Abstract
Hyperuricemia is a significant risk factor for cardiovascular morbidity and chronic kidney disease progression. IgA nephropathy (IgAN) is a well-known primary glomerular nephropathy. Hyperuricemia is associated with a poor prognosis in IgAN patients. We evaluated the association of hyperuricemia with the histopathological severity of IgAN in male and female patients; 658 patients diagnosed with IgAN via kidney biopsy were initially included. Baseline patient data were collected by eight university hospitals affiliated with the College of Medicine of the Catholic University of Korea. Pathological features were independently evaluated by eight expert pathologists working in the hospitals, and the consensus was reached. Of the initial 658 patients, 517 were finally included (253 males and 264 females). Hyperuricemia was defined as a serum uric acid (UA) level >7.0 mg/dL for males and >5.6 mg/dL for females; 108 (42.7%) males and 95 (35.9%) females exhibited hyperuricemia. Compared to the patients with normal UA levels, the global glomerulosclerosis, segmental sclerosis, mesangial matrix expansion (MME), endocapillary proliferation (ECP), interstitial fibrosis (IF), and tubular atrophy (TA) scores were higher in hyperuricemic males and females. In multivariable linear regression, the serum UA level correlated significantly with the MME, ECP, IF, and TA scores of female IgAN patients only.
1. Introduction
Immunoglobulin A nephropathy (IgAN) is the most common primary glomerular nephropathy, both in Korea and worldwide [1]. The clinical outcomes include asymptomatic hematuria, proteinuria, kidney failure, and even end-stage renal disease (ESRD) requiring renal replacement therapy [2,3,4]. Approximately 20–40% of IgAN nephropathy patients progress to ESRD within 10–20 years of diagnosis [5,6]. The prevalence of IgAN differs between males and females. The male:female ratio is almost 1:1 in Asian populations, compared to 6:1 in Europe and the United States [7,8,9]. The risk factors for IgAN progression to chronic renal failure or ESRD include an elevated serum creatinine level, hypertension, proteinuria, dyslipidemia, and hyperuricemia [7,10].
The definition of hyperuricemia differs between males and females. Females usually have lower serum uric acid (UA) levels than men. Male sex is a significant risk factor for hyperuricemia and gout; males have been affected four-fold more frequently than females [11,12,13,14]. Hyperuricemia predicts mortality from heart failure, cerebrovascular and cardiac ischemic events, hypertension, and metabolic syndrome. An elevated serum UA level has been shown to be an independent risk factor for chronic kidney disease and renal function failure [15,16,17]. In IgAN patients, hyperuricemia was an independent risk factor for a poor renal outcomes and all-cause mortality [18]. Both histopathological parameters and the serum UA level play roles in IgAN; hyperuricemia is associated with tubular atrophy (TA) and interstitial fibrosis (IF) [19,20]. Nagasawa et al. and Oh et al. recently reported that hyperuricemic females with IgAN had a worse prognosis than males [21,22]. We aimed to determine whether hyperuricemia was associated with more severe IgAN histopathological features in females than males.
