Endoscopic Management of Pancreatic Fluid Collections
Abstract
1. Introduction
2. Classification of Pancreatic Fluid Collections
3. Current Approach to Management of Infected Pancreatic Fluid Collections
4. Endoscopic Ultrasound-Guided Transmural Drainage Versus Conventional Transmural Drainage
5. Types of Stents for Drainage of Pancreatic Fluid Collections
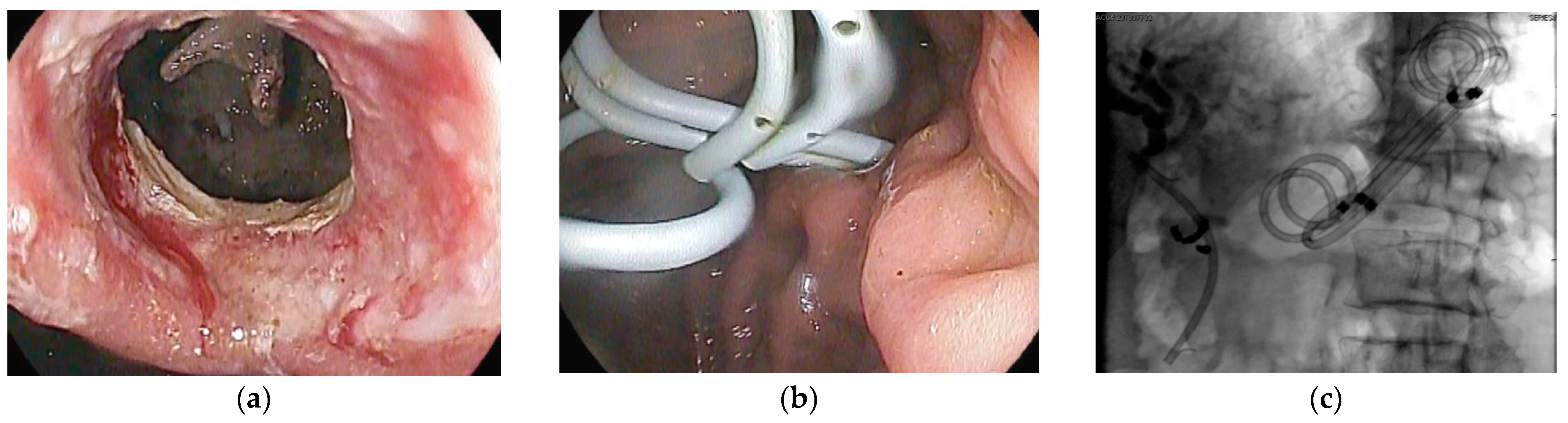
6. Endoscopic Management of Pseudocysts
7. Endoscopic Management of Walled-Off Pancreatic Necrosis
8. Direct Endoscopic Necrosectomy
9. Dual-Modality Drainage
10. Disconnected Duct Syndrome
11. Multiple Transluminal Gateway Technique
12. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tyberg, A.; Karia, K.; Gabr, M.; Desai, A.; Doshi, R.; Gaidhane, M.; Sharaiha, R.Z.; Kahaleh, M. Management of pancreatic fluid collections: A comprehensive review of the literature. World J. Gastroenterol. 2016, 22, 2256. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yu, Y.; Li, P.; Zhang, S.-T. Advancements in the endoscopic treatment of pancreatic fluid collections. Chronic Dis. Transl. Med. 2020, 6, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Bradley, E.L., 3rd. The natural and unnatural history of pancreatic fluid collections associated with acute pancreatitis. Dig. Dis. Sci. 2014, 59, 908–910. [Google Scholar] [CrossRef] [PubMed]
- Bradley, E.L., 3rd; Clements, J.L., Jr. Implications of diagnostic ultrasound in the surgical management of pancreatic pseudocysts. Am. J. Surg. 1974, 127, 163–173. [Google Scholar] [CrossRef]
- Bradley, E.L. A clinically based classification system for acute pancreatitis: Summary of the International Symposium on Acute Pancreatitis, Atlanta, Ga, September 11 through 13, 1992. Arch. Surg. 1993, 128, 586–590. [Google Scholar] [CrossRef]
- Banks, P.A.; Bollen, T.L.; Dervenis, C.; Gooszen, H.G.; Johnson, C.D.; Sarr, M.G.; Tsiotos, G.G.; Vege, S.S. Classification of acute pancreatitis—2012: Revision of the Atlanta classification and definitions by international consensus. Gut 2013, 62, 102–111. [Google Scholar] [CrossRef]
- Cui, M.L.; Kim, K.H.; Kim, H.G.; Han, J.; Kim, H.; Cho, K.B.; Jung, M.K.; Cho, C.M.; Kim, T.N. Incidence, risk factors and clinical course of pancreatic fluid collections in acute pancreatitis. Dig. Dis. Sci. 2014, 59, 1055–1062. [Google Scholar] [CrossRef]
