The Safety of Pharmacological and Surgical Treatment of Diabetes in Patients with Diabetic Retinopathy—A Review
Abstract
:1. Introduction
2. Material and Methods
3. Results and Discussion
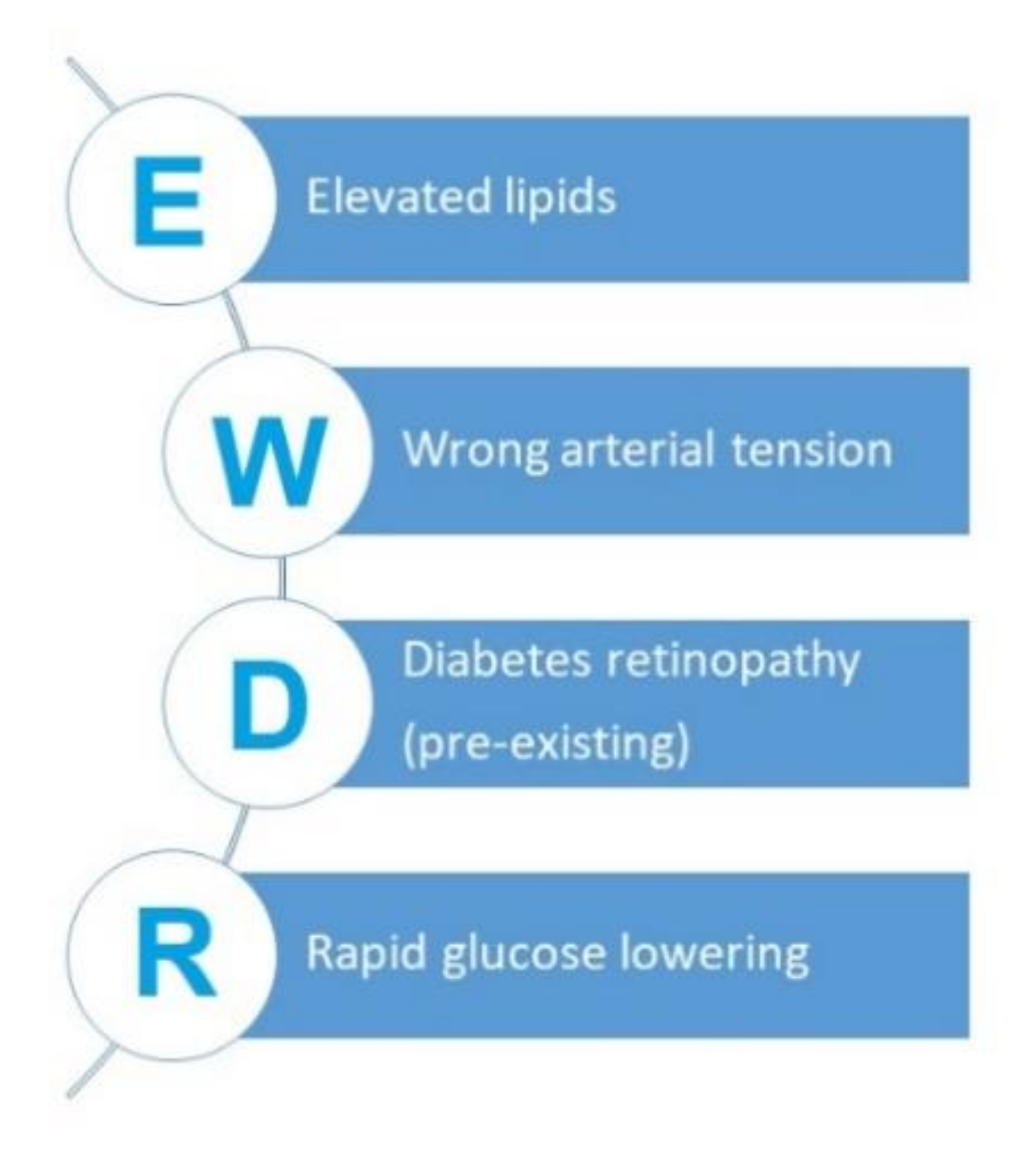
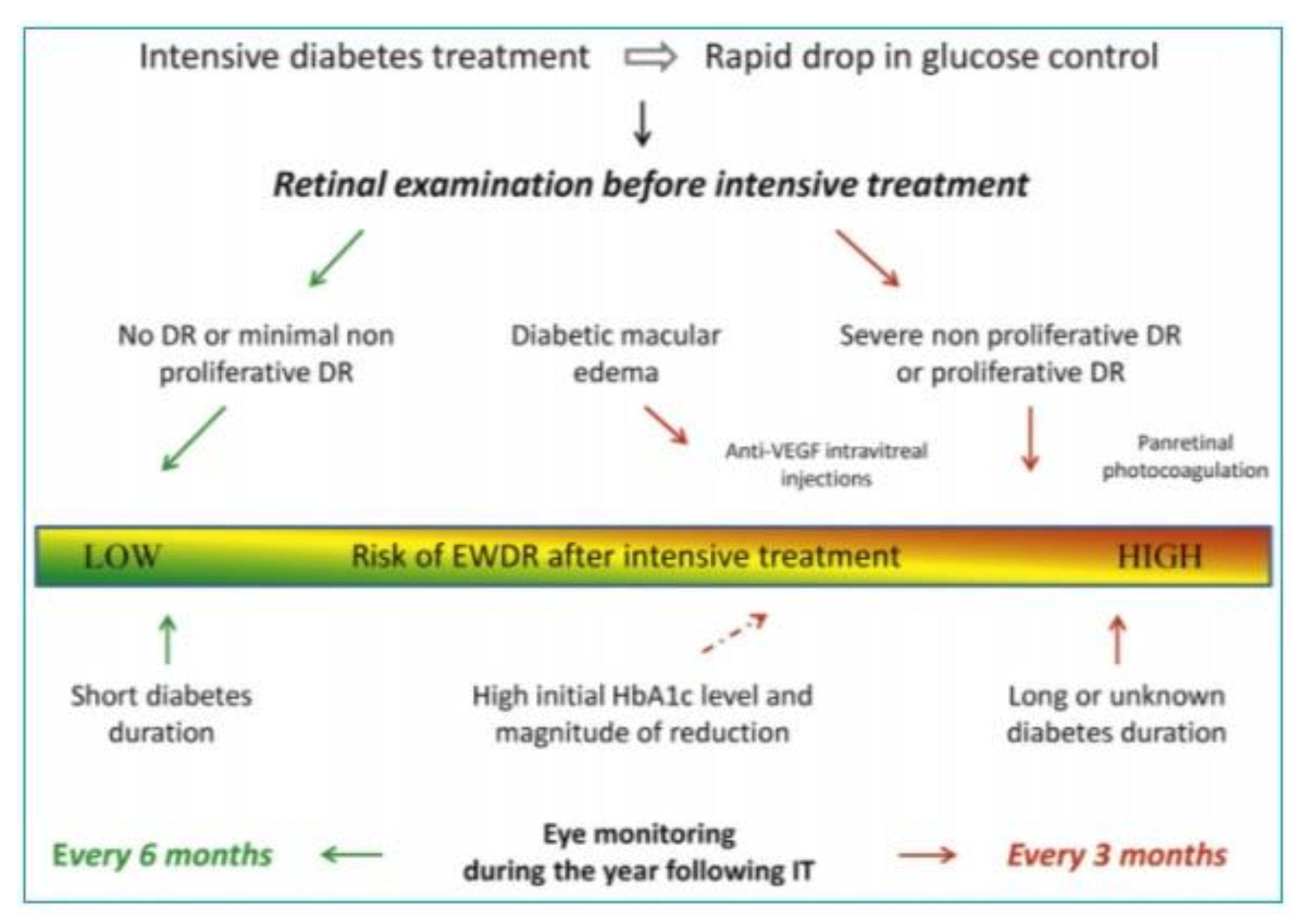
3.1. The Phenomenon of Early Worsening of Diabetic Retinopathy
3.2. Mechanisms of EWDR Development
3.3. Pharmacological Treatment
3.3.1. Insulin
3.3.2. Metformin
3.3.3. Sulfonylurea Derivatives
3.3.4. Thiazolidinediones
3.4. New Antihyperglycaemic Drugs
3.4.1. Incretin Agents
I-DPP4
GLP-1RA
3.4.2. Flozins
3.5. Surgical Treatment of Diabetes
3.5.1. Pancreatic Islet Transplantation
3.5.2. Metabolic Surgery
3.6. Lifestyle Changes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Definition, Diagnosis and Classification of Diabetes Mellitus and Its Complications. Report of a WHO Consultation. Part 1. Diagnosis and Classification of Diabetes Mellitus; WHO: Geneva, Switzerland, 1999. [Google Scholar]
- International Diabetes Federation. Diabetes Atlas 2017 8th Edition. Available online: http://diabetesatlas.org (accessed on 1 February 2020).
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; IDF Diabetes Atlas Committee; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th ed. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [Green Version]
- Solomon, S.D.; Chew, E.; Duh, E.J.; Sobrin, L.; Sun, J.K.; Van der Beek, B.L.; Wykoff, C.C.; Gardner, T.W. Diabetic Retinopathy: A Position Statement by the American Diabetes Association. Diabetes Care 2018. [Google Scholar] [CrossRef] [Green Version]
- Aiello, L.P.; Gardner, T.W.; King, G.L. Diabetic retinopathy. Diabetes Care 1998, 21, 143–156. [Google Scholar] [CrossRef]
- Solomon, S.D.; Chew, E.; Duh, E.J.; Sobrin, L.; Sun, J.K.; Van der Beek, B.L.; Wykoff, C.C.; Gardner, T.W. Diabetic Retinopathy: A Position Statement by the American Diabetes Association. Diabetes Care 2017, 40, 412–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dahl-Jorgensen, K. Near-normoglycemia and late diabetic complications. Oslo Study. Acta Endocrinol. Suppl. 1987, 284, 1–38. [Google Scholar]
- Early worsening of diabetic retinopathy in the Diabetes Control and Complications Trial [published correction appears in Arch Ophthalmol 1998 Nov;116(11):1469]. Arch. Ophthalmol. 1998, 116, 874–886. [CrossRef]
- Viswanath, K.; McGavin, D.D. Diabetic retinopathy: Clinical findings and management. Commun. Eye Health 2003, 16, 21–24. [Google Scholar]
- Bain, S.C.; Klufas, M.A.; Ho, A.; Matthews, D.R. Worsening of diabetic retinopathy with rapid improvement in systemic glucose control: A review. Diabetes Obes. Metab. 2019, 21, 454–466. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.Y.; Cheung, C.M.; Larsen, M.; Sharma, S.; Simo, R. Diabetic retinopathy. Nat. Rev. Dis. Primers. 2016, 2, 16012. [Google Scholar] [CrossRef]
- UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ 1998, 317, 703–713. [Google Scholar] [CrossRef] [Green Version]
- Matthews, D.R.; Stratton, I.M.; Aldington, S.J.; Holman, R.R.; Kohner, E.M. Risks of progression of retinopathy and vision loss related to tight blood pressure control in type 2 diabetes mellitus: UKPDS 69. Arch. Ophthalmol. 2004, 122, 1631–1640. [Google Scholar]
- Liu, L.; Quang, N.D.; Banu, R.; Kumar, H.; Tham, Y.-C.; Cheng, C.-Y.; Wong, T.Y.; Sabanayagam, C. Hypertension, blood pressure control and diabetic retinopathy in a large population-based study. PLoS ONE 2020, 15, e0229665. [Google Scholar] [CrossRef] [PubMed]
- The Early Treatment Diabetic Retinopathy Study Research Group. Association of abstract serum lipid levels with retinal hard exudate in diabetic retinopathy: ETDRS report no. 22. Arch. Ophthalmol. 1987, 94, 1614–1620. [Google Scholar]
- Gordon, B.; Chang, S.; Kavanagh, B.M.; Yannuzi, L.; Robertson, C.; Drexler, A. The effects of lipid lowering on diabetic retinopathy. Am. J. Ophthalmol. 1991, 112, 385–391. [Google Scholar] [CrossRef]
- Cusick, M.; Chew, E.Y.; Chan, C.C.; Kruth, H.S.; Murphy, R.P.; Ferris, F.L., III. Histopathology and regression of retinal hard exudates in diabetic retinopathy after reduction of elevated serum lipid levels. Ophthalmology 2003, 110, 2126–2133. [Google Scholar] [CrossRef] [PubMed]
- Miljanovic, B.; Glynn, R.J.; Nathan, D.M. A prospective study of serum lipids and risk of diabetic macular edema in type 1 diabetes. Diabetes 2004, 53, 2883–2892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Wang, J.; Ying, G. Serum lipids and other risk factors for diabetic retinopathy in Chinese type 2 diabetic patients. J. Zhejiang Univ. 2013, 5, 392–399. [Google Scholar] [CrossRef] [Green Version]
