Date of Admission during COVID-19 Pandemic Impacted Patient Outcomes in Addition to the Higher Efficacy of Tocilizumab Plus High-Dose Corticosteroid Therapy Compared to Tocilizumab Alone
Abstract
:1. Introduction
2. Materials and Methods
2.1. Population and Study Design
2.2. Data Collection
2.3. Univariate Analysis
2.4. Machine Learning Severity Score Comparison by Means of COVID-19 Salamanca Risk Score
2.5. Multivariate Analysis through Machine Learning Methodology
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef] [Green Version]
- Wu, Z.; McGoogan, J.M. Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72314 Cases from the Chinese Center for Disease Control and Prevention. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Gustine, J.N.; Jones, D. Immunopathology of Hyperinflammation in COVID-19. Am. J. Pathol. 2021, 191, 4–17. [Google Scholar] [CrossRef] [PubMed]
- Dorward, D.A.; Russell, C.D.; Um, I.H.; Elshani, M.; Armstrong, S.D.; Penrice-Randal, R.; Millar, T.; Lerpiniere, C.E.B.; Tagliavini, G.; Hartley, C.S.; et al. Tissue-Specific Immunopathology in Fatal COVID-19. Am. J. Respir. Crit. Care Med. 2021, 203, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Hertanto, D.M.; Wiratama, B.S.; Sutanto, H.; Wungu, C.D.K. Immunomodulation as a Potent COVID-19 Pharmacotherapy: Past, Present and Future. J. Inflamm. Res. 2021, 14, 3419–3428. [Google Scholar] [CrossRef] [PubMed]
- Sterne, J.A.C.; Murthy, S.; Diaz, J.V.; Slutsky, A.S.; Villar, J.; Angus, D.C.; Annane, D.; Azevedo, L.C.P.; Berwanger, O.; Cavalcanti, A.B.; et al. Association between Administration of Systemic Corticosteroids and Mortality among Critically Ill Patients with COVID-19: A Meta-analysis. JAMA J. Am. Med. Assoc. 2020, 324, 1330–1341. [Google Scholar] [CrossRef]
- Ghosn, L.; Chaimani, A.; Evrenoglou, T.; Davidson, M.; Graña, C.; Schmucker, C.; Bollig, C.; Henschke, N.; Sguassero, Y.; Nejstgaard, C.H.; et al. Interleukin-6 blocking agents for treating COVID-19: A living systematic review. Cochrane Database Syst. Rev. 2021, 2021, CD013881. [Google Scholar] [CrossRef]
- The WHO Rapid Evidence Appraisal for COVID-19 Therapies (REACT) Working Group; Domingo, P.; Mur, I.; Mateo, G.M.; Gutierrez, M.D.M.; Pomar, V.; de Benito, N.; Corbacho, N.; Herrera, S.; Millan, L.; et al. Association between Administration of IL-6 Antagonists and Mortality among Patients Hospitalized for COVID-19: A Meta-analysis. JAMA 2021, 326, 449–518. [Google Scholar] [CrossRef]
