Radiological Evaluation of Cervical Spine Involvement in Rheumatoid Arthritis: A Cross-Sectional Retrospective Study
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wasserman, B.R.; Moskovich, R.; Razi, A.E. Rheumatoid arthritis of the cervical spine--clinical considerations. Bull. NYU Hosp. Jt. Dis. 2011, 69, 136–148. [Google Scholar] [PubMed]
- Neva, M.H.; Myllykangas-Luosujärvi, R.; Kautiainen, H.; Kauppi, M. Mortality associated with cervical spine disorders: A population-based study of 1666 patients with rheumatoid arthritis who died in Finland in 1989. Rheumatology 2001, 40, 123–127. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ahn, J.K.; Hwang, J.-W.; Oh, J.-M.; Lee, J.; Lee, Y.S.; Jeon, C.H.; Cha, H.-S.; Koh, E.-M. Risk factors for development and progression of atlantoaxial subluxation in Korean patients with rheumatoid arthritis. Rheumatol. Int. 2011, 31, 1363–1368. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Xu, W.; Luo, Y.; Zhao, Y.; Liu, Y. Cervical spine involvement risk factors in rheumatoid arthritis: A meta-analysis. Int. J. Rheum. Dis. 2017, 20, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Pope, J. Cervical spine involvement in rheumatoid arthritis over time: Results from a meta-analysis. Arthritis Res. Ther. 2015, 17, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Younes, M.; Belghali, S.; Kriâa, S.; Zrour, S.; Bejia, I.; Touzi, M.; Bergaoui, N. Compared imaging of the rheumatoid cervical spine: Prevalence study and associated factors. Jt. Bone Spine 2009, 76, 361–368. [Google Scholar] [CrossRef] [PubMed]
- McGregor, M. The Significance of Certain Measurements of the Skull in the Diagnosis of Basilar Impression. Br. J. Radiol. 1948, 21, 171–181. [Google Scholar] [CrossRef]
- Yurube, T.; Sumi, M.; Nishida, K.; Miyamoto, H.; Kohyama, K.; Matsubara, T.; Doita, M. Incidence and aggravation of cervical spine instabilities in rheumatoid arthritis: A prospective minimum 5-year follow-up study of patients initially without cervical involvement. Spine 2012, 37, 2136–2144. [Google Scholar] [CrossRef]