- Muhammad, B.S.; Tien, Y.; Wong, T.T.; Nguyen, R.K.; Alicia, J.; Jonathan, S.; Jie, J.W. Serum Apolipoprotein AI and B Are Stronger Biomarkers of Diabetic Retinopathy Than Traditional Lipids. Diabetes Care Feb. 2011, 34, 474–479. [Google Scholar] [CrossRef] [Green Version]
- Littmann, K.; Wodaje, T.; Alvarsson, M.; Bottai, M.; Eriksson, M.; Parini, P.; Brinck, J. The Association of Lipoprotein(a) plasma levels with prevalence of cardiovascular disease and metabolic control status in patients with type 1 diabetes. Diabetes Care 2019, 43, 1851–1858. [Google Scholar] [CrossRef] [PubMed]
- Koschinsky, M.L.; Marcovina, S.M. The relationship between lipoprotein(a) and the complications of diabetes mellitus. Acta Diabetol. 2003, 40, 65–76. [Google Scholar] [CrossRef]
- Feldman-Billard, S.É.; Larger, P.M. Early worsening of diabetic retinopathy after rapid improvement of blood glucose control in patients with diabetes. Diabetes Metab. 2018, 44, 4–14. [Google Scholar] [CrossRef]
- Zapuskalov, I.V.; Filippova, S.V.; Shilova, O.G.; Krivosheina, O.I. Rol’ osmoticheskogo davleniia krovi v patogeneze diabeticheskikh izmeneniĭ setchatki [The role of the osmotic pressure of the blood in the pathogenesis of diabetic changes in the retina]. Vestn. Oftalmol. 2000, 116, 32–34. [Google Scholar]
- Tarr, J.M.; Kaul, K.; Chopra, M.; Kohner, E.M.; Chibber, R. Pathophysiology of Diabetic Retinopathy. Int. Sch. Res. Not. 2013, 2013, 13. [Google Scholar] [CrossRef] [Green Version]
- Willermain, F.; Scifo, L.; Weber, C.; Caspers, L.; Perret, J.; Delporte, C. Potential Interplay between Hyperosmolarity and Inflammation on Retinal Pigmented Epithelium in Pathogenesis of Diabetic Retinopathy. Int. J. Mol. Sci. 2018, 19, 1056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eshaq, R.S.; Aldalati, A.M.Z.; Alexander, J.S.; Harris, N.R. Diabetic retinopathy: Breaking the barrier. Pathophysiology 2017, 24, 229–241. [Google Scholar] [CrossRef]
- Altmann, C.; Schmidt, M.H.H. The Role of Microglia in Diabetic Retinopathy: Inflammation, Microvasculature Defects and Neurodegeneration. Int. J. Mol. Sci. 2018, 19, 110. [Google Scholar] [CrossRef] [Green Version]
- Min-Heui, Y.; Young, H.Y.; Hyewon, C.; Kyung, S.; Cho, J.-Y.K. Insulin increases retinal hemorrage in mild oxygen-Induced retinopathy in the rat: Inhibition by riluzole. Investig. Ophthalmol. Vis. Sci. 2007, 48, 1. [Google Scholar]
- Jingi, M.A.; Tankeu, T.A.; Ateba, N.A.; Noubiap, J.J. Mechanism of worsening diabetic retinopathy with rapid loweringt of blood glucose: The synergistic hypothesis. Bmc Endocr. Disord. 2017, 17, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, D.; Mei, A.; Liu, J.; Kang, X.; Shi, X.; Qian, R.; Chen, S. NADPH Oxidase 4 Mediates Insulin-Stimulated HIF-1α and VEGF Expression, and Angiogenesis In Vitro. PLoS ONE 2012, 7, e48393. [Google Scholar] [CrossRef]
- Dhoot, D.S.; Avery, R.L. Vascular endothelial growth factor inhibitors for diabetic retinopathy. Curr. Diab. Rep. 2016, 16, 122. [Google Scholar] [CrossRef]
- Simo, R.; Sundstrom, J.M.; Antonetti, D.A. Ocular anti-VEGF therapy for diabetic retinopathy: The role of VEGF in the pathogenesis of diabetic retinopathy. Diabetes Care 2014, 37, 893–899. [Google Scholar] [CrossRef] [Green Version]
- Clemmons, D.R. The relative roles of growth hormone and IGF-1 in controlling insulin sensitivity. J. Clin. Investig. 2004, 113, 25–27. [Google Scholar] [CrossRef] [Green Version]
- Bronson, S.K.; Reiter, C.E.N.; Gardner, T.W. An eye on insulin. J. Clin. Investig. 2003, 111, 1817–1819. [Google Scholar] [CrossRef]
- Ruberte, J.; Ayuso, E.; Navarro, M.; Carretero, A.; Nacher, V.; Haurigot, V.; George, M.; Llombart, C.; Casellas, A.; Costa, C.; et al. Increased ocular levels of IGF-1 in transgenic mice lead to diabetes-like eye disease. J. Clin. Investig. 2004, 113, 1149–1157. [Google Scholar] [CrossRef] [Green Version]
- Ann, H.; Björn, C.; Aimon, N.; Karin, S.; Margaret, B.L.L.; Martin, S.; Elisabeth, S.; Lois, S.; Dov, W.; Kerstin, A.; et al. IGF-I Is Critical for Normal Vascularization of the Human Retina. J. Clin. Endocrinol. Metab. 2002, 87, 3413–3416. [Google Scholar] [CrossRef] [Green Version]
