The Relationship between Mucins and Ulcerative Colitis: A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Review Questions
- Does mucin expression and activity change in patients with UC compared to healthy controls?
- How do changes in mucin expression effect mucus structure?
- What impact do these changes have on host-microbiome interactions and inflammation?
2.2. Search Strategy
- Colitis AND mucin *
- (IBD OR Inflammatory Bowel Disease) AND mucin *
- (IBD OR Inflammatory Bowel Disease) AND mucin * degradationTo optimise the retrieval of articles in databases, Boolean operators were used (AND, OR). Using ‘AND’ allows for citations containing all search terms to be retrieved, whereas using ‘OR’ can produce citations containing at least one of a group of search terms. Therefore, ‘OR’ was used here where words were similar/related e.g. IBD and Inflammatory Bowel Disease. Mucin was also truncated (truncation is indicated by Asterix – *) in order to find citations including mucin and all words with the stem ‘mucin’ e.g. mucin, mucins, mucinase.
2.3. Study Selection and Items Collected
2.4. Inclusion & Exclusion Criteria
2.5. Study Quality Assessment
2.6. Data Synthesis
3. Results
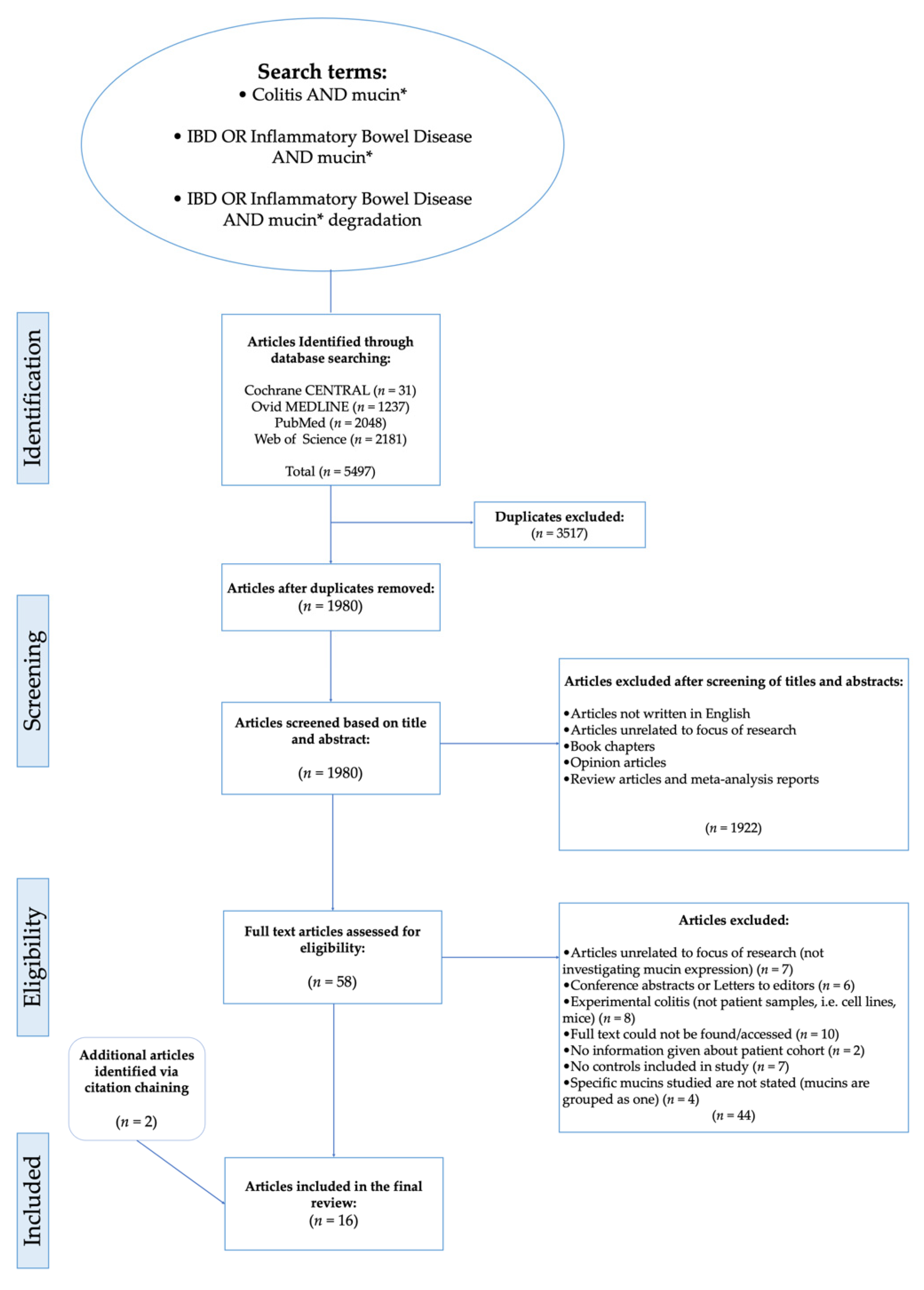
3.1. Search Results
3.2. Overview of Studies Selected for Analysis
3.3. Critical Appraisal of Included Studies
3.4. MUC2 Expression in Colitis
3.4.1. MUC2 mRNA Expression
3.4.2. MUC2 Protein Expression
3.4.3. Goblet Cells and MUC2
3.5. Membrane-Associated Mucins in Colitis
3.6. MUC5AC in Colitis
4. Discussion
5. Conclusions
Authors Contribution
Funding
Conflicts of Interest
References
- Strous, G.J.; Dekker, J. Mucin-Type Glycoproteins. Crit. Rev. Biochem. Mol. Biol. 1992, 27, 57–92. [Google Scholar] [CrossRef] [PubMed]
- Corfield, A.P. Mucins: A Biologically Relevant Glycan Barrier in Mucosal Protection. Biochim. Biophys. Acta 2015, 1850, 236–252. [Google Scholar] [CrossRef] [PubMed]
- Derrien, M.; van Passel, M.W.; van de Bovenkamp, J.H.; Schipper, R.G.; de Vos, W.M.; Dekker, J. Mucin-Bacterial Interactions in the Human Oral Cavity and Digestive Tract. Gut Microbes 2010, 1, 254–268. [Google Scholar] [CrossRef] [Green Version]
- Gendler, S.J.; Spicer, A.P. Epithelial Mucin Genes. Annu. Rev. Physiol. 1995, 57, 607–634. [Google Scholar] [CrossRef]
- Wagner, C.E.; Wheeler, K.M.; Ribbeck, K. Mucins and Their Role in Shaping the Functions of Mucus Barriers. Annu. Rev. Cell Dev. Biol. 2018, 34, 189–215. [Google Scholar] [CrossRef] [Green Version]
- Pearson, J.P.; Chater, P.I.; Wilcox, M.D. The Properties of the Mucus Barrier, a Unique Gel—How Can Nanoparticles Cross It? Ther. Deliv. 2016, 7, 229–244. [Google Scholar] [CrossRef]
- Bansil, R.; Turner, B.S. The Biology of Mucus: Composition, Synthesis and Organization. Adv. Drug Deliv. Rev. 2018, 124, 3–15. [Google Scholar] [CrossRef]
- Martinez-Saez, N.; Peregrina, J.M.; Corzana, F. Principles of Mucin Structure: Implications for the Rational Design of Cancer Vaccines Derived from Muc1-Glycopeptides. Chem. Soc. Rev. 2017, 46, 7154–7175. [Google Scholar] [CrossRef] [Green Version]