2. Materials and Methods
2.1. Study Design and Data
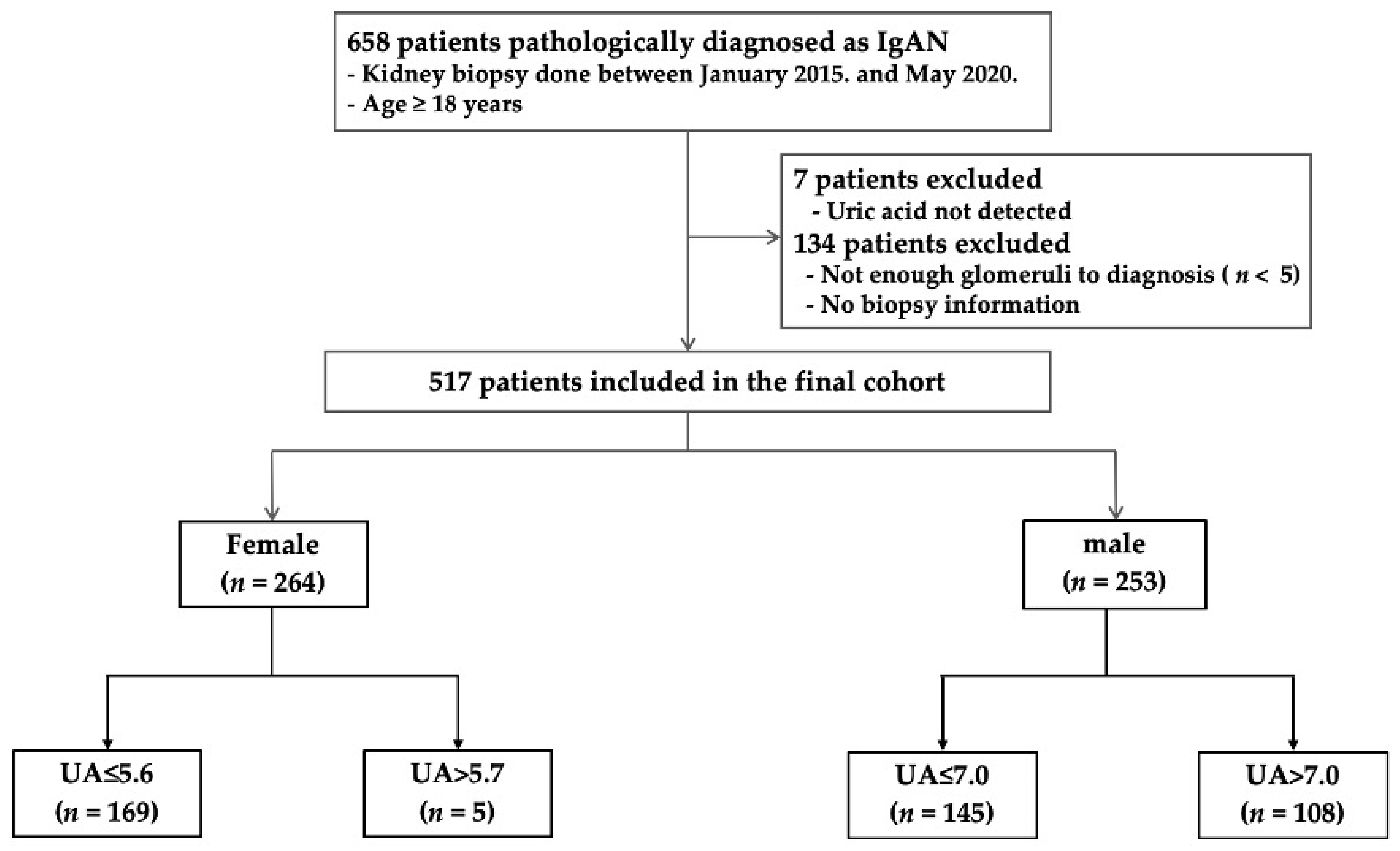
This multicenter cross-sectional cohort study recruited IgAN patients who underwent kidney biopsies between January 2015 and May 2020 in eight university hospitals affiliated with the College of Medicine of the Catholic University of Korea. The study adhered to all relevant tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of the College of Medicine, Catholic University of Korea (IRB no. XC19OEDI0025). Written informed consent was obtained from all patients before the biopsy. A total of 658 IgAN patients diagnosed via kidney biopsy were included. We excluded patients aged < 18 years, as well as 6 patients whose serum UA levels were not measured, and 134 patients whose histological data were incomplete or who had insufficient glomeruli for a diagnosis to be made.
2.2. Data Collection, Definitions, and Measurements
Kidney biopsy data were uploaded to the Kidney Biopsy Registry of the Catholic Medical Center, as were baseline clinical and laboratory data. Hyperuricemia was defined as a serum UA level > 7.0 mg/dL for males and >5.6 mg/dL for females [23]. Proteinuria was assessed via a spot test that yielded the urine protein:creatinine ratio [24]. The estimated glomerular filtration rate (eGFR) was calculated using the equation from the modification of diet in renal disease study [25]. Urinary red blood cell (RBC) levels were graded as follows: <3 RBCs/high-power field (HPF), 0; 3–5 RBCs/HPF, 1; 5–9 RBCs/HPF, 2; 10–19 RBCs/HPF, 3; and >19 RBCs/HPF, 4.
2.3. Histopathological Parameters
Renal samples were reviewed by eight expert pathologists working in the eight hospitals using the following grading system. Under a light microscope, global glomerulosclerosis (GS), segmental sclerosis (SS), and crescent formation (or capsular adhesion, CA) were calculated as percentages (involved glomeruli/total glomeruli). Mesangial matrix expansion (MME), mesangial cell proliferation (MCP), endocapillary proliferation (ECP), interstitial fibrosis (IF), and tubular atrophy (TA) were all graded from 0 to 4, as follows: 0, absent; 1, trace (<20%); 2, mild (20–40%); 3, moderate (40–70%); and 4, severe (≥70%). Based on immunofluorescence microscopy, the extent of mesangial deposition of IgA, C3, and C4d was graded as 0 (absent), +1 (trace), +2 (mild), +3 (moderate), or +4 (marked). Renal biopsy data were also assessed using the World Health Organization classification (grades I–VI) for IgA nephropathy [26].