- Baron, T.H.; DiMaio, C.J.; Wang, A.Y.; Morgan, K.A. American Gastroenterological Association Clinical Practice Update: Management of Pancreatic Necrosis. Gastroenterology 2020, 158, 67–75.e1. [Google Scholar] [CrossRef]
- Freeman, M.L.; Werner, J.; Van Santvoort, H.C.; Baron, T.H.; Besselink, M.G.; Windsor, J.A.; Horvath, K.D.; Bollen, T.L.; Vege, S.S. Interventions for necrotizing pancreatitis: Summary of a multidisciplinary consensus conference. Pancreas 2012, 41, 1176–1194. [Google Scholar] [CrossRef]
- van Baal, M.C.; Bollen, T.L.; Bakker, O.J.; van Goor, H.; Boermeester, M.A.; Dejong, C.H.; Gooszen, H.G.; van der Harst, E.; van Eijck, C.H.; van Santvoort, H.C. The role of routine fine-needle aspiration in the diagnosis of infected necrotizing pancreatitis. Surgery 2014, 155, 442–448. [Google Scholar] [CrossRef]
- Wolbrink, D.R.; Kolwijck, E.; Ten Oever, J.; Horvath, K.D.; Bouwense, S.A.; Schouten, J.A. Management of infected pancreatic necrosis in the intensive care unit: A narrative review. Clin. Microbiol. Infect. 2020, 26, 18–25. [Google Scholar] [CrossRef] [PubMed]
- van Grinsven, J.; van Brunschot, S.; van Baal, M.C.; Besselink, M.G.; Fockens, P.; van Goor, H.; van Santvoort, H.C.; Bollen, T.L.; Group, D.P.S. Natural history of gas configurations and encapsulation in necrotic collections during necrotizing pancreatitis. J. Gastrointest. Surg. 2018, 22, 1557–1564. [Google Scholar] [CrossRef] [PubMed]
- van Grinsven, J.; Van Dijk, S.M.; Dijkgraaf, M.G.; Boermeester, M.A.; Bollen, T.L.; Bruno, M.J.; Van Brunschot, S.; Dejong, C.H.; Van Eijck, C.H.; Van Lienden, K.P. Postponed or immediate drainage of infected necrotizing pancreatitis (POINTER trial): Study protocol for a randomized controlled trial. Trials 2019, 20, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Van Santvoort, H.; Besselink, M.; Cirkel, G.; Gooszen, H. A nationwide Dutch study into the optimal treatment of patients with infected necrotising pancreatitis: The PANTER trial. Ned. Tijdschr. Voor Geneeskd. 2006, 150, 1844–1846. [Google Scholar]
- Bakker, O.J.; van Santvoort, H.C.; van Brunschot, S.; Geskus, R.B.; Besselink, M.G.; Bollen, T.L.; van Eijck, C.H.; Fockens, P.; Hazebroek, E.J.; Nijmeijer, R.M. Endoscopic transgastric vs surgical necrosectomy for infected necrotizing pancreatitis: A randomized trial. JAMA 2012, 307, 1053–1061. [Google Scholar] [CrossRef]
