Enhanced Medical and Community Face Masks with Antimicrobial Properties: A Systematic Review
Abstract
1. Introduction
2. Methods
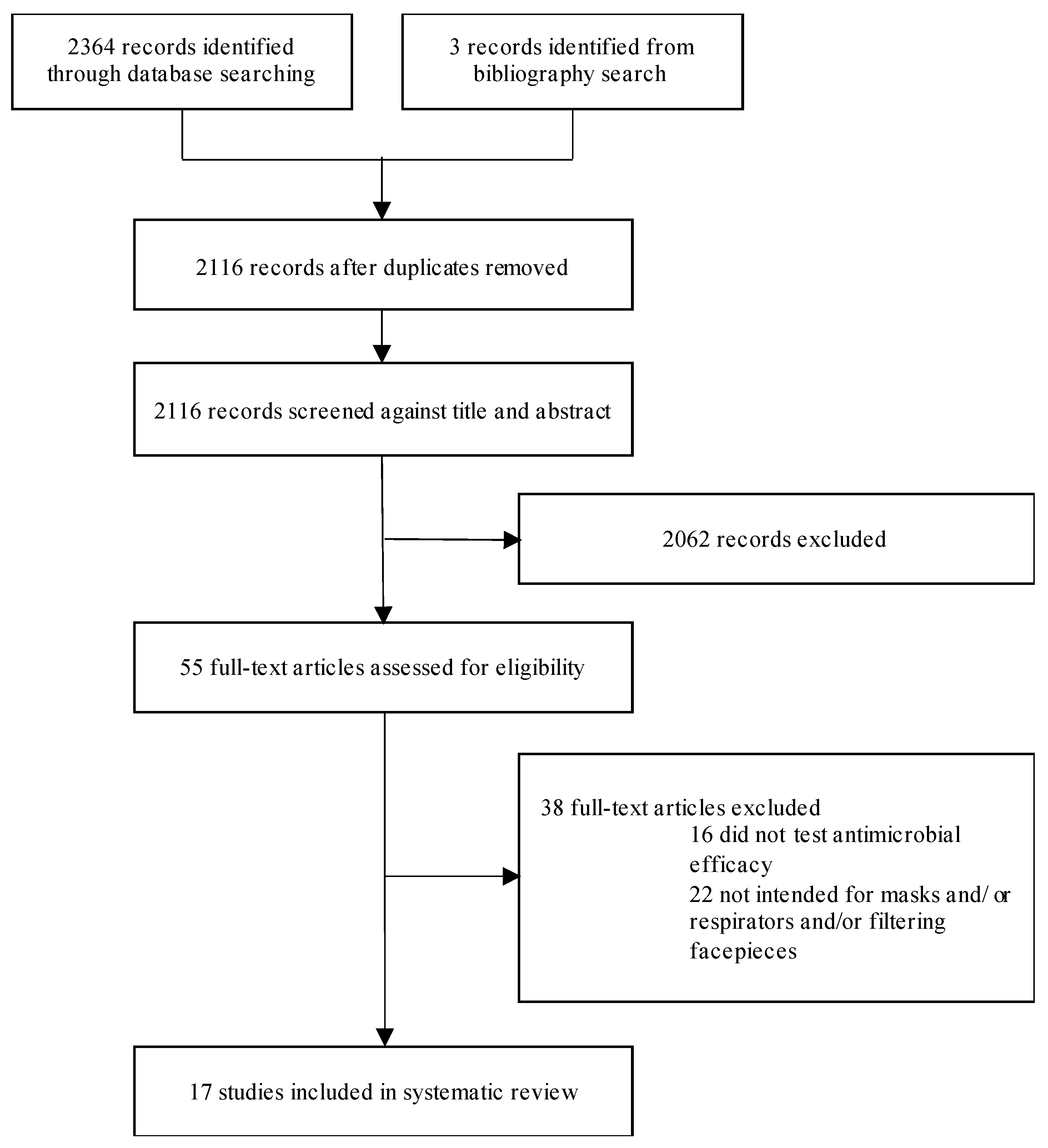
2.1. Search Strategy and Selection Criteria
2.2. Data Analysis
2.3. Outcomes
2.4. Risk of Bias
2.5. Role of Funding Source
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rowan, N.J.; Laffey, J.G. Unlocking the surge in demand for personal and protective equipment (PPE) and improvised face coverings arising from coronavirus disease (COVID-19) pandemic-Implications for efficacy, re-use and sustainable waste management. Sci. Total Environ. 2021, 752, 142259. [Google Scholar] [CrossRef]
- World Health Organization. Rational Use of Personal Protective Equipment for Coronavirus Disease (COVID-19): Interim Guidance, 27 February 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- WHO. Advice on the Use of Masks in the Context of COVID-19: Interim Guidance, 5 June 2020; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Pecchia, L.; Piaggio, D.; Maccaro, A.; Formisano, C.; Iadanza, E. The Inadequacy of Regulatory Frameworks in Time of Crisis and in Low-Resource Settings: Personal Protective Equipment and COVID-19. Health Technol. 2020, 10, 1375–1383. [Google Scholar] [CrossRef]
- Aljabo, A.; Mueller, E.; Abdul-Azeez, D.; Hoare, T.; Jain, A. Gravity steam reprocessing in healthcare facilities for the reuse of N95 respirators. J. Hosp. Infect. 2020, 106, 698–708. [Google Scholar] [CrossRef] [PubMed]
- O’Hearn, K.; Gertsman, S.; Webster, R.; Tsampalieros, A.; Ng, R.; Gibson, J.; Sampson, M.; Sikora, L.; McNally, J.D. Efficacy and safety of disinfectants for decontamination of N95 and SN95 filtering facepiece respirators: A systematic review. J. Hosp. Infect. 2020, 106, 504–521. [Google Scholar] [CrossRef] [PubMed]
- Gertsman, S.; Agarwal, A.; O’Hearn, K.; Webster, R.; Tsampalieros, A.; Barrowman, N.; Sampson, M.; Sikora, L.; Staykov, E.; Ng, R.; et al. Microwave- and heat-based decontamination of N95 filtering facepiece respirators: A systematic review. J. Hosp. Infect. 2020, 106, 536–553. [Google Scholar] [CrossRef] [PubMed]
- Pullangott, G.; Kannan, U.; Gayathri, S.; Kiran, D.V.; Maliyekkal, S.M. A comprehensive review on antimicrobial face masks: An emerging weapon in fighting pandemics. RSC Adv. 2021, 11, 6544–6576. [Google Scholar] [CrossRef]
- Levine, C.; Grady, C.; Block, T.; Hurley, H.; Russo, R.; Peixoto, B.; Frees, A.; Ruiz, A.; Alland, D. Use, re-use or discard? Quantitatively defined variance in the functional integrity of N95 respirators following vaporized hydrogen peroxide decontamination during the COVID-19 pandemic. J. Hosp. Infect. 2021, 107, 50–56. [Google Scholar] [CrossRef]