- Perez-Vilar, J.; Hill, R.L. The Structure and Assembly of Secreted Mucins. J. Biol. Chem. 1999, 274, 31751–31754. [Google Scholar] [CrossRef] [Green Version]
- Dekker, J.; Rossen, J.W.; Büller, H.A.; Einerhand, A.W. The Muc Family: An Obituary. Trends Biochem. Sci. 2002, 27, 126–131. [Google Scholar] [CrossRef]
- Dhanisha, S.S.; Guruvayoorappan, C.; Drishya, S.; Abeesh, P. Mucins: Structural Diversity, Biosynthesis, Its Role in Pathogenesis and as Possible Therapeutic Targets. Crit. Rev. Oncol. Hematol. 2018, 122, 98–122. [Google Scholar] [CrossRef]
- Lang, T.; Klasson, S.; Larsson, E.; Johansson, M.E.; Hansson, G.C.; Samuelsson, T. Searching the Evolutionary Origin of Epithelial Mucus Protein Components-Mucins and Fcgbp. Mol. Biol. Evol. 2016, 33, 1921–1936. [Google Scholar] [CrossRef] [PubMed]
- Noguera, M.E.; Jakoncic, J.; Ermacora, M.R. High-Resolution Structure of Intramolecularly Proteolyzed Human Mucin-1 Sea Domain. Biochim. Biophys. Acta Proteins Proteom. 2020, 1868, 140361. [Google Scholar] [CrossRef] [PubMed]
- Appella, E.; Weber, I.T.; Blasi, F. Structure and Function of Epidermal Growth Factor-Like Regions in Proteins. FEBS Lett. 1988, 231, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Ciccarelli, F.D.; Doerks, T.; Bork, P. Amop, a Protein Module Alternatively Spliced in Cancer Cells. Trends Biochem. Sci. 2002, 27, 113–115. [Google Scholar] [CrossRef]
- Xu, D.; Pavlidis, P.; Thamadilok, S.; Redwood, E.; Fox, S.; Blekhman, R.; Ruhl, S.; Gokcumen, O. Recent Evolution of the Salivary Mucin Muc7. Sci. Rep. 2016, 6, 31791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Javitt, G.; Khmelnitsky, L.; Albert, L.; Bigman, L.S.; Elad, N.; Morgenstern, D.; Ilani, T.; Levy, Y.; Diskin, R.; Fass, D. Assembly Mechanism of Mucin and Von Willebrand Factor Polymers. Cell 2020, 183, 717–729.e16. [Google Scholar] [CrossRef]
- Van Klinken, B.J.; Dekker, J.; Buller, H.A.; Einerhand, A.W. Mucin Gene Structure and Expression: Protection Vs. Adhesion. Am. J. Physiol. 1995, 269 Pt 1, G613–G627. [Google Scholar] [CrossRef]
- Sicard, J.F.; le Bihan, G.; Vogeleer, P.; Jacques, M.; Harel, J. Interactions of Intestinal Bacteria with Components of the Intestinal Mucus. Front. Cell. Infect. Microbiol. 2017, 7, 387. [Google Scholar] [CrossRef]
- Podolsky, D.K. Inflammatory Bowel Disease. N. Engl. J. Med. 1991, 325, 928–937. [Google Scholar] [CrossRef]
- Guan, Q. A Comprehensive Review and Update on the Pathogenesis of Inflammatory Bowel Disease. J. Immunol. Res. 2019, 2019, 7247238. [Google Scholar] [CrossRef] [Green Version]
- Roda, G.; Chien Ng, S.; Kotze, P.G.; Argollo, M.; Panaccione, R.; Spinelli, A.; Kaser, A.; Peyrin-Biroulet, L.; Danese, S. Crohn’s Disease. Nat. Rev. Dis. Primers 2020, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Jairath, V.; Feagan, B.G. Global Burden of Inflammatory Bowel Disease. Lancet Gastroenterol. Hepatol. 2020, 5, 2–3. [Google Scholar] [CrossRef] [Green Version]