- van Brunschot, S.; van Grinsven, J.; van Santvoort, H.C.; Bakker, O.J.; Besselink, M.G.; Boermeester, M.A.; Bollen, T.L.; Bosscha, K.; Bouwense, S.A.; Bruno, M.J. Endoscopic or surgical step-up approach for infected necrotising pancreatitis: A multicentre randomised trial. Lancet 2018, 391, 51–58. [Google Scholar] [CrossRef]
- Cremer, M.; Deviere, J.; Engelholm, L. Endoscopic management of cysts and pseudocysts in chronic pancreatitis: Long-term follow-up after 7 years of experience. Gastrointest. Endosc. 1989, 35, 1–9. [Google Scholar] [CrossRef]
- Binmoeller, K.F.; Seifert, H.; Walter, A.; Soehendra, N. Transpapillary and transmural drainage of pancreatic pseudocysts. Gastrointest. Endosc. 1995, 42, 219–224. [Google Scholar] [CrossRef]
- Grimm, H.; Binmoeller, K.F.; Soehendra, N. Endosonography-guided drainage of a pancreatic pseudocyst. Gastrointest. Endosc. 1992, 38, 170–171. [Google Scholar] [CrossRef]
- Kahaleh, M.; Shami, V.M.; Conaway, M.R.; Tokar, J.; Rockoff, T.; De La Rue, S.A.; de Lange, E.; Bassignani, M.; Gay, S.; Adams, R.B.; et al. Endoscopic ultrasound drainage of pancreatic pseudocyst: A prospective comparison with conventional endoscopic drainage. Endoscopy 2006, 38, 355–359. [Google Scholar] [CrossRef]
- Varadarajulu, S.; Christein, J.D.; Tamhane, A.; Drelichman, E.R.; Wilcox, C.M. Prospective randomized trial comparing EUS and EGD for transmural drainage of pancreatic pseudocysts (with videos). Gastrointest. Endosc. 2008, 68, 1102–1111. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Lee, S.; Moon, S.-H.; Choi, S.; Jung, S.; Seo, D.; Kim, M.-H. Endoscopic ultrasound-guided versus conventional transmural drainage for pancreatic pseudocysts: A prospective randomized trial. Endoscopy 2009, 41, 842–848. [Google Scholar] [CrossRef] [PubMed]
- Talreja, J.P.; Shami, V.M.; Ku, J.; Morris, T.D.; Ellen, K.; Kahaleh, M. Transenteric drainage of pancreatic-fluid collections with fully covered self-expanding metallic stents (with video). Gastrointest. Endosc. 2008, 68, 1199–1203. [Google Scholar] [CrossRef] [PubMed]
- Penn, D.E.; Draganov, P.V.; Wagh, M.S.; Forsmark, C.E.; Gupte, A.R.; Chauhan, S.S. Prospective evaluation of the use of fully covered self-expanding metal stents for EUS-guided transmural drainage of pancreatic pseudocysts. Gastrointest. Endosc. 2012, 76, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Sarkaria, S.; Sethi, A.; Rondon, C.; Lieberman, M.; Srinivasan, I.; Weaver, K.; Turner, B.G.; Sundararajan, S.; Berlin, D.; Gaidhane, M.; et al. Pancreatic necrosectomy using covered esophageal stents: A novel approach. J. Clin. Gastroenterol. 2014, 48, 145–152. [Google Scholar] [CrossRef]
