Role of 18F-FDG PET/CT Radiomics Features in the Differential Diagnosis of Solitary Pulmonary Nodules: Diagnostic Accuracy and Comparison between Two Different PET/CT Scanners
Abstract
:1. Introduction
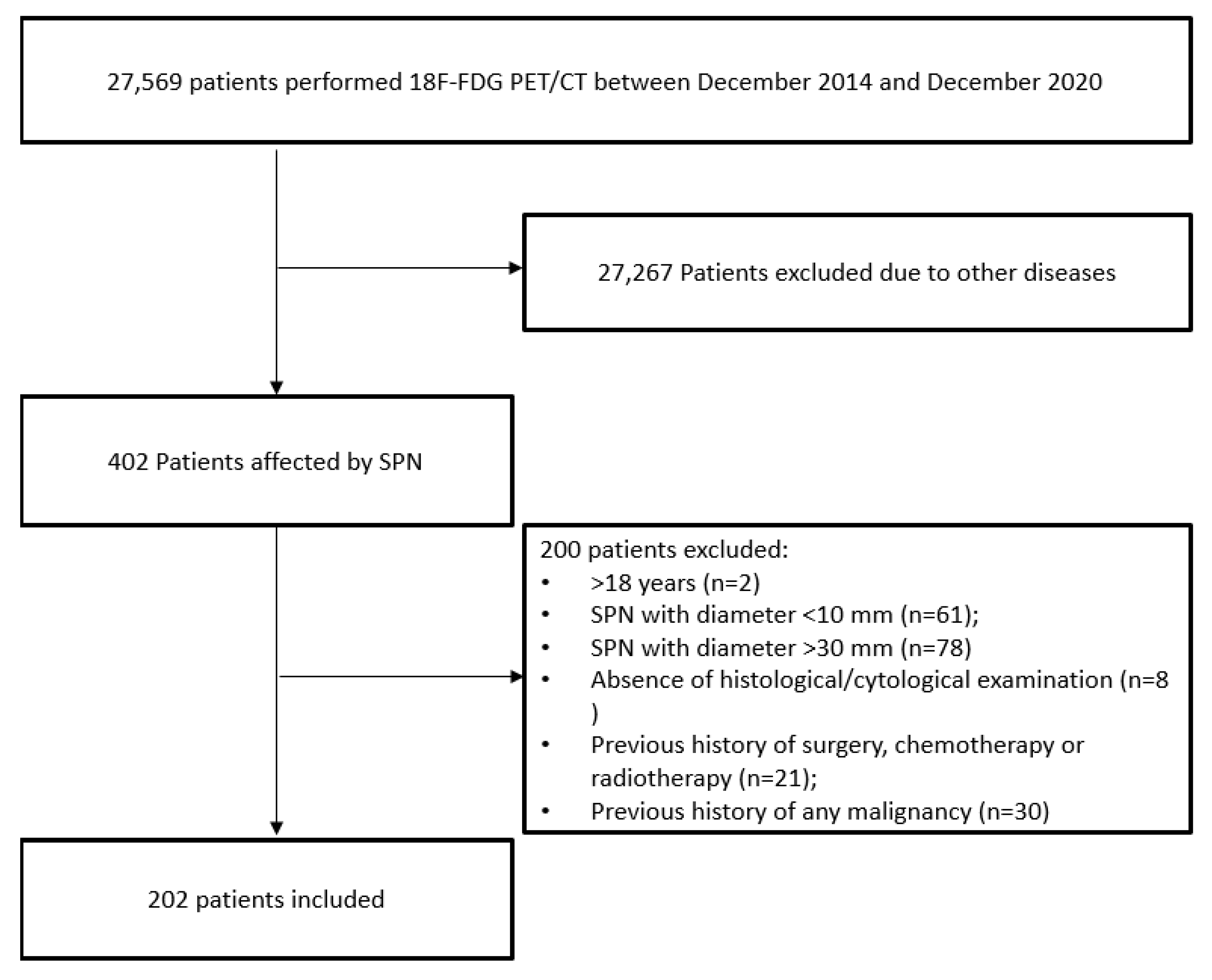
2. Materials and Methods
2.1. Patients
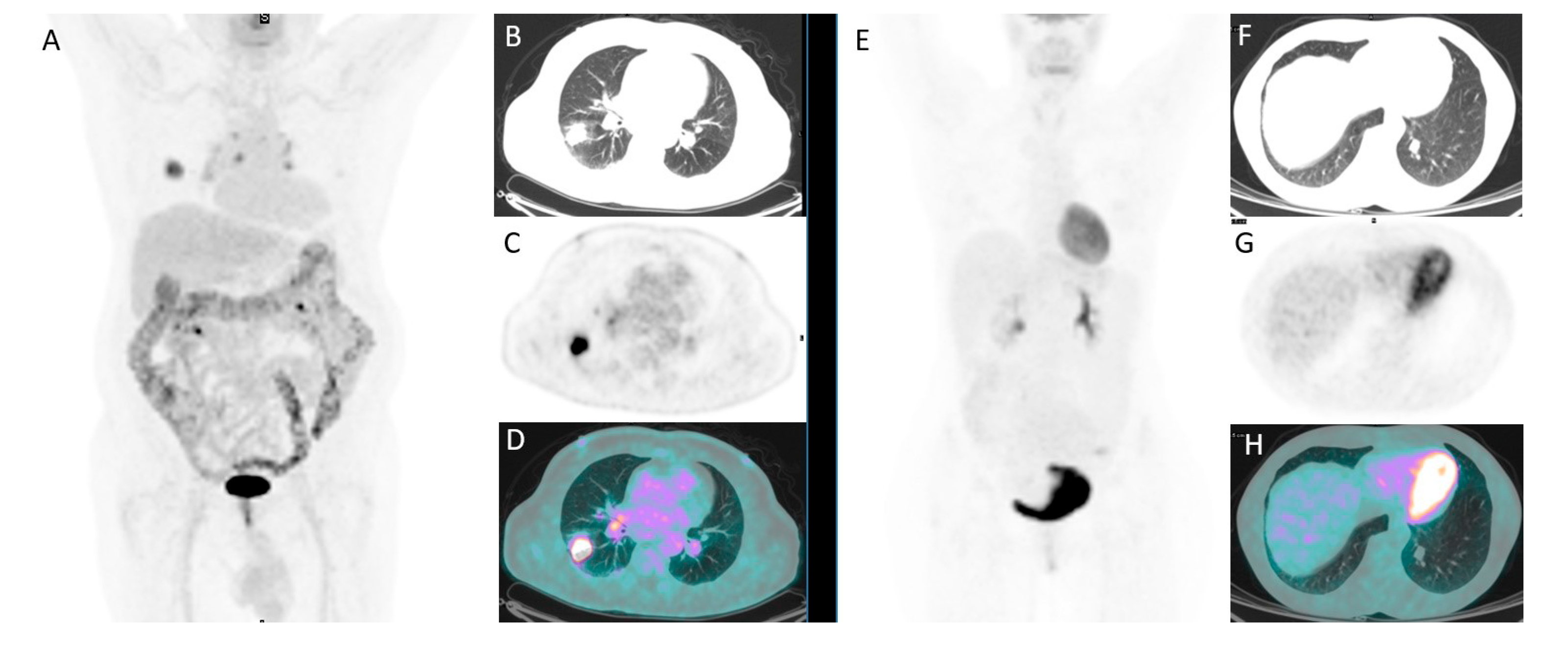
2.2. 18F-FDG PET/CT Imaging and Interpretation
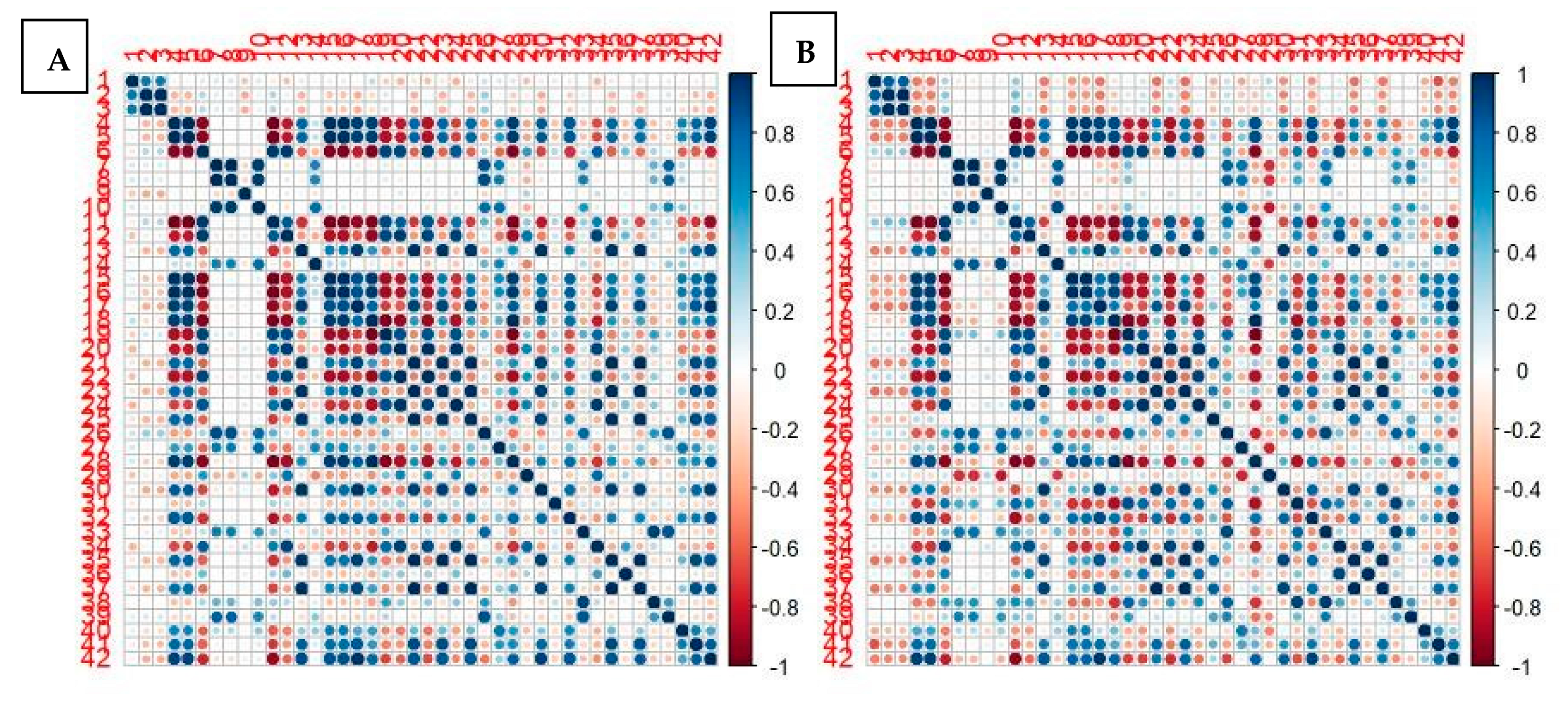
2.3. Texture Feature Extraction
2.4. Statistical Analysis
- -
- Univariate analysis: a univariate analysis was performed on three different sets; the set of patients were treated with Scanner 1, Scanner 2, and the entire dataset. The aim of this step was to figure out how different technologies can affect the relationship between each RF and the clinical outcome.
- -
- Bivariate analysis: with the aim of developing three predictive models (Scanner 1, Scanner 2, and for both the scanners), we analyzed the entire set of the possible couples of variables (the Cartesian product of the radiomics and the main clinical features, such as age, gender, nodule size, side). For each couple of variables, we calculated the bivariate logistic regression model and then we ranked them on the basis of the area under the curve (AUC) under the receiving operator curve (ROC) after a 10-cross fold validation training/testing test.
- -
- Model selection: the best bivariate logistic regression model was selected for Scanner 1, Scanner 2, and Scanner 1 + 2 on the basis of the highest AUC.
3. Results
3.1. Patients Characteristics
3.2. Comparison between the Two PET/CT Scanners
3.3. Prediction Accuracy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Ost, D.; Fein, A.; Feinsilver, S. The solitary pulmonary nodule. N. Engl. J. Med. 2003, 348, 2535–2542. [Google Scholar] [CrossRef]
- Sim, Y.T.; Poon, F.W. Imaging of solitary pulmonary nodule—A clinical review. Quant. Imaging Med. Surg. 2013, 3, 316–326. [Google Scholar]
- Gould, M.; Tang, T.; Liu, I.L.; Lee, J.; Zheng, C.; Danforth, K.; Kosco, A.; Di Fiore, J.; Suh, D. Recent trends in the identification of incidental pulmonary nodules. Am. J. Respir. Crit. Care Med. 2015, 192, 1208–1214. [Google Scholar] [CrossRef] [PubMed]
- Kikano, G.E.; Fabien, A.; Schilz, R. Evaluation of the solitary pulmonary nodule. Am. Fam. Phys. 2015, 92, 1084–1091. [Google Scholar]