2.4. Statistical Analysis
Continuous variables were compared using the t-test. Categorical variables are expressed as numbers with percentages and were compared using the chi-squared test. Linear regression analysis was performed to evaluate the associations between the serum UA level and histopathological parameters stratified using clinical and laboratory scoring systems. A p-value < 0.05 was considered statistically significant. All statistical analyses were performed using SPSS software (ver. 23.0; IBM Corporation, New York, NY, USA).
3. Results
3.1. Baseline Characteristics
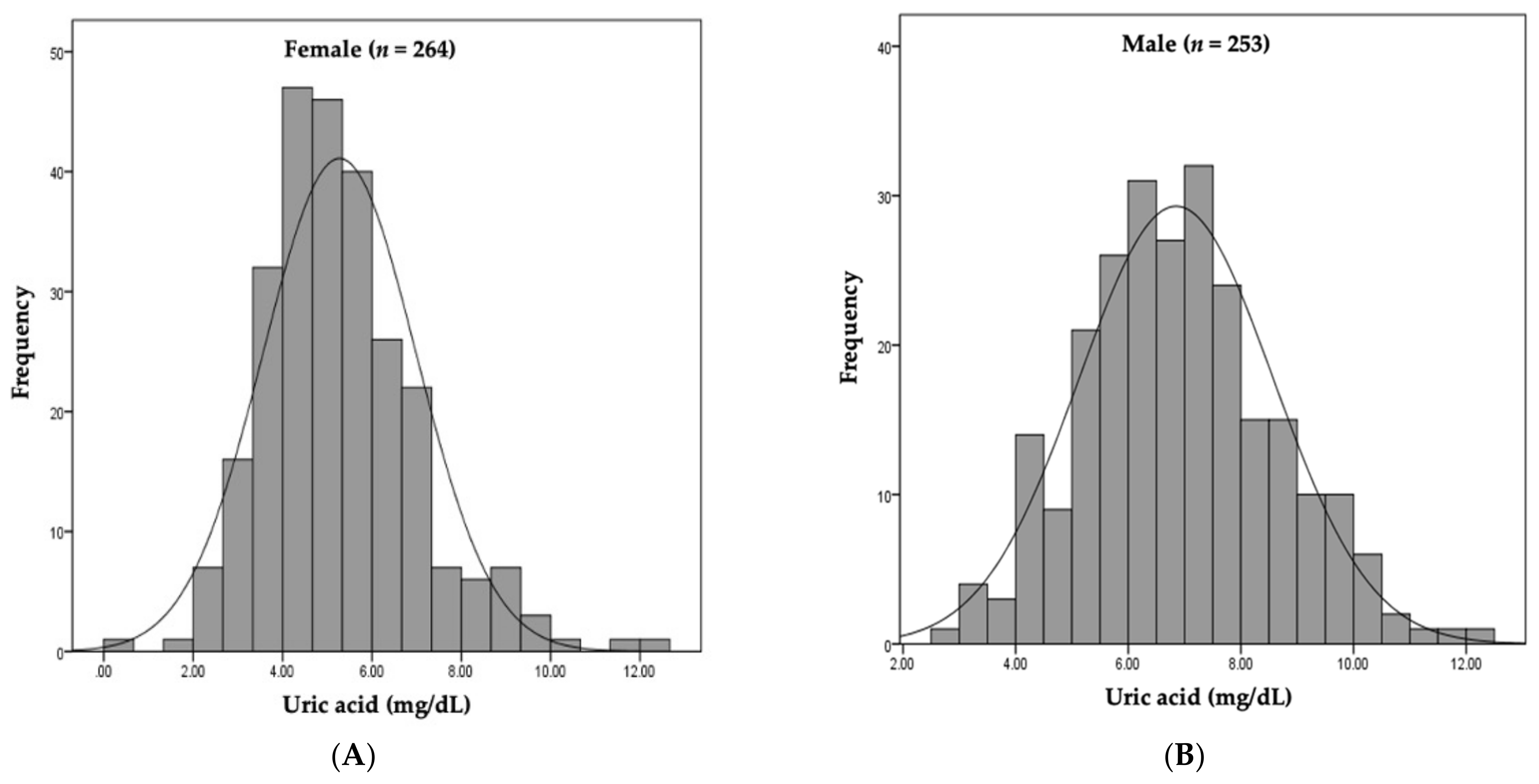
A total of 517 patients were retrospectively analyzed (Figure 1). Patients with normal UA levels were compared to hyperuricemic patients. The mean patient age was 42.0 ± 14.6 years. There were 253 (48.9%) males and 264 (51.0%) females. The median serum UA level was 5.27 mg/dL in females and 6.84 mg/dL in males (Figure 2). The baseline characteristics of all patients, and of females and males separately, are listed in Table 1. Hyperuricemia was defined as a UA level > 5.6 mg/dL in females and >7 mg/dL in males. Of all hyperuricemic patients, 108 (42.6%) were male, and 95 (35.9%) were female. The baseline clinical characteristics of all patients are shown in Table 2. No significant difference in age, alcohol consumption, smoking status, or hypertension status was seen between hyperuricemic patients and those with normal UA levels. The prevalence of diabetes mellitus was significantly higher in hyperuricemic patients, including body mass index and total cholesterol, triglyceride, and creatinine levels. The high-density lipoprotein cholesterol level and eGFR were lower in both male and female hyperuricemic patients than in the patients with normal UA levels. The systolic/diastolic blood pressure was significantly elevated in females, but not males, with hyperuricemia compared to patients with normal UA levels. Female, but not male, hyperuricemic patients exhibited significantly lower serum albumin and higher serum IgA level than patients with normal UA levels. Table 1 summarizes the clinical characteristics of males and females with normal and elevated UA levels.