- Ruiz-Antorán, B.; Sancho-López, A.; Torres, F.; Moreno-Torres, V.; de Pablo-López, I.; García-López, P.; Abad-Santos, F.; Rosso-Fernández, C.M.; Aldea-Perona, A.; Montané, E.; et al. Combination of Tocilizumab and Steroids to Improve Mortality in Patients with Severe COVID-19 Infection: A Spanish, Multicenter, Cohort Study. Infect. Dis. Ther. 2021, 10, 347–362. [Google Scholar] [CrossRef] [PubMed]
- Abani, O.; Abbas, A.; Abbas, F.; Abbas, M.; Abbasi, S.; Abbass, H.; Abbott, A.; Abdallah, N.; Abdelaziz, A.; Abdelfattah, M.; et al. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): A randomised, controlled, open-label, platform trial. Lancet 2021, 397, 1637–1645. [Google Scholar] [CrossRef]
- Dennis, J.M.; McGovern, A.P.; Vollmer, S.J.; Mateen, B.A. Improving Survival of Critical Care Patients with Coronavirus Disease 2019 in England: A National Cohort Study, March to June 2020. Crit. Care Med. 2021, 49, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Xiong, X.; Wai, A.K.C.; Wong, J.Y.H.; Tang, E.H.M.; Chu, O.C.K.; Wong, C.K.H.; Rainer, T.H. Impact of varying wave periods of COVID-19 on in-hospital mortality and length of stay for admission through emergency department: A territory-wide observational cohort study. Influ. Other Respir. Viruses 2021. [Google Scholar] [CrossRef] [PubMed]
- Marcos, M.; Belhassen-García, M.; Sánchez-Puente, A.; Sampedro-Gomez, J.; Azibeiro, R.; Dorado-Díaz, P.-I.; Marcano-Millán, E.; García-Vidal, C.; Moreiro-Barroso, M.-T.; Cubino-Bóveda, N.; et al. Development of a severity of disease score and classification model by machine learning for hospitalized COVID-19 patients. PLoS ONE 2021, 16, e0240200. [Google Scholar] [CrossRef] [PubMed]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stuart, E. Matching methods for causal inference: A review and a look forward. Stat. Sci. 2010, 25, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Beretta, L.; Santaniello, A. Nearest neighbor imputation algorithms: A critical evaluation. BMC Med. Inform. Decis. Mak. 2016, 16, 197–208. [Google Scholar] [CrossRef] [Green Version]
- Chen, T.; Guestrin, C. XGBoost: A scalable tree boosting system. In Proceedings of the ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13–17 August 2016; Association for Computing Machinery: New York, NY, USA, 2016; pp. 785–794. [Google Scholar]