- Nasim, F.; Ost, D. Management of the solitary pulmonary nodule. Curr. Opin. Pulm. Med. 2019, 25, 344–353. [Google Scholar] [CrossRef]
- Chan, E.; Gaur, P.; Ge, Y.; Kopas, L.; Santacruz, J.; Gupta, N.; Munden, R.; Cagle, P.; Kim, M. Management of the solitary pulmonary nodule. Arch. Pathol. Lab. Med. 2017, 141, 927–931. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- MacMahon, H.; Naidich, D.; Goo, J.; Lee, K.; Leung, A.; Mayo, J.; Mehta, A.; Ohno, Y.; Powell, C.; Prokop, M.; et al. Guidelines for management of incidental pulmonary nodules detected on CT images: From the Fleischner Society 2017. Radiology 2017, 284, 228–243. [Google Scholar] [CrossRef] [Green Version]
- Li, W.; Pang, H.; Liu, Q.; Zhou, J. The role of 18F-FDG PET or18F-FDG-PET/CT in the evaluation of solitary pulmonary nodule. Eur. J. Radiol. 2015, 84, 2032–2037. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.Z.; Huang, Y.L.; Song, H.J.; Wang, Y.J.; Huang, Y. The value of 18F-FDG-PET/CT in the diagnosis of solitary pulmonary nodules. A meta-analysis. Medicina 2018, 97, e0130. [Google Scholar]
- Wilson, R.; Devaraj, A. Radiomics of pulmonary nodules and lung cancer. Transl. Lung Cancer Res. 2017, 6, 86–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krewer, H.; Geiger, B.; Hall, L.; Goldgof, D.; Gu, Y.; Tockman, M.; Gillies, R. Effect of texture features in computer aided diagnosis of pulmonary nodules in low-dose computed tomography. In Proceedings of the IEEE International Conference on Systems, Man, and Cybernetics, Manchester, UK, 13–16 October 2013; pp. 3887–3891. [Google Scholar]
- Dhara, A.; Mukhopadhyay, S.; Dutta, A.; Garg, M.; Khandelwal, N. A Combination of Shape and Texture Features for Classification of Pulmonary Nodules in Lung CT Images. J. Digit. Imaging 2016, 29, 466–475. [Google Scholar] [CrossRef] [Green Version]
- Hawkins, S.; Wang, H.; Liu, Y.; Garcia, A.; Stringfield, O.; Krewer, H.; Li, Q.; Cherezov, D.; Gatenby, R.; Balagurunathan, Y.; et al. Predicting Malignant Nodules from Screening CT Scans. J. Thorac. Oncol. 2016, 11, 2120–2128. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, J.J.; Oliveira, M.; de Azevedo-Marques, P. Characterization of Pulmonary Nodules Based on Features of margin Sharpness and Texture. J. Digit. Imaging 2018, 31, 451–463. [Google Scholar] [CrossRef]
