Current Status of Clinical and Genetic Screening of Hereditary Hemorrhagic Telangiectasia Families in Hungary
1. Introduction
2. Patients and Methods
2.1. Patient Recruitment
2.2. Clinical Evaluation
2.3. Mutation Analysis
2.4. Variant Assessment
2.5. The Algorithm of Cascade Family Screening
2.6. Statistical Analysis
3. Results
3.1. Demographical Data
3.2. Mutation Analysis
3.3. Genotype–Phenotype Correlations
3.4. Cascade Family Screening
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Borst, A.J.; Nakano, T.A.; Blei, F.; Adams, D.M.; Duis, J. A primer on a comprehensive genetic approach to vascular anomalies. Front. Pediatr. 2020, 8, 579591. [Google Scholar] [CrossRef]
- Sharathkumar, A.A.; Shapiro, A. Hereditary haemorrhagic telangiectasia. Haemophilia 2008, 14, 1269–1280. [Google Scholar] [CrossRef]
- ISSVA Classification for Vascular Anomalies. Available online: https://www.issva.org/UserFiles/file/ISSVA-Classification-2018.pdf (accessed on 5 June 2021).
- Bayrak-Toydemir, P.; McDonald, J.; Markewitz, B.; Lewin, S.; Miller, F.; Chou, L.; Gedge, F.; Tang, W.; Coon, H.; Mao, R. Genotype-phenotype correlation in hereditary hemorrhagic telangiectasia: Mutations and manifestations. Am. J. Med. Genet. 2006, 140, 463–470. [Google Scholar] [CrossRef]
- Shovlin, C.L.; Guttmacher, A.E.; Buscarini, E.; Faughnan, M.E.; Hyland, R.H.; Westermann, C.J.; Kjeldsen, A.D.; Plauchu, H. Diagnostic criteria for hereditary hemorrhagic telangiectasia (Rendu-Osler-Weber syndrome). Am. J. Med. Genet. 2000, 91, 66–67. [Google Scholar] [CrossRef]
- Govani, F.S.; Shovlin, C.L. Hereditary haemorrhagic telangiectasia: A clinical and scientific review. Eur. J. Hum. Genet. 2009, 17, 860–871. [Google Scholar] [CrossRef] [Green Version]
- Shovlin, C.L.; Hughes, J.M.; Tuddenham, E.G.; Temperley, I.; Perembelon, Y.F.; Scott, J.; Seidman, C.E.; Seidman, J.G. A gene for hereditary haemorrhagic telangiectasia maps to chromosome 9q3. Nat. Genet. 1994, 2, 205–209. [Google Scholar] [CrossRef]
- Johnson, D.W.; Berg, J.N.; Baldwin, M.A.; Gallione, C.J.; Marondel, I.; Yoon, S.J.; Stenzel, T.T.; Speer, M.; Pericak-Vance, M.A.; Diamond, A.; et al. Mutations in the activin receptor-like kinase 1 gene in hereditary haemorrhagic telangiectasia type 2. Nat. Genet. 1996, 13, 189–195. [Google Scholar] [CrossRef]
- Gallione, C.J.; Repetto, G.M.; Legius, E.; Rustgi, A.K.; Schelley, S.L.; Tejpar, S.; Mitchell, G.; Drouin, E.; Westermann, C.J.; Marchuk, D.A. A combined syndrome of juvenile polyposis and hereditary haemorrhagic telangiectasia associated with mutations in MADH4 (SMAD4). Lancet 2004, 9412, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Wooderchak-Donahue, W.L.; McDonald, J.; O’Fallon, B.; Upton, P.D.; Li, W.; Roman, B.L.; Young, S.; Plant, P.; Fulop, G.T.; Langa, C.; et al. BMP9 Mutations cause a vascular-anomaly syndrome with phenotypic overlap with hereditary hemorrhagic telangiectasia. Am. J. Hum. Genet. 2013, 93, 530–537. [Google Scholar] [CrossRef] [Green Version]
- McDonald, J.; Wooderchak-Donahue, W.; Van Sant Webb, C.; Whitehead, K.; Stevenson, D.A.; Bayrak-Toydemir, P. Hereditary hemorrhagic telangiectasia: Genetics and molecular diagnostics in a new era. Front. Genet. 2015, 6, 1–8. [Google Scholar] [CrossRef]
- HHT Mutation Database. Available online: https://arup.utah.edu/database/HHT/ (accessed on 5 June 2021).
- Tørring, P.M.; Brusgaard, K.; Ousager, L.B.; Andersen, P.E.; Kjeldsen, A.D. National mutation study among Danish patients with hereditary haemorrhagic telangiectasia. Clin. Genet. 2013, 86, 123–133. [Google Scholar] [CrossRef]
- Pierucci, P.; Lenato, G.M.; Suppressa, P.; Lastella, P.; Triggiani, V.; Valerio, R.; Comelli, M.; Salvante, D.; Stella, A.; Resta, N.; et al. A long diagnostic delay in patients with hereditary haemorrhagic telangiectasia: A questionnaire-based retrospective study. Orphanet J. Rare Dis. 2012, 7, 33. [Google Scholar] [CrossRef] [Green Version]
- Major, T.; Gindele, R.; Szabó, Z.; Kis, Z.; Bora, L.; Jóni, N.; Bárdossy, P.; Rácz, T.; Bereczky, Z. The stratified population screening of hereditary hemorrhagic telangiectasia. Pathol. Oncol. Res. 2020, 26, 2783–2788. [Google Scholar] [CrossRef]
- Major, T.; Gindele, R.; Szabó, Z.; Alef, T.; Thiele, B.; Bora, L.; Kis, Z.; Bárdossy, P.; Rácz, T.; Havacs, I.; et al. Evidence for the founder effect of a novel ACVRL1 splice-site mutation in Hungarian hereditary hemorrhagic telangiectasia families. Clin. Genet. 2016, 90, 466–467. [Google Scholar] [CrossRef]
- Gindele, R.; Kerényi, A.; Kállai, J.; Pfliegler, G.; Schlammadinger, Á.; Szegedi, I.; Major, T.; Szabó, Z.; Bagoly, Z.; Kiss, C.; et al. Resolving differential diagnostic problems in von willebrand disease, in fibrinogen disorders, in prekallikrein deficiency and in hereditary hemorrhagic telangiectasia by next-generation sequencing. Life 2021, 11, 202. [Google Scholar] [CrossRef]
- Széles, G.; Vokó, Z.; Jenei, T.; Kardos, L.; Pocsai, Z.; Bajtay, A.; Papp, E.; Pásti, G.; Kósa, Z.; Molnár, I.; et al. A preliminary evaluation of a health monitoring programme in Hungary. Eur. J. Public Health 2005, 15, 26–32. [Google Scholar] [CrossRef]
- The Human Gene Mutation Database. Available online: http://www.hgmd.cf.ac.uk/ac/index.php (accessed on 5 June 2021).
