Food-Related Brain Activation Measured by fMRI in Adults with Prader–Willi Syndrome
Abstract
:1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Healthy Controls (Siblings)
2.3. Laboratory Tests
2.4. Intelligence
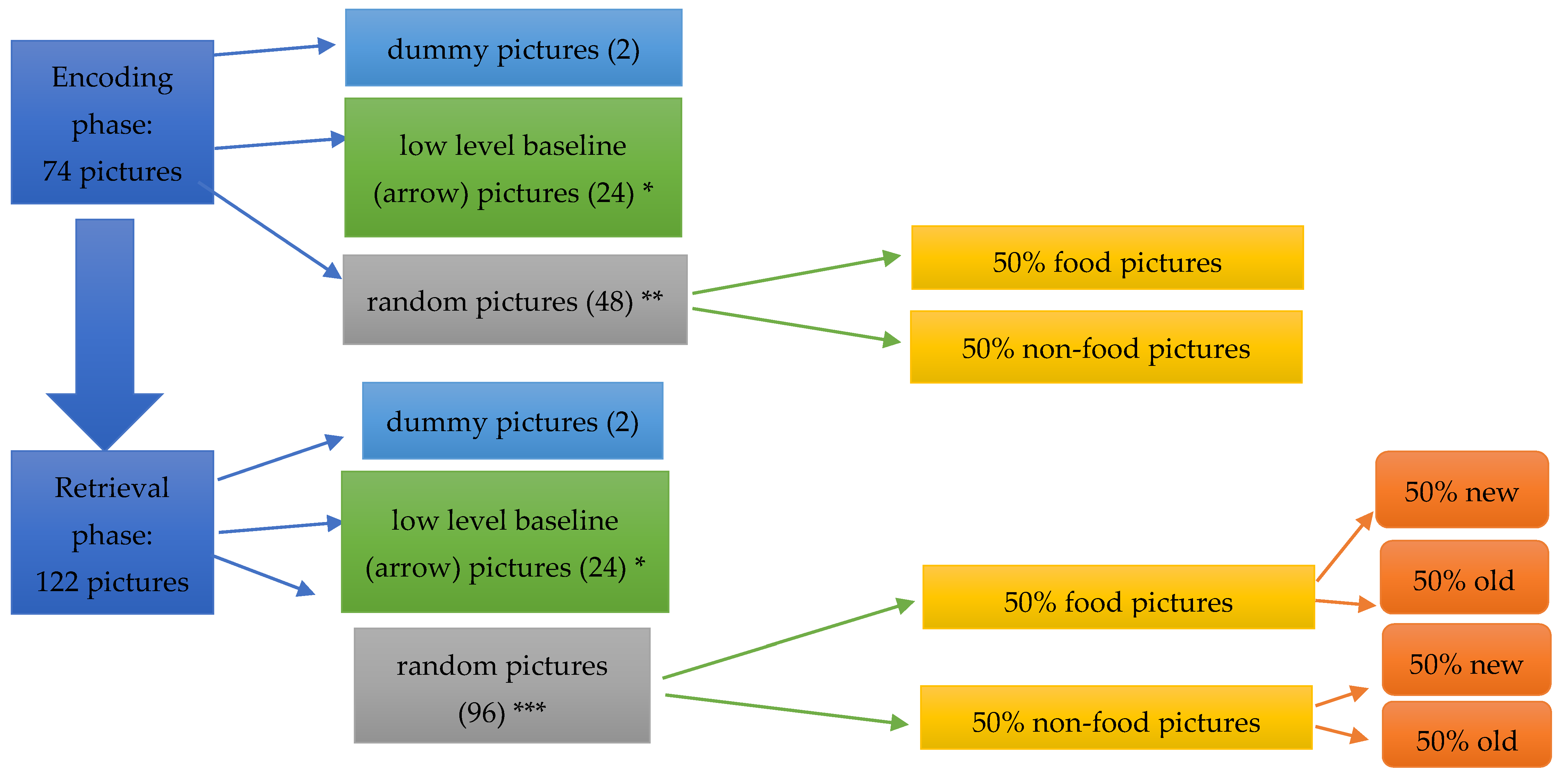
2.5. Experimental Tasks
2.6. Statistical Analysis
3. Results
3.1. Psychometric Measurements fMRI Task
3.2. Imaging Data
3.3. Imaging Data Combined with Endocrine and Metabolic Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Laboratory Parameter | Lab Method | Company | Lower Limit of Quantitation | Mean intra-Assay % CV | Reference Values |
|---|---|---|---|---|---|
| IGF-1 and IGFBP-3 | Immunometric assay, Luminescence | Immulite 2500 ® laboratory assay, Siemens Medical Solutions Diagnostics, USA | sex- and age-specific | ||
| Insulin | Immunometric assay, Luminescence | Advia Centaur, Siemens Medical Solutions Diagnostics, USA | 10 pmol/L | 20 pmol/L: 4% 500 pmol/L: 3% 500 pmol/L: 4% | 12–96 pmol/L * |
| Adiponectin | Radioimmunoassay | Linco Research Inc., St. Charles, MO, USA | 0.5 mg/L | whole range: 5% | 0.8–48 mg/L |
| Ghrelin | Radioimmunoassay | Linco Research Inc., St. Charles, MO, USA | 240 ng/L | whole range: 4% | 800–3000 ng/L |
| Leptin | Radioimmunoassay | Linco Research Inc., St. Charles, MO, USA | 0.5 μg/L | 5 μg/L: 8%25 μg/L: 3% | Men **: 2.5–8 μg/L Women **: 4.5–16 μg/L |
| Resistin | Immnometric assay (colorimetric) | BioVendor Laboratory Medicine, INC, Modrice, Czech Republic | 0.8 ng/ml | whole range: 5% | 4.1–12.1 ng/ml |
References
- Emerick, J.E.; Vogt, K.S. Endocrine manifestations and management of Prader-Willi syndrome. Int. J. Pediatr. Endocrinol. 2013, 2013, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butler, M.G.; Miller, J.L.; Forster, J.L. Prader-Willi Syndrome–Clinical Genetics, Diagnosis and Treatment Approaches: An Update. Curr. Pediatr. Rev. 2019, 15, 207–244. [Google Scholar] [CrossRef] [PubMed]
- Gunay-Aygun, M.; Schwartz, S.; Heeger, S.; O’Riordan, M.A.; Cassidy, S.B. The changing purpose of Prader-Willi syndrome clinical diagnostic criteria and proposed revised criteria. Pediatrics 2001, 108, E92. [Google Scholar] [CrossRef] [Green Version]
- Holm, V.A.; Cassidy, S.B.; Butler, M.G.; Hanchett, J.M.; Greenswag, L.R.; Whitman, B.Y. Prader-Willi syndrome: Consensus diagnostic criteria. Pediatrics 1993, 91, 398–402. [Google Scholar] [PubMed]
- Brambilla, P.; Bosio, L.; Manzoni, P.; Pietrobelli, A.; Beccaria, L.; Chiumello, G. Peculiar body composition in patients with Prader-Labhart-Willi syndrome. Am. J. Clin. Nutr. 1997, 65, 1369–1374. [Google Scholar] [CrossRef] [Green Version]