- Rodriguez-Martinez, C.E.; Sossa-Briceño, M.P.; Cortés, J.A. Decontamination and reuse of N95 filtering facemask respirators: A systematic review of the literature. Am. J. Infect. Control. 2020, 48, 1520–1532. [Google Scholar] [CrossRef]
- Karim, N.; Afroj, S.; Lloyd, K.; Oaten, L.C.; Andreeva, D.V.; Carr, C.; Farmery, A.D.; Kim, I.-D.; Novoselov, K.S. Sustainable Personal Protective Clothing for Healthcare Applications: A Review. ACS Nano 2020, 14, 12313–12340. [Google Scholar] [CrossRef]
- Ahmed, M.K.; Afifi, M.; Uskoković, V. Protecting healthcare workers during COVID-19 pandemic with nanotechnology: A protocol for a new device from Egypt. J. Infect. Public Health 2020, 13, 1243–1246. [Google Scholar] [CrossRef]
- Chowdhury, M.A.; Shuvho, B.A.; Shahid, A.; Haque, A.M.; Kashem, M.A.; Lam, S.S.; Ong, H.C.; Uddin, A.; Mofijur, M. Prospect of biobased antiviral face mask to limit the coronavirus outbreak. Environ. Res. 2020, 192, 110294. [Google Scholar] [CrossRef]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ Br. Med. J. 2015, 349, g7647. [Google Scholar] [CrossRef]
- Zorko, D.; Gertsman, S.; O’Hearn, K.; Timmerman, N.; Ambu-Ali, N.; Dinh, T.; Sampson, M.; Sikora, L.; McNally, J.; Choong, K. Decontamination interventions for the reuse of surgical mask personal protective equipment: A systematic review. J. Hosp. Infect. 2020, 106, 283–294. [Google Scholar] [CrossRef]
- Rengasamy, S.; Fisher, E.; Shaffer, R.E. Evaluation of the survivability of MS2 viral aerosols deposited on filtering face piece respirator samples incorporating antimicrobial technologies. Am. J. Infect. Control 2010, 38, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Borkow, G.; Zhou, S.S.; Page, T.; Gabbay, J. A Novel Anti-Influenza Copper Oxide Containing Respiratory Face Mask. PLoS ONE 2010, 5, e11295. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Leung, P.; Yao, L.; Song, Q.; Newton, E. Antimicrobial effect of surgical masks coated with nanoparticles. J. Hosp. Infect. 2006, 62, 58–63. [Google Scholar] [CrossRef]
- Tseng, C.-C.; Pan, Z.-M.; Chang, C.-H. Application of a quaternary ammonium agent on surgical face masks before use for pre-decontamination of nosocomial infection-related bioaerosols. Aerosol Sci. Technol. 2016, 50, 199–210. [Google Scholar] [CrossRef]
- Hiragond, C.B.; Kshirsagar, A.; Dhapte, V.V.; Khanna, T.; Joshi, P.; More, P.V. Enhanced anti-microbial response of commercial face mask using colloidal silver nanoparticles. Vacuum 2018, 156, 475–482. [Google Scholar] [CrossRef]
- Xiong, S.-W.; Fu, P.-G.; Zou, Q.; Chen, L.-Y.; Jiang, M.-Y.; Zhang, P.; Wang, Z.-G.; Cui, L.-S.; Guo, H.; Gai, J.-G. Heat Conduction and Antibacterial Hexagonal Boron Nitride/Polypropylene Nanocomposite Fibrous Membranes for Face Masks with Long-Time Wearing Performance. ACS Appl. Mater. Interfaces 2020, 13, 196–206. [Google Scholar] [CrossRef]