- Pavlov, H.; Torg, J.S.; Robie, B.; Jahre, C. Cervical spinal stenosis: Determination with vertebral body ratio method. Radiology 1987, 164, 771–775. [Google Scholar] [CrossRef]
- Ulbrich, E.J.; Schraner, C.; Boesch, C.; Hodler, J.; Busato, A.; Anderson, S.E.; Eigenheer, S.; Zimmermann, H.; Sturzenegger, M. Normative MR Cervical Spinal Canal Dimensions. Radiology 2014, 271, 172–182. [Google Scholar] [CrossRef]
- McHugh, M.L. Interrater reliability: The kappa statistic. Biochem. Med. 2012, 22, 276–282. [Google Scholar] [CrossRef]
- Baek, I.-W.; Bin Joo, Y.; Park, K.-S.; Kim, K.-J. Risk factors for cervical spine instability in patients with rheumatoid arthritis. Clin. Rheumatol. 2021, 40, 547–555. [Google Scholar] [CrossRef]
- Oláh, C.; Kardos, Z.; Kostyál, L.; Hodosi, K.; Tamási, L.; Bereczki, D.; Szekanecz, Z. Assessment of cervical spine involvement in rheumatoid arthritis patients in the era of biologics: A real-life, cross-sectional MRI study. Rheumatol. Int. 2020, 40, 915–921. [Google Scholar] [CrossRef]
- Gillick, J.; Wainwright, J.; Das, K. Rheumatoid Arthritis and the Cervical Spine: A Review on the Role of Surgery. Int. J. Rheumatol. 2015, 2015, 1–12. [Google Scholar] [CrossRef]
- Yonezawa, T.; Tsuji, H.; Matsui, H.; Hirano, N. Subaxial lesions in rheumatoid arthritis. Radiographic factors suggestive of lower cervical myelopathy. Spine 1995, 20, 208–215. [Google Scholar] [CrossRef]
- Chamberlain, W.E. Basilar Impression (Platybasia): A Bizarre Developmental Anomaly of the Occipital Bone and Upper Cervical Spine with Striking and Misleading Neurologic Manifestations. Yale J. Biol. Med. 1939, 11, 487. [Google Scholar]
- Clark, C.R.; Goetz, D.D.; Menezes, A.H. Arthrodesis of the cervical spine in rheumatoid arthritis. J. Bone Jt. Surg. Am. Vol. 1989, 71, 381–392. [Google Scholar] [CrossRef]