- Matijasic, M.; Mestrovic, T.; Peric, M.; Paljetak, H.C.; Panek, M.; Bender, D.V.; Kelecic, D.L.; Krznaric, Z.; Verbanac, D. Modulating Composition and Metabolic Activity of the Gut Microbiota in Ibd Patients. Int. J. Mol. Sci. 2016, 17, 578. [Google Scholar] [CrossRef]
- Tytgat, K.M.; Opdam, F.J.; Einerhand, A.W.; Buller, H.A.; Dekker, J. Muc2 Is the Prominent Colonic Mucin Expressed in Ulcerative Colitis. Gut 1996, 38, 554–563. [Google Scholar] [CrossRef] [Green Version]
- Pullan, R.D.; Thomas, G.A.; Rhodes, M.; Newcombe, R.G.; Williams, G.T.; Allen, A.; Rhodes, J. Thickness of Adherent Mucus Gel on Colonic Mucosa in Humans and Its Relevance to Colitis. Gut 1994, 35, 353–359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Prisma Group. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The Prisma Statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef] [Green Version]
- Dorofeyev, A.E.; Vasilenko, I.V.; Rassokhina, O.A.; Kondratiuk, R.B. Mucosal Barrier in Ulcerative Colitis and Crohn’s Disease. Gastroenterol. Res. Pract. 2013, 2013, 431231. [Google Scholar] [CrossRef]
- Hensel, K.O.; Boland, V.; Postberg, J.; Zilbaur, M.; Heuschkel, R.; Vogel, S.; Gödde, D.; Wirth, S.; Jenke, A.C. Differential Expression of Mucosal Trefoil Factors and Mucins in Pediatric Inflammatory Bowel Diseases. Sci. Rep. 2014, 4, 7343. [Google Scholar] [CrossRef] [Green Version]
- Swallow, D.M.; Vinall, L.E.; Gum, J.R.; Kim, Y.S.; Yang, H.; Rotter, J.I.; Mirza, M.; Lee, J.C.; Lennard-Jones, J.E. Ulcerative Colitis Is Not Associated with Differences in Muc2 Mucin Allele Length. J. Med. Genet. 1999, 36, 859–860. [Google Scholar]
- Kyo, K.; Parkes, M.; Takei, Y.; Nishimori, H.; Vyas, P.; Satsangi, J.; Simmons, J.; Nagawa, H.; Baba, S.; Jewell, D.; et al. Association of Ulcerative Colitis with Rare Vntr Alleles of the Human Intestinal Mucin Gene, Muc3. Hum. Mol. Genet. 1999, 8, 307–311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiss, A.A.; Babyatsky, M.W.; Ogata, S.; Chen, A.; Itzkowitz, S.H. Expression of Muc2 and Muc3 Mrna in Human Normal, Malignant, and Inflammatory Intestinal Tissues. J. Histochem. Cytochem. 1996, 44, 1161–1166. [Google Scholar] [CrossRef] [Green Version]
- Tytgat, K.M.; van der Wal, J.W.; Einerhand, A.W.; Buller, H.A.; Dekker, J. Quantitative Analysis of Muc2 Synthesis in Ulcerative Colitis. Biochem. Biophys. Res. Commun. 1996, 224, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Hinoda, Y.; Akashi, H.; Suwa, T.; Itoh, F.; Adachi, M.; Endo, T.; Satoh, M.; Xing, P.X.; Imai, K. Immunohistochemical Detection of Muc2 Mucin Core Protein in Ulcerative Colitis. J. Clin. Lab. Anal. 1998, 12, 150–153. [Google Scholar] [CrossRef]
- Van Klinken, B.J.; van der Wal, J.W.; Einerhand, A.W.; Buller, H.A.; Dekker, J. Sulphation and Secretion of the Predominant Secretory Human Colonic Mucin Muc2 in Ulcerative Colitis. Gut 1999, 44, 387–393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanski, C.; Born, M.; Foss, H.D.; Marowski, B.; Mansmann, U.; Arasteh, K.; Bachler, B.; Papenfuss, M.; Niedobitek, F. Defective Post-Transcriptional Processing of Muc2 Mucin in Ulcerative Colitis and in Crohn’s Disease Increases Detectability of the Muc2 Protein Core. J. Pathol. 1999, 188, 304–311. [Google Scholar] [CrossRef]