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef] [Green Version]
- Airola, A.; Pahikkala, T.; Waegeman, W.; De Baets, B.; Salakoski, T. An experimental comparison of cross-validation techniques for estimating the area under the ROC curve. Comput. Stat. Data Anal. 2011, 55, 1828–1844. [Google Scholar] [CrossRef]
- Nadeau, C.; Bengio, Y. Inference for the generalization error. Mach. Learn. 2003, 52, 239–281. [Google Scholar] [CrossRef] [Green Version]
- Bouckaert, R.R. Choosing between two learning algorithms based on calibrated tests. In Proceedings of the Twentieth International Conference on Machine Learning (ICML, 2003), Washington, DC, USA, 21–24 August 2003; pp. 51–58. [Google Scholar]
- Ramiro, S.; Mostard, R.L.M.; Magro-Checa, C.; Van Dongen, C.M.P.; Dormans, T.; Buijs, J.; Gronenschild, M.; De Kruif, M.D.; Van Haren, E.H.J.; Van Kraaij, T.; et al. Historically controlled comparison of glucocorticoids with or without tocilizumab versus supportive care only in patients with COVID-19-associated cytokine storm syndrome: Results of the CHIC study. Ann. Rheum. Dis. 2020, 79, 1143–1151. [Google Scholar] [CrossRef]
- Van den Eynde, E.; Gasch, O.; Oliva, J.C.; Prieto, E.; Calzado, S.; Gomila, A.; Machado, M.L.; Falgueras, L.; Ortonobes, S.; Morón, A.; et al. Corticosteroids and tocilizumab reduce in-hospital mortality in severe COVID-19 pneumonia: A retrospective study in a Spanish hospital. Infect. Dis. 2021, 53, 291–302. [Google Scholar] [CrossRef]
- López-Medrano, F.; Asín, M.A.P.-J.; Fernández-Ruiz, M.; Carretero, O.; Lalueza, A.; de la Calle, G.M.; Caro, J.M.; de la Calle, C.; Catalán, M.; García-García, R.; et al. Combination therapy with tocilizumab and corticosteroids for aged patients with severe COVID-19 pneumonia: A single-center retrospective study. Int. J. Infect. Dis. 2021, 105, 487–494. [Google Scholar] [CrossRef]
- Rodríguez-Baño, J.; Pachón, J.; Carratala, J.; Ryan, P.; Jarrín, I.; Yllescas, M.; Arribas, J.R.; Berenguer, J.; Muñoz, E.A.; Gil Divasson, P.; et al. Treatment with tocilizumab or corticosteroids for COVID-19 patients with hyperinflammatory state: A multicentre cohort study (SAM-COVID-19). Clin. Microbiol. Infect. 2021, 27, 244–252. [Google Scholar] [CrossRef]
- Delgado-Rodríguez, M.; Llorca, J. Bias. J. Epidemiol. Community Health 2004, 58, 635–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ward, T.; Johnsen, A. Understanding an evolving pandemic: An analysis of the clinical time delay distributions of COVID-19 in the United Kingdom. PLoS ONE 2021, 16, e0257978. [Google Scholar] [CrossRef] [PubMed]
- Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Phil, D.; Staplin, N.; Brightling, C.; Med, F.; et al. Dexamethasone in Hospitalized Patients with Covid-19. N. Engl. J. Med. 2021, 384, 693–704. [Google Scholar] [CrossRef]
- Ruiz-Irastorza, G.; Pijoan, J.-I.; Bereciartua, E.; Dunder, S.; Dominguez, J.; Garcia-Escudero, P.; Rodrigo, A.; Gomez-Carballo, C.; Varona, J.; Guio, L.; et al. Second week methyl-prednisolone pulses improve prognosis in patients with severe coronavirus disease 2019 pneumonia: An observational comparative study using routine care data. PLoS ONE 2020, 15, e0239401. [Google Scholar] [CrossRef]
- Cusacovich, I.; Aparisi, Á.; Marcos, M.; Ybarra-Falcón, C.; Iglesias-Echevarria, C.; Lopez-Veloso, M.; Barraza-Vengoechea, J.; Dueñas, C.; Martínez, S.A.J.; Rodríguez-Alonso, B.; et al. Corticosteroid Pulses for Hospitalized Patients with COVID-19: Effects on Mortality. Mediat. Inflamm. 2021, 2021, 6637227. [Google Scholar] [CrossRef] [PubMed]
- Edalatifard, M.; Akhtari, M.; Salehi, M.; Naderi, Z.; Jamshidi, A.; Mostafaei, S.; Najafizadeh, S.R.; Farhadi, E.; Jalili, N.; Esfahani, M.; et al. Intravenous methylprednisolone pulse as a treatment for hospitalised severe COVID-19 patients: Results from a randomised controlled clinical trial. Eur. Respir. J. 2020, 56, 2002808. [Google Scholar] [CrossRef]
- Li, J.; Liao, X.; Zhou, Y.; Wang, L.; Yang, H.; Zhang, W.; Zhang, Z.; Kang, Y. Comparison of Associations between Glucocorticoids Treatment and Mortality in COVID-19 Patients and SARS Patients. Shock, 2021; in press. [Google Scholar] [CrossRef]


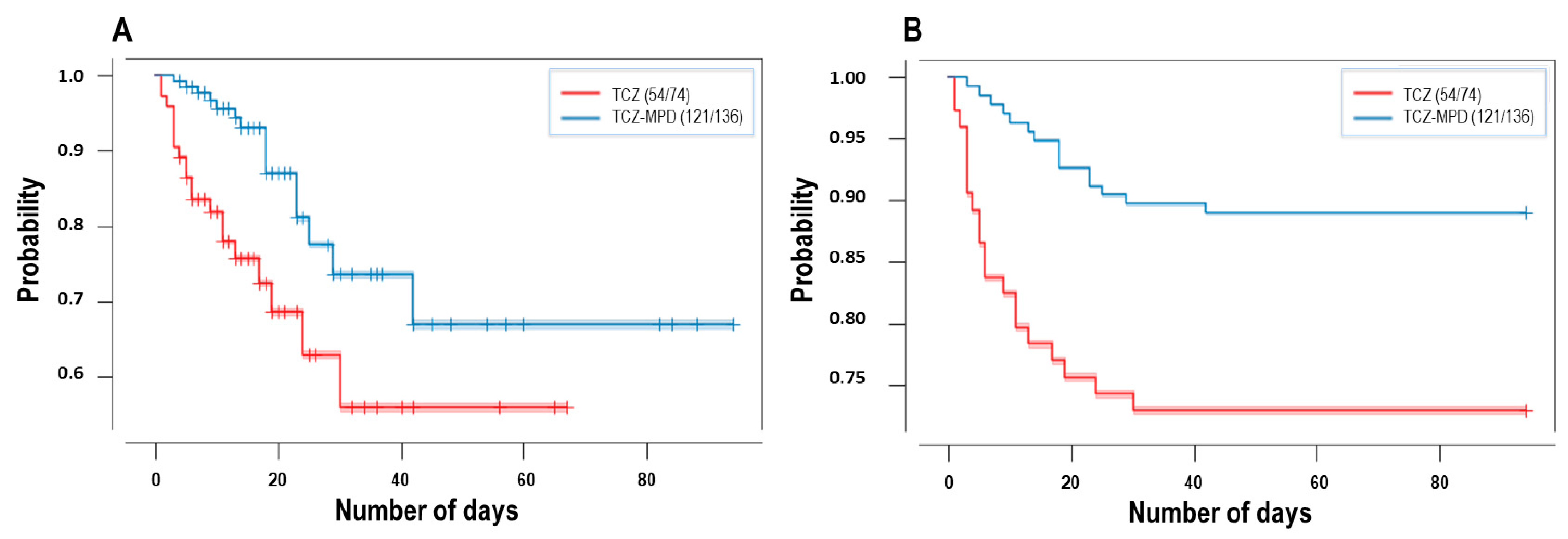

| Tocilizumab and Glucocorticoids | Tocilizumab | ||||
|---|---|---|---|---|---|
| Name | n | Mean | n | Mean | p-Value |
| Age, years (mean ± SD) | 136 | 64.3 ± 11.7 | 74 | 65.2 ± 10.9 | 0.578 |
| Male, n (%) | 136 | 92 (67.6%) | 74 | 51 (68.9%) | 0.878 |
| COMORBIDITIES | |||||
| Classic Charlson comorbidity index, n (mean ± SD) | 136 | 1.0 ± 1.6 | 74 | 0.9 ± 1.3 | 0.553 |
| Updated Charlson comorbidity index, n (mean ± SD) | 136 | 0.6 ± 1.4 | 74 | 0.5 ± 1.1 | 0.616 |
| Myocardial infarction, n (%) | 136 | 10 (7.4%) | 74 | 6 (8.1%) | 1 |
| Congestive heart failure, n (%) | 136 | 7 (5.1%) | 74 | 5 (6.8%) | 0.757 |