- Møller, P.; Clark, N.; Mæhle, L. A SImplified method for segregation analysis (SISA) to determine penetrance and expression of a genetic variant in a family. Hum. Mutat. 2011, 32, 568–571. [Google Scholar] [CrossRef]
- Franklin by Genoox. Available online: https://franklin.genoox.com/clinical-db/home (accessed on 14 June 2021).
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the american college of medical genetics and genomics and the association for molecular pathology. Genet. Med. 2015, 17, 405–423. [Google Scholar] [CrossRef] [PubMed]
- Major, T.; Gindele, R.; Szabó, Z.; Jóni, N.; Kis, Z.; Bora, L.; Bárdossy, P.; Rácz, T.; Karosi, T.; Bereczky, Z. A herediter haemorrhagiás teleangiectasia (Osler–Weber–Rendu-kór) genetikai diagnosztikája. Orv. Hetil. 2019, 160, 710–719. [Google Scholar] [CrossRef]
- Major, T.; Csobay-Novák, C.; Gindele, R.; Szabó, Z.; Bora, L.; Jóni, N.; Rácz, T.; Karosi, T.; Bereczky, Z. Pitfalls of delaying the diagnosis of hereditary haemorrhagic telangiectasia. J. Int. Med. Res. 2020, 48, 300060519860971. [Google Scholar] [CrossRef]
- Lesca, G.; Olivieri, C.; Burnichon, N.; Pagella, F.; Carette, M.F.; Gilbert-Dussardier, B.; Goizet, C.; Roume, J.; Rabilloud, M.; Saurin, J.C.; et al. French-Italian-Rendu-Osler Network. Genotype-phenotype correlations in hereditary hemorrhagic telangiectasia: Data from the French-Italian HHT network. Genet. Med. 2007, 9, 14–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westermann, C.J.J.; Rosina, A.F.; de Vries, V.; de Coteau, P.A. The prevalence and manifestations of hereditary hemorrhagic telangiectasia in the Afro-Caribbean population of the Netherlands Antilles: A family screening. Am. J. Med. Genet. 2003, 116, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, J.W.; McKeever, T.M.; Hall, I.P.; Hubbard, R.B.; Fogarty, A.W. The UK prevalence of hereditary haemorrhagic telangiectasia and its association with sex, socioeconomic status and region of residence: A population-based study. Thorax 2014, 69, 161–677. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grosse, S.D.; Boulet, S.L.; Grant, A.M.; Hulihan, M.M.; Faughnan, M.E. The use of US health insurance data for surveillance of rare disorders: Hereditary hemorrhagic telangiectasia. Genet. Med. 2014, 16, 33–39. [Google Scholar] [CrossRef] [Green Version]
- Bossler, A.D.; Richards, J.; George, C.; Godmilow, L.; Ganguly, A. Novel mutations in ENG and ACVRL1 identified in a series of 200 individuals undergoing clinical genetic testing for hereditary hemorrhagic telangiectasia (HHT): Correlation of genotype with phenotype. Hum. Mutat. 2006, 27, 667–675. [Google Scholar] [CrossRef]
