguez, Óscar Cano-Valderrama, Ignacio Cueto-Hernández, Javier Ruiz-Labarta, María del Mar Muñoz-Chápuli, Óscar Martínez-Pérez, and et al. 2021. "Influence of the Human Development Index on the Maternal–Perinatal Morbidity and Mortality of Pregnant Women with SARS-CoV-2 Infection: Importance for Personalized Medical Care" Journal of Clinical Medicine 10, no. 16: 3631. https://doi.org/10.3390/jcm10163631
APA StyleCuñarro-López, Y., Larroca, S. G.-T., Pintado-Recarte, P., Hernández-Martín, C., Prats-Rodríguez, P., Cano-Valderrama, Ó., Cueto-Hernández, I., Ruiz-Labarta, J., Muñoz-Chápuli, M. d. M., Martínez-Pérez, Ó., Ortega, M. A., & De León-Luis, J. A. (2021). Influence of the Human Development Index on the Maternal–Perinatal Morbidity and Mortality of Pregnant Women with SARS-CoV-2 Infection: Importance for Personalized Medical Care. Journal of Clinical Medicine, 10(16), 3631. https://doi.org/10.3390/jcm10163631