| Peripheral vascular disease, n (%) | 136 | 4 (2.9%) | 74 | 3 (4.1%) | 0.699 |
| Arrhythmia, n (%) | 136 | 10 (7.4%) | 74 | 3 (4.1%) | 0.55 |
| Cerebrovascular accident, n (%) | 136 | 5 (3.7%) | 74 | 3 (4.1%) | 1 |
| Cognitive impairment, n (%) | 136 | 2 (1.5%) | 74 | 3 (4.1%) | 0.348 |
| Other central nervous system diseases, n (%) | 136 | 3 (2.2%) | 74 | 2 (2.7%) | 1 |
| Chronic obstructive pulmonary disease, n (%) | 136 | 4 (2.9%) | 74 | 5 (6.8%) | 0.284 |
| Asthma, n (%) | 136 | 5 (3.7%) | 74 | 2 (2.7%) | 1 |
| Other chronic pulmonary disease, n (%) | 136 | 7 (5.1%) | 74 | 6 (8.1%) | 0.388 |
| Rheumatological disorder, n (%) | 136 | 7 (5.1%) | 74 | 3 (4.1%) | 1 |
| Peptic ulcer disease, n (%) | 136 | 4 (2.9%) | 74 | 2 (2.7%) | 1 |
| Hemiplegia/paraplegia, n (%) | 136 | 0 (0.0%) | 74 | 1 (1.4%) | 0.352 |
| Chronic kidney disease (eGFR < 30), n (%) | 136 | 1 (0.7%) | 74 | 1 (1.4%) | 1 |
| Solid tumor, n (%) | 136 | 11 (8.1%) | 74 | 3 (4.1%) | 0.387 |
| Neoplasia without metastasis, n (%) | 136 | 10 (7.4%) | 74 | 1 (1.4%) | 0.101 |
| Solid tumor metastasis, n (%) | 136 | 1 (0.7%) | 74 | 0 (0.0%) | 1 |
| Hematologic neoplasm, n (%) | 136 | 3 (2.2%) | 74 | 3 (4.1%) | 0.427 |
| Chronic acute leukemia, n (%) | 136 | 1 (0.7%) | 74 | 2 (2.7%) | 0.284 |
| Lymphoma, n (%) | 136 | 3 (2.2%) | 74 | 1 (1.4%) | 1 |
| Hypertension, n (%) | 136 | 55 (40.4%) | 74 | 32 (43.2%) | 0.77 |
| Other endocrine disease, n (%) | 136 | 12 (8.8%) | 74 | 9 (12.2%) | 0.475 |
| Inflammatory autoimmune disease, n (%) | 136 | 9 (6.6%) | 74 | 4 (5.4%) | 1 |
| Transplant recipient, n (%) | 136 | 0 (0.0%) | 74 | 1 (1.4%) | 0.352 |
| Obesity, n (%) | 105 | 23 (21.9%) | 60 | 19 (31.7%) | 0.195 |
| Dyslipidemia, n (%) | 136 | 43 (31.6%) | 74 | 34 (45.9%) | 0.051 |
| Current smoking, n (%) | 116 | 9 (7.8%) | 72 | 4 (5.6%) | 0.769 |
| Former/current smoking, n (%) | 116 | 33 (28.4%) | 72 | 14 (19.4%) | 0.225 |
| Diabetes, n (%) | 136 | 31 (22.8%) | 74 | 17 (23.0%) | 1 |
| Cancer, n (%) | 136 | 13 (9.6%) | 74 | 6 (8.1%) | 0.806 |
| PREVIOUS MEDICATIONS, n (%) | |||||
| Angiotensin-converting enzyme inhibitors | 135 | 16 (11.9%) | 73 | 7 (9.6%) | 0.817 |
| Angiotensin II receptor blockers | 135 | 23 (17.0%) | 71 | 20 (28.2%) | 0.072 |
| Chemotherapy | 136 | 3 (2.2%) | 74 | 0 (0.0%) | 0.554 |
| Immunosuppressants | 136 | 4 (2.9%) | 73 | 3 (4.1%) | 0.697 |
| Systemic corticosteroids | 136 | 3 (2.2%) | 73 | 4 (5.5%) | 0.242 |