- Höybye, C.; Hilding, A.; Jacobsson, H.; Thorén, M. Metabolic profile and body composition in adults with Prader-Willi syndrome and severe obesity. J. Clin. Endocrinol. Metab. 2002, 87, 3590–3597. [Google Scholar] [CrossRef] [Green Version]
- Sode-Carlsen, R.; Farholt, S.; Rabben, K.F.; Bollerslev, J.; Schreiner, T.; Jurik, A.G.; Christiansen, J.S.; Höybye, C. Body composition, endocrine and metabolic profiles in adults with Prader-Willi syndrome. Growth Horm. IGF Res. 2010, 20, 179–184. [Google Scholar] [CrossRef]
- Van Nieuwpoort, I.C.; Twisk, J.W.R.; Curfs, L.M.G.; Lips, P.; Drent, M.L. Body composition, adipokines, bone mineral density and bone remodeling markers in relation to IGF-1 levels in adults with Prader-Willi syndrome. Int. J. Pediatr. Endocrinol. 2018, 2018, 1. [Google Scholar] [CrossRef] [Green Version]
- Butler, M.G.; Theodoro, M.F.; Bittel, D.C.; Donnelly, J.E. Energy expenditure and physical activity in Prader-Willi syndrome: Comparison with obese subjects. Am. J. Med. Genet. A. 2007, 143a, 449–459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hill, A.J.; Rogers, P.J.; Blundell, J.E. Techniques for the experimental measurement of human eating behaviour and food intake: A practical guide. Int. J. Obes. Relat. Metab. Disord. 1995, 19, 361–375. [Google Scholar]
- Jakobsdottir, S.; De Ruiter, M.; Deijen, J.B.; Veltman, D.J.; Drent, M.L. Brain Activation by Visual Food-Related Stimuli and Correlations with Metabolic and Hormonal Parameters: A fMRI Study. Open Neuroendocrinol. J. 2012, 5, 5–12. [Google Scholar] [CrossRef]
- Jakobsdottir, S.; Van Nieuwpoort, I.C.; Van Bunderen, C.C.; De Ruiter, M.B.; Twisk, J.W.; Deijen, J.B.; Drent, M.L. Acute and short-term effects of caloric restriction on metabolic profile and brain activation in obese, postmenopausal women. Int. J. Obes. 2016, 40, 1671–1678. [Google Scholar] [CrossRef]
- LaBar, K.S.; Gitelman, D.R.; Parrish, T.B.; Kim, Y.H.; Nobre, A.C.; Mesulam, M.M. Hunger selectively modulates corticolimbic activation to food stimuli in humans. Behav. Neurosci. 2001, 115, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.S.; Dolan, R.J. Involvement of human amygdala and orbitofrontal cortex in hunger-enhanced memory for food stimuli. J. Neurosci. 2001, 21, 5304–5310. [Google Scholar] [CrossRef] [Green Version]
- Stoeckel, L.E.; Kim, J.; Weller, R.E.; Cox, J.E.; Cook, E.W., 3rd; Horwitz, B. Effective connectivity of a reward network in obese women. Brain Res. Bull. 2009, 79, 388–395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Killgore, W.D.; Young, A.D.; Femia, L.A.; Bogorodzki, P.; Rogowska, J.; Yurgelun-Todd, D.A. Cortical and limbic activation during viewing of high- versus low-calorie foods. Neuroimage 2003, 19, 1381–1394. [Google Scholar] [CrossRef]