- Shaoul, R.; Okada, Y.; Cutz, E.; Marcon, M.A. Colonic Expression of Muc2, Muc5ac, and Tff1 in Inflammatory Bowel Disease in Children. J. Pediatr. Gastroenterol. Nutr. 2004, 38, 488–493. [Google Scholar] [CrossRef]
- Longman, R.J.; Poulsom, R.; Corfield, A.P.; Warren, B.F.; Wright, N.A.; Thomas, M.G. Alterations in the Composition of the Supramucosal Defense Barrier in Relation to Disease Severity of Ulcerative Colitis. J. Histochem. Cytochem. 2006, 54, 1335–1348. [Google Scholar] [CrossRef] [Green Version]
- Moehle, C.; Ackermann, N.; Langmann, T.; Aslanidis, C.; Kel, A.; Kel-Margoulis, O.; Schmitz-Madry, A.; Zahn, A.; Stremmel, W.; Schmitz, G. Aberrant Intestinal Expression and Allelic Variants of Mucin Genes Associated with Inflammatory Bowel Disease. J. Mol. Med. 2006, 84, 1055–1066. [Google Scholar] [CrossRef]
- Furr, A.E.; Ranganathan, S.; Finn, O.J. Aberrant Expression of Muc1 Mucin in Pediatric Inflammatory Bowel Disease. Pediatr Dev. Pathol. 2010, 13, 24–31. [Google Scholar] [CrossRef]
- Larsson, J.M.; Karlsson, H.; Crespo, J.G.; Johansson, M.E.; Eklund, L.; Sjovall, H.; Hansson, C.G. Altered O-Glycosylation Profile of Muc2 Mucin Occurs in Active Ulcerative Colitis and Is Associated with Increased Inflammation. Inflamm. Bowel Dis. 2011, 17, 2299–2307. [Google Scholar] [CrossRef] [PubMed]
- Kini, A.T.; Thangaraj, K.R.; Simon, E.; Shivappagowdar, A.; Thiagarajan, D.; Abbas, S.; Ramachandran, A.; Venkatraman, A. Aberrant Niche Signaling in the Etiopathogenesis of Ulcerative Colitis. Inflamm. Bowel Dis. 2015, 21, 549–2561. [Google Scholar] [CrossRef]
- Yamamoto-Furusho, J.K.; Ascano-Gutierrez, I.; Furuzawa-Carballeda, J.; Fonseca-Camarillo, G. Differential Expression of Muc12, Muc16, and Muc20 in Patients with Active and Remission Ulcerative Colitis. Mediat. Inflamm. 2015, 2015, 659018. [Google Scholar] [CrossRef] [Green Version]
- Alipour, M.; Zaidi, D.; Valcheva, R.; Jovel, J.; Martinez, I.; Sergi, C.; Walter, J.; Mason, A.L.; Wong, G.K.; Dieleman, L.A.; et al. Mucosal Barrier Depletion and Loss of Bacterial Diversity Are Primary Abnormalities in Paediatric Ulcerative Colitis. J. Crohn’s Colitis 2016, 10, 462–471. [Google Scholar] [CrossRef] [PubMed]
- van der Post, S.; Jabbar, K.S.; Birchenough, G.; Arike, L.; Akhtar, N.; Sjovall, H.; Johansson, M.E.V.; Hansson, G.C. Structural Weakening of the Colonic Mucus Barrier Is an Early Event in Ulcerative Colitis Pathogenesis. Gut 2019, 68, 2142–2151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gersemann, M.; Becker, S.; Kübler, I.; Koslowski, M.; Wang, G.; Herrlinger, K.R.; Griger, J.; Fritz, P.; Fellermann, K.; Schwab, M.; et al. Differences in Goblet cell Differentiation between Crohn’s Disease and Ulcerative Colitis. Differentiation 2009, 77, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Senapati, S.; Ho, S.B.; Sharma, P.; Das, S.; Chakraborty, S.; Kaur, S.; Niehans, G.; Batra, S.K. Expression of Intestinal MUC17 Membrane-bound Mucin in Inflammatory and Neoplastic Diseases of the Colon. J. Clin. Pathol. 