Predicted Versus Non-Predicted Opioid Administration Using Preoperative Pain Sensitivity in Patients Undergoing Gynecological Surgery: A Randomized-Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
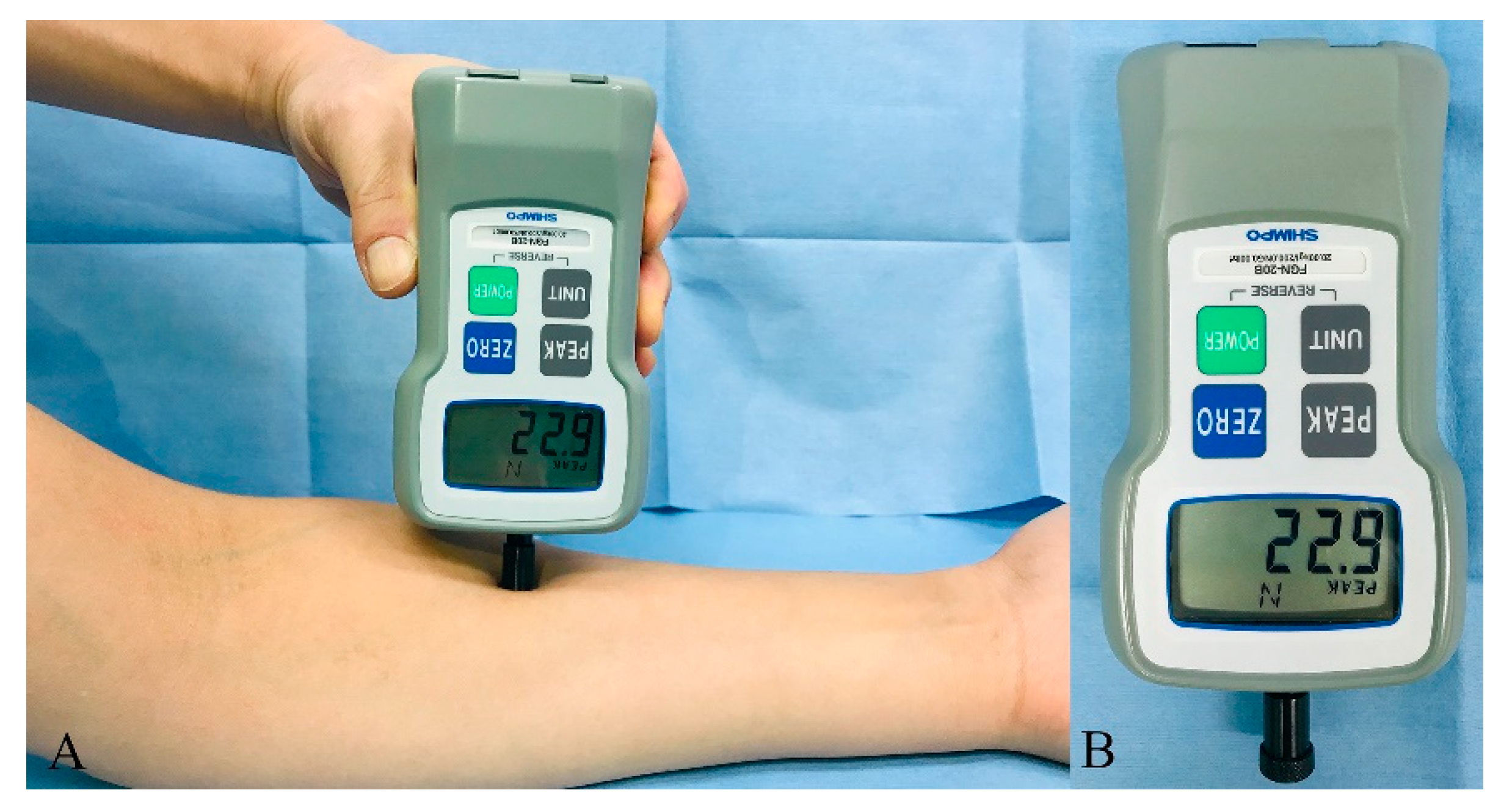
2.2. Preoperative Assessment of Pain Sensitivity
2.3. Randomization and Minimization of Bias
2.4. Anesthetic Procedures
2.5. Postoperative Management Protocol
2.6. Outcome Assessments
2.7. Statistical Analysis
2.8. Sample Size
3. Results
3.1. Opioid Use, Pain Scores and Opioid-Related Side Effects in Predicted versus Non-Predicted Groups
3.2. Effect of the Opioid Dose Given to Sensitive Patients
3.3. Effect of the Opioid Dose Given to Non-Sensitive Patients
3.4. Exploratory Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aubrun, F.; Zahr, N.; Langeron, O.; Boccheciampe, N.; Cozic, N.; Belin, L.; Hulot, J.S.; Khiami, F.; Riou, B. Opioid-related genetic polymorphisms do not influence postoperative opioid requirement: A prospective observational study. Eur. J. Anaesthesiol. 2018, 35, 496–504. [Google Scholar] [CrossRef]
- Abrishami, A.; Chan, J.; Chung, F.; Wong, J. Preoperative pain sensitivity and its correlation with postoperative pain and analgesic consumption: A qualitative systematic review. Anesthesiology 2011, 114, 445–457. [Google Scholar] [CrossRef] [PubMed]
- Aubrun, F.; Mazoit, J.X.; Riou, B. Postoperative intravenous morphine titration. Br. J. Anaesth. 2012, 108, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Nelson, G.; Bakkum-Gamez, J.; Kalogera, E.; Glaser, G.; Altman, A.; Meyer, L.A.; Taylor, J.S.; Iniesta, M.; Lasala, J.; Mena, G.; et al. Guidelines for perioperative care in gynecologic/oncology: Enhanced Recovery After Surgery (ERAS) Society recommendations-2019 update. Int. J. Gynecol. Cancer 2019. [Google Scholar] [CrossRef] [PubMed]
- Rawal, N. Current issues in postoperative pain management. Eur. J. Anaesthesiol. 2016, 33, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Oderda, G.M.; Gan, T.J.; Johnson, B.H.; Robinson, S.B. Effect of opioid-related adverse events on outcomes in selected surgical patients. J. Pain Palliat. Care Pharmacother. 2013, 27, 62–70. [Google Scholar] [CrossRef]
- Shim, H.; Gan, T.J. Side effect profiles of different opioids in the perioperative setting: Are they different and can we reduce them? Br. J. Anaesth. 2019, 123, 266–268. [Google Scholar] [CrossRef]
- Benyamin, R.; Trescot, A.M.; Datta, S.; Buenaventura, R.; Adlaka, R.; Sehgal, N.; Glaser, S.E.; Vallejo, R. Opioid complications and side effects. Pain Phys. 2008, 11, S105–S120. [Google Scholar]
- Florence, C.S.; Zhou, C.; Luo, F.; Xu, L. The Economic Burden of Prescription Opioid Overdose, Abuse, and Dependence in the United States, 2013. Med. Care 2016, 54, 901–906. [Google Scholar] [CrossRef] [PubMed]
- Oderda, G.M.; Said, Q.; Evans, R.S.; Stoddard, G.J.; Lloyd, J.; Jackson, K.; Rublee, D.; Samore, M.H. Opioid-related adverse drug events in surgical hospitalizations: Impact on costs and length of stay. Ann. Pharmacother. 2007, 41, 400–406. [Google Scholar] [CrossRef]