- Tan, S.; Zhong, C.; Ren, Y.; Luo, X.; Xu, J.; Peng, Y.; Fu, X.; Tang, X. Are Lumen-Apposing Metal Stents More Effective Than Plastic Stents for the Management of Pancreatic Fluid Collections: An Updated Systematic Review and Meta-analysis. Gastroenterol. Res. Pract. 2020, 2020, 4952721. [Google Scholar] [CrossRef]
- Hammad, T.; Khan, M.A.; Alastal, Y.; Lee, W.; Nawras, A.; Ismail, M.K.; Kahaleh, M. Efficacy and Safety of Lumen-Apposing Metal Stents in Management of Pancreatic Fluid Collections: Are They Better Than Plastic Stents? A Systematic Review and Meta-Analysis. Dig. Dis. Sci. 2018, 63, 289–301. [Google Scholar] [CrossRef]
- Fugazza, A.; Sethi, A.; Trindade, A.J.; Troncone, E.; Devlin, J.; Khashab, M.A.; Vleggaar, F.P.; Bogte, A.; Tarantino, I.; Deprez, P.H. International multicenter comprehensive analysis of adverse events associated with lumen-apposing metal stent placement for pancreatic fluid collection drainage. Gastrointest. Endosc. 2020, 91, 574–583. [Google Scholar] [CrossRef]
- Aburajab, M.; Smith, Z.; Khan, A.; Dua, K. Safety and efficacy of lumen-apposing metal stents with and without simultaneous double-pigtail plastic stents for draining pancreatic pseudocyst. Gastrointest. Endosc. 2018, 87, 1248–1255. [Google Scholar] [CrossRef]
- Yang, J.; Chen, Y.I.; Friedland, S.; Holmes, I.; Paiji, C.; Law, R.; Hosmer, A.; Stevens, T.; Matheus, F.; Pawa, R.; et al. Lumen-apposing stents versus plastic stents in the management of pancreatic pseudocysts: A large, comparative, international, multicenter study. Endoscopy 2019, 51, 1035–1043. [Google Scholar]
- Chen, Y.-I.; Khashab, M.A.; Adam, V.; Bai, G.; Singh, V.K.; Bukhari, M.; Gutierrez, O.B.; Elmunzer, B.J.; Moran, R.A.; Fayad, L. Plastic stents are more cost-effective than lumen-apposing metal stents in management of pancreatic pseudocysts. Endosc. Int. Open 2018, 6, E780. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.A.; Kowalski, T.E.; Loren, D.E.; Khalid, A.; Soomro, A.; Mazhar, S.M.; Isby, L.; Kahaleh, M.; Karia, K.; Yoo, J.; et al. Fully covered self-expanding metal stents versus lumen-apposing fully covered self-expanding metal stent versus plastic stents for endoscopic drainage of pancreatic walled-off necrosis: Clinical outcomes and success. Gastrointest. Endosc. 2017, 85, 758–765. [Google Scholar] [CrossRef]