2010, 63, 702–707. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cumpston, M.; Li, T.; Page, M.J.; Chandler, J.; Welch, V.A.; Higgins, J.P.; Thomas, J. Updated Guidance for Trusted Systematic Reviews: A New Edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst. Rev. 2019, 10, ED000142. [Google Scholar] [CrossRef] [Green Version]
- Grondin, J.A.; Kwon, Y.H.; Far, P.M.; Haq, S.; Khan, W.I. Mucins in Intestinal Mucosal Defense and Inflammation: Learning from Clinical and Experimental Studies. Front. Immunol. 2020, 11, 2054. [Google Scholar] [CrossRef]
- Parikh, K.; Antanaviciute, A.; Fawkner-Corbett, D.; Jagielowicz, M.; Aulicino, A.; Lagerholm, C.; Davis, S.; Kinchen, J.; Chen, H.H.; Alham, N.K.; et al. Colonic Epithelial Cell Diversity in Health and Inflammatory Bowel Disease. Nature 2019, 567, 49–55. [Google Scholar] [CrossRef]
- Forgue-Lafitte, M.E.; Fabiani, B.; Levy, P.P.; Maurin, N.; Flejou, J.F.; Bara, J. Abnormal Expression of M1/Muc5ac Mucin in Distal Colon of Patients with Diverticulitis, Ulcerative Colitis and Cancer. Int. J. Cancer 2007, 121, 1543–1549. [Google Scholar] [CrossRef]
- Rehman, S.; Grigoryeva, L.S.; Richardson, K.H.; Corsini, P.; White, R.C.; Shaw, R.; Portlock, T.J.; Dorgan, B.; Zanjani, Z.S.; Fornili, A.; et al. Structure and Functional Analysis of the Legionella Pneumophila Chitinase Chia Reveals a Novel Mechanism of Metal-Dependent Mucin Degradation. PLoS Pathog. 2020, 16, e1008342. [Google Scholar] [CrossRef]
- Wong, E.; Vaaje-Kolstad, G.; Ghosh, A.; Hurtado-Guerrero, R.; Konarev, P.V.; Ibrahim, A.F.; Svergun, D.I.; Eijsink, V.G.; Chatterjee, N.S.; van Aalten, D.M. The Vibrio Cholerae Colonization Factor Gbpa Possesses a Modular Structure That Governs Binding to Different Host Surfaces. PLoS Pathog. 2012, 8, e1002373. [Google Scholar] [CrossRef]
- Atanasova, K.R.; Reznikov, L.R. Strategies for measuring airway mucus and mucins. Respir. Res. 2019, 20, 261. [Google Scholar] [CrossRef]




| Identification | Population | Methods Used | |||||
|---|---|---|---|---|---|---|---|
| First Author, Reference | Year | Country | Control Samples | Nature of Control Samples | Patient Samples | Diagnosis-Disease Severity/Scoring Indices | Study Design |
| Weiss, [32] | 1996 | USA | 1 | Resected tissue from patient with colon diverticular disease | 8 | Method of Diagnosis/ Scoring Indices used are not stated | Case–control study |
| Tytgat, [33] | 1996 | Netherlands | 7 | Negative history of IBD (colon appeared normal endoscopically and histologically) | Active UC: 6, Inactive UC (remission): 6 | Truelove and Richards Index | Case–control study |
| Hinoda, [34] | 1998 | Japan | 14 | Normal colonic tissue obtained post-mortem (from autopsies) | 31 | Matts Score | Case–control study |