- Heimdal, K.; Dalhus, B.; Rødningen, O.K.; Kroken, M.; Eiklid, K.; Dheyauldeen, S.; Røysland, T.; Andersen, R.; Kulseth, M.A. Mutation analysis in Norwegian families with hereditary hemorrhagic telangiectasia: Founder mutations inACVRL1. Clin. Genet. 2015, 89, 182–186. [Google Scholar] [CrossRef]
- Lesca, G.; Plauchu, H.; Coulet, F.; Lefebvre, S.; Plessis, G.; Odent, S.; Rivière, S.; Leheup, B.; Goizet, C.; Carette, M.-F.; et al. Molecular screening ofALK1/ACVRL1andENGgenes in hereditary hemorrhagic telangiectasia in France. Hum. Mutat. 2004, 23, 289–299. [Google Scholar] [CrossRef]
- Lesca, G.; Burnichon, N.; Raux, G.; Tosi, M.; Pinson, S.; Marion, M.J.; Babin, E.; Gilbert-Dussardier, B.; Rivière, S.; Goizet, C.; et al. French rendu-osler network. distribution of ENG and ACVRL1 (ALK1) mutations in French HHT patients. Hum. Mutat. 2006, 27, 598. [Google Scholar] [CrossRef]
- Sabbà, C.; Pasculli, G.; Lenato, G.M.; Suppressa, P.; Lastella, P.; Memeo, M.; Dicuonzo, F.; Guant, G. Hereditary hemorrhagic telangictasia: Clinical features in ENG and ALK1 mutation carriers. J. Thromb. Haemost. 2007, 6, 1149–1157. [Google Scholar] [CrossRef]
- Letteboer, T.G.W.; Zewald, R.A.; Kamping, E.J.; de Haas, G.; Mager, J.J.; Snijder, R.J.; Lindhout, D.; Hennekam, F.A.M.; Westermann, C.J.J.; Ploos van Amstel, J.K. Hereditary hemorrhagic telangiectasia: ENG and ALK-1 mutations in Dutch patients. Hum. Genet. 2004, 116, 8–16. [Google Scholar] [CrossRef]
- Letteboer, T.G.; Mager, J.J.; Snijder, R.J.; Koeleman, B.P.; Lindhout, D.; Ploos van Amstel, J.K.; Westermann, C.J. Genotype-phenotype relationship in hereditary haemorrhagic telangiectasia. J. Med. Genet. 2006, 43, 371–377. [Google Scholar] [CrossRef] [Green Version]
- Olivieri, C.; Pagella, F.; Semino, L.; Lanzarini, L.; Valacca, C.; Pilotto, A.; Corno, S.; Scappaticci, S.; Manfredi, G.; Buscarini, E.; et al. Analysis of ENG and ACVRL1 genes in 137 HHT Italian families identifies 76 different mutations (24 novel). Comparison with other European studies. J. Hum. Genet. 2007, 52, 820–829. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sánchez-Martínez, R.; Iriarte, A.; Mora-Luján, J.M.; Patier, J.L.; López-Wolf, D.; Ojeda, A.; Torralba, M.A.; Juyol, M.C.; Gil, R.; Añón, S.; et al. RiHHTa Investigators of the rare diseases working group from the spanish society of internal medicine. Current HHT genetic overview in Spain and its phenotypic correlation: Data from RiHHTa registry. Orphanet J. Rare Dis. 2020, 15, 138. [Google Scholar] [CrossRef] [PubMed]
- McDonald, J.; Bayrak-Toydemir, P.; DeMille, D.; Wooderchak-Donahue, W.; Whitehead, K. Curaçao diagnostic criteria for hereditary hemorrhagic telangiectasia is highly predictive of a pathogenic variant in ENG or ACVRL1 (HHT1 and HHT2). Genet. Med. 2020, 22, 1201–1205. [Google Scholar] [CrossRef] [PubMed]