- Del Parigi, A.; Gautier, J.F.; Chen, K.; Salbe, A.D.; Ravussin, E.; Reiman, E.; Tataranni, P.A. Neuroimaging and obesity: Mapping the brain responses to hunger and satiation in humans using positron emission tomography. Ann. N. Y. Acad. Sci. 2002, 967, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Tataranni, P.A.; Gautier, J.F.; Chen, K.; Uecker, A.; Bandy, D.; Salbe, A.D.; Pratley, R.E.; Lawson, M.; Reiman, E.M.; Ravussin, E. Neuroanatomical correlates of hunger and satiation in humans using positron emission tomography. Proc. Natl. Acad. Sci. USA 1999, 96, 4569–4574. [Google Scholar] [CrossRef] [Green Version]
- Shapira, N.A.; Lessig, M.C.; He, A.G.; James, G.A.; Driscoll, D.J.; Liu, Y. Satiety dysfunction in Prader-Willi syndrome demonstrated by fMRI. J. Neurol. Neurosurg. Psychiatry 2005, 76, 260–262. [Google Scholar] [CrossRef] [PubMed]
- Dimitropoulos, A.; Schultz, R.T. Food-related neural circuitry in Prader-Willi syndrome: Response to high- versus low-calorie foods. J. Autism Dev. Disord. 2008, 38, 1642–1653. [Google Scholar] [CrossRef] [PubMed]
- Holsen, L.M.; Savage, C.R.; Martin, L.E.; Bruce, A.S.; Lepping, R.J.; Ko, E.; Brooks, W.M.; Butler, G.M.; Zarcone, J.R.; Goldstein, J.M. Importance of reward and prefrontal circuitry in hunger and satiety: Prader-Willi syndrome vs simple obesity. Int. J. Obes. 2012, 36, 638–647. [Google Scholar] [CrossRef] [Green Version]
- Holsen, L.M.; Zarcone, J.R.; Brooks, W.M.; Butler, M.G.; Thompson, T.I.; Ahluwalia, J.S.; Nollen, L.N.; Savage, R.C. Neural mechanisms underlying hyperphagia in Prader-Willi syndrome. Obesity 2006, 14, 1028–1037. [Google Scholar] [CrossRef] [Green Version]
- Miller, J.L.; James, G.A.; Goldstone, A.P.; Couch, J.A.; He, G.; Driscoll, D.J.; Liu, Y. Enhanced activation of reward mediating prefrontal regions in response to food stimuli in Prader-Willi syndrome. J. Neurol. Neurosurg. Psychiatry 2007, 78, 615–619. [Google Scholar] [CrossRef] [Green Version]
- Ogura, K.; Fujii, T.; Abe, N.; Hosokai, Y.; Shinohara, M.; Fukuda, H.; Mori, E. Regional cerebral blood flow and abnormal eating behavior in Prader-Willi syndrome. Brain Dev. 2013, 35, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, M.; Parigi, A.D.; Chen, K.; Reiman, E.M.; Thiyyagura, P.; Krakoff, J.; Hohenadel, M.G.; Le, D.S.N.T.; Weise, C.M. Deactivation of the left dorsolateral prefrontal cortex in Prader-Willi syndrome after meal consumption. Int. J. Obes. 2016, 40, 1360–1368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Wang, J.; Zhang, G.; Zhu, Q.; Cai, W.; Tian, J.; Zhang, E.Y.; Miller, L.J.; Wen, X.; Ding, M.; et al. The neurobiological drive for overeating implicated in Prader-Willi syndrome. Brain Res. 2015, 1620, 72–80. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, H.; Qiu, S.; Tian, J.; Wen, X.; Miller, J.L.; von Deneen, M.K.; Zhou, Z.; Gold, S.M.; Liu, Y. Altered functional brain networks in Prader-Willi syndrome. NMR Biomed. 2013, 26, 622–629. [Google Scholar] [CrossRef]
- DelParigi, A.; Tschop, M.; Heiman, M.L.; Salbe, A.D.; Vozarova, B.; Sell, S.M.; Bunt, C.J.; Tataranni, P.A. High circulating ghrelin: A potential cause for hyperphagia and obesity in Prader-Willi syndrome. J. Clin. Endocrinol. Metab. 2002, 87, 5461–5464. [Google Scholar] [CrossRef] [Green Version]
- Haqq, A.M.; Farooqi, I.S.; O’Rahilly, S.; Stadler, D.D.; Rosenfeld, R.G.; Pratt, K.L.; LaFranchi, H.S.; Purnell, Q.J. Serum ghrelin levels are inversely correlated with body mass index, age and insulin concentrations in normal children and are markedly increased in Prader-Willi syndrome. J. Clin. Endocrinol. Metab. 2003, 88, 174–178. [Google Scholar] [CrossRef]
- Cummings, D.E.; Clement, K.; Purnell, J.Q.; Vaisse, C.; Foster, K.E.; Frayo, R.S.; Schwartz, W.M.; Basdevant, A.; Weigle, S.D. Elevated plasma ghrelin levels in Prader-Willi syndrome. Nat. Med. 2002, 8, 643–644. [Google Scholar] [CrossRef] [PubMed]
- Schuster, D.P.; Osei, K.; Zipf, W.B. Characterization of alterations in glucose and insulin metabolism in Prader-Willi subjects. Metabolism 1996, 45, 1514–1520. [Google Scholar] [CrossRef]
- Pagano, C.; Marin, O.; Calcagno, A.; Schiapelli, P.; Pilon, C.; Milan, G.; Bertelli, M.; Fanin, E.; Andrighetto, G.; Federspil, G.; et al. Increased serum resistin in adults with Prader-Willi syndrome is related to obesity and not to insulin resistance. J. Clin. Endocrinol. Metab. 2005, 90, 4335–4340. [Google Scholar] [CrossRef] [Green Version]
- Dimitropoulus, A.; Feurer, I.D.; Roof, E.; Stone, W.; Butler, M.G.; Sutcliffe, J.; Thompson, T. Appetite behavior, compulsivity and neurochemistry in Prader Willi syndrome. Ment. Retard. Dev. Disabil. Res. Rev. 2000, 6, 125–130. [Google Scholar] [CrossRef]
- Cummings, D.E.; Overduin, J. Gastrointestinal regulation of food intake. J. Clin. Investig. 2007, 117, 13–23. [Google Scholar] [CrossRef]
- Morton, G.J.; Cummings, D.E.; Baskin, D.G.; Barsh, G.S.; Schwartz, M.W. Central nervous system control of food intake and body weight. Nature 2006, 443, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Schloegl, H.; Percik, R.; Horstmann, A.; Villringer, A.; Stumvoll, M. Peptide hormones regulating appetite—Focus on neuroimaging studies in humans. Diabetes Metab. Res. Rev. 2011, 27, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.W.; Woods, S.C.; Porte, D., Jr.; Seeley, R.J.; Baskin, D.G. Central nervous system control of food intake. Nature 2000, 404, 661–671. [Google Scholar] [CrossRef]