| Van Klinken, [35] | 1999 | Netherlands | 12 | Tissue resected from patients diagnosed with Irritable Bowel Syndrome, hyperplastic polyps or diverticulosis but with negative history of IBD | Active UC: 13, Inactive UC (remission): 12 | Truelove and Richards | Case–control study |
| Hanski, [36] | 1999 | Germany | 13 | Histologically normal mucosae samples | 70 | Matts Score (degree of inflammation), Remmele and Stegner (immunoreactive score for immunohistochemistry) | Case–control study |
| Shaoul, [37] | 2004 | Japan | 5 | Samples taken from study groups histology library (original source of controls is not stated) | 5 | Method of Diagnosis/Scoring Indices used are not stated | Case–control study |
| Longman, [38] | 2006 | UK | 17 | Tissue resected from patients undergoing elective colorectal resection surgery but with negative history of IBD | 40 | Truelove and Witts Criteria | Case–control study |
| Moehle, [39] | 2006 | Germany | 14 | Tissue resected from patients following colonoscopy (8 with no tissue abnormalities; remaining samples from patients with diverticulitis, polyposis coli, lymphoid tissue-lymphoma, carcinoma, diverticulosis, eosinophilic colitis, infectious colitis | Active UC: 14, Inactive UC (remission): 5 | Diagnosis based on clinical features and radiologic/endoscopic findings | Case–control study |
| Gersemann, [46] | 2009 | Germany | 21 | Tissue resected from patients undergoing routine colonoscopy (specific reasons for colonoscopy are not outlined) | Active UC: 25Inactive UC: 15 | Method of Diagnosis/Scoring Indices used are not stated | Case–control study |
| Furr, [40] | 2010 | USA | 21 | Tissues randomly resected from patients undergoing colonic biopsy but with negative history of IBD | 14 | Diagnosis based on medical records | Case–control study |
| Senapati, [47] | 2010 | USA | 12 | Patients undergoing colonoscopy whose colonoscopic exams/histology were normal | 25 | Diagnosis based on medical records | Case–control study |
| Larsson, [41] | 2011 | Sweden | 25 | Tissue resected from patients during colonoscopy for polyp surveillance, investigation of anaemia or rectal bleeding (normal colonic mucosa, no signs of inflammation) | Active UC: 15, Inactive UC (remission): 13 | Sandborn’s histological activity score | Case–control study |
| Kini, [42] | 2015 | India | 12 | Tissue resected from patients with IBS undergoing routine colonoscopy or during polyp surveillance colonoscopy | 22 | Truelove and Witts (clinical disease severity), Ulcerative Colitis Disease Activity Index (endoscopic severity) | Case–control study |
| Yamamoto-Furusho, [43] | 2015 | México | 30 | Tissue resected from patients during colonoscopy for polyp surveillance/screening and evaluation for anaemia (normal colonic mucosa, no signs of inflammation) | Active UC: 20, Inactive UC (remission): 20 | Mayo score (clinical and endoscopic activity evaluation), Riley score (histological activity evaluation) | Case–control study |