- Major, T.; Gindele, R.; Balogh, G.; Bárdossy, P.; Bereczky, Z. Founder effects in hereditary hemorrhagic telangiectasia. J. Clin. Med. 2021, 10, 1682. [Google Scholar] [CrossRef]
- Lesca, G.; Genin, E.; Blachier, C.; Olivieri, C.; Coulet, F.; Brunet, G.; Dupuis-Girod, S.; Buscarini, E.; Soubrier, F.; Calender, A.; et al. Hereditary hemorrhagic telangiectasia: Evidence for regional founder effects of ACVRL1 mutations in French and Italian patients. Eur. J. Hum. Genet. 2008, 16, 742–749. [Google Scholar] [CrossRef] [Green Version]
- Plauchu, H.; De Chadarévian, J.-P.; Bideau, A.; Robert, J.-M. Age-related clinical profile of hereditary hemorrhagic telangiectasia in an epidemiologically recruited population. Am. J. Med. Genet. 1989, 32, 291–297. [Google Scholar] [CrossRef]
- Faughnan, M.E.; Palda, V.A.; Garcia-Tsao, G.; Geisthoff, U.W.; McDonald, J.; Proctor, D.D.; Spears, J.; Brown, D.H.; Buscarini, E.; Chesnutt, M.S.; et al. HHT foundation international—Guidelines working group. International guidelines for the diagnosis and management of hereditary haemorrhagic telangiectasia. J. Med. Genet. 2011, 48, 73–87. [Google Scholar] [CrossRef]
- Faughnan, M.E.; Mager, J.J.; Hetts, S.W.; Palda, V.A.; Lang-Robertson, K.; Buscarini, E.; Deslandres, E.; Kasthuri, R.S.; Lausman, A.; Poetker, D.; et al. Second International guidelines for the diagnosis and management of hereditary hemorrhagic telangiectasia. Ann. Intern. Med. 2020, 15, 989–1001. [Google Scholar] [CrossRef]
- Kritharis, A.; Al-Samkari, H.; Kuter, D.J. Hereditary hemorrhagic telangiectasia: Diagnosis and management from the hematologist’s perspective. Haematologica 2018, 103, 1433–1443. [Google Scholar] [CrossRef]
- Shovlin, C.L.; Buscarini, E.; Kjeldsen, A.D.; Mager, H.J.; Sabbà, C.; Droege, F.; Geisthoff, U.; Ugolini, S.; Dupuis-Girod, S. European reference network for rare vascular diseases (VASCERN) outcome measures for hereditary haemorrhagic telangiectasia (HHT). Orphanet J. Rare Dis. 2018, 13, 136. [Google Scholar] [CrossRef] [PubMed]
- Ingrosso, M.; Sabbà, C.; Pisani, A.; Principi, M.; Gallitelli, M.; Cirulli, A.; Francavilla, A. Evidence of small-bowel involvement in hereditary hemorrhagic telangiectasia: A capsule-endoscopic study. Endoscopy 2004, 36, 1074–1079. [Google Scholar] [CrossRef]