| Inhaled corticosteroids | 136 | 6 (4.4%) | 73 | 2 (2.7%) | 0.716 |
| Acenocumarol | 136 | 4 (2.9%) | 74 | 1 (1.4%) | 0.659 |
| Low-molecular-weight heparin | 136 | 5 (3.7%) | 74 | 3 (4.1%) | 1 |
| Direct oral anticoagulants | 136 | 10 (7.4%) | 74 | 5 (6.8%) | 1 |
| New oral anticoagulants | 136 | 1 (0.7%) | 74 | 1 (1.4%) | 1 |
| Androgen antagonists | 136 | 1 (0.7%) | 74 | 0 (0.0%) | 1 |
| Hydroxychloroquine treatment prior to admission, n (%) | 136 | 22 (16.2%) | 74 | 5 (6.8%) | 0.055 |
| Azithromycin treatment prior to admission, n (%) | 136 | 39 (28.7%) | 74 | 10 (13.5%) | 0.016 |
| SYMPTOMS/SIGNS | |||||
| Duration of symptoms before admissionguifen(days), n (mean ± SD) | 127 | 7.2 ± 4.5 | 71 | 7.2 ± 5.1 | 0.939 |
| Fever, n (%) | 136 | 101 (74.3%) | 74 | 66 (89.2%) | 0.012 |
| Duration of fever before admissionguifen(days), n (mean ± SD) | 90 | 6.7 ± 4.0 | 64 | 6.2 ± 3.5 | 0.396 |
| Maximum temperature, n (mean ± SD) | 92 | 38.3 ± 0.6 | 60 | 37.9 ± 4.1 | 0.357 |
| Dry cough, n (%) | 136 | 82 (60.3%) | 74 | 48 (64.9%) | 0.554 |
| Productive cough, n (%) | 136 | 8 (5.9%) | 74 | 10 (13.5%) | 0.072 |
| Chest Pain, n (%) | 136 | 15 (11.0%) | 74 | 9 (12.2%) | 0.823 |
| Dyspnea, n (%) | 136 | 84 (61.8%) | 74 | 53 (71.6%) | 0.174 |
| Diminished level of consciousness, n (%) | 136 | 9 (6.6%) | 74 | 7 (9.5%) | 0.587 |
| Seizures, n (%) | 136 | 1 (0.7%) | 74 | 0 (0.0%) | 1 |
| Asthenia, n (%) | 136 | 55 (40.4%) | 74 | 33 (44.6%) | 0.562 |
| Myalgia/arthralgia, n (%) | 136 | 28 (20.6%) | 74 | 20 (27.0%) | 0.306 |
| Anosmia, n (%) | 136 | 6 (4.4%) | 74 | 2 (2.7%) | 0.715 |
| Ageusia, n (%) | 136 | 9 (6.6%) | 74 | 2 (2.7%) | 0.335 |
| Conjunctivitis, n (%) | 136 | 1 (0.7%) | 74 | 0 (0.0%) | 1 |
| Nasal congestion, n (%) | 136 | 1 (0.7%) | 74 | 5 (6.8%) | 0.021 |
| Headache, n (%) | 136 | 12 (8.8%) | 74 | 6 (8.1%) | 1 |
| Odynophagia, n (%) | 136 | 5 (3.7%) | 74 | 3 (4.1%) | 1 |
| Hemoptysis, n (%) | 136 | 0 (0.0%) | 74 | 5 (6.8%) | 0.005 |
| Nausea/vomiting, n (%) | 136 | 19 (14.0%) | 74 | 6 (8.1%) | 0.267 |
| Abdominal pain, n (%) | 136 | 7 (5.1%) | 74 | 1 (1.4%) | 0.265 |
| Diarrhea, n (%) | 136 | 34 (25.0%) | 74 | 15 (20.3%) | 0.497 |
| BASELINE CHARACTERISTICS | |||||
| COVID-19 Salamanca Risk Score (n) | 136 | 28.6 ± 23.4 | 74 | 30.8 ± 26.0 | 0.543 |
| SOFA Score (n) | 136 | 1.0 ± 1.2 | 74 | 1.4 ± 1.6 | 0.045 |
| Pneumonia, (%) | 136 | 130 (95.6%) | 74 | 70 (94.6%) | 0.744 |