- Balagurunathan, Y.; Schabath, M.; Wang, H.; Liu, Y.; Gillies, R. Quantitative Imaging features Improve Discrimination of Malignancy in Pulmonary nodules. Sci. Rep. 2019, 9, 8528. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, N.; Tian, S.; Li, X.; Huang, J.; Wang, J.; Chen, S.; Ma, Y.; Liu, X.; Guo, X. Three-Dimensional Texture Feature Analysis of Pulmonary Nodules in CT Images: Lung Cancer Predictive Models Based on Support Vector Machine Classifier. J. Digit. Imaging 2019, 33, 414–422. [Google Scholar] [CrossRef]
- Wu, W.; Pierce, L.; Zhang, Y.; Pipavath, S.; Randolph, T.; Lastwika, K.; Lampe, P.; Houghton, A.; Liu, H.; Xia, L.; et al. Comparison of prediction models with radiological semantic features and radiomics in lung cancer diagnosis of the pulmonary nodules: A case-control study. Eur. Radiol. 2019, 29, 6100–6108. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Hu, H.; Gong, J.; Li, X.; Huang, G.; Nie, S. Malignant-benign classification of pulmonary nodules based on random forest aided by clustering analysis. Phys. Med. Biol. 2019, 64, 035017. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Y.; Tang, K.; Lin, J.; Zhang, H. The Value of 18F-FDG PET/CT Mathematical Prediction Model in Diagnosis of Solitary Pulmonary Nodules. Biomed Res. Int. 2018, 2018, 9453967. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Harmon, S.; Perk, T.; Li, X.; Chen, M.; Li, Y.; Jeraj, R. Using neighborhood gray tone difference matrix texture features on dual time point PET/CT images to differentiate malignant from benign FDG-avid solitary pulmonary nodules. Cancer Imaging 2019, 19, 56. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, B.; Bianconi, F.; Palumbo, I.; Fravolini, M.L.; Minestrini, M.; Nuvoli, S.; Stazza, M.L.; Rondini, M.; Spanu, A. Value of Shape and Texture Features from 18F-FDG PET/CT to Discriminate between Benign and Malignant Solitary Pulmonary Nodules: An Experimental Evaluation. Diagnostics 2020, 10, 96. [Google Scholar] [CrossRef]
- Miwa, K.; Inubushi, M.; Wagatsuma, K.; Nagao, M.; Murata, T.; Koyama, M.; Koizumi, M.; Sasaki, M. FDG uptake heterogeneity evaluated by fractal analysis improves the differential diagnosis of pulmonary nodules. Eur. J. Radiol. 2014, 83, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Harmon, S.; Perk, T.; Li, X.; Chen, M.; Li, Y.; Jeraj, R. Diagnostic classification of solitary pulmonary nodules using dual time 18 FFDG PET/CT image texture features in granuloma-endemic regions. Sci. Rep. 2017, 7, 9370. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Ma, G.; Cheng, J.; Song, S.; Zhang, Y.; Shi, L.Q. Diagnostic classification of solitary pulmonary nodules using support vector machine model based on 2-[18F]fluoro-2-deoxy-D-glucose PET/computed tomography texture features. Nucl. Med. Commun. 2019, 41, 560–566. [Google Scholar] [CrossRef] [PubMed]
- Cook, G.; Goh, V. A Role for FDG PET Radiomics in Personalized Medicine? Semin. Nucl. Med. 2020, 50, 532–540. [Google Scholar] [CrossRef] [PubMed]
- Pfaehler, E.; van Sluis, J.; Merema, B.B.J.; van Ooijen, P.; Berendsen, R.C.M.; van Velden, F.H.P.; Boellaard, R. Experimental Multicenter and Multivendor Evaluation of the Performance of PET Radiomic Features Using 3-Dimensionally Printed Phantom Inserts. J. Nucl. Med. 2020, 61, 469–476. [Google Scholar] [CrossRef]
- Zwanenburg, A. Radiomics in nuclear medicine: Robustness, reproducibility, standardization, and how to avoid data analysis traps and replication crisis. Eur. J. Nucl. Med. 2019, 46, 2638–2655. [Google Scholar] [CrossRef]
- Boellaard, R.; Delgado-Bolton, R.; Oyen, W.J.; Giammarile, F.; Tatsch, K.; Eschner, W.; Verzijlbergen, F.J.; Barrington, S.F.; Pike, L.C.; Weber, W.A.; et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: Version 2.0. Eur. J. Nucl. Med. Mol. Imaging 2014, 42, 328–354. [Google Scholar] [CrossRef]
- Nioche, C.; Orlhac, F.; Boughdad, S.; Reuzè, S.; Goya-Outi, J.; Robert, C.; Pellot-Barakt, C.; Soussan, M.; Frouin, F.; Buvat, I. LIFEx: A Freeware for Radiomic Feature Calculation in Multimodality Imaging to Accelerate Advances in the Characterization of Tumor Heterogeneity. Cancer Res. 2018, 78, 4786–4789. [Google Scholar] [CrossRef] [Green Version]
- Durmo, R.; Lechiara, M.; Benetti, D.; Rodella, C.; Camoni, L.; Albano, D.; Bertagna, F.; Giubbini, R. Radioguided lung lesion localization: Introducing a fluoroscopy system in a SPECT/CT scan. Nucl. Med. Commun. 2019, 40, 597–603. [Google Scholar] [CrossRef]
- Sollini, M.; Cozzi, L.; Antunovic, L.; Chiti, A.; Kirienko, M. PET Radiomics in NSCLC: State of the art and a proposal for harmonization of methodology. Sci. Rep. 2017, 7, 358. [Google Scholar] [CrossRef] [Green Version]