- Park, S.K.; Yoo, S.; Kim, W.H.; Lim, Y.J.; Bahk, J.H.; Kim, J.T. Association of nefopam use with postoperative nausea and vomiting in gynecological patients receiving prophylactic ramosetron: A retrospective study. PLoS ONE 2018, 13, e0199930. [Google Scholar]
- Li, S.C.; Wang, Y.; Choi, S.J.; Jung, Y.S.; Han, K.H.; Chung, I.B.; Lee, S.H. Scheduled injection of ramosetron for prevention of nausea and vomiting following single-port access total laparoscopic hysterectomy: A prospective randomized study. Obstet. Gynecol. Sci. 2019, 62, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Stadler, M.; Bardiau, F.; Seidel, L.; Albert, A.; Boogaerts, J.G. Difference in risk factors for postoperative nausea and vomiting. Anesthesiology 2003, 98, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Oh, C.S.; Jung, E.; Lee, S.J.; Kim, S.H. Effect of nefopam- versus fentanyl-based patient-controlled analgesia on postoperative nausea and vomiting in patients undergoing gynecological laparoscopic surgery: A prospective double-blind randomized controlled trial. Curr. Med. Res. Opin. 2015, 31, 1599–1607. [Google Scholar] [CrossRef]
- Carvalho, B.; Sutton, C.D.; Kowalczyk, J.J.; Flood, P.D. Impact of patient choice for different postcesarean delivery analgesic protocols on opioid consumption: A randomized prospective clinical trial. Reg. Anesth. Pain Med. 2019, 44, 578–585. [Google Scholar] [CrossRef]
- Van Dorp, E.L.; Romberg, R.; Sarton, E.; Bovill, J.G.; Dahan, A. Morphine-6-glucuronide: Morphine’s successor for postoperative pain relief? Anesth. Analg. 2006, 102, 1789–1797. [Google Scholar] [CrossRef]
- Abou Hammoud, H.; Simon, N.; Urien, S.; Riou, B.; Lechat, P.; Aubrun, F. Intravenous morphine titration in immediate postoperative pain management: Population kinetic-pharmacodynamic and logistic regression analysis. Pain 2009, 144, 139–146. [Google Scholar] [CrossRef]
- Weissman-Fogel, I.; Granovsky, Y.; Crispel, Y.; Ben-Nun, A.; Best, L.A.; Yarnitsky, D.; Granot, M. Enhanced presurgical pain temporal summation response predicts post-thoracotomy pain intensity during the acute postoperative phase. J. Pain 2009, 10, 628–636. [Google Scholar] [CrossRef]
- Rudin, A.; Wolner-Hanssen, P.; Hellbom, M.; Werner, M.U. Prediction of post-operative pain after a laparoscopic tubal ligation procedure. Acta Anaesthesiol. Scand. 2008, 52, 938–945. [Google Scholar] [CrossRef]
- Hsu, Y.W.; Somma, J.; Hung, Y.C.; Tsai, P.S.; Yang, C.H.; Chen, C.C. Predicting postoperative pain by preoperative pressure pain assessment. Anesthesiology 2005, 103, 613–618. [Google Scholar] [CrossRef]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef] [PubMed]