- Chen, H.; Wu, T.; Hsiao, L.; Lin, S. Interaction between glycaemic control and serum insulin-like growth factor 1 on the risk of retinopathy in type 2 diabetes. Eur. J. Clin. Investig. 2012, 42, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Growth Hormone Antagonist for Proliferative Diabetic Retinopathy Study Group. The effect of a growth hormone receptor antagonist drug on proliferative diabetic retinopathy. Ophthalmology 2001, 108, 2266–2271. [Google Scholar] [CrossRef]
- Ceriello, A. The emerging challenge in diabetes: The “metabolic memory”. Vascul. Pharmacol. 2012, 57, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Ceriello, A.; Ihnat, M.A. ‘Glycaemic variability’: A new therapeutic challenge in diabetes and the critical care setting. Diabet. Med. 2010, 27, 862–867. [Google Scholar] [CrossRef]
- Prattichizzo, F.; Giuliani, A.; Ceka, A.; Rippo, M.R.; Bonfigli, A.R.; Testa, R.; Procopio, A.D.; Olivieri, F. Epigenetic mechanisms of endothelial dysfunction in type 2 diabetes. Clin. Epigenet. 2015, 7, 56. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Q.; Kowluru, R.A. Epigenetic modification of Sod2 in the development of diabetic retinopathy and in the metabolic memory: Role of histone methylation. Investig. Ophthalmol. Vis. Sci. 2012, 54, 244–250. [Google Scholar] [CrossRef] [Green Version]
- Zhong, Q.; Kowluru, R.A. Epigenetic changes in mitochondrial superoxide dismutase in the retina and the development of diabetic retinopathy. Diabetes 2011, 60, 1304–1313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fish, J.E.; Matouk, C.C.; Rachlis, A.; Lin, S.; Tai, S.C.; D’Abreo, C.; Marsden, P.A. The expression of endothelial nitric-oxide synthase is controlled by a cell-specific histone code. J. Biol. Chem. 2005, 280, 24824–24838. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salzmann, J.; Limb, G.A.; Khaw, P.T.; Gregor, Z.J.; Webster, L.; Chignell, A.H.; Charteris, D.G. Matrix metalloproteinases and their natural inhibitors in fibrovascular membranes of proliferative diabetic retinopathy. Br. J. Ophthalmol. 2000, 84, 1091–1096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kowluru, R.A. Role of matrix metalloproteinase-9 in the development of diabetic retinopathy and its regulation by H-Ras. Investig. Ophthalmol. Vis. Sci. 2010, 51, 4320–4326. [Google Scholar] [CrossRef] [Green Version]
- Kowluru, R.A.; Mohammad, G.; dos Santos, J.M.; Zhong, Q. Abrogation of MMP-9 gene protects against the development of retinopathy in diabetic mice by preventing mitochondrial damage. Diabetes 2011, 60, 3023–3033. [Google Scholar] [CrossRef] [Green Version]
- Mohammad, G.; Kowluru, R.A. Diabetic retinopathy and signalling mechanism for activation of matrix metalloproteinase-9. J. Cell. Physiol. 2011, 227, 1052–1061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arguelles, A.O.; Meruvu, S.; Bowman, J.D.; Choudhury, M. Are epigenetic drugs for diabetes and obesity at our door step? Drug Discov. Today 2016, 21, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanyam, K.; Altaf, M.; Varier, R.A.; Swaminathan, V.; Ravindran, A.; Sadhale, P.P.; Kundu, T.K. Polyisoprenylated benzophenone, garcinol, a natural histone acetyltransferase inhibitor, represses chromatin transcription and alters global gene expression. J. Biol. Chem. 2004, 279, 33716–33726. [Google Scholar] [CrossRef] [Green Version]
- Kadiyala, C.S.; Zheng, L.; Du, Y.; Yohannes, E.; Kao, H.Y.; Miyagi, M.; Kern, T.S. Acetylation of retinal histones in diabetes increases inflammatory proteins: Effects of minocycline and manipulation of histone acetyltransferase (HAT) and histone deacetylase (HDAC). J. Biol. Chem. 2012, 287, 25869–25880. [Google Scholar] [CrossRef] [Green Version]
- Yun, J.M.; Jialal, I.; Devaraj, S. Epigenetic regulation of high glucose-induced proinflammatory cytokine production in monocytes by curcumin. J. Nutr. Biochem. 2011, 22, 450–458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wickenberg, J.; Ingemansson, S.L.; Hlebowicz, J. Effects of Curcuma longa (turmeric) on postprandial plasma glucose and insulin in healthy subjects. Nutr. J. 2010, 9, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chous, A.P.; Richer, S.P.; Gerson, J.D.; Kowluru, R.A. The Diabetes Visual Function Supplement Study (DiVFuSS). Br. J. Ophthalmol. 2016, 100, 227–234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reddy, M.A.; Natarajan, R. Role of epigenetic mechanisms in the vascular complications of diabetes. Subcell. Biochem. 2012, 61, 435–454. [Google Scholar]