- Demir, B.; Cerkez, I.; Worley, S.D.; Broughton, R.M.; Huang, T.-S. N-Halamine-Modified Antimicrobial Polypropylene Nonwoven Fabrics for Use against Airborne Bacteria. ACS Appl. Mater. Interfaces 2015, 7, 1752–1757. [Google Scholar] [CrossRef]
- Majchrzycka, K.; Gutarowska, B.; Brochocka, A.; Brycki, B. New Filtering Antimicrobial Nonwovens With Various Carriers for Biocides as Respiratory Protective Materials Against Bioaerosol. Int. J. Occup. Saf. Ergon. 2012, 18, 375–385. [Google Scholar] [CrossRef]
- Zheng, C.R.; Li, S.; Ye, C.; Li, X.; Zhang, C.; Yu, X. Particulate Respirators Functionalized with Silver Nanoparticles Showed Excellent Real-Time Antimicrobial Effects against Pathogens. Environ. Sci. Technol. 2016, 50, 7144–7151. [Google Scholar] [CrossRef]
- Lore, M.B.; Sebastian, J.M.; Brown, T.L.; Viner, A.S.; McCullough, N.V.; Hinrichs, S.H. Performance of Conventional and Antimicrobial-Treated Filtering Facepiece Respirators Challenged with Biological Aerosols. J. Occup. Environ. Hyg. 2012, 9, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Karmacharya, M.; Joshi, S.R.; Gulenko, O.; Park, J.; Kim, G.-H.; Cho, Y.-K. Photoactive Antiviral Face Mask with Self-Sterilization and Reusability. Nano Lett. 2020, 21, 337–343. [Google Scholar] [CrossRef]
- Rubino, I.; Oh, E.; Han, S.; Kaleem, S.; Hornig, A.; Lee, S.H.; Kang, H.J.; Lee, D.H.; Chu, K.B.; Kumaran, S.; et al. Salt coatings functionalize inert membranes into high-performing filters against infectious respiratory diseases. Sci. Rep. 2020, 10, 13875. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Xu, S.; Wang, Z.; Xue, K.; Su, J.; Song, Y.; Chen, S.; Zhu, C.; Tang, B.Z.; Ye, R. Self-Reporting and Photothermal Enhanced Rapid Bacterial Killing on Laser-Induced Graphene Mask. ACS Nano 2020, 14, 12045–12053. [Google Scholar] [CrossRef]
- Duong-Quy, S.; Ngo-Minh, X.; Tang-Le-Quynh, T.; Tang-Thi-Thao, T.; Nguyen-Quoc, B.; Le-Quang, K.; Tran-Thanh, D.; Doan-Thi-Quynh, N.; Canty, E.; Do, T.; et al. The use of exhaled nitric oxide and peak expiratory flow to demonstrate improved breathability and antimicrobial properties of novel face mask made with sustainable filter paper and Folium Plectranthii amboinicii oil: Additional option for mask shortage during COVID-19 pandemic. Multidiscip. Respir. Med. 2020, 15, 664. [Google Scholar] [PubMed]
- Quan, F.-S.; Rubino, I.; Lee, S.-H.; Koch, B.; Choi, H.-J. Universal and reusable virus deactivation system for respiratory protection. Sci. Rep. 2017, 7, 39956. [Google Scholar] [CrossRef]
- Woo, M.-H.; Grippin, A.; Wu, C.-Y.; Baney, R.H. Use of dialdehyde starch treated filters for protection against airborne viruses. J. Aerosol Sci. 2012, 46, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Ren, T.; Dormitorio, T.V.; Qiao, M.; Huang, T.-S.; Weese, J. N-halamine incorporated antimicrobial nonwoven fabrics for use against avian influenza virus. Vet. Microbiol. 2018, 218, 78–83. [Google Scholar] [CrossRef]