- Canzonieri, C.; Centenara, L.; Ornati, F.; Pagella, F.; Matti, E.; Alvisi, C.; Danesino, C.; Perego, M.; Olivieri, C. Endoscopic evaluation of gastrointestinal tract in patients with hereditary hemorrhagic telangiectasia and correlation with their genotypes. Genet. Med. 2014, 16, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Pahl, K.S.; Choudhury, A.; Wusik, K.; Hammill, A.; White, A.; Henderson, K.; Pollak, J.; Kasthuri, R.S. Applicability of the curaçao criteria for the diagnosis of hereditary hemorrhagic telangiectasia in the pediatric population. J. Pediatr. 2018, 197, 207–213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kroon, S.; Snijder, R.J.; Faughnan, M.E.; Mager, H.J. Systematic screening in hereditary hemorrhagic telangiectasia: A review. Curr. Opin. Pulm. Med. 2018, 24, 260–268. [Google Scholar] [CrossRef] [PubMed]

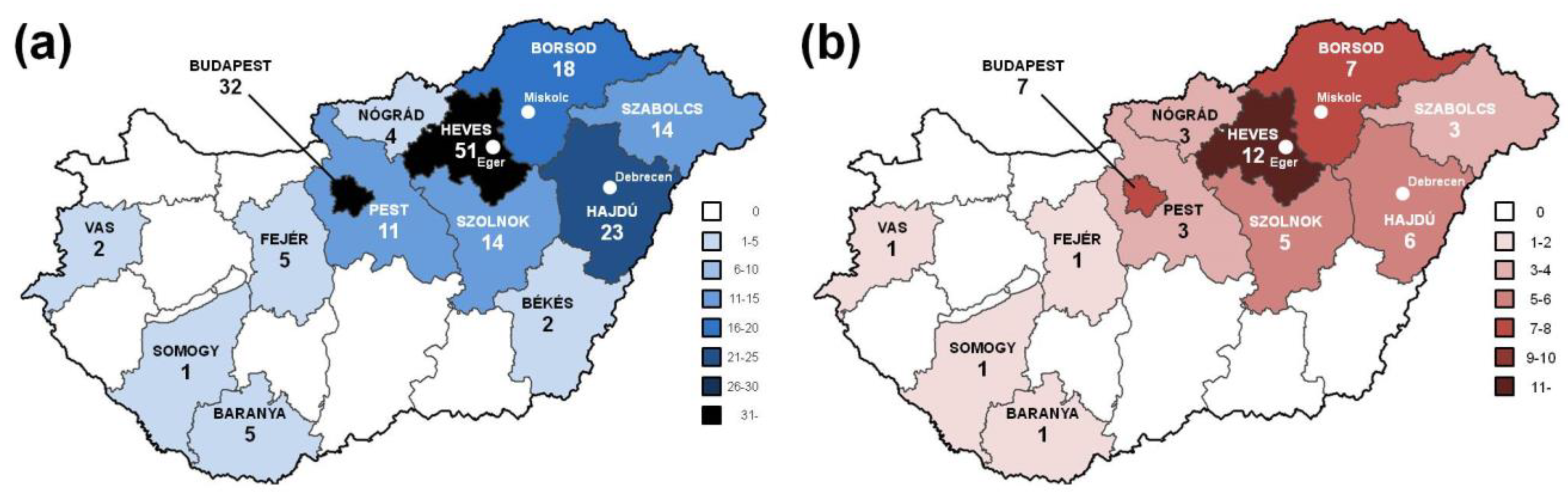
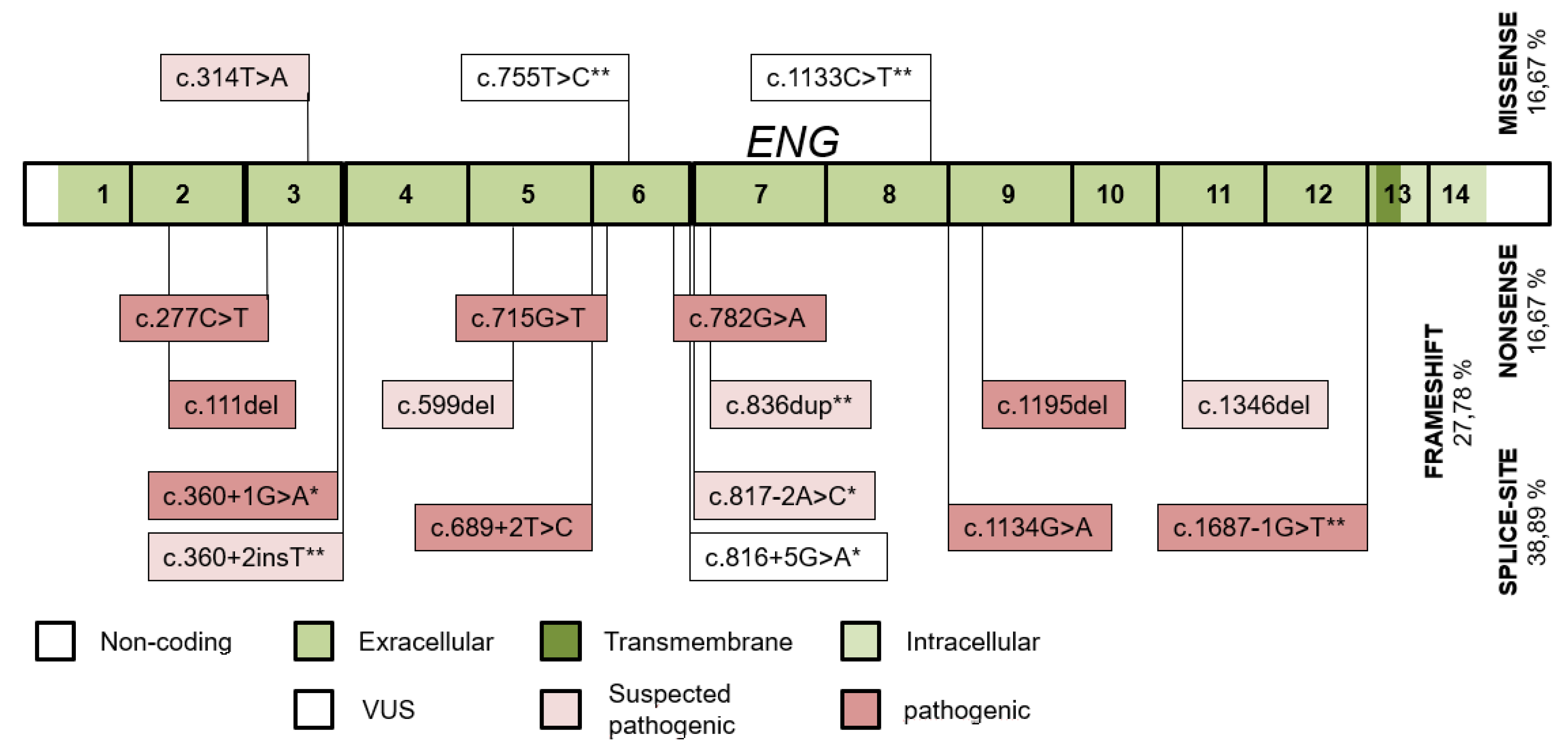
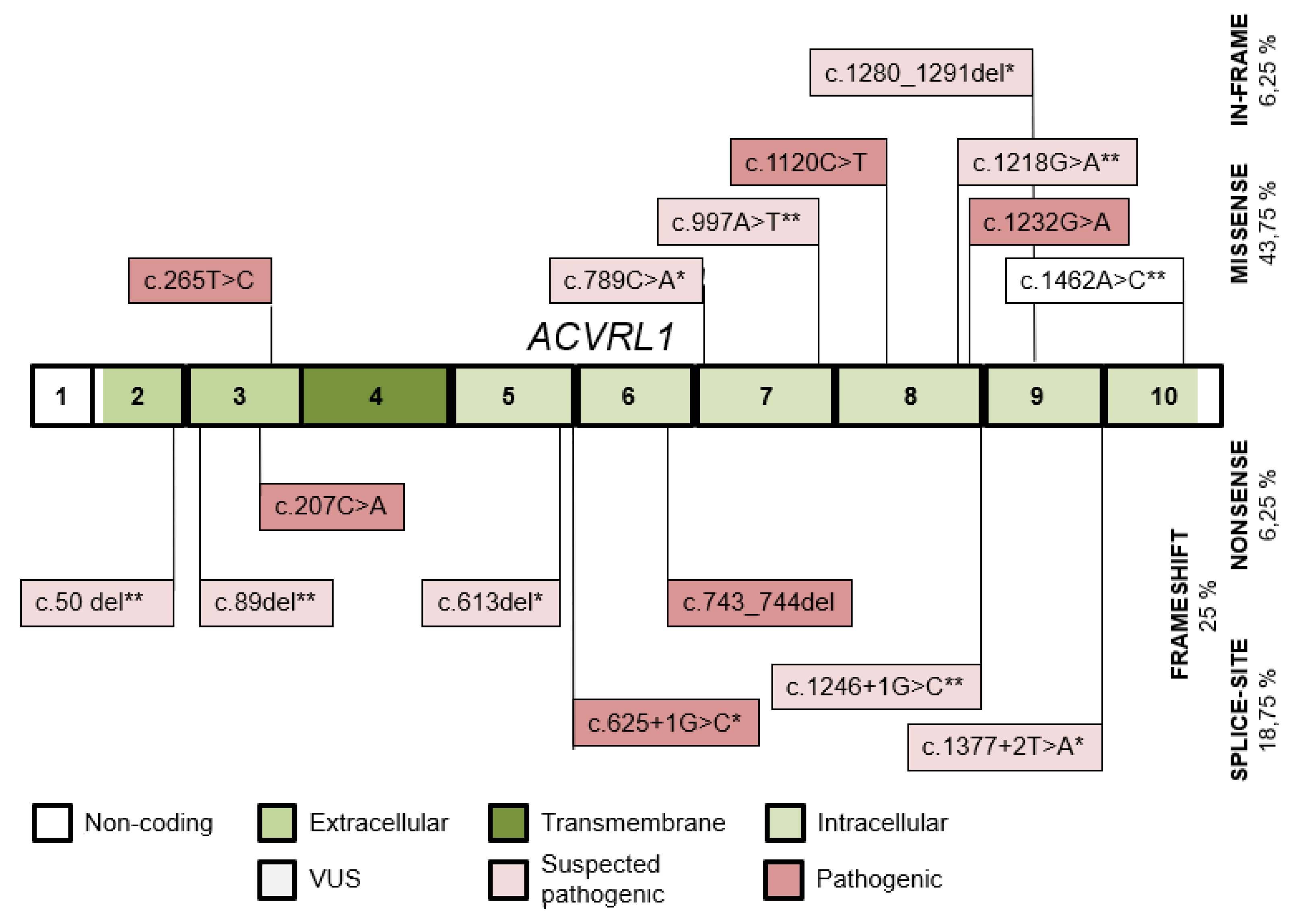
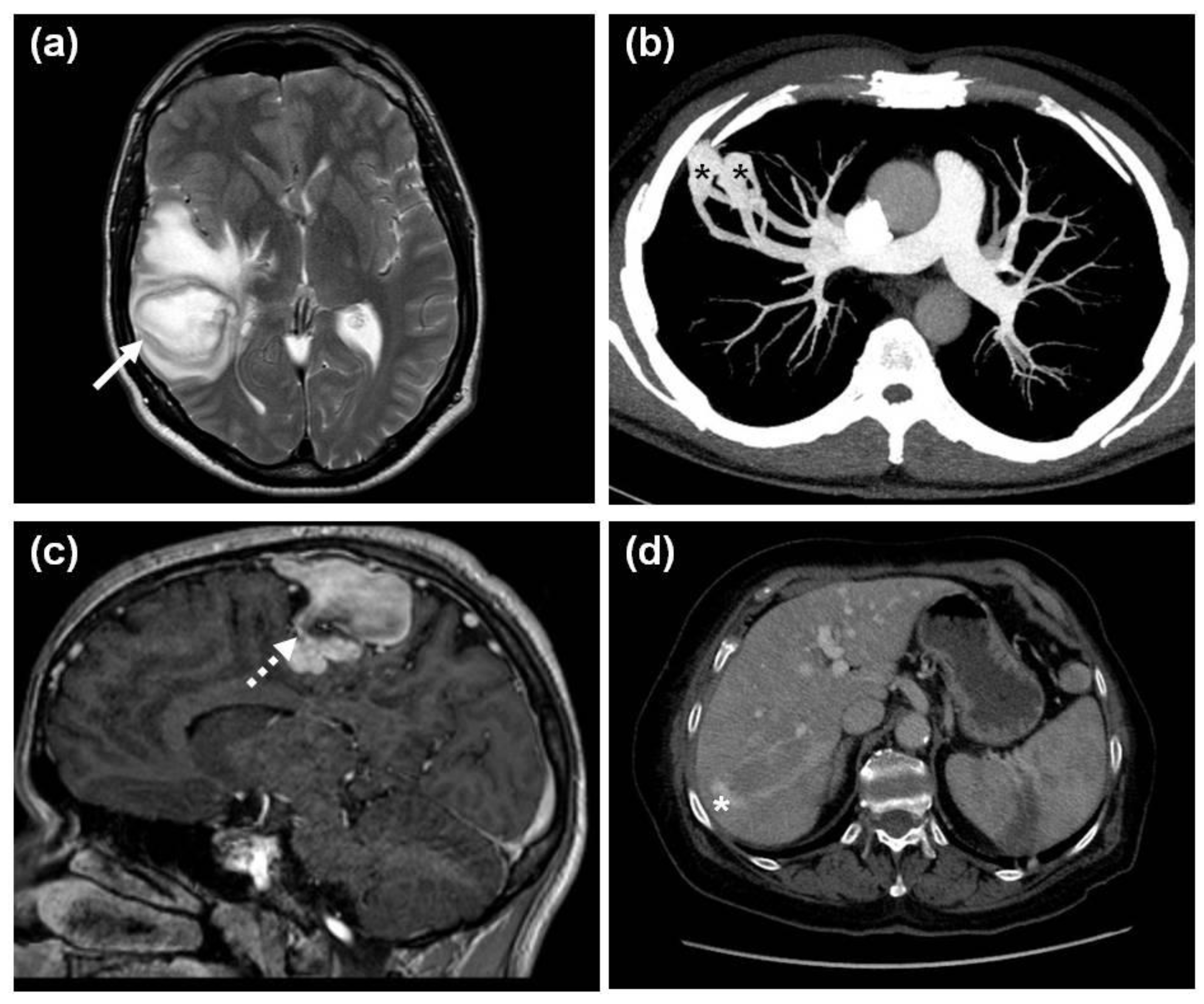
| Variant | Protein Change | Type | Population dbSNP 1000 Genomes HMSSP | Co-Segregation | SISA | In Silico Prediction Modeling Software | ACMG § |
|---|---|---|---|---|---|---|---|
| ENG | |||||||
| c.360+2insT | SS | none | M: 1D | NA | LP | ||
| c.755T>C | p.Ile252Thr | MS | none | M: 2D | PolyPhen2 HumDiv *: possibly damaging PolyPhen2 HumVar *: probably damaging MutPred2 *: non-pathogenic/borderline SIFT **: affecting protein function | VUS | |
| c.836dup | p.Cys279fs | FS | none | M: 1D | NA | LP | |
| c.1133C>T | p.Ala378Val | MS | none | M: 1S I; w: 1NL | PolyPhen2 HumDiv *: possibly damaging PolyPhen2 HumVar *: benign MutPred2 *: non-pathogenic SIFT **: tolerated | VUS | |
| c.1687-1G>T | SS | none | M: 3D + 1S PED | ≥87.5% | NA | P | |
| ACVRL1 | |||||||
| c.50del # | p.Leu17Trpfs *2 | FS | none | NA | LP | ||
| c.89del | p.Pro30Argfs *3 | FS | none | M: 1S I | NA | LP | |
| c.997A>T # | p.Ser333Cys | MS | none | M: 4D w: 1NL | ≥87.5% | PolyPhen2 HumDiv *: probably damaging PolyPhen2 HumVar *: probably damaging MutPred2 *: likely pathogenic SIFT **: affecting protein function | LP |
| c.1218G>A # | p.Trp406 * | NS | none | M: 2D w: 2NL | NA | LP | |
| c.1246+1G>C # | SS | none | M: 1D + 1NL I; w: 2NL | NA | LP | ||
| c.1462A>C | p.Thr488Pro | MS | none | M: 2D + 1S PED | PolyPhen2 HumDiv *: probably damaging PolyPhen2 HumVar *: probably damaging MutPred2 *: likely pathogenic SIFT **: affecting protein function | VUS |
| HHT1 | HHT2 | HHT1+HHT2 | |
|---|---|---|---|
| Overall | |||
| No. (male/female) | 53 (31/22) | 63 (24/39) | 116 (55/61) |
| Age (years) ± SD | 46.1 ± 18.0 | 43.5 ± 19.9 | 44.7 ± 19.1 |
| 3 or 4 clinical criteria exist | |||
| No. (male/female) | 44 (24/20) | 47 (14/33) | 91 (38/53) |
| Age (years) ± SD | 49.2 ± 16.4 | 50.0 ± 15.7 | 49.6 ± 16.1 |
| 2 clinical criteria exist | |||
| No. (male/female) | 9 (7/2) | 11 (7/4) | 20 (14/6) |
| Age (years) ± SD | 31.1 ± 17.8 | 30.0 ± 18.1 | 30.5 ± 18.0 |
| 1 clinical criterion exists | |||