- Rosenfeld, L. Insulin: Discovery and controversy. Clin. Chem. 2002, 48, 2270–2288. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hooymans, J.M.; Ballegooie, E.V.; Schweitzer, N.M.; Doorebos, H.; Reitsma, W.D.; Slutter, W.J. Worsening of diabetic retinopathy with strict control of blood sugar. Lancet 1982, 2, 438. [Google Scholar] [CrossRef]
- The Kroc Collaborative Study Group. Blood glucose control and the evolution of diabetic retinopathy and albuminuria. A preliminary multicenter trial. N. Engl. J. Med. 1984, 311, 365–372. [Google Scholar] [CrossRef]
- Brinchmann-Hansen, O.; Dahl-Jorgensen, K.; Hanssen, K.F.; Sandvik, L. Effects of intensified insulin treatment on various lesions of diabetic retinopathy. Am. J. Ophthalmol. 1985, 100, 644–653. [Google Scholar] [PubMed]
- Zhao, C.; Wang, W.; Xu, D.; Li, H.; Li, M.; Wang, F.; Li, H.; Li, M.; Wang, F. Insulin and risk of diabetic retinopathy in patients with type 2 diabetes mellitus: Data from a metaanalysis of seven cohort studies. Diagn. Pathol. 2014, 9, 130. [Google Scholar] [CrossRef] [Green Version]
- Henricsson, M.; Berntorp, K.; Fernlund, P.; Sundkvist, G. Progression of retinopathy in inulin treated type 2 diabetic patients. Diabetes Care 2002, 25, 381–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Poulaki, V.; Joussen, A.M.; Mitsiades, N.; Mitsiades, C.S.; Iliaki, E.F.; Adamis, A.P. Insulin-like growth factor-I plays a pathogenetic role in diabetic retinopathy. Am. J. Pathol. 2004, 165, 457–469. [Google Scholar] [CrossRef] [Green Version]
- Wilkinson-Berka, J.L.; Wraight, C.; Werther, G. The role of growth hormone, insulin-like growth factor and somatostatin in diabetic retinopathy. Curr. Med. Chem. 2006, 13, 3307–3317. [Google Scholar] [CrossRef]
- Wang, H.; Xu, J.; Chen, J.; Little, P.J.; Zheng, W. Role of IGF-1 signaling in the pathology of diabetic retinopathy. Ther. Targets Neurol. Dis. 2015, 2, 785–793. [Google Scholar]
- Kiepe, D.; Tönshoff, B. Insulin-Like Growth Factors in Normal and Diseased Kidney. Endocrinol. Metab. Clin. 2015, 41, 351–374. [Google Scholar] [CrossRef]
- Rosenstock, J.; Fonseca, V.; McGill, J.B.; Riddle, M.; Hallé, J.P.; Hramiak, I.; Johnston, P.; Davis, M. Similar progression of diabetic retinopathy with insulin glargine and neutral protamine Hagedorn (NPH) insulin in patients with type 2 diabetes: A long-term, randomised, open-label study. Diabetologia 2009, 52, 1778–1788. [Google Scholar] [CrossRef] [Green Version]
- American Diabetes Association. Standard of Medical Care in Diabetes-2020. Am. Diabetes Assoc. 2020, 43 (Suppl. S1), 152–163. [Google Scholar]
- Davies, M.J.; D’Alessio, D.A.; Fradkin, J.; Kernan, W.N.; Mathieu, C.; Mingrone, G.; Rossing, P.; Tsapas, A.; Wexler, D.J.; Buse, J.B. Management of hyperglycaemia in type 2 diabetes, 2018. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2018, 61, 2461–2498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- UK Prospective Diabetes Study (UKPDS) Group. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). Lancet 1998, 352, 854–865. [Google Scholar] [CrossRef]
- Li, Y.; Ryu, C.; Munie, M.; Noorula, S.; Rana, S.; Edwards, P.; Gao, H.; Qiao, X. Association of Metformin Treatment with Reduced Severity of Diabetic Retinopathy in Type 2 Diabetic Patients. Hindawi J. Diabetes Res. 2018, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryu, C.; Munie, M.; Noorulla, S.; Edwards, P.; Qiao, X.; Gao, H. Effect of Metformin on the Development of Diabetic Retinopathy. Investig. Ophthalmol. Vis. Sci. 2018, 54, 2449. [Google Scholar]
- Han, J.; Li, Y.; Liu, X.; Zhou, T.; Sun, H.; Edwards, P.; Gao, H.; Yu, F.-S.; Qiao, X. Metformin suppresses retinal angiogenesis and inflammation in vitro and in vivo. PLoS ONE 2018, 13, e0193031. [Google Scholar] [CrossRef] [Green Version]
- Quan-Yong, Y.; Gang, D.; Nan, C.; Zhi-Sha, B.; Jian-Shu, Y.; Guo-Hai, W.; Yu-Wen, W.; Shan-Jun, W. Metformin inhibits development of diabetic retinopathy through inducing alternative splicing of VEGF-A. Am. J. Transl. Res. 2016, 8, 3947–3954. [Google Scholar]
- Yi, Z.; Fei, C.; Liang, W. Metformin inhibits development of diabetic retinopathy through microRNA-497a-5p. Am. J. Transl. Res. 2017, 9, 5558–5566. [Google Scholar]
- Loubatières-Mariani, M.M. La découverte des sulfamides hypoglycémiants. The discovery of hypoglycemic sulfonamides. J. Soc. Biol. 2007, 201, 121–125. [Google Scholar] [CrossRef]
- Sola, D.; Rossi, L.; Schianca, G.P.C.; Maffioli, P.; Bigliocca, M.; Mella, R.; Corlianò, F.; Fra, G.P.; Bartoli, E.; Derosa, G. Sulfonylureas and their use in clinical practice. Arch. Med. Sci. 2015, 11, 840–848. [Google Scholar] [CrossRef]