- Yin, X.; Zhang, J.; Xu, J.; Tian, M.; Li, L.; Tan, L.; Li, Z. Fast-acting and highly rechargeable antibacterial composite nanofibrous membrane for protective applications. Compos. Sci. Technol. 2020, 202, 108574. [Google Scholar] [CrossRef]
- Wang, J.; Planz, V.; Vukosavljevic, B.; Windbergs, M. Multifunctional electrospun nanofibers for wound application—Novel insights into the control of drug release and antimicrobial activity. Eur. J. Pharm. Biopharm. 2018, 129, 175–183. [Google Scholar] [CrossRef]
- Simões, D.; Miguel, S.A.P.; Ribeiro, M.; Coutinho, P.; Mendonça, A.; Correia, I.J. Recent advances on antimicrobial wound dressing: A review. Eur. J. Pharm. Biopharm. 2018, 127, 130–141. [Google Scholar] [CrossRef] [PubMed]
- Ungur, G.; Hrůza, J. Modified polyurethane nanofibers as antibacterial filters for air and water purification. RSC Adv. 2017, 7, 49177–49187. [Google Scholar] [CrossRef]
- Fahimirad, S.; Fahimirad, Z.; Sillanpää, M. Efficient removal of water bacteria and viruses using electrospun nanofibers. Sci. Total Environ. 2021, 751, 141673. [Google Scholar] [CrossRef] [PubMed]
- Joe, Y.H.; Park, D.H.; Hwang, J. Evaluation of Ag nanoparticle coated air filter against aerosolized virus: Anti-viral efficiency with dust loading. J. Hazard. Mater. 2016, 301, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.H.; Hwang, G.B.; Lee, J.E.; Bae, G.N. Preparation of Airborne Ag/CNT Hybrid Nanoparticles Using an Aerosol Process and Their Application to Antimicrobial Air Filtration. Langmuir 2011, 27, 10256–10264. [Google Scholar] [CrossRef]
- Adlhart, C.; Verran, J.; Azevedo, N.; Olmez, H.; Keinänen-Toivola, M.M.; Gouveia, I.C.; Melo, L.F.; Crijns, F. Surface modifications for antimicrobial effects in the healthcare setting: A critical overview. J. Hosp. Infect. 2018, 99, 239–249. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; Communicable Diseases Cluster. Avian Influenza: Assessing the Pandemic Threat; World Health Organization: Geneva, Switzerland, 2005. [Google Scholar]
- WHO. WHO Recommendations for the Post-Pandemic Period 2010. Cited 2021. Available online: https://www.who.int/csr/disease/swineflu/notes/briefing_20100810/en/ (accessed on 4 August 2021).
- Sohrabi, C.; Alsafi, Z.; O’Neill, N.; Khan, M.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, R. World Health Organization declares global emergency: A review of the 2019 novel coronavirus (COVID-19). Int. J. Surg. 2020, 76, 71–76. [Google Scholar] [CrossRef]
- WHO. WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19–11 March 2020. Cited 2021. Available online: https://www.who.int/director-general/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 4 August 2021).