- Bang, J.Y.; Navaneethan, U.; Hasan, M.K.; Sutton, B.; Hawes, R.; Varadarajulu, S. Non-superiority of lumen-apposing metal stents over plastic stents for drainage of walled-off necrosis in a randomised trial. Gut 2019, 68, 1200–1209. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.I.; Yang, J.; Friedland, S.; Holmes, I.; Law, R.; Hosmer, A.; Stevens, T.; Franco, M.C.; Jang, S.; Pawa, R.; et al. Lumen apposing metal stents are superior to plastic stents in pancreatic walled-off necrosis: A large international multicenter study. Endosc. Int. Open 2019, 7, E347–E354. [Google Scholar] [CrossRef] [PubMed]
- Hori, Y.; Yoshida, M.; Hayashi, K.; Naitoh, I.; Kato, A.; Miyabe, K.; Kataoka, H. Endoscopic drainage using a lumen-apposing metal stent under contrast-enhanced harmonic endoscopic ultrasonography guidance. Endoscopy 2019, 51, E187–E188. [Google Scholar] [CrossRef]
- Parsa, N.; Nieto, J.M.; Powers, P.; Mitsuhashi, S.; Abdelqader, A.; Hadzinakos, G.; Anderloni, A.A.; Fugazza, A.; James, T.W.; Arlt, A.; et al. Endoscopic ultrasound-guided drainage of pancreatic walled-off necrosis using 20-mm versus 15-mm lumen-apposing metal stents: An international, multicenter, case-matched study. Endoscopy 2020, 52, 211–219. [Google Scholar] [CrossRef]
- Hyun, J.J.; Sahar, N.; Singla, A.; Ross, A.S.; Irani, S.S.; Gan, S.I.; Larsen, M.C.; Kozarek, R.A.; Gluck, M. Outcomes of Infected versus Symptomatic Sterile Walled-Off Pancreatic Necrosis Treated with a Minimally Invasive Therapy. Gut Liver 2019, 13, 215–222. [Google Scholar] [CrossRef]
- Thompson, C.C.; Kumar, N.; Slattery, J.; Clancy, T.E.; Ryan, M.B.; Ryou, M.; Swanson, R.S.; Banks, P.A.; Conwell, D.L. A standardized method for endoscopic necrosectomy improves complication and mortality rates. Pancreatology 2016, 16, 66–72. [Google Scholar] [CrossRef]
- Baron, T.H.; Thaggard, W.G.; Morgan, D.E.; Stanley, R.J. Endoscopic therapy for organized pancreatic necrosis. Gastroenterology 1996, 111, 755–764. [Google Scholar] [CrossRef]