- Lebovitz, H.E.; Feinglos, M.N. Sulfonylurea drugs: Mechanism of antidiabetic action and therapeutic usefulness. Diabetes Care 1978, 1, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Baba, S.; Nakagawa, S.; Takebe, K.; Goto, Y.; Maezawa, H.; Takeda, R.; Sakamoto, N.; Fukui, I. Comparison of gliclazyde and glibenclamide treatment in non-insulin-dependent diabetes. Tohoku J. Exp. Med. 1983, 141, 693–706. [Google Scholar] [CrossRef] [Green Version]
- Minami, N.; Ikeda, Y.; Abe, M. Preventive and therapeutic effects of gliclazyde on diabetic retinopathy: Comparison with gliblenclamide treatment. Toboku J. Exp. Med. 1983, 141, 707–711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diabetic Retinopathy Program Research Group. Clinical comparative study on the therapeutic effects of oral hypoglycemic agents in patients with diabetic retinopathy. J. Jpn. Diabet. Soc. 1983, 26, 531–570. [Google Scholar]
- Tang, H.; Li, G.; Zhao, Y.; Wang, F.; Gower, E.W.; Shi, L.; Wang, T. Comparisons of diabetic retinopathy events associated with glucoselowering drugs in patients with type 2 diabetes mellitus: A network metaanalysis. Diabetes Obes. Metab. 2018, 20, 1262–1279. [Google Scholar] [CrossRef]
- Schernthaner, G.; Grimaldi, A.; Di Mario, U.; Drzewoski, J.; Kempler, P.; Kvapil, M.; Novials, A.; Rottiers, R.; Rutten, G.E.H.M.; Shaw, K.M. GUIDE study: Double-blind comparison of once-daily gliclazide MR and glimepiride in type 2 diabetic patients. Eur. J. Clin. Invest. 2004, 34, 535–542. [Google Scholar] [CrossRef]
- Konya, H.; Hasegawa, Y.; Hamaguchi, T.; Satani, K.; Umehara, A.; Katsuno, T.; Ishikawa, T.; Miuchi, M.; Kohri, K.; Suehiro, A.; et al. Effects of gliclazide on platelet aggregation and the plasminogen activator inhibitor type 1 level in patients with type 2 diabetes mellitus. Metabolism 2010, 59, 1294–1299. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.M.; Moore, L.B.; Smith-Oliver, T.A.; Wilkison, W.O.; Willson, T.M.; Kliewer, S.A. An antidiabetic thiazolidinedione is a high affinity ligand for peroxisome proliferator-activated receptor gamma (PPAR gamma). J. Biol. Chem. 1995, 270, 953–956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzuki, S.; Arnold, L.L.; Pennington, K.L. Effects of pioglitazone, a peroxisome proliferator-activated receptor gamma agonist, on the urine and urothelium of the rat. Toxicol. Sci. 2010, 113, 349–357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gale, E.A.M. Lessons from the glitazones: A story of drug development. Lancet 2001, 357, 1870–1875. [Google Scholar] [CrossRef]
- Idris, I.; Warren, G.; Donnelly, R. Association between thiazolidinedione treatment and risk of macular edema among patients with type 2 diabetes. Arch. Intern. Med. 2012, 172, 1005–1011. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fong, D.S.; Contreras, R. Glitazone use associated with diabetic macular edema. Am. J. Ophthalmol. 2009, 147, 583–586. [Google Scholar] [CrossRef]
- Shen, L.Q.; Child, A.; Weber, G.M.; Folkman, J.; Aiello, L.P. Rosiglitazone and Delayed Onset of Proliferative Diabetic Retinopathy. Arch. Ophthalmol. 2008, 126, 793–799. [Google Scholar] [CrossRef] [Green Version]
- ACCORD Study Group. Lack of Association between Thiazolidinediones and Macular Edema in Type 2 Diabetes: The ACCORD Eye Study. Arch. Ophthalmol. 2010, 128, 312–318. [Google Scholar] [CrossRef] [Green Version]
- Mcintyren, N.; Holdsworth, C.D.; Turner, D.S. New Interpretation of Oral Glucose Tolerance. Lancet 1964, 2, 20–21. [Google Scholar] [CrossRef]
- Scirica, B.M.; Bhatt, D.L.; Braunwald, E. Saxagliptin and Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus. N. Engl. J. Med. 2013, 369, 1317–1326. [Google Scholar] [CrossRef] [Green Version]
- White, W.B.; Cannon, C.P.; Heller, S.R. Alogliptin after Acute Coronary Syndrome in Patients with Type 2 Diabetes. N. Engl. J. Med. 2013, 369, 1327–1335. [Google Scholar] [CrossRef] [Green Version]
- Green, J.B.; Bethel, M.A.; Armstrong, P.W.; Buse, J.B.; Engel, S.S.; Garg, J.; Josse, R.; Kaufman, K.D.; Koglin, J.; TECOS Study Group; et al. Effect of Sitagliptin on Cardiovascular Outcomes in Type 2 Diabetes. N Engl. J. Med. 2015, 373, 232–242. [Google Scholar] [CrossRef] [Green Version]
- Marx, N.; Rosenstock, J.; Kahn, S.E.; Zinman, B.; Kastelein, J.J.; Lachin, J.M.; Espeland, M.A.; Bluhmki, E.; Mattheus, M.; Ryckaert, B.; et al. Design and baseline characteristics of the CARdiovascular Outcome Trial of LINAgliptin Versus Glimepiride in Type 2 Diabetes (CAROLINA®). Diabets Vasc. Dis. Res. 2015, 12, 164–174. [Google Scholar] [CrossRef] [Green Version]