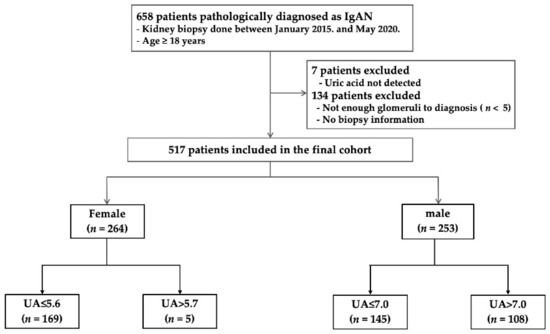
Figure 1.
Flow diagram for the study population.
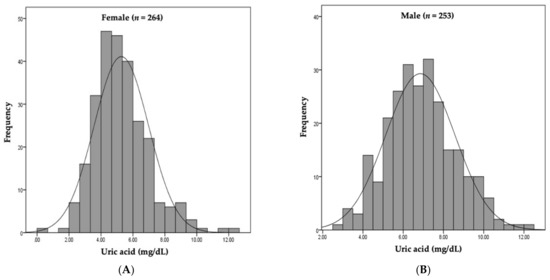
Figure 2.
Distribution of serum uric acid by sex. (A) Median serum UA level was 5.27 mg/dL in females. (B) Median serum UA level was 6.84 mg/dL in males.

Table 1.
Baseline clinical variables of the total patient and each sex group at the time of renal biopsy.

Table 2.
Baseline clinical variables of the uric acid group at the time of renal biopsy. Comparison between normal UA and hyperuricemia in each female and male group measured.
3.2. Histopathological Characteristics According to UA Levels in Males and Females
The histopathological findings of hyperuricemic patients and those with normal UA levels are shown in Table 3. Hyperuricemic females exhibited significantly higher rates of GS, SS, and CA (p < 0.001, p = 0.001, and p = 0.0.002, respectively). The MME (p = 0.023), IF (p < 0.001), and TA (p < 0.001) scores were also significantly higher in female hyperuricemic patients than in those with normal UA levels. Hyperuricemic males also exhibited significantly higher GS, SS, and CA scores than those with normal UA levels (p = 0.004, p = 0.002 and p = 0.004, respectively). The MME (p = 0.037), ECP (p = 0.001), IF (p < 0.001), and TA (p < 0.001) scores were also significantly higher in males with than without hyperuricemia. Immunofluorescence microscopy revealed that the mesangial IgA, C3, and C4d grades were not associated with hyperuricemia in either females or males. The WHO IgAN grades were significantly higher in both female and male hyperuricemic patients than those with normal UA levels (p < 0.001, p = 0.002, respectively).

Table 3.
Histopathological characteristics between the uric acid group.
3.3. Associations between Serum UA Levels and Histopathological Parameters
We used linear regression analysis to test for associations between the UA level and histopathological severity (Table 4 and Table 5). For all patients, the GS (p < 0.001), SS (p < 0.001), MME (p < 0.001), ECP (p < 0.001), IF (p < 0.001), TA (p < 0.001), and C3 mesangial deposition (p = 0.008) scores were positively correlated with the serum UA level in univariate analysis, as were the GS (p = 0.002), SS (p = 0.004), MME (p < 0.001), ECP (p = 0.026), IF (p < 0.001), TA (p < 0.001), and C3 mesangial deposition (p = 0.037) scores in multivariate analysis. In females, the GS (p < 0.001), SS (p < 0.001), CA (p = 0.003), MME (p = 0.001), ECP (p < 0.001), IF (p < 0.001), TA (p < 0.001), and C3 mesangial deposition (p = 0.036) scores positively correlated with the serum UA level in univariate analysis. In multivariate analysis, the MME (p = 0.002), ECP (p = 0.001), IF (p = 0.023), and TA (p = 0.002) scores were positively associated with the serum UA level in females. In contrast, the GS (p = 0.005), SS (p = 0.013), CA (p = 0.002), ECP (p = 0.014), IF (p < 0.001), and TA (p < 0.001) scores were positively associated with the serum UA level of males in univariate analysis. However, multivariate analysis revealed no significant association between any histopathological parameter and the serum UA level in males.