| No. (male/female) | 0 | 5 (3/2) | 5 (3/2) |
| Age (years) ± SD | 12.0 ± 11.6 | 12.0 ± 11.6 |
| HHT1 | HHT2 | p Value | HHT1 + HHT2 | |
|---|---|---|---|---|
| Epistaxis | ||||
| Overall | 53/53 (100%) | 55/63 (87.3%) | 0.007 | 108/116 (93.1%) |
| Definite | 44/44 (100%) | 46/47 (97.9%) | NS | 90/91 (98.9%) |
| Suspected | 9/9 (100%) | 9/11 (81.8%) | NS | 18/20 (90%) |
| Telangiectasia | ||||
| Overall | 33/53 (62.3%) | 48/63 (76.2%) | NS | 81/116 (69.8%) |
| Definite | 33/44 (75%) | 46/47 (97.9%) | 0.001 | 79/91 (86.8%) |
| Suspected | 0/9 (0%) | 2/11 (18.2%) | NS | 2/20 (10%) |
| PAVM overall 1 | 22/41 (53.7%) 2 | 2/46 (4.3%) 2 | <0.001 | 24/87 (27.6%) 2 |
| CAVM overall 3 | 4/21 (19.0%) 2 | 1/36 (2.8%) 2 | NS | 5/57 (8.8%) 2 |
| HAVM overall 3 | 7/29 (24.1%) 2 | 19/39 (48.7%) 2 | 0.047 | 26/68 (38.2%) 2 |
| GI telangiectasia 4 | 9/53 (17.0%) | 7/63 (11.1%) 2 | NS | 16/116 (13.8%) 2 |
| Kindreds | Male | Female |
|---|---|---|
| HHT1 family members (n = 8) | ||
| Individuals with ENG mutation | ||
| 3 or 4 clinical criteria exist | - | - |
| 2 clinical criteria exist | 12y (E); 13y (E) | - |
| 1 clinical criterion exists | - | - |
| Wild-type individuals | 4y, 11y, 12y | 6y, 7y, 17y |
| HHT2 family members (n = 16) | ||
| Individuals with ACVRL1 mutation | ||
| 3 or 4 clinical criteria exist | - | 9y (E,T) |
| 2 clinical criteria exist | 1y (E), 11y (T) | 15y (E) |
| 1 clinical criterion exists | 0y, 4y | 3y |
| Wild-type individual | 5y, 7y, 8y, 11y, 13y | 1y, 11y, 11y, 14y |
| Gene | Variant | M+ | w | Non-Testable | Refused Screening | Obligately Testable | Facultatively Testable | ||
|---|---|---|---|---|---|---|---|---|---|
| ≥3 1 | 2 1 | 1 1 | |||||||
| ENG | c.111del | 1 | 0 | 0 | 2 | 0 | 0 | 0 | 0 |
| c.314T>A | 1 | 0 | 0 | 1 | 0 | 0 | 2 | 0 | |
| c.360+1G>A | 3 | 0 | 0 | 8 | 0 | 1 | 0 | 0 | |
| c.816+5G>A § | 6 | 2 | 0 | 6 | 0 | 7 | 5 | 0 | |
| c.817-2A>C (2) | 11 | 1 | 0 | 10 | 0 | 7 | 7 | 0 | |
| c.1134G>A | 1 | 1 | 0 | 0 | 0 | 0 | 2 | 0 | |
| c.1195del | 1 | 0 | 0 | 0 | 0 | 0 | 12 | 2 | |
| c.1346del | 1 | 0 | 0 | 1 | 0 | 0 | 2 | 2 | |
| c.1687-1G>T | 1 | 1 | 0 | 0 | 0 | 5 | 0 | 0 | |
| ACVRL1 | c.50del | 3 | 1 | 0 | 0 | 0 | 0 | 2 | 2 |
| c.207C>A | 1 | 0 | 0 | 6 | 7 | 0 | 3 | 0 | |
| c.265T>C (3) | 3 | 0 | 0 | 0 | 0 | 0 | 41 | 3 | |
| c.613del | 1 | 0 | 0 | 1 | 0 | 0 | 3 | 0 | |
| c.625+1G>C (6) | 16 | 4 | 3 | 14 | 2 | 3 | 6 | 0 | |
| c.743_744del | 1 | 0 | 0 | 0 | 0 | 0 | 8 | 0 | |
| c.789C>A | 1 | 1 | 1 | 0 | 0 | 0 | 3 | 0 | |
| c.997A>T | 4 | 0 | 0 | 1 | 1 | 0 | 17 | 7 | |
| c.1120C>T | 3 | 0 | 0 | 0 | 0 | 0 | 13 | 1 | |
| c.1218G>A | 2 | 0 | 0 | 2 | 0 | 0 | 2 | 0 | |
| c.1232G>A | 2 | 0 | 0 | 2 | 0 | 0 | 3 | 3 | |
| c.1246+1G>C | 2 | 0 | 0 | 2 | 0 | 0 | 6 | 4 | |