- Farooqi, I.S.; Bullmore, E.; Keogh, J.; Gillard, J.; O’Rahilly, S.; Fletcher, P.C. Leptin regulates striatal regions and human eating behavior. Science 2007, 317, 1355. [Google Scholar] [CrossRef] [Green Version]
- Van Nieuwpoort, I.C.; Sinnema, M.; Castelijns, J.A.; Twisk, J.W.; Curfs, L.M.; Drent, M.L. The GH/IGF-I axis and pituitary function and size in adults with Prader-Willi syndrome. Horm. Res. Paediatr. 2011, 75, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Burman, P.; Ritzén, E.M.; Lindgren, A.C. Endocrine dysfunction in Prader-Willi syndrome: A review with special reference to GH. Endocr. Rev. 2001, 22, 787–799. [Google Scholar] [CrossRef]
- Ashpole, N.M.; Sanders, J.E.; Hodges, E.L.; Yan, H.; Sonntag, W.E. Growth hormone, insulin-like growth factor-1 and the aging brain. Exp. Gerontol. 2015, 68, 76–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Nieuwpoort, I.C.; Drent, M.L. Cognition in the adult with childhood-onset GH deficiency. Eur. J. Endocrinol. 2008, 159, S53–S57. [Google Scholar] [CrossRef] [Green Version]
- Wasinski, F.; Frazão, R.; Donato, J., Jr. Effects of growth hormone in the central nervous system. Arch. Endocrinol. Metab. 2019, 63, 549–556. [Google Scholar] [CrossRef]
- Raven, J.; Raven, J.C.; Court, J.H. Manual for Raven’s Progressive Matrices and Vocabulary Scales; Harcourt Assessment: San Antonio, TX, USA, 1998; Updated 2003. [Google Scholar]
- Van Nieuwpoort, I.C.; Deijen, J.B.; Curfs, L.M.; Drent, M.L. The relationship between IGF-I concentration, cognitive function and quality of life in adults with Prader-Willi syndrome. Horm. Behav. 2011, 59, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Luteijn, F.; Van der Ploeg, F. Handleiding Groninger Intelligentietest (GIT) [Manual Groningen Intelligence Test]; Swets and Zeitlinger: Leiden, The Netherlands, 1983. [Google Scholar]
- Rugg, M.D.; Johnson, J.D.; Park, H.; Uncapher, M.R. Encoding-retrieval overlap in human episodic memory: A functional neuroimaging perspective. Prog. Brain Res. 2008, 169, 339–352. [Google Scholar]
- Kim, H. Neural correlates of explicit and implicit memory at encoding and retrieval: A unified framework and meta-analysis of functional neuroimaging studies. Biol. Psychol. 2019, 145, 96–111. [Google Scholar] [CrossRef]