- Lautenbacher, S.; Huber, C.; Kunz, M.; Parthum, A.; Weber, P.G.; Griessinger, N.; Sittl, R. Hypervigilance as predictor of postoperative acute pain: Its predictive potency compared with experimental pain sensitivity, cortisol reactivity, and affective state. Clin. J. Pain 2009, 25, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Melia, M.; Schmidt, M.; Geissler, B.; Konig, J.; Krahn, U.; Ottersbach, H.J.; Letzel, S.; Muttray, A. Measuring mechanical pain: The refinement and standardization of pressure pain threshold measurements. Behav. Res. Methods 2015, 47, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Apfel, C.C.; Laara, E.; Koivuranta, M.; Greim, C.A.; Roewer, N. A simplified risk score for predicting postoperative nausea and vomiting: Conclusions from cross-validations between two centers. Anesthesiology 1999, 91, 693–700. [Google Scholar] [CrossRef]
- Brandsborg, B.; Dueholm, M.; Kehlet, H.; Jensen, T.S.; Nikolajsen, L. Mechanosensitivity before and after hysterectomy: A prospective study on the prediction of acute and chronic postoperative pain. Br. J. Anaesth. 2011, 107, 940–947. [Google Scholar] [CrossRef] [PubMed]
- Gamez, B.H.; Habib, A.S. Predicting Severity of Acute Pain After Cesarean Delivery: A Narrative Review. Anesth. Analg. 2018, 126, 1606–1614. [Google Scholar] [CrossRef] [PubMed]


| Characteristics | Predicted Group (n = 50) | Control Group (n = 59) | p Value |
|---|---|---|---|
| Age (y) | 48.7 (14.5) | 48.6 (13.3) | 0.995 |
| Weight (kg) | 59.1 (9.1) | 59.5 (8.8) | 0.840 |
| Height (cm) | 157.3 (6.4) | 158.5 (5.1) | 0.272 |
| Body mass index (kg/m2) | 23.9 (3.5) | 23.7 (3.3) | 0.723 |
| ASA physical status (I/II) | 37/13 (74%/26%) | 52/7 (88%/12%) | 0.082 |
| Type of gynecological surgery | 0.611 | ||
| Laparoscopic | 30 (60%) | 40 (68%) | |
| Abdominal | 11 (22%) | 12 (20%) | |
| Vaginal | 9 (18%) | 7 (12%) | |
| Operative time (min) | 93 (54–151) | 95 (65–145) | 0.692 |
| Anesthesia time (min) | 130 (85–185) | 135 (105–175) | 0.293 |
| Intraoperative remifentanil use (µg) | 725 (500–1000) | 1000 (550–1000) | 0.063 |
| Pressure pain threshold (N) | 28.2 (9.2) | 28.6 (9.8) | 0.857 |
| Sensitive/Non-sensitive | 29/21 (58%/42%) | 30/29 (51%/49%) | 0.455 |
| Higher dose/Lower dose | 29/21 (58%/42%) | 39/20 (66%/34%) | 0.502 |
| Use of the correspond regimen to sensitivity | 50 (100%) | 34 (58%) | < 0.0001 |
| Hospital stays (d) | 4 (3–5) | 4 (3–5) | 0.905 |
| Outcomes | Predicted Group (n = 50) | Control Group (n = 59) | p Value |
|---|---|---|---|
| Pain score at 3 h postoperatively (NRS) | 5 (2–7) | 5 (2–7) | 0.946 |
| Pain score at 24 h postoperatively (NRS) | 3 (2–4.3) | 3 (2–5) | 0.232 |
| Pain score at 48 h postoperatively (NRS) | 3 (2–3.3) | 2 (1–4) | 0.491 |
| Mean pain score, during the 48 h postoperative period (NRS) | 3.3 (1.8) | 3.5 (1.8) | 0.691 |
| Fentanyl consumption via IV PCA, within the first 3 h postoperatively (µg) | 68.5 (45–117.8) | 88 (60–119) | 0.166 |
| Fentanyl consumption via IV PCA, 3–24 h postoperatively (µg) | 214.8 (145.5–294.8) | 264 (152–352.5) | 0.170 |
| Fentanyl consumption via IV PCA, 24–48 h postoperatively (µg) | 64 (0–246.8) | 222 (0–307.5) | 0.089 |
| Total cumulative fentanyl consumption via IV PCA, for the first 48 h period (µg) | 406 (309.8–614.6) | 526.5 (370.5–718.5) | 0.042 |