- Reynes-Llompart, G.; Sabatè-Llobera, A.; Linares-Tello, E.; Martì-Climent, J.; Gamez-Cenzano, C. Image quality evaluation in a modern PET system: Impact of new reconstructions methods and a radiomics approach. Sci. Rep. 2019, 9, 10640. [Google Scholar] [CrossRef]
- Ha, S.; Choi, H.; Paeng, J.C.; Cheon, G.J. Radiomics in Oncological PET/CT: A Methodological Overview. Nucl. Med. Mol. Imaging 2019, 53, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Kamibayashi, T.; Tsuchida, T.; Demura, Y.; Tsujikawa, T.; Okazawa, H.; Kudoh, T.; Kimura, H. Reproducibility of semi-quantitative parameters in FDG-PET using two different PET scanners: Influence of attenuation correction method and examination interval. Mol. Imaging Biol. 2008, 10, 162–166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lodge, M.A. Repeatability of SUV in Oncologic 18F-FDG PET. J. Nucl. Med. 2017, 58, 523–532. [Google Scholar] [CrossRef] [Green Version]
- Berenguer, R.; Del Rosario Pasto-Juan, M.; Canales-Vazquez, J.; Castro-Garcia, M.; Villas, M.V.; Masilla Legorburo, F.; Sabater, S. Radiomics of CT Features May Be Nonreproducible and Redundant: Influence of CT Acquisition Parameters. Radiology 2018, 288, 407–415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Espinasse, M.; Pitre-Champagnat, S.; Charmettant, B.; Bidault, F.; Volk, A.; Balleyguier, C.; Lassau, N.; Caramella, C. CT Texture Analysis Challenges: Influence of Acquisition and Reconstruction Parameters: A Comprehensive Review. Diagnsotics 2020, 10, 258. [Google Scholar] [CrossRef]

| n (%) | |
|---|---|
| Age, mean ± SD (range) | 68 ± 11 (37–86) |
| Male/female | 117:85 |
| Lung side right/left | 128:74 |
| Lung localization Upper lobe Inferior lobe Medium lobe Lingula | 110 (54%) 68 (34%) 20 (10%) 4 (2%) |
| Diameter max mm, mean ± SD (range) | 20.6 ± 6.5 (10–29) |
| Volume mm3, mean ± SD (range) | 3861 ± 3578 (197–17,342) |
| Scanner PET Discovery 690 Discovery ST | 90 (45%) 112 (55%) |
| PET/CT visual result Positive Negative | 140 (69%) 62 (31%) |
| Mediastinal nodes FDG positive | 60 (30%) |
| SUVmax, mean ± SD (range) | 7 ± 7.3 (0,52–61.4) |
| SUVmean, mean ± SD (range) | 4.6 ± 5.5 (0.32–52) |
| SUVlbm, mean ± SD (range) | 5.3 ± 5.5 (0.34–45) |
| SUVbsa, mean ± SD (range) | 1.8 ± 1,8 (0.13–14) |
| Lesion to liver SUV ratio, mean ± SD (range) | 2.5 ± 2.5 (0.17–19.13) |
| Lesion to liver SUV ratio, mean ± SD (range) | 3,1 ± 3.2 (0.22–23.1) |