- McRae, D.L.; Barnum, A.S. Occipitalization of the atlas. Am. J. Roentgenol. Radium Ther. Nucl. Med. 1953, 70, 23–46. [Google Scholar]
- Redlund-Johnell, I.; Pettersson, H. Radiographic measurements of the cranio-vertebral region. Designed for evaluation of abnormalities in rheumatoid arthritis. Acta Radiol. Diagn. 1984, 25, 23–28. [Google Scholar] [CrossRef]
- Ranawat, C.S.; O’Leary, P.; Pellicci, P.; Tsairis, P.; Marchisello, P.; Dorr, L. Cervical spine fusion in rheumatoid arthritis. J. Bone Jt. Surg. Am. 1979, 61, 1003–1010. [Google Scholar] [CrossRef]
- Fischgold, H.; Metzger, J. Radio-tomography of the impression fractures of the cranial basis. Rev. Du Rhum. Et Des Mal. Osteo-artic. 1952, 19, 261–264. [Google Scholar]
- Wackenheim, A. Roentgen Diagnosis of the Craniovertebral Region; Springer: New York, NY, USA, 1974. [Google Scholar]
- Kauppi, M.; Sakaguchi, M.; Konttinen, Y.T.; Hämäläinen, M. A new method of screening for vertical atlantoaxial dislocation. J. Rheumatol. 1990, 17, 167–172. [Google Scholar] [PubMed]
- Riew, K.D.; Hilibrand, A.S.; Palumbo, M.A.; Sethi, N.; Bohlman, H.H. Diagnosing Basilar Invagination in the Rheumatoid Patient. J. Bone Jt. Surg. Am. Vol. 2001, 83, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Boden, S.D.; Dodge, L.D.; Bohlman, H.H.; Rechtine, G.R. Rheumatoid arthritis of the cervical spine. A long-term analysis with predictors of paralysis and recovery. J. Bone Jt. Surg. Am. 1993, 75, 1282–1297. [Google Scholar] [CrossRef]
- Carotti, M.; Salaffi, F.; Di Carlo, M.; Sessa, F.; Giovagnoni, A. Magnetic resonance imaging of the craniovertebral junction in early rheumatoid arthritis. Skelet. Radiol. 2019, 48, 553–561. [Google Scholar] [CrossRef]
- Han, M.H.; Ryu, J.I.; Kim, C.H.; Cheong, J.H.; Bak, K.H.; Chun, H.J. Influence of systemic bone mineral density on atlantoaxial subluxation in patients with rheumatoid arthritis. Osteoporos. Int. 2017, 37, 1938–2136. [Google Scholar] [CrossRef]