Table 4.
Linear regression for uric acid and the histopathologic parameters in all patients.

Table 5.
Linear regression for uric acid and the histopathologic parameters in sex differences (a) univariable analysis (b) multivariable analysis.
4. Discussion
This study showed that the UA level is associated with poor histopathological findings, such as MME, ECP, IF, and TA in female IgAN patients, but not males. Hyperuricemia is becoming more common, attributed to lifestyle and dietary changes and the aging of the population [27]. There is no consensus threshold serum UA level for hyperuricemia diagnosis. In addition, serum UA levels differ between males and females. We defined hyperuricemia as a serum UA level >7.0 mg/dL for males and >5.6 mg/dL for females [23].
To evaluate the prognosis of IgAN, WHO [26], Haas [28], and Oxford [29] classification have been used. The key histological damage is reflected in the MCP(M), ECP(E), GS(S), TF(T) and cresent(C) [30]. MEST-C scoring system was recently used for evaluation of IgAN prognosis. However, the MEST-C scoring system roughly reflects the extent of tissue damage in a way that scores with 0 and 1 (M, E, S) or 0, 1, and 2 (T) for each item. For this reason, we analyze key histological damage mentioned above with a more specialized hospital consensus scoring system [31].
A few studies have investigated the UA levels and histological findings of IgAN patients, but they focused only on the TA and IF scores [20,23,32]. Recent studies reported that mesangial C3 deposition was associated with active inflammation and poor outcomes for IgAN patients [33,34]. Hyperuricemia and C3 deposition were independent risk factors for IgAN, as were the Oxford T-score and a declining eGFR [35]. In the univariate analysis conducted in the present study, mesangial C3 deposition was correlated with the serum UA level of female patients. UA may activate the complement system and cause inflammation in association with IgAN.
In our study, the GS, SS, MME, ECP, IF, and TA scores were higher in both male and female hyperuricemic patients than in patients with normal UA levels. In the linear regression analysis, the GS, SS, MME, ECP, IF, TA and C3 mesangial deposition scores were associated with the UA level in all patients and in females only; however, only the GS, CA, IF, and TA scores showed an association with the UA level in males. The MME, ECP, IF, and TA scores were correlated with the serum UA level in females but not in males. Oh, et al. found that the serum UA level was an independent risk factor for IgAN progression, particularly in females [22]. However, histological findings were not analyzed.
Regarding how UA could affect the kidney, hyperuricemia can lead to GS, interstitial injury, and fibrosis [36,37], as well as oxidative stress. Many factors are involved in endothelial dysfunction, renal arteriopathy, and renovascular constriction [16,38,39,40]. It remains unclear why the serum UA level is more important in females than males, although it is known that estrogen inhibits urate transporter 1, reduces the serum UA level, and promotes urinary UA excretion [41]. The serum UA level increases in postmenopausal females, while the level of estrogen and of the transcriptional factors that it, directly and indirectly, regulates decreases [13]. Estrogen plays a crucial role in reno-protection. Estrogen negatively regulates TGF-ß synthesis; estrogen deficiency and ovariectomy accelerate the progression of glomerular injury and may contribute to the observed gender differences in IgAN [42]. Endogenous estradiol was shown to reduce the serum UA level [43].
This study had several limitations. First, it used a cross-sectional, retrospective design, so there were no long-term follow-up data. Second, the study involved eight university hospitals, and renal biopsy specimens were reviewed by eight different renal pathologists; thus, there may have been a degree of interobserver variability. However, all hospitals used identical biopsy criteria, and all pathologists were trained by the same international expert.
5. Conclusions
The serum UA level may affect more severe renal histopathology of female IgAN patients than male patients. A large prospective study is needed to validate this and to assess whether appropriate management of the UA levels of female patients can prevent IgAN-related histological damage.