| Labored breathing, n (mean ± SD) | 135 | 41 (30.4%) | 74 | 26 (35.1%) | 0.536 |
| Heart rate, beats/min, n (mean ± SD) | 136 | 87.3 ± 16.3 | 74 | 89.6 ± 16.2 | 0.339 |
| Mean arterial pressure, n (mean ± SD) | 136 | 87.6 ± 13.6 | 74 | 91.5 ± 13.5 | 0.048 |
| Glasgow Coma Scale, n (mean ± SD) | 136 | 14.8 ± 1.0 | 74 | 14.8 ± 0.9 | 0.903 |
| Temperature, n (mean ± SD) | 136 | 37.1 ± 0.9 | 74 | 37.1 ± 1.1 | 0.739 |
| SpO2/FiO2 ratio, n (mean ± SD) | 136 | 368.5 ± 90.5 | 74 | 342.6 ± 116.6 | 0.076 |
| Oxygen supplementation, n (mean ± SD) | 136 | 67 (49.3%) | 74 | 40 (54.1%) | 0.564 |
| Pulmonary infiltrates on chest X-ray, n (mean ± SD) | 136 | 131 (96.3%) | 74 | 71 (95.9%) | 1 |
| Bilateral pulmonary infiltrate, n (mean ± SD) | 136 | 121 (89.0%) | 74 | 65 (87.8%) | 0.823 |
| Lopinavir/ritonavir treatment | 136 | 128 (94.1%) | 74 | 72 (97.3%) | 0.5 |
| Laboratory Findings | |||||
|---|---|---|---|---|---|
| Name | n | Mean | n | Mean | p-Value |
| Glucose (mg/dL) | 128 | 136.4 ± 61.7 | 71 | 126.5 ± 35.2 | 0.216 |
| Urea (mg/dL) | 131 | 44.1 ± 30.0 | 72 | 43.8 ± 27.6 | 0.936 |
| Urate (mg/dL) | 112 | 4.9 ± 2.0 | 62 | 4.8 ± 1.8 | 0.95 |
| eGFR (mL/min/1.73 m2) | 133 | 70.9 ± 21.4 | 74 | 71.6 ± 20.8 | 0.813 |
| Calcium (mg/dL) | 125 | 9.0 ± 0.6 | 63 | 8.8 ± 0.5 | 0.003 |
| Magnesium (mmol/L) | 125 | 2.1 ± 0.3 | 63 | 2.0 ± 0.2 | 0.05 |
| Sodium (mmol/L) | 130 | 136.0 ± 3.3 | 71 | 136.5 ± 3.3 | 0.348 |
| Potassium (mmol/L) | 130 | 4.0 ± 0.5 | 71 | 4.0 ± 0.4 | 0.74 |
| Alanine Aminotransferase (U/L) | 129 | 49.5 ± 73.8 | 70 | 41.8 ± 37.7 | 0.418 |
| Aspartate Aminotransferase (U/L) | 107 | 54.4 ± 46.7 | 37 | 68.6 ± 51.3 | 0.123 |
| Alkaline phosphatase (U/L) | 126 | 87.6 ± 99.0 | 70 | 70.3 ± 28.2 | 0.153 |
| Gamma-glutamyl transferase (U/L) | 127 | 108.8 ± 287.9 | 70 | 67.1 ± 52.1 | 0.232 |
| Lactate dehydrogenase (U/L) | 128 | 394.4 ± 149.9 | 71 | 397.0 ± 142.4 | 0.905 |
| Proteins (g/L) | 125 | 7.6 ± 0.6 | 64 | 7.4 ± 0.5 | 0.012 |
| Albumin (g/L) | 122 | 3.8 ± 0.4 | 64 | 3.8 ± 0.4 | 0.956 |
| Creatine kinase (U/L) | 123 | 140.0 ± 136.3 | 63 | 215.1 ± 283.7 | 0.016 |
| Procalcitonin (ng/mL) | 82 | 0.4 ± 1.1 | 67 | 0.6 ± 1.7 | 0.548 |
| Prothrombine time (%) | 116 | 86.5 ± 16.9 | 67 | 84.6 ± 17.6 | 0.488 |
| INR | 115 | 1.2 ± 0.5 | 67 | 1.3 ± 1.2 | 0.391 |
| Activated partial thromboplastine time (s) | 63 | 34.7 ± 6.8 | 59 | 34.6 ± 4.9 | 0.904 |
| Fibrinogen levels (mg/dL) | 112 | 693.1 ± 192.3 | 61 | 626.2 ± 189.8 | 0.03 |