- Schweizer, A.; Dejager, S.; Foley, J.E.; Couturier, A.; Ligueros-Saylan, M.; Kothny, W. Assessing the cardio-cerebrovascular safety of vildagliptin: Meta-analysis of adjudicated events from a large Phase III type 2 diabetes population. Diabetes Obes. Metab. 2010, 12, 485–494. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.; Leal, E.; Paiva, A.; Teixeira Lemos, E.; Teixeira, F.; Ribeiro, C.F.; Reis, F.; Ambrosio, A.F.; Fernandes, R. Protective effects of the dipeptidyl peptidase IV inhibitor sitagliptin in the blood-retinal barrier in a type 2 diabetes animal model. Diabetes Obes. Metab. 2012, 14, 454–463. [Google Scholar] [CrossRef]
- Maeda, S.; Yamagishi, S.; Matsui, T.; Nakashima, S.; Ojima, A.; Maeda, S.; Nishino, Y.; Ishibashi, Y.; Yoshida, Y.; Yamakawa, R. Beneficial effects of vildagliptin on retinal injury in obese type 2 diabetic rats. Ophthalmic Res. 2013, 50, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Ott, C.; Raff, U.; Schmidt, S.; Friedrich, S.; Bramlage, P.; Harazny, J.M.; Schmeider, R.E. Effects of saxagliptin on early microvascular changes in patients with type 2 diabetes. Cardiovasc. Diabetol. 2014, 13, 107. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, N.H.; Choi, J.; Kim, N.H.; Choi, K.M.; Baik, S.H.; Lee, J.; Kim, S.G. Dipeptidyl peptidase-4 inhibitor use and risk of diabetic retinopathy: A population-based study. Diabetes Metab. 2018, 44, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Yoo-Ri, C.; Kyoung, H.H.; Hyeon, C.K.; Sang, J.P.; Kihwang, L.; Dae, J.K. Dipeptidyl Peptidase-4 Inhibitors versus Other Antidiabetic Drugs Added to Metformin Monotherapy in Diabetic Retinopathy Progression: A Real WorldBased Cohort Study. Diabetes Metab. J. 2019, 43, 640–648. [Google Scholar]
- Heng, L.; Junjun, Z.; Li, L.; Lishuai, X. Vascular protection of DPP-4 inhibitors in retinal endothelial cells in in vitro culture. Int. Immunopharmacol. 2019, 66, 162–168. [Google Scholar]
- Bell, G.I.; Santerre, R.F.; Mullenbach, G.T. Hamster preproglucagon contains the sequence of glucagon and two related peptides. Nature 1983, 302, 716–718. [Google Scholar] [CrossRef]
- Holman, R.R.; Bethel, M.A.; Mentz, R.J.; Thompson, V.P.; Lokhnygina, Y.; Buse, J.B.; Chan, J.C.; Choi, J.; Gustavson, S.M.; EXSEL Study Group; et al. Effects of Once-Weekly Exenatide on Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2017, 377, 1228–1239. [Google Scholar] [CrossRef]
- Drucker, D.J.; Habener, J.F.; Holst, J.J. Discovery, characterization, and clinical development of the glucagon-like peptides. J. Clin. Investig. 2017, 127, 4217–4227. [Google Scholar] [CrossRef] [PubMed]
- Marso, S.P.; Daniels, G.H.; Brown-Frandsen, K.; Kristensen, P.; Mann, J.F.; Nauck, M.A.; Nissen, S.E.; Pocock, S.; LEADER Steering Committee; LEADER Trial Investigators; et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 311–322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marso, S.P.; Bain, S.C.; Consoli, A.; Eliaschewitz, F.G.; Jódar, E.; Leiter, L.A.; Lingvay, I.; Rosenstock, J.; Seufert, J.; SUSTAIN-6 Investigators; et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 1834–1844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vilsbøll, T.; Bain, S.C.; Leiter, L.A.; Lingvay, I.; Matthews, D.; Simó, R.; Helmark, I.C.; Wijayasinghe, N.; Larsen, M. Semaglutide, reduction in glycated haemoglobin and the risk of diabetic retinopathy. Diabetes Obes. Metab. 2018, 20, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Simo, R.; Bogdanov, P.; Ramos, H.; Huerta, J.; Hernandez, C. Effects of topical administartion (eye drops) of semaglutide on retinal neuroinflammation and vascular leakage in experimenatl diabetes. In Proceedings of the EASD 56th Annual Meeting, Virtual Meeting, 21–25 September 2020. [Google Scholar]
- Hernandez, A.F.; Green, J.B.; Janmohamed, S.; D’Agostino, R.B., Sr.; Granger, C.B.; Jones, N.P.; Leiter, L.A.; Rosenberg, A.E.; Sigmon, K.N.; Harmony Outcomes Committees and Investigators; et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (Harmony Outcomes): A double-blind, randomised placebo-controlled trial. Lancet 2018, 392, 1519–1529. [Google Scholar] [CrossRef] [Green Version]
- Gerstein, H.C.; Colhoun, H.M.; Dagenais, G.R.; Diaz, R.; Lakshmanan, M.; Pais, P.; Probstfield, J.; Riesmeyer, J.S.; Riddle, M.C.; REWIND Investigators. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): A double-blind, randomised placebo-controlled trial. Lancet 2019, 394, 121–130. [Google Scholar] [CrossRef]