- Hinton, E.; Holland, A.; Gellatly, M.; Soni, S.; Patterson, M.; Ghatei, M.A.; Owen, A.M. Neural representations of hunger and satiety in Prader–Willi syndrome. J. Eat. Disord. 2005, 20, 313–321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uddin, L.Q.; Nomi, J.S.; Hébert-Seropian, B.; Ghaziri, J.; Boucher, O. Structure and Function of the Human Insula. J. Clin. Neurophysiol. 2017, 34, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Clark, L.; Bechara, A.; Damasio, H.; Aitken, M.R.F.; Sahakian, B.J.; Robbins, T.W. Differential effects of insular and ventromedial prefrontal cortex lesions on risky decision-making. Brain 2008, 131, 1311–1322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mak, Y.E.; Simmons, K.B.; Gitelman, D.R.; Small, D.M. Taste and olfactory intensity perception changes following left insular stroke. Behav. Neurosci. 2005, 119, 1693–1700. [Google Scholar] [CrossRef]
- Blanco-Hinojo, L.; Pujol, J.; Esteba-Castillo, S.; Martinez-Vilavella, G.; Gimenez-Palop, O.; Gabau, E.; Casamitjana, L.; Deus, J.; Novell, R.; Caixàsd, A. Lack of response to disgusting food in the hypothalamus and related structures in Prader Willi syndrome. Neuroimage Clin. 2019, 21, 101662. [Google Scholar] [CrossRef]
- Dykens, E.M. Contaminated and unusual food combinations: What do people with Prader-Willi syndrome choose? Ment. Retard. 2000, 38, 163–171. [Google Scholar] [CrossRef]
- Murray, E.A.; Izquierdo, A. Orbitofrontal cortex and amygdala contributions to affect and action in primates. Ann. N. Y. Acad. Sci. 2007, 1121, 273–296. [Google Scholar] [CrossRef] [Green Version]
- Siep, S.; Roefs, A.; Roebroeck, A.; Havermans, R.; Bonte, M.L.; Jansen, A. Hunger is the best spice: An fMRI study of the effects of attention, hunger and calorie content on food reward processing in the amygdala and orbitofrontal cortex. Behav. Brain Res. 2009, 198, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Jastreboff, A.M.; Lacadie, C.; Seo, D.; Kubat, J.; Van Name, M.A.; Giannini, C.; Savoye, M.; Constable, R.T.; Sherwin, S.R.; Caprio, S.; et al. Leptin Is Associated With Exaggerated Brain Reward and Emotion Responses to Food Images in Adolescent Obesity. Diabetes Care Nov. 2014, 37, 3061–3068. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchez-Ortiga, R.; Klibanski, A.; Tritos, N.A. Effects of recombinant human growth hormone therapy in adults with Prader-Willi syndrome: A meta-analysis. Clin. Endocrinol. 2012, 77, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Cavaglia, M.; Dombrowski, S.M.; Drazba, J.; Vasanji, A.; Boksech, P.M.; Janigro, D. Regional variation in brain capillary density and vascular response to ischemia. Brain Res. J. 2001, 910, 81–93. [Google Scholar] [CrossRef]