- Imani, S.M.; Ladouceur, L.; Marshall, T.; Maclachlan, R.; Soleymani, L.; Didar, T.F. Antimicrobial Nanomaterials and Coatings: Current Mechanisms and Future Perspectives to Control the Spread of Viruses Including SARS-CoV-2. ACS Nano 2020, 14, 12341–12369. [Google Scholar] [CrossRef]
- Cortes, A.A.; Zuñiga, J.M. The use of copper to help prevent transmission of SARS-coronavirus and influenza viruses. A general review. Diagn. Microbiol. Infect. Dis. 2020, 98, 115176. [Google Scholar] [CrossRef]
- Han, J.; Chen, L.; Duan, S.-M.; Yang, Q.-X.; Yang, M.; Gao, C.; Zhang, B.-Y.; He, H.; Dong, X.-P. Efficient and quick inactivation of SARS coronavirus and other microbes exposed to the surfaces of some metal catalysts. Biomed. Environ. Sci. 2005, 18, 176–180. [Google Scholar]
- Codita, I.; Caplan, D.M.; Drăgulescu, E.-C.; Lixandru, B.-E.; Coldea, I.L.; Dragomirescu, C.C.; Surdu-Bob, C.; Badulescu, M. Antimicrobial activity of copper and silver nanofilms on nosocomial bacterial species. Roum. Arch. Microbiol. Immunol. 2010, 69, 204–212. [Google Scholar]
- Hui, F.; Debiemme-Chouvy, C. Antimicrobial N-Halamine Polymers and Coatings: A Review of Their Synthesis, Characterization, and Applications. Biomacromolecules 2013, 14, 585–601. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, M.; Dadashpour, M.; Hejazi, M.; Hasanzadeh, M.; Behnam, B.; de la Guardia, M.; Shadjou, N.; Mokhtarzadeh, A. Anti-bacterial activity of graphene oxide as a new weapon nanomaterial to combat multidrug-resistance bacteria. Mater. Sci. Eng. C 2017, 74, 568–581. [Google Scholar] [CrossRef] [PubMed]
- Seifi, T.; Kamali, A.R. Anti-pathogenic activity of graphene nanomaterials: A review. Colloids Surf. B Biointerfaces 2020, 199, 111509. [Google Scholar] [CrossRef]
- Zhang, Z.; El-Moghazy, A.Y.; Wisuthiphaet, N.; Nitin, N.; Castillo, D.; Murphy, B.G.; Sun, G. Daylight-Induced Antibacterial and Antiviral Nanofibrous Membranes Containing Vitamin K Derivatives for Personal Protective Equipment. ACS Appl. Mater. Interfaces 2020, 12, 49416–49430. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, W.; Niu, J.; Chen, Y. Mechanism of Photogenerated Reactive Oxygen Species and Correlation with the Antibacterial Properties of Engineered Metal-Oxide Nanoparticles. ACS Nano 2012, 6, 5164–5173. [Google Scholar] [CrossRef]


| Substrate | Antimicrobial System | Antimicrobial Efficacy Testing Methods | Pathogen(s) Used in Testing | Comparators/Controls | Antimicrobial Efficacy Results | |
|---|---|---|---|---|---|---|
| Borkow et al. (2010) [17] | NIOSH N95 FFP | Copper oxide | Bioaerosol challenge, bacterial Filtration Efficacy | Viral | Untreated | Significant, higher, direct contact inactivation in test masks than control |
| Li et al. (2006) [18] | FFP | Silver nitrate, titanium dioxide nanoparticles | Inoculation of fabric with pathogens | Bacterial | Sterile FFP | Control mask: Increase in viable bacteria. Test mask: 100% reduction in viable bacteria |
| Zheng et al. (2016) [24] | NIOSH 3M N95 | Silver nanoparticles | Bacterial growth track, FESEM | Bacterial | Untreated | Bacterial growth effectively inhibited. FESEM: Few bacterial cells intact, debris on treated surface |
| Hiragond et al. (2018) [20] | Surgical face mask | Silver nanoparticles | Well diffusion assay | Bacterial | Untreated | Inhibition zone of treated masks significantly higher than control |
| Rengasamy et al. (2010) [16] | 4 FFRs | Silver-copper, EvixO3-Shield, Iodine, Titanium dioxide | Bioaerosol challenge. Conditions: (1) 22 °C 30% RH for 0, 8, 20 h, (2) 37 °C 80% RH for 0, 2, 4 h | Viral | Equivalent FFR | Conditions: (1) no significant difference to control (2) Silver-copper and EvixO3-Shield technology higher log10 reduction than control. Highest: EvixO3-Shield technology |