| MTV, mean ± SD (range) | 5.5 ± 5.2 (0.6–34,2) |
| TLG, mean ± SD (range) | 22.2 ± 39.2 (0.32–432) |
| Final diagnosis Benign Malignant Indeterminate | 64 (32%) 127 (63%) 11 (5%) |
| n (%) | |
|---|---|
| MALIGNANT n = 127 | |
| Adenocarcinoma | 86 (68%) |
| Squamous cell carcinoma | 17 (13%) |
| Large cell carcinoma | 1 (1%) |
| Neuroendocrine tumor | 12 (9%) |
| Sarcomatoid carcinoma | 1 (1%) |
| Unspecified | 10 (8%) |
| BENIGN n = 64 | |
| Hamartoma | 7 (11%) |
| Inflammation | 6 (9%) |
| Solitary fibrous tumor | 2 (3%) |
| Granuloma | 2 (3%) |
| Hamatochondroma | 1 (1.5%) |
| Angioma | 1 (1.5%) |
| Lipoma | 1 (1.5%) |
| Active tuberculosis | 1 (1.5%) |
| Fibrosis | 1 (1.5%) |
| Negative cytological examination | 42 (66.5%) |
| SCANNER D690 vs. D-STE | |||
|---|---|---|---|
| Clinical-Epidemiological Features | p-Value | Second-Order Statistics | p-Value |
| Age | 0.659 | GLCM entropy_log10 | <0.001 |
| Gender | 0.659 | GLCM entropy_log2 | <0.001 |
| Size | 0.746 | GLCM dissimilarity | <0.001 |
| “Conventional” PET features | GLRLM SRE | <0.001 | |
| SUVmax | <0.001 | GLRLM LRE | <0.001 |
| SUVmean | <0.001 | GLRLM LGRE | 0.001 |
| SUVlbm | <0.001 | GLRLM HGRE | <0.001 |
| SUVbsa | <0.001 | GLRLM SRLGE | 0.002 |
| L-L SUV R | 0.003 | GLRLM SRHGE | <0.001 |
| L-BP SUV R | 0.009 | GLRLM LRLGE | <0.001 |
| MTV | <0.001 | GLRLM LRHGE | 0.006 |
| TLG | <0.001 | GLRLM GLNU | <0.001 |
| First-order statistics | GLRLM RLNU | 0.052 | |
| Histo skewness | 0.316 | GLRLM RP | <0.001 |
| Histo kurtosis | 0.758 | NGLDM coarseness | 0.799 |
| Histo excess kurtosis | 0.758 | NGLDM contrast | <0.001 |
| Histo entropy_log10 | <0.001 | NGLDM busyness | <0.001 |
| Histo entropy_log2 | <0.001 | GLZLM SZE | <0.001 |
| Histo energy | <0.001 | GLZLM LZE | <0.001 |
| Shape volume_mL | 0.917 | GLZLM LGZE | 0.003 |
| Shape volume_vx | 0.917 | GLZLM HGZE | <0.001 |
| Shape sphericity | 0.037 | GLZLM SZLGE | 0.877 |
| Shape compacity | 0.859 | GLZLM SZHGE | <0.001 |
| Second-order statistics | GLZLM LZLGE | <0.001 | |
| GLCM homogeneity | <0.001 | GLZLM LZHGE | 0.002 |
| GLCM energy | <0.001 | GLZLM GLNU | 0.803 |
| GLCM contrast | < 0.001 | GLZLM ZLNU | <0.001 |
| GLCM correlation | 0.052 | GLZLM ZP | < 0.001 |
| Mean AUC | |||
|---|---|---|---|
| Scanner 1 | Scanner 2 | Scanner 1 + 2 | |
| “Conventional” PET features | |||
| SUV-related | |||