- Rossini, M.; Adami, G.; Viapiana, O.; Idolazzi, L.; Orsolini, G.; Fassio, A.; Giollo, A.; Gatti, D. Osteoporosis: An Independent Determinant of Bone Erosions in Rheumatoid Arthritis? J. Bone Miner. Res. 2017, 32, 2142–2143. [Google Scholar] [CrossRef]
- Blavnsfeldt, A.-B.G.; de Thurah, A.; Thomsen, M.D.; Tarp, S.; Langdahl, B.; Hauge, E.-M. The effect of glucocorticoids on bone mineral density in patients with rheumatoid arthritis: A systematic review and meta-analysis of randomized, controlled trials. Bone 2018, 114, 172–180. [Google Scholar] [CrossRef]
- Iizuka, H.; Nishinome, M.; Sorimachi, Y.; Ara, T.; Nakajima, T.; Iizuka, Y.; Takagishi, K. The characteristics of bony ankylosis of the facet joint of the upper cervical spine in rheumatoid arthritis patients. Eur. Spine J. 2009, 18, 1130–1134. [Google Scholar] [CrossRef]
- Parish, D.C.; Clark, J.A.; Liebowitz, S.M.; Hicks, W.C. Sudden death in rheumatoid arthritis from vertical subluxation of the odontoid process. J. Natl. Med. Assoc. 1990, 82, 297–304. [Google Scholar]
- Sunahara, N.; Matsunaga, S.; Mori, T.; Ijiri, K.; Sakou, T. Clinical Course of Conservatively Managed Rheumatoid Arthritis Patients With Myelopathy. Spine 1997, 22, 2603–2607, discussion 2608. [Google Scholar] [CrossRef]
- Delamarter, R.B.; Bohlman, H.H. Postmortem Osseous and Neuropathologic Analysis of the Rheumatoid Cervical Spine. Spine 1994, 19, 2267–2274. [Google Scholar] [CrossRef]
- Mańczak, M.; Pracoń, G.; Sudoł-Szopińska, I.; Gasik, R. Apparent diffusion coefficient as an indicator of spinal cord compression due to anterior atlanto-axial subluxation in rheumatoid arthritis patients. Eur. Spine J. 2019, 28, 2352–2358. [Google Scholar] [CrossRef]
- Orr, C.K.; Najm, A.; Young, F.; McGarry, T.; Biniecka, M.; Fearon, U.; Veale, D. The Utility and Limitations of CRP, ESR and DAS28-CRP in Appraising Disease Activity in Rheumatoid Arthritis. Front. Med. 2018, 5, 185. [Google Scholar] [CrossRef]