- Nauck, M.A.; Quast, D.R.; Wefers, J.; Meier, J.J. GLP-1 receptor agonists in the treatment of type 2 diabetes–state-of-the-art Molecular. Metabolism 2020. [Google Scholar] [CrossRef]
- Available online: https://www.clinicaltrials.gov/ct2/show/NCT03811561?term=NN9535–4352&draw=2&rank=1 (accessed on 3 January 2021).
- Dicembrini, I.; Nreu, B.; Scatena, A.; Andreozzi, F.; Sesti, G.; Mannucci, E.; Monami, M. Microvascular effects of glucagon-like peptide-1 receptor agonists in type 2 diabetes: A meta-analysis of randomized controlled trials. Acta Diabetol. 2017, 54, 933–941. [Google Scholar] [CrossRef]
- Tiansheng, W.; Jin-Liern, H.; Emily, W.; Gower, V.P.; Seema, G.; John, B.; Buse, T.S. Incretin-Based Therapies and Diabetic Retinopathy: Real-World Evidence in Older, U.S. Adults. Diabetes Care Sep. 2018, 41, 1998–2009. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Lu, W.; Tang, H.; Buse, J.B.; Stürmer, T.; Gower, E.W. Assessing the Association Between GLP-1 Receptor Agonist Use and Diabetic Retinopathy Through the FDA Adverse Event Reporting System. Diabetes Care 2019, 42, e21–e23. [Google Scholar] [CrossRef] [PubMed]
- Ehrenkranz, R.R.L.; Lewis, N.G.; Kahn, C.R.; Roth, J. Phlorizin: A review. Diabetes Metab. Res. Rev. 2005, 21, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Cosentino, F.; Grant, P.J.; Aboyans, V.; Bailey, C.J.; Ceriello, A.; Delgado, V.; Federici, M.; Filippatos, G.; Grobbee, D.E.; ESC Scientific Document Group; et al. 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 2020, 41, 255–323. [Google Scholar] [CrossRef] [Green Version]
- Kanai, Y.; Lee, W.S.; You, G.; Brown, D.M.A.; Hediger, M.A. The human kidney low affinity Na+/glucose cotransporter SGLT2. Delineation of the major renal reabsorptive mechanism for D-glucose. J. Clin. Investig. 1994, 93, 397–404. [Google Scholar] [CrossRef]
- Wakisaka, M.; Yoshinari, M.; Nakamura, S.; Asano, T.; Sonoki, K.; Shi, A.; Iwase, M.; Takata, Y.; Fujishima, M. Suppression of sodium-dependent glucose uptake by captopril improves high-glucose-induced morphological and functional changes of cultured bovine retinal pericytes. Microvasc. Res. 1999, 58, 215–223. [Google Scholar] [CrossRef]
- Oelze, M.; Kroller-Schon, S.; Welschof, P.; Jansen, T.; Hausding, M.; Mikhed, Y.; Stamm, P.; Mader, M.; Zinßius, E.; Agdauletova, S.; et al. The sodium-glucose co-transporter 2 inhibitor empagliflozin improves diabetes-induced vascular dysfunction in the streptozotocin diabetes rat model by interfering with oxidative stress and glucotoxicity. PLoS ONE 2014, 9, e112394. [Google Scholar] [CrossRef]
- Thorp, A.A.; Schlaich, M.P. Relevance of sympathetic nervous system activation in obesity and metabolic syndrome. J. Diabetes Res. 2015, 2015, 341583. [Google Scholar] [CrossRef] [Green Version]
- Herat, L.Y.; Matthews, V.B.; Rakoczy, P.E.; Carnagarin, R.; Schlaich, M. Focusing on Sodium Glucose Cotransporter-2 and the Sympathetic Nervous System: Potential Impact in Diabetic Retinopathy. Hindawi Int. J. Endocrinol. 2018, 8. [Google Scholar] [CrossRef] [Green Version]
- Inzucchi, S.E.; Wanner, C.; Hehnke, U.; Zwiener, I.; Kaspers, S.; Clark, D.; George, J.T.; Zinman, B. Retinopathy outcomes with empagliflozin versus placebo in the EMPA-REG OUTCOME trial. Diabetes Care 2019, 42, e53–e55. [Google Scholar] [CrossRef] [Green Version]
- Ott, C.; Jumar, A.; Striepe, K.; Friedrich, S.; Karg, M.V.; Bramlage, P.; Schmieder, R.E. A randomised study of the impact of the SGLT2 inhibitor dapagliflozin on microvascular and macrovascular circulation. Cardiovasc. Diabetol. 2017, 16, 26. [Google Scholar] [CrossRef] [Green Version]
- Packer, M.; Anker, S.D.; Butler, J.; Filippatos, G.; Pocock, S.J.; Carson, P.; Januzzi, J.; Verma, S.; Tsutsui, H.; Brueckmann, M.; et al. On behalf of the EMPEROR-Reduced Trial Investigators. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure. N. Engl. J. Med. 2020, 383, 1413–1424. [Google Scholar] [CrossRef]
- McMurray, J.J.; Solomon, S.D.; Inzucchi, S.E.; Køber, L.; Kosiborod, M.N.; Martinez, F.A.; Ponikowski, P.; Sabatine, M.S.; Anand, I.S.; DAPA-HF Trial Committees and Investigators; et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N. Engl. J. Med. 2019, 381, 1995–2008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tenth Annual Report, Collaborative Islet Transplant Registry, January 2017. Available online: www.CITRegistry.org (accessed on 10 January 2021).