| Kumar et al. (2021) [26] | PP nonwoven | Copper nanoparticles | Bacterial inoculation, bacterial live dead assay, bioaerosol challenge | Bacterial, viral | Untreated | 4-log reduction in E. coli CFUs. Live/dead assay indicates >99.99% reduction of E. coli. VLP concentration decreased by 2–3 log |
| Lore et al. (2012) [25] | 4 NIOSH FFRs | Iodine-based | Bioaerosol challenges | Bacterial, viral | Equivalent FFRs | No detectable antimicrobial properties in test masks compared with conventional |
| Rubino et al. (2020) [27] | Surgical masks | Sodium chloride, potassium sulphate potassium chloride | Bioaerosol challenge, TEM, in vivo mouse model | Bacterial | Untreated | Physical damage to pathogens. Time-dependent bacterial inactivation. Infected mice lost less body weight and had lower concentrations of lung bacteria than those infected from control |
| Quan et al. (2017) [30] | PP microfiber filter | Salt: Sodium chloride | Bioaerosol filtration efficiency, TEM | Viral | Untreated | All challenge viruses were inactivated. Evidence that this is due to hyperosmotic stress on viral envelope |
| Huang et al. (2020) [28] | Prototype | Inherent from LIG | Bacterial live/dead assay, SEM, bioaerosol collection | Bacterial | Commercial filter layer | Antibacterial activity against E. coli: LIG: 8157%, ACF: 2.00%, MBF: 9.13%. SEM: surface disruption bacteria. Aerosolized bacterial efficiency 88.89%. E. coli viability: 0.73 log reduction |
| Tseng et al. (2006) [19] | Surgical mask | Goldshield 5 | Bioaerosol challenge | Bacterial | Untreated | >99.3% antimicrobial efficiency against bacteria on mask surface for all test pathogens |
| Xiong et al. (2021) [21] | Prototype | QAC with boron nitride nanoparticles | Incubation with bacteria | Bacterial | Untreated | Antibacterial rate 99.3% for E. coli and 96.1% for S. aureus through ‘contact killing’ mechanism |
| Majchrzycka et al. (2012) [23] | PP nonwoven | Alkylammonium | Incubation with bacteria, bioaerosol filtration efficiency | Bacterial | Untreated | Biobentonite carrier: no antimicrobial activity. Bioperlite carrier: inoculation and bioaerosol tests 95% of E. coli, 65.5% of S. aureus ‘blocked’ |
| Ren et al. (2018) [32] | NIOSH N95 FFR | N-halamine: MC | Incubation with bacteria, bioaerosol challenge | Viral | Ethanol-soaked fabric | Virus undetectable after 30 min contact. As effective as sodium hypochlorite |
| Demir et al. (2015) [22] | PP nonwoven | N-halamine: MC | Incubation with bacteria, bioaerosol challenge | Bacterial | Untreated | No viable bacteria recovered from treated fabrics or pores |
| Duong-Quy (2020) [29] | Prototype | Plectranthii amboinicii plant oil extract | Subject mask wearing: Bacterial inhibition, aerobic microbial test | Recovered bacteria | Conventional surgical mask | Both conventional and LMC showed sterile rings indicating both resistant to bacteria, no significant difference in radius. Antibacterial ability greater for aerobic microbial testing |
| Woo et al. (2012) [31] | Filters | DAS | Bioaerosol challenge | Viral | Untreated | Very low survivability of MS2 on all filter types treated with DAS. Higher concentration of DAS associated with lower survivability |
| Safety | Breathability | Filtration Efficiency | Stability/Durability | Reusability | Cost/Production | |
|---|---|---|---|---|---|---|
| Borkow et al. (2010) [17] | Copper eluted to air from test mask in 5 h: 0.467 ± 0.47 pg (<105 folds lower than permissible exposure limit) | Filtration efficiency unaffected by treatment | Statement: copper oxide layer does not add ‘significant costs’ | |||
| Li et al. (2006) [18] | No sign of skin allergy/irritation after 1 h 15 min wearing (20 volunteers) | |||||
| Zheng et al. (2016) [24] | ||||||
| Hiragond et al. (2018) [20] | Statement: starch is abundant and low cost | |||||
| Rengasamy et al. (2010) [16] | Antiviral activity only observed at high temp and RH | |||||