Author Contributions
Conceptualization, W.J.C. and Y.-K.C.; methodology, W.J.C., Y.A.H., H.D.K. and T.H.B.; formal analysis, W.J.C. and E.S.K.; funding acquisition, W.J.C.; investigation, Y.S.K., Y.K.K. and S.J.S.; data curation, Y.A.H., J.W.M., E.S.K., H.D.K. and T.H.B.; visualization, W.J.C.; writing—original draft preparation, W.J.C.; writing—review and editing, Y.-K.C.; supervision, S.Y.K., Y.O.K., C.W.Y. and Y.-K.C. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by a Clinical Research Institute Grant funded by The Catholic University of Korea Daejeon St. Mary’s Hospital (CMCDJ-P-2021-016).
Institutional Review Board Statement
This study adhered to all relevant tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of the College of Medicine, Catholic University of Korea (IRB no. XC19OEDI0025).
Informed Consent Statement
The need for patient consent was waived given the retrospective nature of the study.
Data Availability Statement
All data are reported in the article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chang, J.H.; Kim, D.K.; Kim, H.W.; Park, S.Y.; Yoo, T.H.; Kim, B.S.; Kang, S.W.; Choi, K.H.; Han, D.S.; Jeong, H.J.; et al. Changing prevalence of glomerular diseases in Korean adults: A review of 20 years of experience. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2009, 24, 2406–2410. [Google Scholar] [CrossRef] [PubMed]
- Canetta, P.A.; Kiryluk, K.; Appel, G.B. Glomerular diseases: Emerging tests and therapies for IgA nephropathy. Clin. J. Am. Soc. Nephrol. CJASN 2014, 9, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Barbour, S.J.; Reich, H.N. Risk stratification of patients with IgA nephropathy. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2012, 59, 865–873. [Google Scholar] [CrossRef]
- Eiro, M.; Katoh, T.; Kuriki, M.; Asano, K.; Watanabe, K.; Watanabe, T. The product of duration and amount of proteinuria (proteinuria index) is a possible marker for glomerular and tubulointerstitial damage in IgA nephropathy. Nephron 2002, 90, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Radford, M.G., Jr.; Donadio, J.V., Jr.; Bergstralh, E.J.; Grande, J.P. Predicting renal outcome in IgA nephropathy. J. Am. Soc. Nephrol. JASN 1997, 8, 199–207. [Google Scholar] [CrossRef]
- D’Amico, G.; Colasanti, G.; Barbiano di Belgioioso, G.; Fellin, G.; Ragni, A.; Egidi, F.; Radaelli, L.; Fogazzi, G.; Ponticelli, C.; Minetti, L. Long-term follow-up of IgA mesangial nephropathy: Clinico-histological study in 374 patients. Semin. Nephrol. 1987, 7, 355–358. [Google Scholar]
- Goto, M.; Wakai, K.; Kawamura, T.; Ando, M.; Endoh, M.; Tomino, Y. A scoring system to predict renal outcome in IgA nephropathy: A nationwide 10-year prospective cohort study. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2009, 24, 3068–3074. [Google Scholar] [CrossRef] [PubMed]
- Duan, Z.Y.; Cai, G.Y.; Chen, Y.Z.; Liang, S.; Liu, S.W.; Wu, J.; Qiu, Q.; Lin, S.P.; Zhang, X.G.; Chen, X.M. Aging promotes progression of IgA nephropathy: A systematic review and meta-analysis. Am. J. Nephrol. 2013, 38, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Geddes, C.C.; Rauta, V.; Gronhagen-Riska, C.; Bartosik, L.P.; Jardine, A.G.; Ibels, L.S.; Pei, Y.; Cattran, D.C. A tricontinental view of IgA nephropathy. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2003, 18, 1541–1548. [Google Scholar] [CrossRef]
- Syrjänen, J.; Mustonen, J.; Pasternack, A. Hypertriglyceridaemia and hyperuricaemia are risk factors for progression of IgA nephropathy. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2000, 15, 34–42. [Google Scholar] [CrossRef]
- Kawabe, M.; Sato, A.; Hoshi, T.; Sakai, S.; Hiraya, D.; Watabe, H.; Kakefuda, Y.; Ishibashi, M.; Abe, D.; Takeyasu, N.; et al. Gender differences in the association between serum uric acid and prognosis in patients with acute coronary syndrome. J. Cardiol. 2016, 67, 170–176. [Google Scholar] [CrossRef]