- Seifert, H.; Biermer, M.; Schmitt, W.; Jürgensen, C.; Will, U.; Gerlach, R.; Kreitmair, C.; Meining, A.; Wehrmann, T.; Rösch, T. Transluminal endoscopic necrosectomy after acute pancreatitis: A multicentre study with long-term follow-up (the GEPARD Study). Gut 2009, 58, 1260–1266. [Google Scholar] [CrossRef]
- Bazarbashi, A.N.; Ge, P.S.; de Moura, D.T.H.; Thompson, C.C. A novel endoscopic morcellator device to facilitate direct necrosectomy of solid walled-off necrosis. Endoscopy 2019, 51, E396–E397. [Google Scholar] [CrossRef] [PubMed]
- Rizzatti, G.; Rimbaș, M.; De Riso, M.; Impagnatiello, M.; Costamagna, G.; Larghi, A. Endorotor-based endoscopic necrosectomy avoiding the superior mesenteric artery. Endoscopy 2020, 52, E420–E421. [Google Scholar] [CrossRef] [PubMed]
- van der Wiel, S.E.; May, A.; Poley, J.W.; Grubben, M.; Wetzka, J.; Bruno, M.J.; Koch, A.D. Preliminary report on the safety and utility of a novel automated mechanical endoscopic tissue resection tool for endoscopic necrosectomy: A case series. Endosc. Int. Open 2020, 8, E274–E280. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.S.; Irani, S.; Gan, S.I.; Rocha, F.; Siegal, J.; Fotoohi, M.; Hauptmann, E.; Robinson, D.; Crane, R.; Kozarek, R.; et al. Dual-modality drainage of infected and symptomatic walled-off pancreatic necrosis: Long-term clinical outcomes. Gastrointest. Endosc. 2014, 79, 929–935. [Google Scholar] [CrossRef]
- Gluck, M.; Ross, A.; Irani, S.; Lin, O.; Gan, S.I.; Fotoohi, M.; Hauptmann, E.; Crane, R.; Siegal, J.; Robinson, D.H.; et al. Dual modality drainage for symptomatic walled-off pancreatic necrosis reduces length of hospitalization, radiological procedures, and number of endoscopies compared to standard percutaneous drainage. J. Gastrointest. Surg. 2012, 16, 248–256; discussion 256–257. [Google Scholar] [CrossRef]
- Yokoi, Y.; Kikuyama, M.; Kurokami, T.; Sato, T. Early dual drainage combining transpapillary endotherapy and percutaneous catheter drainage in patients with pancreatic fistula associated with severe acute pancreatitis. Pancreatology 2016, 16, 497–507. [Google Scholar] [CrossRef]
- Bang, J.Y.; Wilcox, C.M.; Navaneethan, U.; Hasan, M.K.; Peter, S.; Christein, J.; Hawes, R.; Varadarajulu, S. Impact of Disconnected Pancreatic Duct Syndrome on the Endoscopic Management of Pancreatic Fluid Collections. Ann. Surg. 2018, 267, 561–568. [Google Scholar] [CrossRef]