- Weinblatt, M.E. Methotrexate in Rheumatoid Arthritis: A Quarter Century of Development. Trans. Am. Clin. Clim. Assoc. 2013, 124, 16–25. [Google Scholar]
- Kauppi, M.J.; Neva, M.H.; Laiho, K.; Kautiainen, H.; Luukkainen, R.; Karjalainen, A.; Hannonen, P.J.; Leirisalo-Repo, M.; Korpela, M.; Ilva, K.; et al. Rheumatoid Atlantoaxial Subluxation Can Be Prevented by Intensive Use of Traditional Disease Modifying Antirheumatic Drugs. J. Rheumatol. 2009, 36, 273–278. [Google Scholar] [CrossRef]
- Salli, A.; Sahin, N.; Paksoy, Y.; Kucuksarac, S.; Ugurlu, H. Treatment of Periodontoid Pannus With Infliximab in a Patient With Rheumatoid Arthritis. JCR: J. Clin. Rheumatol. 2009, 15, 250–251. [Google Scholar] [CrossRef]
- Sandström, T.; Rantalaiho, V.; Yli-Kerttula, T.; Kautiainen, H.; Malmi, T.; Karjalainen, A. Cervical Spine Involvement is very rare in Patients with Rheumatoid Arthritis Treated actively with Treat to Target Strategy. Ten-Year Results of the NEORACo Study. J. Rheumatol. 2019, 19013. [Google Scholar] [CrossRef]
- Takahashi, S.; Suzuki, A.; Koike, T.; Yamada, K.; Yasuda, H.; Tada, M.; Sugioka, Y.; Okano, T.; Nakamura, H. Current prevalence and characteristics of cervical spine instability in patients with rheumatoid arthritis in the era of biologics. Mod. Rheumatol. 2014, 24, 904–909. [Google Scholar] [CrossRef]
- Kaito, T.; Hosono, N.; Ohshima, S.; Ohwaki, H.; Takenaka, S.; Fujiwara, H.; Makino, T.; Yonenobu, K. Effect of Biological Agents on Cervical Spine Lesions in Rheumatoid Arthritis. Spine 2012, 37, 1742–1746. [Google Scholar] [CrossRef]
- Kaito, T.; Ohshima, S.; Fujiwara, H.; Makino, T.; Yonenobu, K. Predictors for the progression of cervical lesion in rheumatoid arthritis under the treatment of biological agents. Spine 2013, 38, 2258–2263. [Google Scholar] [CrossRef]
- Kauppi, M.; Neva, M.H. Sensitivity of lateral view cervical spine radiographs taken in the neutral position in atlantoaxial subluxation in rheumatic diseases. Clin. Rheumatol. 1998, 17, 511–514. [Google Scholar] [CrossRef]