- Lee, T.C.; Barshes, N.R.; O’Mahony, C.A.; Nguyen, L.; Brunicardi, F.C.; Ricordi, C.; Alejandro, R.; Schock, A.P.; Mote, A.; Goss, J.A. The effect of pancreatic islet transplantation on progression of diabetic retinopathy and neuropathy. Transpl. Proc. 2005, 37, 2263–2265. [Google Scholar] [CrossRef]
- Kim, Y.J.; Shin, S.; Han, D.J.; Kim, Y.H.; Lee, J.Y.; Yoon, Y.H.; Kim, J.-G. Long-term effects of pancreas transplantation on diabetic retinopathy and incidence and predictive risk factors for early worsening. Transplantation 2018, 102, e30–e38. [Google Scholar] [CrossRef]
- Tsai, F.-Y.; Lau, I.; Li, A.-F.; Chen, S.-J.; Wang, S.-E.; Lee, F.-L.; Liu, C.J.-J.; Shyr, Y.-M.J. Acute macular edema and peripapillary soft exudate after pancreas transplantation with accelerated progression of diabetic retinopathy. Chin. Med. Assoc. 2017, 80, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Voglová, B.; Hladíková, Z.; Nemétová, L.; Zahradnická, M.; Kesslerová, K.; Sosna, T.; Lipár, K.; Kožnarová, R.; Girman, P.; Saudek, F. Early worsening of diabetic retinopathy after simultaneous pancreas and kidney transplantation—Myth or reality? Am. J. Transpl. 2020, 20, 2832–2841. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Standards of Medical Care in Diabetes. Diabetes Care 2016, 34 (Suppl. S1), S23–S99. [Google Scholar]
- Milewicz, A. Polskie Towarzystwo Endokrynologiczne. Wrocław 2012. In Endokrynologia Kliniczna; Tom, I.R., Ed.; Ikamed: Wrocław, Poland, 2016; Volume 7, pp. 360–363. [Google Scholar]
- Gorman, D.M.; le Roux, C.W.; Docherty, N.G. The effect of bariatric surgery on diabetic retinopathy: Good, bad, or both? Diabetes Metab. J. 2016, 40, 354–364. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Laybourne, J.P.; Sandinha, M.T.; de Alwis, N.M.W.; Avery, P.; Steel, D.H. Does bariatric surgery prevent progression of diabetic retinopathy? Eye 2017, 31, 1131–1139. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.; Jiang, Y.; Booth, M.; Babor, R.; MacCormick, A.; Hammodat, H.; Beban, G.; Barnes, R.M.; Vincent, A.L. Progression of diabetic retinopathy after bariatric surgery. Diabet. Med. 2015, 32, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Merlotti, C.; Ceriani, V.; Morabito, A.; Pontiroli, A.E. Bariatric surgery and diabetic retinopathy: A systematic review and meta-analysis of controlled clinical studies. Obes. Rev. 2017, 18, 309–316. [Google Scholar] [CrossRef] [Green Version]
- Cundiff, D.K.; Nigg, C.R. Diet and diabetic retinopathy: Insights from the Diabetes Control and Complications Trial (DCCT). Med. Gen. Med. 2005, 7, 3. [Google Scholar]
- Aro, A.; Kauppinen, A.; Kivinen, N.; Selander, T.; Kinnunen, K.; Tuomilehto, J.; Keinänen-Kiukaanniemi, S.; Lindström, J.; Uusitupa, M.; Kaarniranta, K. Life Style Intervention Improves Retinopathy Status-The Finnish Diabetes Prevention Study. Nutrients 2019, 23, 1691. [Google Scholar] [CrossRef] [Green Version]
- Praidou, A.; Harris, M.; Niakas, D.; Labiris, G. Physical activity and its correlation to diabetic retinopathy. J. Diabetes Complicat. 2017, 31, 456–461. [Google Scholar] [CrossRef]
- Ren, C.; Liu, W.; Li., J.; Cao, Y.; Xu, J.; Lu, P. Physical activity and risk of diabetic retinopathy: A systematic review and meta-analysis. Acta Diabetol. 2019, 56, 823–837. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matuszewski, W.; Baranowska-Jurkun, A.; Stefanowicz-Rutkowska, M.M.; Gontarz-Nowak, K.; Gątarska, E.; Bandurska-Stankiewicz, E. The Safety of Pharmacological and Surgical Treatment of Diabetes in Patients with Diabetic Retinopathy—A Review. J. Clin. Med. 2021, 10, 705. https://doi.org/10.3390/jcm10040705
Matuszewski W, Baranowska-Jurkun A, Stefanowicz-Rutkowska MM, Gontarz-Nowak K, Gątarska E, Bandurska-Stankiewicz E. The Safety of Pharmacological and Surgical Treatment of Diabetes in Patients with Diabetic Retinopathy—A Review. Journal of Clinical Medicine. 2021; 10(4):705. https://doi.org/10.3390/jcm10040705
Chicago/Turabian StyleMatuszewski, Wojciech, Angelika Baranowska-Jurkun, Magdalena Maria Stefanowicz-Rutkowska, Katarzyna Gontarz-Nowak, Ewa Gątarska, and Elżbieta Bandurska-Stankiewicz. 2021. "The Safety of Pharmacological and Surgical Treatment of Diabetes in Patients with Diabetic Retinopathy—A Review" Journal of Clinical Medicine 10, no. 4: 705. https://doi.org/10.3390/jcm10040705