| c.1280_1291del | 1 | 1 | 0 | 0 | 0 | 0 | 8 | 2 | |
| c.1377+2T>A (2) | 3 | 1 | 0 | 4 | 0 | 1 | 6 | 0 | |
| Total: | 69 | 13 | 4 | 60 | 10 | 24 | 151 | 26 | |
| Screening status | 156 clarified | 24 refuses | 177 testable | ||||||
| Reference | Population 1 HHT Centre 2 | Molecular Analysis | Probands with Mutations/Total | ENG/ACVRL1 Mutation Rate | ENG/ACVRL1 Family Rate | ENG/ACVRL1 Pt Rate | Pts/Family 3 Inds/Family 4 |
|---|---|---|---|---|---|---|---|
| [4] | Utah, US 2 | ENG/ACVRL1 SS | 26/34 (76.5%) | 14/10 (1.4) | 14/12 (1.17) | 61/50 (1.22) | 111/26 (4.27) 3 |
| [29] | Multicentric (US, Australia, Canada, Japan) 2 | ENG/ACVRL1 SS + 5′UTR SS + LDI | 137/200 (68.5%) | 71/42 (1.69) | 77/50 (1.54) | ND | ND |
| [30] | Norwegian national study 1 | ENG/ACVRL1/SMAD4 SS + LDI | total: 105/113 (92.9%) LP+P: 97/113 (85.8%) | total: 30/27 (1.11) LP+P: 30/23 (1.30) SMAD4: 0 | total: 42/63 (0.67) LP+P: 39/58 (0.67) | ND | 237/97 (2.44) 3 423/113 (3.74) 4 |
| [31,32] | French HHT network 1 | ENG/ACVRL1 SS + 5′UTR SS + LDI, SMAD4 SS | D: 119/136 (87.5%) | D: 42/56 (0.75) SMAD4: 0 | D: 40/79 (0.51) | ||
| [25] | French–Italian HHT network 1 | 91/250 (0.36) | 343/135 (2.54) 3 | ||||
| [33] | Bari, Italy 2 | 135/65 (2.08) 3 | |||||
| [34] | Utrecht, The Netherlands 2 | ENG/ACVRL1 SS + LDI | 97/104 (93.3%) | 40/31 (1.29) | 55/42 (1.31) | ||
| [35] | 63/40 (1.58) | 508/103 (4.93) 3 | |||||
| [36] | Pavia-Crema, Italy 2 | ENG/ACVRL1 SS | 101/137 (73.7%) | 26/50 (0.52) | 29/72 (0.40) | 263/101 (2.60) 3 457/101 (4.52) 4 | |
| [37] | Spanish RiHHTa registry 1 | ENG/ACVRL1/SMAD4 SS, NGS + LDI | 16/25 (0.64) | 36/77 (0.47) | |||
| [13] | Danish national study 1 | ENG/ACVRL1/SMAD4 SS + LDI | 95/107 (88.8%) | 29/32 (0.91) SMAD4: 3 fam | 47/45 (1.04) SMAD4: 3 fam | 151/132 (1.14) SMAD4: 5 pts | 320/107 (3.06) 3 |
| This study | Debrecen, Hungary 2 | ENG/ACVRL1/SMAD4 SS, NGS + LDI | 48/50 (96%) | 18/16 (1.13) SMAD4: 1 fam | 21/26 (0.81) SMAD4: 1 fam | 53/63 (0.84) SMAD4: 1 pt | 121/50 (2.42) 3 186/50 (3.72) 4 |
| Ref. | Cohort | PAVM | CAVM | HAVM | GI Lesion | ||||
|---|---|---|---|---|---|---|---|---|---|
| HHT1 | HHT2 | HHT1 | HHT2 | HHT1 | HHT2 | HHT1 | HHT2 | ||
| [4] | Utah, US 2 | 36/61 (59%) | 13/45 (28.9%) | 10/61 (16.4%) | 1/50 (2%) | 1/59 (1.7%) | 13/47 (27.7%) | 7/39 (18%) bleeding | 8/39 (21%) bleeding |
| p = 0.002 | p = 0.012 | p < 0.001 | |||||||
| [29] | Multicentric (US, Australia, Canada, Japan) 2 | 52/77 (67.5%) | 24/50 (48%) | 7/77 (9.1%) | 0 | 2/77 (2.6%) | 7/50 (14%) | 7/77 (9.1%) | 5/50 (10%) |
| [25] | French–Italian HHT network 1 | sy: 32/93 (34.4%) | sy: 13/250 (5.2%) | sy: 2/93 (2.2%) | sy: 3/250 (1.2%) | sy: 0 | sy: 19/250 (7.6%) | 6/93 (6.5%) | 41/250 (16.4%) |