- Hung, S.-C.; Wu, H.-M.; Guo, W.-Y. Revisiting Anterior Atlantoaxial Subluxation with Overlooked Information on MR Images. Am. J. Neuroradiol. 2009, 31, 838–843. [Google Scholar] [CrossRef]
- Sudoł-Szopińska, I.; Jans, L.; Teh, J. Rheumatoid arthritis: What do MRI and ultrasound show. J. Ultrason. 2017, 17, 5–16. [Google Scholar] [CrossRef]
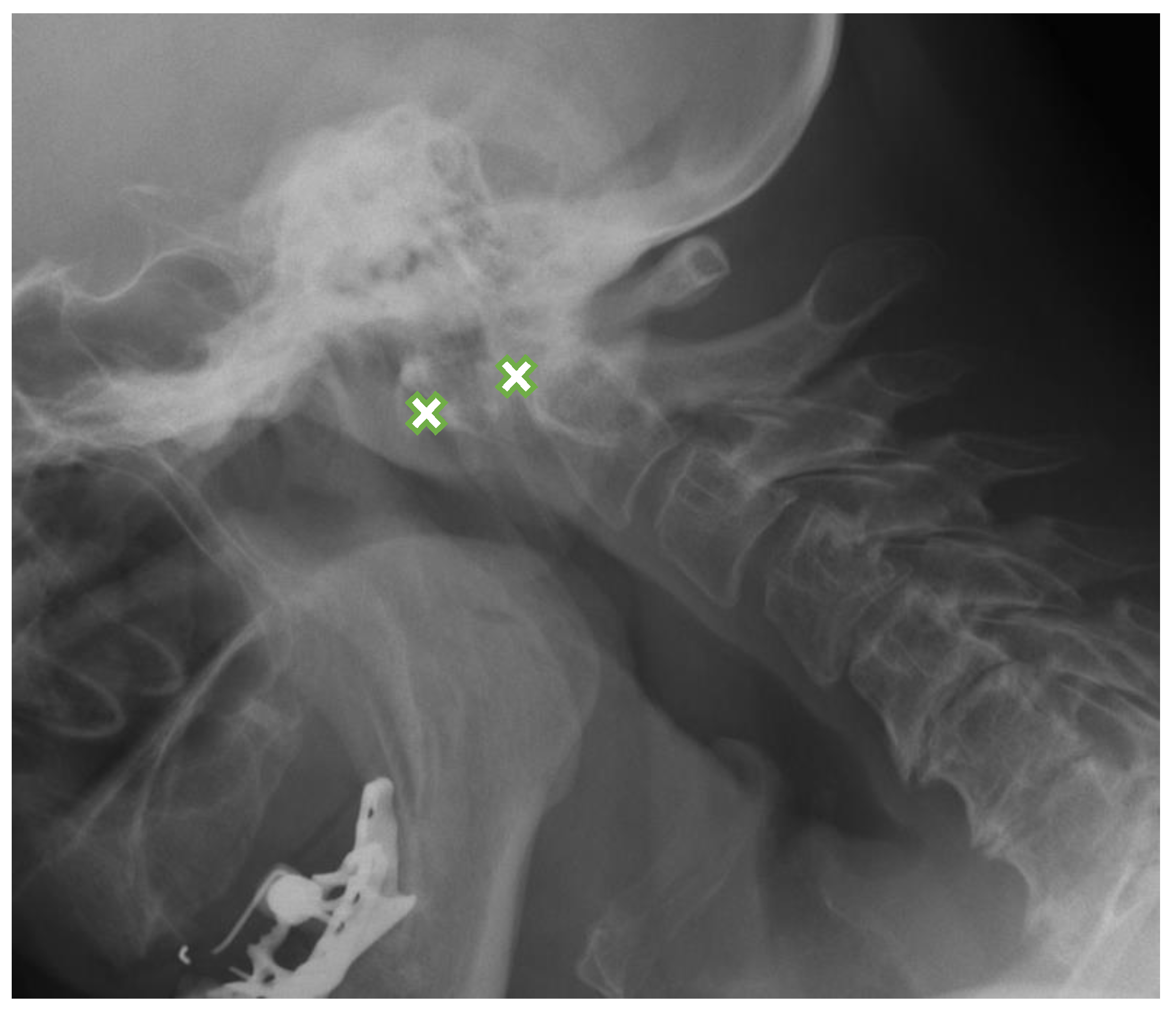
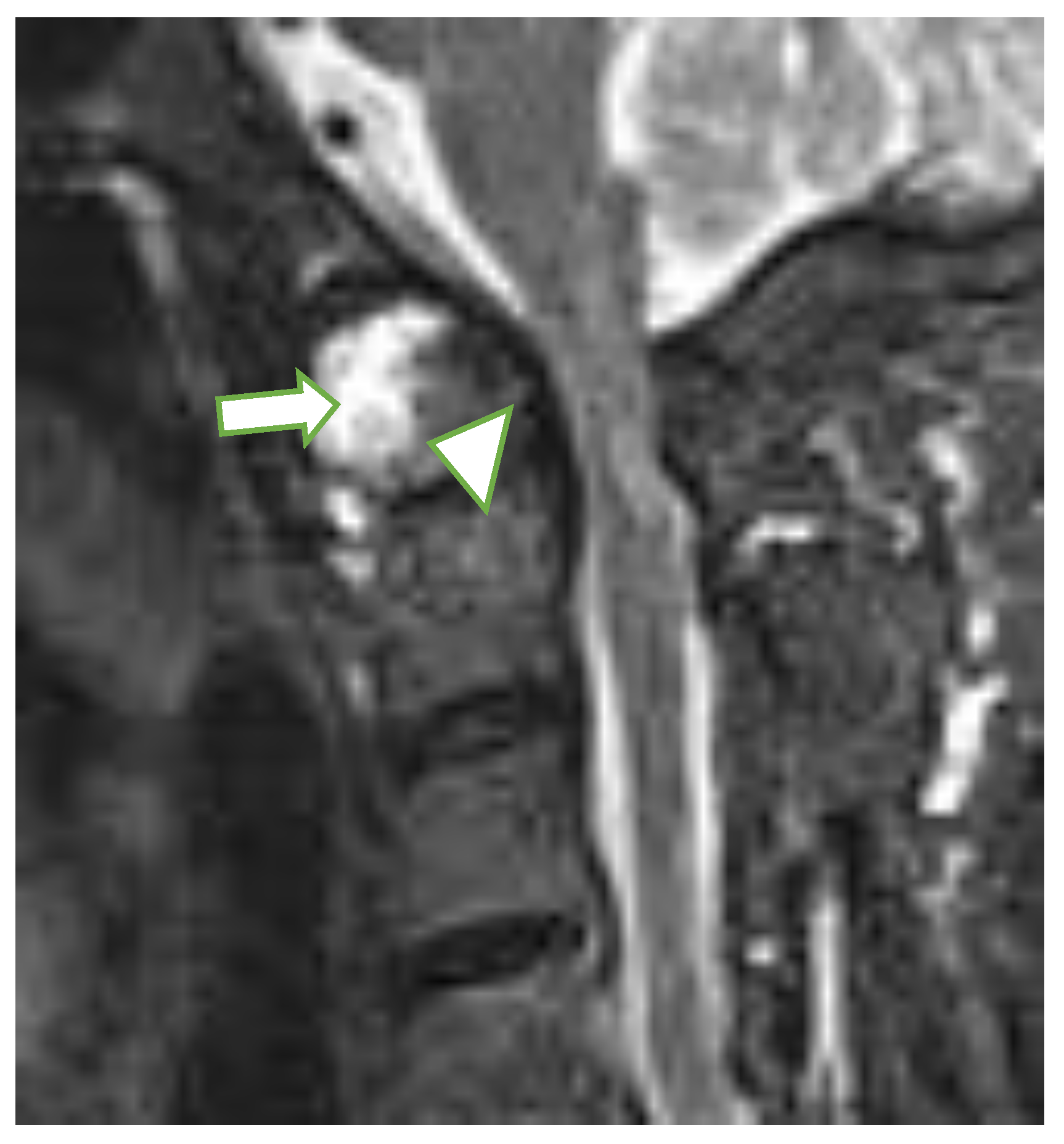
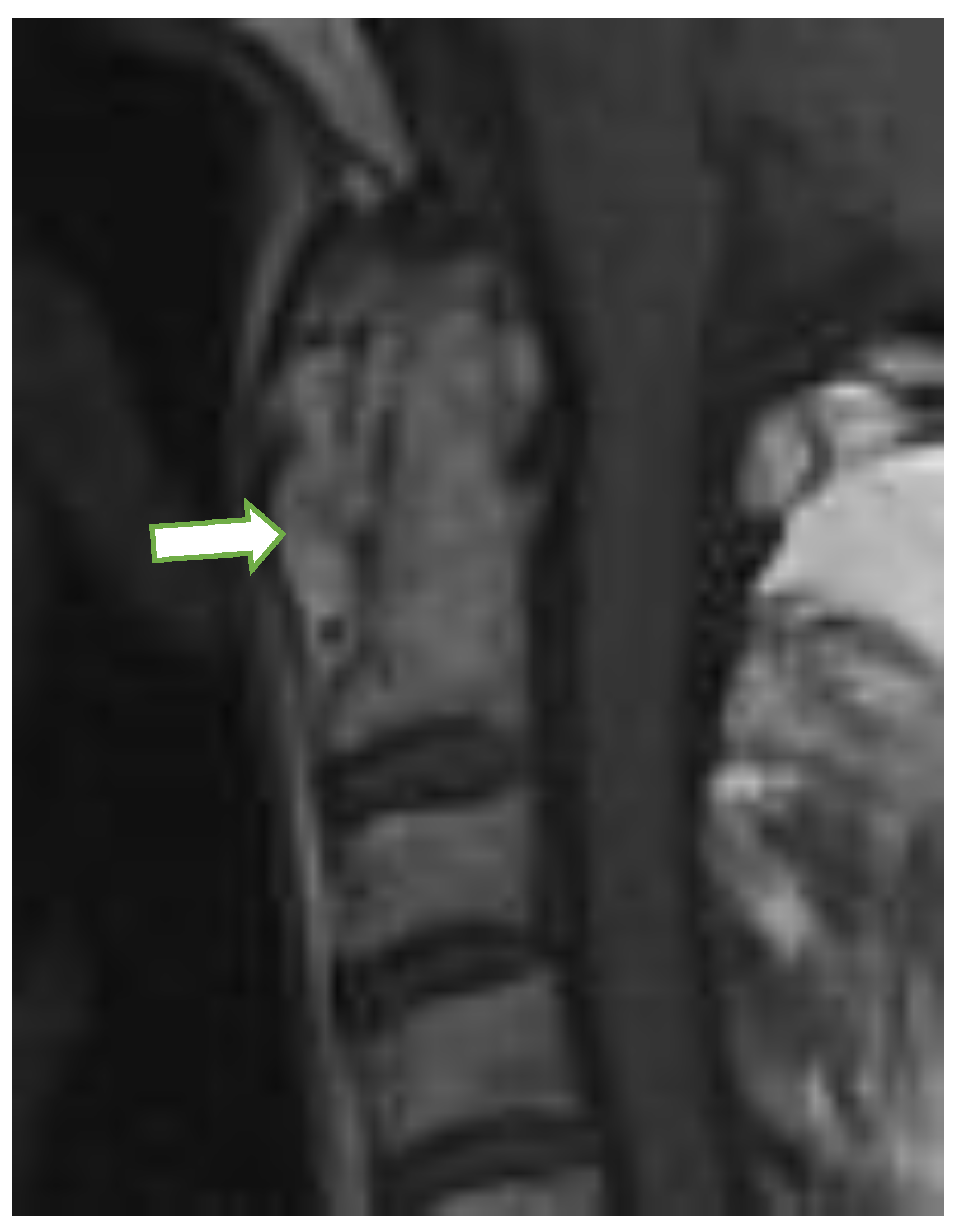
| MRI Lesions | Radiographic Lesions | Percentage | Control n (%) | Interobserver Kappa Value | |
|---|---|---|---|---|---|
| C1/C2 level | |||||
| BME | 11 | not applicable | 11 (5%) | 1 (0.5%) | 0.91 |
| Effusions | 26 | not applicable | 26 (11%) | 5 (2.5%) | 0.89 |
| Pannus | 50 | not applicable | 50 (21%) | 1 (0.5%) | 0.93 |
| Contrast enhancement | 12 | not applicable | 12 (5%) | 0 (0%) | 0.83 |
| Dens erosions | 36 | 11 | 36 (15%) * | 2 (1%) | 0.89 |
| Anterior AAS | 78 | 140 | 140 (58%) # | 7 (3.5%) | 0.79 |
| Posterior AAS | 7 | 0 | 7 (3%) | 0 (0%) | 0.83 |
| Lateral AAS | 11 | not applicable | 11 (5%) | 0 (0%) | 0.84 |
| Vertical AAS | 25 | 27 | 25 (10%) * | 2 (1%) | 0.93 |
| Brain steam compression | 8 | not applicable | 8 (3%) | 2 (1%) ns | 0.95 |
| Subaxial C2-C7 level | |||||
| BME | 11 | not applicable | 11 (5%) | 6 (3%) ns | 0.86 |
| Effusions | 1 | not applicable | 1 (0.4%) | 0 (0%) ns | 0.67 |
| Pannus | 1 | not applicable | 1 (0.4%) | 0 (0%) ns | 0.67 |
| Contrast enhancement | 2 | not applicable | 2 (1%) | 2 (1%) ns | 0.66 |
| SAS | 102 | 139 | 139 (58%) # | 78 (39%) | 0.83 |
| Whole cervical spine C1-C7 | |||||
| Ankylosis | 14 | 19 | 23 (10%) $ | 3 (1.5%) | 0.87 |
| Spinous process erosions | 0 | 14 | 14 (6%) | 0 (0%) | 0.83 |
| Demineralization | not applicable | 114 | 114 (48%) | 35 (18%) | 0.76 |
| Myelopathy | 14 | not applicable | 14 (6%) | 7 (3.5%) ns | 0.92 |
| Cervical spine compression | 72 | not applicable | 72 (30%) | 59 (30%) ns | 0.95 |