| SUVmax | 0.855 | 0.714 | 0.760 |
| SUVmean | 0.840 | 0.714 | 0.752 |
| SUVlbm | 0.851 | 0.730 | 0.767 |
| SUVbsa | 0.859 | 0.724 | 0.769 |
| L-L SUV R | 0.847 | 0.727 | 0.766 |
| L-BP SUV R | 0.860 | 0.727 | 0.776 |
| Metabolic volumes | |||
| MTV | 0.594 | 0.601 | 0.562 |
| TLG | 0.771 | 0.669 | 0.633 |
| First-order statistics | |||
| Histogram-related | |||
| Histo skewness | 0.682 | 0.585 | 0.629 |
| Histo kurtosis | 0.584 | 0.591 | 0.560 |
| Histo excess kurtosis | 0.584 | 0.592 | 0.560 |
| Histo entropy_log10 | 0.869 | 0.718 | 0.763 |
| Histo entropy_log2 | 0.869 | 0.719 | 0.762 |
| Histo energy | 0.852 | 0.704 | 0.743 |
| Shape-related | |||
| Shape volume_mL | 0.656 | 0.626 | 0.542 |
| Shape volume_vx | 0.656 | 0.626 | 0.542 |
| Shape sphericity | 0.615 | 0.641 | 0.580 |
| Shape Compacity | 0.637 | 0.634 | 0.530 |
| Second-order statistics | |||
| GLCM-related | |||
| GLCM homogeneity | 0.846 | 0.692 | 0.733 |
| GLCM energy | 0.846 | 0.702 | 0.743 |
| GLCM contrast | 0.849 | 0.705 | 0.740 |
| GLCM correlation | 0.572 | 0.677 | 0.556 |
| GLCM entropy_log10 | 0.853 | 0.732 | 0.747 |
| GLCM entropy_log2 | 0.850 | 0.734 | 0.747 |
| GLCM dissimilarity | 0.845 | 0.702 | 0.739 |
| GLRLM-related | |||
| GLRLM SRE | 0.843 | 0.692 | 0.731 |
| GLRLM LRE | 0.841 | 0.676 | 0.723 |
| GLRLM LGRE | 0.813 | 0.688 | 0.733 |
| GLRLM HGRE | 0.819 | 0.695 | 0.725 |
| GLRLM SRLGE | 0.799 | 0.687 | 0.729 |
| GLRLM SRHGE | 0.828 | 0.698 | 0.730 |
| GLRLM LRLGE | 0.831 | 0.692 | 0.741 |
| GLRLM LRHGE | 0.762 | 0.678 | 0.686 |
| GLRLM GLNU | 0.752 | 0.752 | 0.663 |
| GLRLM RLNU | 0.719 | 0.705 | 0.642 |
| GLRLM RP | 0.483 | 0.680 | 0.722 |
| NGLDM-related | |||
| NGLDM coarseness | 0.643 | 0.783 | 0.590 |
| NGLDM contrast | 0.808 | 0.808 | 0.721 |
| NGLDM busyness | 0.783 | 0.643 | 0.700 |
| GLZLM-related | |||
| GLZLM SZE | 0.777 | 0.677 | 0.703 |
| GLZLM LZE | 0.821 | 0.821 | 0.704 |
| GLZLM LGZE | 0.785 | 0.785 | 0.722 |
| GLZLM HGZE | 0.793 | 0.689 | 0.722 |
| GLZLM SZLGE | 0.614 | 0.614 | 0.574 |
| GLZLM SZHGE | 0.794 | 0.689 | 0.718 |
| GLZLM LZLGE | 0.837 | 0.697 | 0.725 |
| GLZLM LZHGE | 0.755 | 0.671 | 0.664 |
| GLZLM GLNU | 0.810 | 0.810 | 0.715 |
| GLZLM ZLNU | 0.808 | 0.716 | 0.726 |
| GLZLM ZP | 0.834 | 0.834 | 0.714 |
| Covariate 1 | Covariate 2 | Mean AUC |
|---|---|---|
| Scanner 1 | ||
| GLCM entropy_log10 | GLZLM LZE | 0.861 |