| p < 0.001 | NS | NS | p = 0.017 | ||||||
| asy: 27/50 (54%) | asy: 19/149 (12.8%) | asy: 2/22 (9.1%) | asy: 2/50 (4%) | asy: 20/46 (43.5%) | asy: 87/151 (57.6%) | ||||
| p < 0.0001 | NS | NS | |||||||
| [33] | Bari, Italy 2 | 34/45 (75.5%) | 34/77 (44.1%) | 9/43 (20.9%) | 0 | 27/45 (60%) | 64/77 (83.1%) | 18/30 (60%) | 24/47 (51.1%) |
| p < 0.0005 | p < 0.0002 | p < 0.01 | NS | ||||||
| Large #: 21/34 (61.8%) | Large #: 6/34 (17.6%) | ||||||||
| p < 0.0001 | |||||||||
| [35] | Utrecht, The Netherlands 2 | 167/343 (48.7%) | 6/114 (5.3%) | 38/260 (14.6%) | 1/76 (1.3%) | 11/144 (7.6%) | 13/32 (40.6%) | 56/78 (71.8%) | 19/29 (65.5%) |
| p = 1.2 × 10−16 | p = 0.0015 | p = 8.7 × 10−7 | NS | ||||||
| [37] | Spanish RiHHTa registry 1 | 20/36 (55.5%) | 11/77 (14.3%) | 3/36 (8.3%) | 1/77 (1.3%) | 9/36 (25%) | 33/77 (42.8%) | Upper: 8/36 (22.2%) | 9/77 (11.7%) |
| p < 0.005 | p < 0.005 | NS, p = 0.075 | NS | ||||||
| Lower: 3/36 (8.3%) | 3/77 (3.9%) | ||||||||
| [13] | Danish national study 1 | 79/151 (52.3%) | 17/132 (12.9%) | 2/16 (25%) | 1/7 (14.3%) | 2/5 (40%) | 8/11 (72.7%) | 28/151 (18.5%) | 15/132 (11.4%) |
| p < 0.001 | NS | ||||||||
| This study | Debrecen, Hungary 2 | 22/41 (53.7%) | 2/46 (4.3%) | 4/21 (19.0%) | 1/36 (2.8%) | 7/29 (24.1%) | 19/39 (48.7%) | 9/53 (17%) | 7/63 (11.1%) |
| p < 0.001 | NS, p = 0.056 | p = 0.047 | NS | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Major, T.; Bereczky, Z.; Gindele, R.; Balogh, G.; Rácz, B.; Bora, L.; Kézsmárki, Z.; Brúgós, B.; Pfliegler, G. Current Status of Clinical and Genetic Screening of Hereditary Hemorrhagic Telangiectasia Families in Hungary. J. Clin. Med. 2021, 10, 3774. https://doi.org/10.3390/jcm10173774
Major T, Bereczky Z, Gindele R, Balogh G, Rácz B, Bora L, Kézsmárki Z, Brúgós B, Pfliegler G. Current Status of Clinical and Genetic Screening of Hereditary Hemorrhagic Telangiectasia Families in Hungary. Journal of Clinical Medicine. 2021; 10(17):3774. https://doi.org/10.3390/jcm10173774
Chicago/Turabian StyleMajor, Tamás, Zsuzsanna Bereczky, Réka Gindele, Gábor Balogh, Benedek Rácz, László Bora, Zsolt Kézsmárki, Boglárka Brúgós, and György Pfliegler. 2021. "Current Status of Clinical and Genetic Screening of Hereditary Hemorrhagic Telangiectasia Families in Hungary" Journal of Clinical Medicine 10, no. 17: 3774. https://doi.org/10.3390/jcm10173774
APA StyleMajor, T., Bereczky, Z., Gindele, R., Balogh, G., Rácz, B., Bora, L., Kézsmárki, Z., Brúgós, B., & Pfliegler, G. (2021). Current Status of Clinical and Genetic Screening of Hereditary Hemorrhagic Telangiectasia Families in Hungary. Journal of Clinical Medicine, 10(17), 3774. https://doi.org/10.3390/jcm10173774