- Vanek, P.; Trikudanathan, G.; Freeman, M.L. Diagnosing Disconnected Pancreatic Duct Syndrome: Many Disconnects, Few Answers. Dig. Dis. Sci. 2020, 1–3. [Google Scholar] [CrossRef]
- Jang, J.W.; Kim, M.H.; Oh, D.; Cho, D.H.; Song, T.J.; Park, D.H.; Lee, S.S.; Seo, D.W.; Lee, S.K.; Moon, S.H. Factors and outcomes associated with pancreatic duct disruption in patients with acute necrotizing pancreatitis. Pancreatology 2016, 16, 958–965. [Google Scholar] [CrossRef]
- Kozarek, R.A.; Traverso, L.W. Pancreatic fistulas: Etiology, consequences, and treatment. Gastroenterologist 1996, 4, 238–244. [Google Scholar]
- Maatman, T.K.; Mahajan, S.; Roch, A.M.; Ceppa, E.P.; House, M.G.; Nakeeb, A.; Schmidt, C.M.; Zyromski, N.J. Disconnected pancreatic duct syndrome predicts failure of percutaneous therapy in necrotizing pancreatitis. Pancreatology 2020, 20, 362–368. [Google Scholar] [CrossRef] [PubMed]
- Larsen, M.; Kozarek, R. Management of pancreatic ductal leaks and fistulae. J. Gastroenterol. Hepatol. 2014, 29, 1360–1370. [Google Scholar] [CrossRef] [PubMed]
- Téllez-Aviña, F.I.; Casasola-Sánchez, L.E.; Ramírez-Luna, M.; Saúl, Á.; Murcio-Pérez, E.; Chan, C.; Uscanga, L.; Duarte-Medrano, G.; Valdovinos-Andraca, F. Permanent Indwelling Transmural Stents for Endoscopic Treatment of Patients With Disconnected Pancreatic Duct Syndrome: Long-term Results. J. Clin. Gastroenterol. 2018, 52, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Bang, J.Y.; Mel Wilcox, C.; Arnoletti, J.P.; Varadarajulu, S. Importance of Disconnected Pancreatic Duct Syndrome in Recurrence of Pancreatic Fluid Collections Initially Drained Using Lumen-Apposing Metal Stents. Clin. Gastroenterol. Hepatol. 2020. [Google Scholar] [CrossRef]
- Varadarajulu, S.; Wilcox, C.M. Endoscopic placement of permanent indwelling transmural stents in disconnected pancreatic duct syndrome: Does benefit outweigh the risks? Gastrointest. Endosc. 2011, 74, 1408–1412. [Google Scholar] [CrossRef] [PubMed]
- Rana, S.S.; Sharma, R.; Sharma, V.; Chhabra, P.; Gupta, R.; Bhasin, D.K. Prevention of recurrence of fluid collections in walled off pancreatic necrosis and disconnected pancreatic duct syndrome: Comparative study of one versus two long term transmural stents. Pancreatology 2016, 16, 687–688. [Google Scholar] [CrossRef]