- Sheppard, L.D.; Vernon, P.A. Intelligence and speed of information-processing: A review of 50 years of research. Pers. Individ. Differ. 2008, 44, 535–551. [Google Scholar] [CrossRef]
- Varela, M.C.; Kok, F.; Setian, N.; Kim, C.A.; Koiffmann, C.P. Impact of molecular mechanisms, including deletion size, on Prader–Willi syndrome phenotype: Study of 75 patients. Clin. Genet. 2005, 67, 47–52. [Google Scholar] [CrossRef]
- Van Can, J.; Sloth, B.; Jensen, C.B.; Flint, A.; Blaak, E.E.; Saris, W.H. Effects of the once-daily GLP-1 analog liraglutide on gastric emptying, glycemic parameters, appetite and energy metabolism in obese, non-diabetic adults. Int J. Obes. 2014, 38, 784–793. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| PWS | Controls | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total Group (n = 12) | Men (n = 4) | Women (n = 8) | Total Group (n = 14) | Men (n = 7) | Women (n = 7) | |||||||
| Mdn | IQR | Mdn | IQR | Mdn | IQR | Mdn | IQR | Mdn | IQR | Mdn | IQR | |
| Age (years) | 22.9 | 14.5 | 26.6 | 18.3 | 22.3 | 14.6 | 28.5 | 18.0 | 28.4 | 17.4 | 28.7 | 20.1 |
| Height (m) | 1.57 * | 0.11 | 1.60 | 0.09 | 1.54 | 0.10 | 1.77 | 0.17 | 1.86 | 0.07 | 1.69 | 0.05 |
| Weight (kg) | 69.9 | 36.1 | 67.9 | 32.9 | 77.7 | 39.5 | 73.1 | 15,9 | 79.1 | 15.9 | 65.2 | 14.1 |
| BMI (kg/m²) | 29.8 * | 17.4 | 26.1 | 13.2 | 34.3 | 19.1 | 22.4 | 4.6 | 22.7 | 5.1 | 22.0 | 5.8 |
| IGF-1 (nmol/L) | 15.4 ** | 7.3 | 16.0 | 11.9 | 15.4 | 9.5 | 21.8 | 11.6 | 21.4 | 14.2 | 22.1 | 10.5 |
| IGF-1 Z-score (SDS) | −1.9 ** | 1.1 | −1.5 | 1.24 | −1.95 | 0.90 | −0.79 | 1.1 | −1.00 | 1.1 | −0.76 | 1.7 |
| Adiponectin (mg/L) | 14.2 | 6.3 | 13.2 | 9.1 | 14.2 | 5.1 | ||||||
| Ghrelin (ng/L) | 2118 | 2381 | 2118 | 2201 | 2151 | 2425 | ||||||
| Glucose (mmol/L) | 4.1 | 0.6 | 4.2 | 0.2 | 4.0 | 1.1 | ||||||
| Insulin (pmol/L) | 31.0 | 31.2 | 37.2 | 179.1 | 28.2 | 31.6 | ||||||
| Leptin (µg/L) | 23.2 | 30.7 | 20.1 | 38.7 | 29.0 | 30.7 | ||||||
| Resistin (ng/mL) | 5.1 | 3.8 | 4.3 | 5.7 | 5.1 | 3.7 | ||||||
| Triglycerides (mmol/L) | 0.80 | 0.3 | 1.75 | 2.6 | 0.80 | 0.4 | ||||||
| IQ GIT | 41 ** | 12 | 40 | 3 | 43 | 18 | 87 | 35 | 93 | 48 | 85 | 20 |
| IQ RAVEN | 65 ** | 10 | 70 | 15 | 65 | 10 | 110 | 17 | 110 | 16 | 119 | 25 |
| % correct encoding baseline | 100 | 24 | 100 | 0 | ||||||||
| % correct encoding non-food | 76 ** | 45 | 96 | 5 | ||||||||
| % correct encoding food | 61 ** | 25 | 79 | 16 | ||||||||
| RT correct encoding baseline (ms) | 1209 ** | 654 | 680 | 209 | ||||||||
| RT correct encoding non-food (ms) | 1676 ** | 489 | 1075 | 399 | ||||||||
| RT correct encoding food (ms) | 1747 ** | 615 | 1233 | 658 | ||||||||