| GLCM entropy_log2 | GLZLM LZE | 0.860 |
| Histo entropy_log10 | GLZLM LZE | 0.858 |
| GLCM homogeneity | GLZLM LZE | 0.858 |
| GLCM entropy_log2 | GLZLM LGZE | 0.856 |
| GLCM entropy_log10 | GLZLM LGZE | 0.856 |
| Histo entropy_log2 | GLZLM LZE | 0.856 |
| GLCM entropy_log10 | GLZLM LRE | 0.854 |
| Histo entropy_log2 | GLZLM LGZE | 0.852 |
| Histo entropy_log10 | GLZLM LGZE | 0.852 |
| Scanner 2 | ||
| Histo entropy_log10 | Shape sphericity | 0.734 |
| Histo energy | L-BP SUV R | 0.732 |
| GLCM entropy_log2 | MTV | 0.728 |
| GLCM entropy_log10 | MTV | 0.727 |
| Histo energy | L-L SUV R | 0.727 |
| GLCM entropy_log10 | GLRLM RLNU | 0.727 |
| GLCM entropy_log2 | GLRLM RLNU | 0.726 |
| Histo entropy_log10 | MTV | 0.719 |
| Histo entropy_log10 | Histo entropy_log2 | 0.719 |
| Histo entropy_log2 | MTV | 0.710 |
| Scanner 1 + 2 | ||
| SUVmean | L-BP SUV R | 0.785 |
| SUVmax | L-BP SUV R | 0.780 |
| L-BP SUV R | MTV | 0.774 |
| Histo skewness | L-BP SUV R | 0.771 |
| SUVmean | SUVlbm | 0.770 |
| Histo energy | L-BP SUV R | 0.769 |
| SUVmean | SUVbsa | 0.768 |
| Histo entropy_log2 | MTV | 0.767 |
| Histo entropy_log10 | MTV | 0.767 |
| Histo energy | MTV | 0.753 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Albano, D.; Gatta, R.; Marini, M.; Rodella, C.; Camoni, L.; Dondi, F.; Giubbini, R.; Bertagna, F. Role of 18F-FDG PET/CT Radiomics Features in the Differential Diagnosis of Solitary Pulmonary Nodules: Diagnostic Accuracy and Comparison between Two Different PET/CT Scanners. J. Clin. Med. 2021, 10, 5064. https://doi.org/10.3390/jcm10215064
Albano D, Gatta R, Marini M, Rodella C, Camoni L, Dondi F, Giubbini R, Bertagna F. Role of 18F-FDG PET/CT Radiomics Features in the Differential Diagnosis of Solitary Pulmonary Nodules: Diagnostic Accuracy and Comparison between Two Different PET/CT Scanners. Journal of Clinical Medicine. 2021; 10(21):5064. https://doi.org/10.3390/jcm10215064
Chicago/Turabian StyleAlbano, Domenico, Roberto Gatta, Matteo Marini, Carlo Rodella, Luca Camoni, Francesco Dondi, Raffaele Giubbini, and Francesco Bertagna. 2021. "Role of 18F-FDG PET/CT Radiomics Features in the Differential Diagnosis of Solitary Pulmonary Nodules: Diagnostic Accuracy and Comparison between Two Different PET/CT Scanners" Journal of Clinical Medicine 10, no. 21: 5064. https://doi.org/10.3390/jcm10215064