- Wang, L.; Elhanafi, S.; Storm, A.C.; Topazian, M.D.; Majumder, S.; Abu Dayyeh, B.K.; Levy, M.J.; Petersen, B.T.; Martin, J.A.; Chari, S.T.; et al. Impact of disconnected pancreatic duct syndrome on endoscopic ultrasound-guided drainage of pancreatic fluid collections. Endoscopy 2020. [Google Scholar] [CrossRef] [PubMed]
- Varadarajulu, S.; Phadnis, M.A.; Christein, J.D.; Wilcox, C.M. Multiple transluminal gateway technique for EUS-guided drainage of symptomatic walled-off pancreatic necrosis. Gastrointest. Endosc. 2011, 74, 74–80. [Google Scholar] [CrossRef]
- Bang, J.Y.; Varadarajulu, S. Management of walled-off necrosis using the multiple transluminal gateway technique with the Hot AXIOS System. Dig. Endosc. 2016, 28, 103. [Google Scholar] [CrossRef]
- Bang, J.Y.; Varadarajulu, S. Management of Hemorrhagic Walled-Off Necrosis Using the Multiple Transluminal Gateway Technique. Am. J. Gastroenterol. 2016, 111, 457. [Google Scholar] [CrossRef]
- Binda, C.; Dabizzi, E.; Anderloni, A.; Cennamo, V.; Fiscaletti, M.; Fugazza, A.; Jovine, E.; Ercolani, G.; Gasbarrini, A.; Fabbri, C. Single-step endoscopic ultrasound-guided multiple gateway drainage of complex walled-off necrosis with lumen apposing metal stents. Eur. J. Gastroenterol. Hepatol. 2020, 32, 1401–1404. [Google Scholar] [CrossRef] [PubMed]

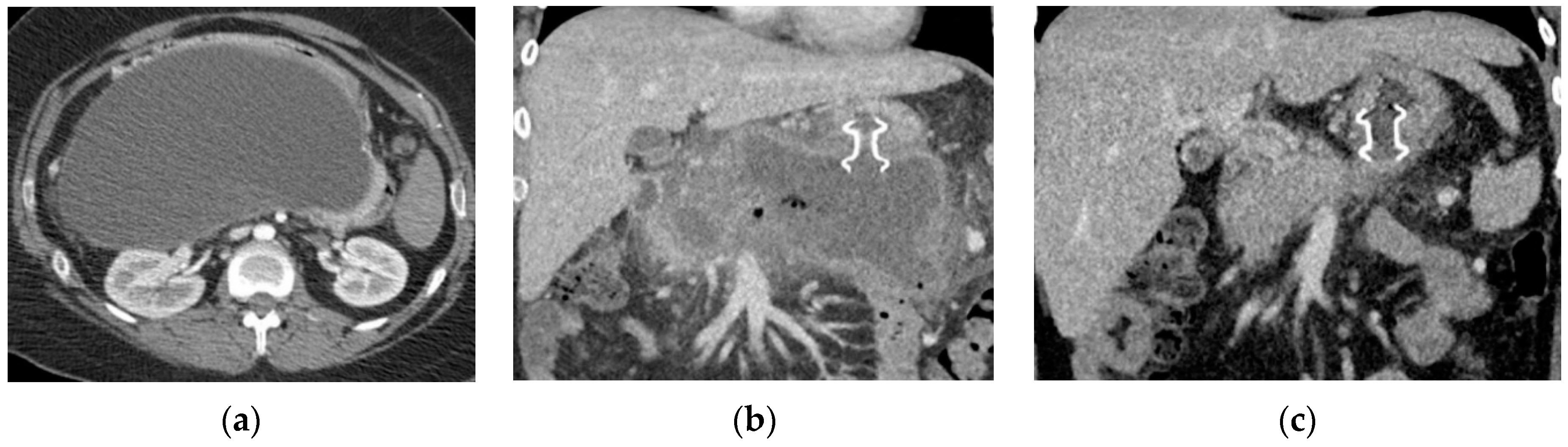
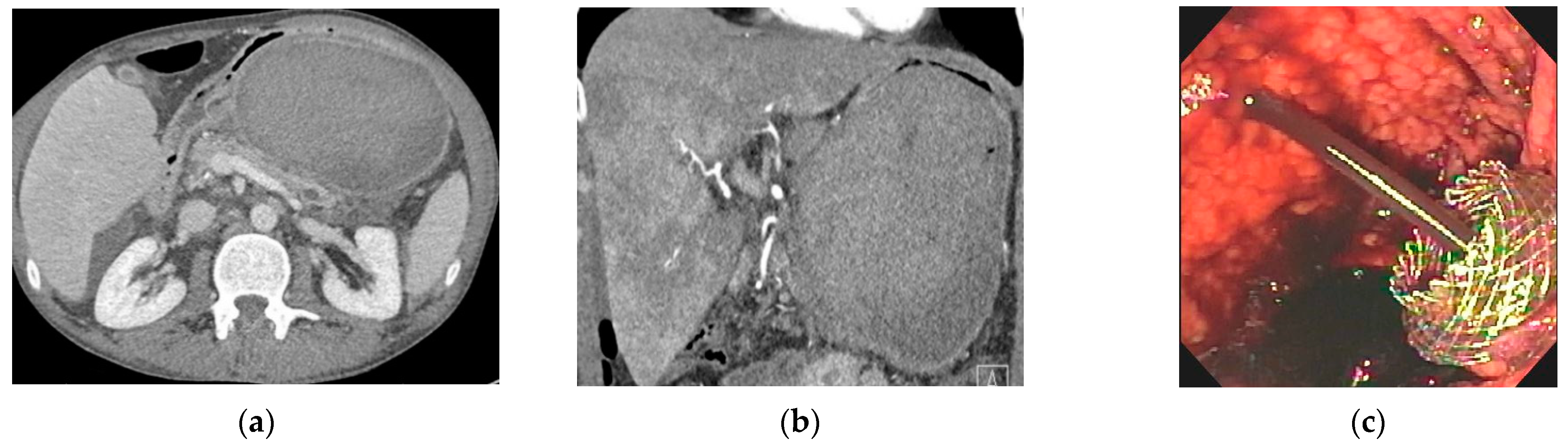
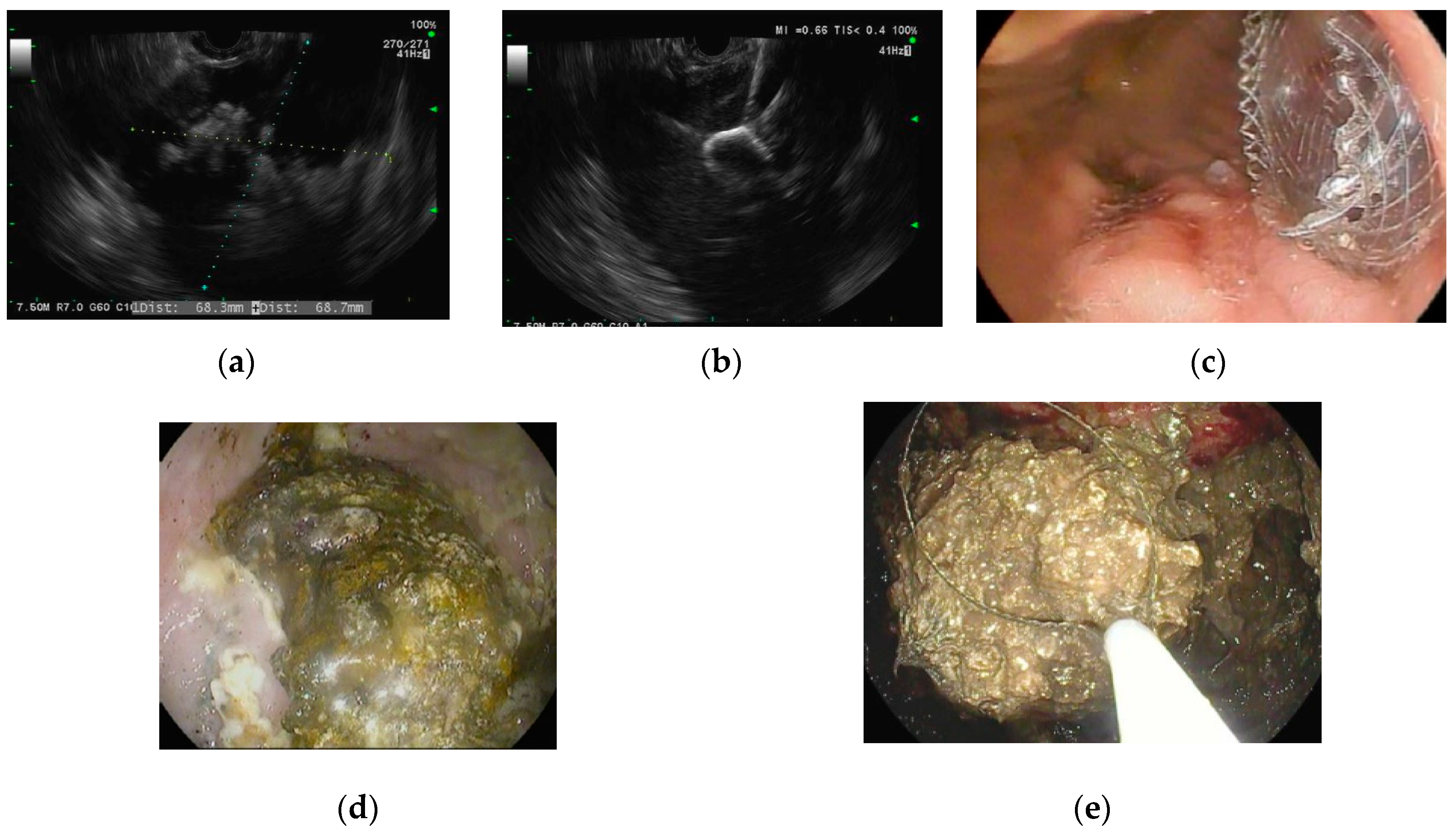
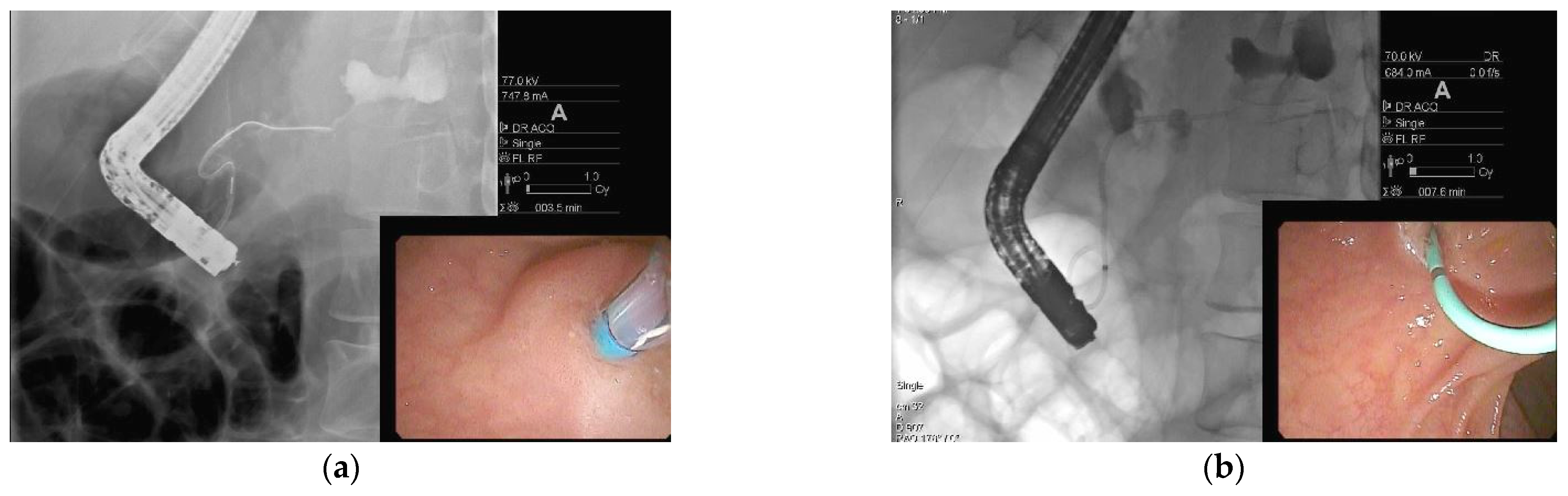
| Type of Collection | Age of Collection | Presence of Necrosis |
|---|---|---|
| Acute peripancreatic fluid collection | ≤4 weeks | No |
| Pseudocyst | >4 weeks | No |
| Acute Necrotic Collection | ≤4 weeks | Yes |
| Walled-off Necrosis | >4 weeks | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dorrell, R.; Pawa, S.; Pawa, R. Endoscopic Management of Pancreatic Fluid Collections. J. Clin. Med. 2021, 10, 284. https://doi.org/10.3390/jcm10020284
Dorrell R, Pawa S, Pawa R. Endoscopic Management of Pancreatic Fluid Collections. Journal of Clinical Medicine. 2021; 10(2):284. https://doi.org/10.3390/jcm10020284
Chicago/Turabian StyleDorrell, Robert, Swati Pawa, and Rishi Pawa. 2021. "Endoscopic Management of Pancreatic Fluid Collections" Journal of Clinical Medicine 10, no. 2: 284. https://doi.org/10.3390/jcm10020284
APA StyleDorrell, R., Pawa, S., & Pawa, R. (2021). Endoscopic Management of Pancreatic Fluid Collections. Journal of Clinical Medicine, 10(2), 284. https://doi.org/10.3390/jcm10020284