| % correct retrieval baseline | 96 ** | 29 | 100 | 0 | ||||||||
| % correct retrieval non-food new | 89 * | 62 | 96 | 4 | ||||||||
| % correct retrieval non-food old | 63 ** | 26 | 83 | 14 | ||||||||
| % correct retrieval food new | 86 | 46 | 94 | 6 | ||||||||
| % correct retrieval food old | 39 ** | 41 | 73 | 35 | ||||||||
| RT correct retrieval baseline (ms) | 1065 ** | 651 | 718 | 148 | ||||||||
| RT correct retrieval non-food new (ms) | 1260 | 317 | 1238 | 444 | ||||||||
| RT correct retrieval non-food old (ms) | 1676 * | 892 | 1353 | 333 | ||||||||
| RT correct retrieval food new (ms) | 1347 | 327 | 1307 | 383 | ||||||||
| RT correct retrieval food old (ms) | 1476 | 1225 | 1621 | 454 | ||||||||
| ROI | MNI-Coordinates | k | Z-Value | p-Value | ||
|---|---|---|---|---|---|---|
| x | y | z | ||||
| R occipital cortex | 39 | –81 | 9 | 5367 | 7.19 | <0.0001 |
| L occipital cortex | –39 | –81 | –12 | 5367 | 6.55 | <0.0001 |
| R fusiform cortex | 33 | –45 | –12 | 5367 | 7.32 | <0.0001 |
| L fusiform cortex | –33 | –51 | –15 | 5367 | 6.63 | <0.0001 |
| R dorsomedial prefrontal cortex | 6 | 21 | 48 | 385 | 5.31 | 0.003 |
| R insula | 36 | 24 | 0 | 443 | 5.07 | 0.009 |
| R dorsolateral prefrontal cortex | 45 | 6 | 33 | 443 | 5.13 | 0.007 |
| L dorsolateral prefrontal cortex | –51 | 9 | 30 | 298 | 5.34 | 0.001 |
| L insula | –30 | 24 | 3 | 199 | 5.29 | 0.003 |
| Controls > PWS Patients | ||||||
|---|---|---|---|---|---|---|
| ROI | MNI-Coordinates | k | Z-Value | p-Value | ||
| x | y | z | ||||
| R fusiform gyrus | 33 | –45 | –18 | 13 | 4.18 | 0.003 |
| L fusiform gyrus | –33 | –57 | –21 | 36 | 3.69 | 0.013 |
| L insula | –33 | 24 | –3 | 20 | 4.06 | 0.004 |
| PWS patients > controls | ||||||
| No significant voxels | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nieuwpoort, I.C.v.; Slagboom, T.N.A.; Jakobsdóttir, S.; Deijen, J.B.; Veltman, D.J.; Curfs, L.M.G.; Drent, M.L. Food-Related Brain Activation Measured by fMRI in Adults with Prader–Willi Syndrome. J. Clin. Med. 2021, 10, 5133. https://doi.org/10.3390/jcm10215133
Nieuwpoort ICv, Slagboom TNA, Jakobsdóttir S, Deijen JB, Veltman DJ, Curfs LMG, Drent ML. Food-Related Brain Activation Measured by fMRI in Adults with Prader–Willi Syndrome. Journal of Clinical Medicine. 2021; 10(21):5133. https://doi.org/10.3390/jcm10215133
Chicago/Turabian StyleNieuwpoort, Ingrid Caroline van, Tessa N. A. Slagboom, Sigridur Jakobsdóttir, Jan Berend Deijen, Dick J. Veltman, Leopold M. G. Curfs, and Madeleine L. Drent. 2021. "Food-Related Brain Activation Measured by fMRI in Adults with Prader–Willi Syndrome" Journal of Clinical Medicine 10, no. 21: 5133. https://doi.org/10.3390/jcm10215133