| Alipour, [44] | 2016 | Canada | 12 | Tissue resected from patients with Irritable bowel syndrome, benign polyps, chronic diarrhoea but negative history of IBD and no signs of inflammation | 10 | Paris Classification and Paediatric Ulcerative Colitis Activity Index (PUCAI) | Case–control study |
| van der Post, [45] | 2019 | Sweden | 47 | Tissues resected from patients during colonoscopy; patients with polyps, diverticulosis but with negative history of IBD | Active UC: 36, Inactive UC (remission): 28 | Mayo score (clinical and endoscopic activity evaluation) and/or Sandborn’s histological activity score | Case–control study |
| First Author, Reference | Purpose of Study | MUC Gene Investigated | Measuring MUC Expression | Key Findings |
|---|---|---|---|---|
| Weiss, [32] | To investigate effect of inflammation on expression of mucin genes at cellular level | MUC2, MUC3 | In-situ hybridisation with RNA probes | MUC2 and MUC3 expression in colonic mucosa is independent of inflammation |
| Tytgat, [33] | To study regulation of MUC2 expression in patients with UC compared with controls | MUC2 | MUC2 precursor quantified by SDS-PAGE, total MUC2 by dot blot, in-situ hybridisation with RNA probes to quantify MUC2 mRNA | Inefficient translation of MUC2 mRNA may lead to the reduction in MUC2 synthesis observed in active UC |
| Hinoda, [34] | To determine if MUC2 protein expression is altered in UC | MUC2 | MUC2 protein detected by Immunohistochemistry | Decreased MUC2 protein production and expression in active UC is associated with undifferentiated goblet cells |
| Van Klinken, [35] | To determine whether there are alterations in MU2 sulphation and secretion in active UC | MUC2 | Analysis and quantification of total MUC2 using SDS-PAGE and dot blotting | Absolute amount of MUC2 secreted is decreased and mucins are under-sulphated in active UC |
| Hanski, [36] | To study alterations in MUC2 expression in UC patient colonic tissue | MUC2 | MUC2 protein detected by immunohistochemistry and MUC2 mRNA detected using in-situ hybridisation | Increase in MUC2 protein detection in UC samples may be related to reduction in post transcriptional modification |
| Shaoul, [37] | To investigate alterations in expression and distribution of MUC2, MUC5AC, trefoil factor 1 (TFF1) in UC | MUC2, MUC5AC | PAS/Alcian blue immunohistochemistry | Immature (poorly glycos-ylated) MUC2 is expressed in UC colonic mucosa depleted of goblet cells |
| Longman, [38] | To investigate alterations in the expression of mucin genes and trefoil peptide genes in UC | MUC1-6 | Immunohistochemistry and in-situ hybridisation | MUC1 expression upregulation is associated with severe UC and there is a reduction of MUC2 expression in UC |
| Moehle, [39] | To characterize changes in mucin expression and identify allelic variants of MUC genes in UC | MUC1-20 | Affymetrix DNA-microarray analysis and RT-PCR | MUC12 mRNA expression is downregulated in UC and is independent of inflammation |