| Kumar et al. (2021) [26] | Pressure drop similar for treated mask at low velocity, slight increase at high velocity | Filtration efficiency unaffected by treatment | Rejection efficiency unchanged after multiple treatment cycles. Nonwetting surface properties grant ‘self-cleaning’ ability | Statement: reliable and suitable for industrial production | ||
| Lore et al. (2012) [25] | Elevated pressure drop | Filtration efficiency unaffected by treatment | ||||
| Rubino et al. (2020) [27] | Statement: Salt types safe | No significant rise in pressure drop | Filtration efficiency improved by treatment | Environmental stability: stored at 37 °C, 70, 80, 90% RH for 5 days, antimicrobial properties improved | Statement: ‘safe reusability without further processing’ | Statement: salt types are inexpensive; production cost would be lower compared with melt blowing methods |
| Quan et al. (2017) [30] | Environmental stability: 37 °C 70% RH storage did not affect efficacy | Statement: reusable at normal environmental conditions | Statement: treatment is low-cost | |||
| Huang et al. (2020) [28] | Pressure drop similar for proposed system and MBF standard | ‘Self-reporting of mask conditions’: through response to moisture | Statement: LIG can be created using wide range of carbon precursors allowing easy supply | |||
| Tseng et al. (2006) [19] | Filtration efficiency unaffected by treatment | ‘Decontamination test’ challenging masks with pathogens repeated 1, 2, 4 or 8 days after coating-Efficacy maintained | ||||
| Xiong et al. (2021) [21] | Air permeability decreased with increasing nanocomposite loading. Acceptable at 10% (114.9 mm/s). PM2.5 removal efficiency >90% | Thermal conductivity as proxy for reusability: maintained after 5 cycles of reuse, PM2.5 removal efficiency also unaffected | ||||
| Majchrzycka et al. (2012) [23] | Filtration efficiency unaffected by treatment | Industrial synthesis: Found to be as effective as laboratory | ||||
| Ren et al. (2018) [32] | Statement: MC has low toxicity | Statement: ‘coating procedure is straightforward and inexpensive’ | ||||
| Demir et al. (2015) [22] | Statement: ‘no issues of biocompatibility or toxicity’ (MC is not volatile and does not emit chlorine gas) | Air permeability not affected by treatment | Antimicrobial system deactivated by fluorescent light—storage implications | |||
| Duong-Quy (2020) [29] | Nitric oxide as a biomarker for respiratory inflammation induced by mask–lower in subjects wearing test mask than conventional mask | Subjects reported higher breathability of test mask than conventional mask | Using natural compound; renewable manufacturing | |||
| Woo et al. (2012) [31] | Statement: DAS does not release toxic chemicals | Pressure drop: air resistance reduced in CFs but not PF |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stokes, K.; Peltrini, R.; Bracale, U.; Trombetta, M.; Pecchia, L.; Basoli, F. Enhanced Medical and Community Face Masks with Antimicrobial Properties: A Systematic Review. J. Clin. Med. 2021, 10, 4066. https://doi.org/10.3390/jcm10184066
Stokes K, Peltrini R, Bracale U, Trombetta M, Pecchia L, Basoli F. Enhanced Medical and Community Face Masks with Antimicrobial Properties: A Systematic Review. Journal of Clinical Medicine. 2021; 10(18):4066. https://doi.org/10.3390/jcm10184066
Chicago/Turabian StyleStokes, Katy, Roberto Peltrini, Umberto Bracale, Marcella Trombetta, Leandro Pecchia, and Francesco Basoli. 2021. "Enhanced Medical and Community Face Masks with Antimicrobial Properties: A Systematic Review" Journal of Clinical Medicine 10, no. 18: 4066. https://doi.org/10.3390/jcm10184066
APA StyleStokes, K., Peltrini, R., Bracale, U., Trombetta, M., Pecchia, L., & Basoli, F. (2021). Enhanced Medical and Community Face Masks with Antimicrobial Properties: A Systematic Review. Journal of Clinical Medicine, 10(18), 4066. https://doi.org/10.3390/jcm10184066