- Harrold, L.R.; Etzel, C.J.; Gibofsky, A.; Kremer, J.M.; Pillinger, M.H.; Saag, K.G.; Schlesinger, N.; Terkeltaub, R.; Cox, V.; Greenberg, J.D. Sex differences in gout characteristics: Tailoring care for women and men. BMC Musculoskelet. Disord. 2017, 18, 108. [Google Scholar] [CrossRef] [PubMed]
- Halperin Kuhns, V.L.; Woodward, O.M. Sex Differences in Urate Handling. Int. J. Mol. Sci. 2020, 21, 4269. [Google Scholar] [CrossRef]
- Kuo, C.F.; Grainge, M.J.; Zhang, W.; Doherty, M. Global epidemiology of gout: Prevalence, incidence and risk factors. Nat. Rev. Rheumatol. 2015, 11, 649–662. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, D.; Unwin, R. The pathophysiology of hyperuricaemia and its possible relationship to cardiovascular disease, morbidity and mortality. BMC Nephrol. 2013, 14, 164. [Google Scholar] [CrossRef]
- Zoccali, C.; Maio, R.; Mallamaci, F.; Sesti, G.; Perticone, F. Uric acid and endothelial dysfunction in essential hypertension. J. Am. Soc. Nephrol. JASN 2006, 17, 1466–1471. [Google Scholar] [CrossRef]
- Li, C.; Hsieh, M.C.; Chang, S.J. Metabolic syndrome, diabetes, and hyperuricemia. Curr. Opin. Rheumatol. 2013, 25, 210–216. [Google Scholar] [CrossRef]
- Tsai, C.W.; Lin, S.Y.; Kuo, C.C.; Huang, C.C. Serum Uric Acid and Progression of Kidney Disease: A Longitudinal Analysis and Mini-Review. PLoS ONE 2017, 12, e0170393. [Google Scholar] [CrossRef]
- Zhou, J.; Chen, Y.; Liu, Y.; Shi, S.; Li, X.; Wang, S.; Zhang, H. Plasma uric acid level indicates tubular interstitial leisions at early stage of IgA nephropathy. BMC Nephrol. 2014, 15, 11. [Google Scholar] [CrossRef] [PubMed]
- Myllymäki, J.; Honkanen, T.; Syrjänen, J.; Helin, H.; Rantala, I.; Pasternack, A.; Mustonen, J. Uric acid correlates with the severity of histopathological parameters in IgA nephropathy. Nephrol. Dial. Transplant. Off. Publ. Eur. Dial. Transpl. Assoc. Eur. Ren. Assoc. 2005, 20, 89–95. [Google Scholar] [CrossRef]
- Nagasawa, Y.; Yamamoto, R.; Shoji, T.; Shinzawa, M.; Hasuike, Y.; Nagatoya, K.; Yamauchi, A.; Hayashi, T.; Kuragano, T.; Moriyama, T.; et al. Serum Uric Acid Level Predicts Progression of IgA Nephropathy in Females but Not in Males. PLoS ONE 2016, 11, e0160828. [Google Scholar] [CrossRef]
- Oh, T.R.; Choi, H.S.; Kim, C.S.; Kang, K.P.; Kwon, Y.J.; Kim, S.G.; Ma, S.K.; Kim, S.W.; Bae, E.H. The Effects of Hyperuricemia on the Prognosis of IgA Nephropathy are More Potent in Females. J. Clin. Med. 2020, 9, 176. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Pandya, B.J.; Choi, H.K. Prevalence of gout and hyperuricemia in the US general population: The National Health and Nutrition Examination Survey 2007–2008. Arthritis Rheum. 2011, 63, 3136–3141. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, J.M.; Chang, B.S.; Matarese, R.A.; Garella, S. Use of single voided urine samples to estimate quantitative proteinuria. N. Engl. J. Med. 1983, 309, 1543–1546. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Bosch, J.P.; Lewis, J.B.; Greene, T.; Rogers, N.; Roth, D. A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Modification of Diet in Renal Disease Study Group. Ann. Intern. Med. 1999, 130, 461–470. [Google Scholar] [CrossRef]
- Sinniah, R. IgA mesangial nephropathy: Berger’s disease. Am. J. Nephrol. 1985, 5, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, E. Reduced glomerular function and prevalence of gout: NHANES 2009–2010. PLoS ONE 2012, 7, e50046. [Google Scholar] [CrossRef]
- Haas, M. Histologic subclassification of IgA nephropathy: A clinicopathologic study of 244 cases. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 1997, 29, 829–842. [Google Scholar] [CrossRef]
- Cattran, D.C.; Coppo, R.; Cook, H.T.; Feehally, J.; Roberts, I.S.; Troyanov, S.; Alpers, C.E.; Amore, A.; Barratt, J.; Berthoux, F.; et al. The Oxford classification of IgA nephropathy: Rationale, clinicopathological correlations, and classification. Kidney Int. 2009, 76, 534–545. [Google Scholar] [CrossRef]