| Spinal stenosis | 93 | 64 | 93 (39%) * | 72 (36%) ns | 0.92 |
| Total | 138 (58%) | 159 (66%) | 179 (75%) | 95 (48%) | |
| Rheumatoid Arthritis | Cervical Spine Lesions on Radiographs and/or MRI (n = 179) | No Abnormalities on Imaging (n = 61) | p |
|---|---|---|---|
| Age (years) | 61.0 [52.0–68.0] | 59.5 [50.5–66.8] | 0.491 |
| Sex (female; %) | 154 (86%) | 53 (87%) | 0.868 |
| Age at onset (years) | 41.8 ± 13.6 | 45.3 ± 16.3 | 0.229 |
| Disease duration (years) | 15.0 [9.0–27.0] | 9.0 [4.0–15.8] | 0.007 |
| CRP (mg/mL) | 19.0 [7.0–32.0] | 11.5 [6.3–19.8] | 0.016 |
| ESR (mm/h) | 35.0 [19.0–58.0] | 28.0 [13.3–48.8] | 0.025 |
| ANA positivity n (%) | 20 (31%) | 14 (39%) | 0.408 |
| RF positivity n (%) | 39 (60%) | 12 (35%) | 0.010 |
| Anti-CCP positivity n (%) | 54 (83%) | 24 (67%) | 0.060 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kotecki, M.; Gasik, R.; Głuszko, P.; Sudoł-Szopińska, I. Radiological Evaluation of Cervical Spine Involvement in Rheumatoid Arthritis: A Cross-Sectional Retrospective Study. J. Clin. Med. 2021, 10, 4587. https://doi.org/10.3390/jcm10194587
Kotecki M, Gasik R, Głuszko P, Sudoł-Szopińska I. Radiological Evaluation of Cervical Spine Involvement in Rheumatoid Arthritis: A Cross-Sectional Retrospective Study. Journal of Clinical Medicine. 2021; 10(19):4587. https://doi.org/10.3390/jcm10194587
Chicago/Turabian StyleKotecki, Mateusz, Robert Gasik, Piotr Głuszko, and Iwona Sudoł-Szopińska. 2021. "Radiological Evaluation of Cervical Spine Involvement in Rheumatoid Arthritis: A Cross-Sectional Retrospective Study" Journal of Clinical Medicine 10, no. 19: 4587. https://doi.org/10.3390/jcm10194587
APA StyleKotecki, M., Gasik, R., Głuszko, P., & Sudoł-Szopińska, I. (2021). Radiological Evaluation of Cervical Spine Involvement in Rheumatoid Arthritis: A Cross-Sectional Retrospective Study. Journal of Clinical Medicine, 10(19), 4587. https://doi.org/10.3390/jcm10194587