| Gersemann, [46] | To understand the mechanisms involved with goblet cell differentiation and mucin production in IBD | MUC1, MUC2, MUC4 | RT-PCR | Impairments in goblet cell differentiation factor induction in UC correlates with a reduction in mucin synthesis. |
| Furr, [40] | To determine whether MUC1 expression is altered in IBD | MUC1 | Immunochemistry using anti-MUC1 anti-bodies | Overexpression and hypoglycosylation of MUC1 observed in a subset of UC patients |
| Senapati, [47] | To determine the subcellular localization of MUC17 in colonic mucosa and to determine whether MUC17 expression is altered in IBD and neoplastic diseases. | MUC17 | Immunohistochemistry using anti-MUC17 polyclonal antibody | MUC17 expression is reduced in colonic mucosa of UC patients |
| Larsson, [41] | To determine whether MUC2 glycosylation is related to degree of mucosal inflammation in UC | MUC2 | SDS-PAGE used to identify and quantify MUC2 | Alterations in MUC2 glycosylation are associated with inflammation |
| Kini, [42] | To determine whether alterations occur in colonic stem cells during the pathogenesis of UC and to determine the impact of such changes on goblet cell development and proteins synthesized by goblet cells | MUC2 | H&E, Alcian blue and PAS immunohistochemistry staining to detect MUC2 protein | A reduction in MUC2 protein within the lower colonic crypt precedes inflammation. |
| Yamamoto-Furusho, [43] | To determine if MUC12, MUC16, MUC20 expression changes in UC | MUC12, MUC16, MUC20 | MUC gene expression measured using RT-PCR and MUC protein expression measured using immunohistochemistry | MUC16, MUC20 expression increase in UC and increase in MUC20 was associated with remission of UC |
| Alipour, [44] | To assess whether mucosal barrier defects are prerequisites to UC | MUC2 | Fluorescence in-situ hybridisation (FISH) and immunofluorescence | Reduction in mucin-containing goblet cells and mucin production in UC patients compared to controls |
| van der Post, [45] | To investigate compositional alterations that occur at the adherent mucus layer in UC | MUC2 | Absolute quantification of MUC2 using Skyline (V.3.6.0.1) following mass spectrometry | Reduction of MUC2 in active UC associated with exhaustion of secretory response of goblet cells to microbes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bankole, E.; Read, E.; Curtis, M.A.; Neves, J.F.; Garnett, J.A. The Relationship between Mucins and Ulcerative Colitis: A Systematic Review. J. Clin. Med. 2021, 10, 1935. https://doi.org/10.3390/jcm10091935
Bankole E, Read E, Curtis MA, Neves JF, Garnett JA. The Relationship between Mucins and Ulcerative Colitis: A Systematic Review. Journal of Clinical Medicine. 2021; 10(9):1935. https://doi.org/10.3390/jcm10091935
Chicago/Turabian StyleBankole, Esther, Emily Read, Michael A. Curtis, Joana F. Neves, and James A. Garnett. 2021. "The Relationship between Mucins and Ulcerative Colitis: A Systematic Review" Journal of Clinical Medicine 10, no. 9: 1935. https://doi.org/10.3390/jcm10091935