- Zeng, C.H.; Le, W.; Ni, Z.; Zhang, M.; Miao, L.; Luo, P.; Wang, R.; Lv, Z.; Chen, J.; Tian, J.; et al. A multicenter application and evaluation of the oxford classification of IgA nephropathy in adult chinese patients. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2012, 60, 812–820. [Google Scholar] [CrossRef]
- Hong, Y.A.; Min, J.W.; Ha, M.A.; Koh, E.S.; Kim, H.D.; Ban, T.H.; Kim, Y.S.; Kim, Y.K.; Kim, D.; Shin, S.J.; et al. The Impact of Obesity on the Severity of Clinicopathologic Parameters in Patients with IgA Nephropathy. J. Clin. Med. 2020, 9, 2824. [Google Scholar] [CrossRef]
- Fan, S.; Zhang, P.; Wang, A.Y.; Wang, X.; Wang, L.; Li, G.; Hong, D. Hyperuricemia and its related histopathological features on renal biopsy. BMC Nephrol. 2019, 20, 95. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Koo, H.M.; Lim, B.J.; Oh, H.J.; Yoo, D.E.; Shin, D.H.; Lee, M.J.; Doh, F.M.; Park, J.T.; Yoo, T.H.; et al. Decreased circulating C3 levels and mesangial C3 deposition predict renal outcome in patients with IgA nephropathy. PLoS ONE 2012, 7, e40495. [Google Scholar] [CrossRef]
- Nam, K.H.; Joo, Y.S.; Lee, C.; Lee, S.; Kim, J.; Yun, H.R.; Park, J.T.; Chang, T.I.; Ryu, D.R.; Yoo, T.H.; et al. Predictive value of mesangial C3 and C4d deposition in IgA nephropathy. Clin. Immunol. 2020, 211, 108331. [Google Scholar] [CrossRef]
- Caliskan, Y.; Ozluk, Y.; Celik, D.; Oztop, N.; Aksoy, A.; Ucar, A.S.; Yazici, H.; Kilicaslan, I.; Sever, M.S. The Clinical Significance of Uric Acid and Complement Activation in the Progression of IgA Nephropathy. Kidney Blood Press. Res. 2016, 41, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Ryu, E.S.; Kim, M.J.; Shin, H.S.; Jang, Y.H.; Choi, H.S.; Jo, I.; Johnson, R.J.; Kang, D.H. Uric acid-induced phenotypic transition of renal tubular cells as a novel mechanism of chronic kidney disease. Am. J. Physiol. Ren. Physiol. 2013, 304, F471–F480. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, T.; Mazzali, M.; Kang, D.H.; Kanellis, J.; Watanabe, S.; Sanchez-Lozada, L.G.; Rodriguez-Iturbe, B.; Herrera-Acosta, J.; Johnson, R.J. Hyperuricemia causes glomerular hypertrophy in the rat. Am. J. Nephrol. 2003, 23, 2–7. [Google Scholar] [CrossRef]
- Sánchez-Lozada, L.G.; Tapia, E.; Santamaría, J.; Avila-Casado, C.; Soto, V.; Nepomuceno, T.; Rodríguez-Iturbe, B.; Johnson, R.J.; Herrera-Acosta, J. Mild hyperuricemia induces vasoconstriction and maintains glomerular hypertension in normal and remnant kidney rats. Kidney Int. 2005, 67, 237–247. [Google Scholar] [CrossRef]
- Corry, D.B.; Eslami, P.; Yamamoto, K.; Nyby, M.D.; Makino, H.; Tuck, M.L. Uric acid stimulates vascular smooth muscle cell proliferation and oxidative stress via the vascular renin-angiotensin system. J. Hypertens. 2008, 26, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Lozada, L.G.; Tapia, E.; Avila-Casado, C.; Soto, V.; Franco, M.; Santamaría, J.; Nakagawa, T.; Rodríguez-Iturbe, B.; Johnson, R.J.; Herrera-Acosta, J. Mild hyperuricemia induces glomerular hypertension in normal rats. Am. J. Physiol. Ren. Physiol. 2002, 283, F1105–F1110. [Google Scholar] [CrossRef]
- Takiue, Y.; Hosoyamada, M.; Kimura, M.; Saito, H. The effect of female hormones upon urate transport systems in the mouse kidney. Nucleosides Nucleic Acids 2011, 30, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Kelimu, A.; Satoshi, H.; Mareki, O.; Yasuhiko, T. Influence of Estrogen on the Progression of Kidney Injury in Murine IgA Nephropathy. Juntendo Med. J. 2017, 63, 178–185. [Google Scholar] [CrossRef]
- Mumford, S.L.; Dasharathy, S.S.; Pollack, A.Z.; Perkins, N.J.; Mattison, D.R.; Cole, S.R.; Wactawski-Wende, J.; Schisterman, E.F. Serum uric acid in relation to endogenous reproductive hormones during the menstrual cycle: Findings from the BioCycle study. Hum. Reprod. 2013, 28, 1853–1862. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).