| Nefopam consumption via IV PCA, within the first 3 h postoperatively (mg) | 4.6 (3.0–6.6) | 5.0 (4.0–7.5) | 0.203 |
| Nefopam consumption via IV PCA, 3–24 h postoperatively (mg) | 14.4 (8.9–17.8) | 14.6 (11.3–20.0) | 0.401 |
| Nefopam consumption via IV PCA, 24–48 h postoperatively (mg) | 5.1 (0.0–18.2) | 14.3 (0.0–19.1) | 0.157 |
| Total cumulative Nefopam consumption via IV PCA, for the first 48 h period (mg) | 28.0 (17.5–36.9) | 35.2 (23.7–41.1) | 0.067 |
| PCA clamping, for the first 48 h period | 6 (12%) | 10 (16.9%) | 0.648 |
| Number of patients requiring rescue analgesics for the first 48 h period | 15 (30%) | 13 (22%) | 0.343 |
| Cumulative dose of ketorolac as rescue analgesics for the first 48 h period (mg) | 16.8 (32.7) | 10.7 (22.1) | 0.347 |
| Satisfaction score for pain managements a | 77 (67–90) | 70 (57–87) | 0.140 |
| Outcomes | Predicted Group (n = 50) | Control Group (n = 59) | p Value |
|---|---|---|---|
| Nausea at 3 h postoperatively | 9 (18%) | 14 (23.7%) | 0.465 |
| Nausea at 24 h postoperatively | 13 (26%) | 20 (33.9%) | 0.371 |
| Nausea at 48 h postoperatively | 3 (6%) | 10 (16.9%) | 0.136 |
| Nausea during the 48 h postoperative period | 20 (40%) | 31 (52.5%) | 0.191 |
| Severity of nausea (NRS) a | 1.3 (0.7–2.8) | 2.3 (1.3–3.0) | 0.076 |
| Vomiting | 5 (10%) | 6 (10.2%) | 1.000 |
| Administration of rescue antiemetic agents | 5 (10%) | 6 (10.2%) | 1.000 |
| Respiratory depression | 3 (6%) | 4 (6.8%) | 1.000 |
| Shivering | 7 (14%) | 6 (10.2%) | 0.567 |
| Pruritus | 1 (2%) | 2 (3.4%) | 1.000 |
| Urinary retention | 5 (10%) | 5 (8.5%) | 1.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.-K.; Kim, H.; Yoo, S.; Kim, W.H.; Lim, Y.-J.; Kim, J.-T. Predicted Versus Non-Predicted Opioid Administration Using Preoperative Pain Sensitivity in Patients Undergoing Gynecological Surgery: A Randomized-Controlled Trial. J. Clin. Med. 2021, 10, 585. https://doi.org/10.3390/jcm10040585
Park S-K, Kim H, Yoo S, Kim WH, Lim Y-J, Kim J-T. Predicted Versus Non-Predicted Opioid Administration Using Preoperative Pain Sensitivity in Patients Undergoing Gynecological Surgery: A Randomized-Controlled Trial. Journal of Clinical Medicine. 2021; 10(4):585. https://doi.org/10.3390/jcm10040585
Chicago/Turabian StylePark, Sun-Kyung, Hansol Kim, Seokha Yoo, Won Ho Kim, Young-Jin Lim, and Jin-Tae Kim. 2021. "Predicted Versus Non-Predicted Opioid Administration Using Preoperative Pain Sensitivity in Patients Undergoing Gynecological Surgery: A Randomized-Controlled Trial" Journal of Clinical Medicine 10, no. 4: 585. https://doi.org/10.3390/jcm10040585
APA StylePark, S.-K., Kim, H., Yoo, S., Kim, W. H., Lim, Y.-J., & Kim, J.-T. (2021). Predicted Versus Non-Predicted Opioid Administration Using Preoperative Pain Sensitivity in Patients Undergoing Gynecological Surgery: A Randomized-Controlled Trial. Journal of Clinical Medicine, 10(4), 585. https://doi.org/10.3390/jcm10040585