| Hemoglobin (g/dL) | 133 | 14.3 ± 1.9 | 72 | 14.3 ± 1.8 | 0.954 |
| White blood cells count (×109/L) | 116 | 11.9 ± 35.9 | 63 | 7.5 ± 6.3 | 0.339 |
| Neutrophil cell count (×109/L) | 131 | 6.2 ± 3.2 | 72 | 6.2 ± 3.7 | 0.948 |
| Lymphocyte count (×109/L) | 132 | 3.0 ± 23.3 | 72 | 1.2 ± 1.1 | 0.511 |
| Monocyte count (×109/L) | 130 | 0.5 ± 0.9 | 71 | 0.5 ± 0.3 | 0.667 |
| C-reactive protein (mg/dL) | 128 | 14.6 ± 11.0 | 71 | 14.5 ± 11.7 | 0.947 |
| Interleukin-6 (pg/mL) | 25 | 89.5 ± 104.9 | 16 | 183.7 ± 382.0 | 0.248 |
| D-dimer level (pg/mL) | 126 | 2.2 ± 7.9 | 64 | 2.9 ± 11.3 | 0.617 |
| Platelet count (×109/L) | 134 | 217.8 ± 90.1 | 74 | 195.7 ± 79.1 | 0.079 |
| Bilirubin (total) (mg/dL) | 132 | 0.6 ± 0.3 | 73 | 0.6 ± 0.3 | 0.738 |
| Creatinine (mg/dL) | 133 | 1.1 ± 0.4 | 74 | 1.1 ± 0.5 | 0.973 |
| Tocilizumab Group | Tocilizumab and Glucocorticoids Group | p-Value | ||||
|---|---|---|---|---|---|---|
| Average COVID-19 Salamanca Risk Score | Death or Intubation | Death | Death or Intubation | Death | Death or Intubation | Death |
| 0–8.8% | 2/16 = 12.5% | 1/16 = 6.3% | 3/26 = 11.5% | 0/26 = 0% | 1.0 | 0.381 |
| 8.8–15.7% | 3/11 = 27.3% | 2/11 = 18.2% | 4/31 = 12.9% | 0/31 = 0% | 0.353 | 0.064 |
| 15.7–28% | 6/16 = 37.5% | 2/16 = 12.5% | 4/26 = 15.4% | 2/26 = 7.7% | 0.142 | 0.628 |
| 28–49.5% | 9/15 = 60% | 4/15 = 26.7% | 7/27 = 25.9% | 5/27 = 18.5% | 0.047 | 0.698 |
| 49.5–100% | 13/16 = 81.3% | 11/16 = 68.8% | 11/26 = 42.3% | 8/26 = 30.8% | 0.024 | 0.026 |
| TOTAL | 33/74 = 44.6% | 20/74 = 27% | 29/136 = 21.3% | 15/136 = 11% | <0.001 | 0.006 |
| Tocilizumab | Tocilizumab and Glucocorticoids | |||||
|---|---|---|---|---|---|---|
| Date of Admission | Average COVID-19 Salamanca Risk Score | Death or Intubation | Death | Average COVID-19 Salamanca Risk Score | Death or Intubation | Death |
| March 1st–March 21st | 33.2% | 22/40 = 55% | 13/40 = 32.5% | 44.4% | 6/12 = 50% | 4/12 = 33.3% |
| March 22nd–March 25th | 34.1% | 9/21 = 42.9% | 7/21 = 33.3% | 27.8% | 5/21 = 23.8% | 2/21 = 9.5% |
| March 26th–March 31st | 25.7% | 2/7 = 28.6% | 0/7 = 0% | 29.9% | 7/39 = 17.9% | 4/39 = 19.3% |
| April 1st–April 7th | 11.5% | 0/4 = 0% | 0/4 = 0% | 24.4% | 5/30 = 16.7% | 1/30 = 3.3% |
| April 8th–April 14th | 4.3% | 0/2 = 0% | 0/2 = 0% | 25.9% | 6/34 = 17.6% | 4/34 = 11.8% |
| TOTAL | 30.8% | 33/74 = 44.6% | 20/74 = 27% | 28.6% | 29/136 = 21.3% | 15/136 = 11% |
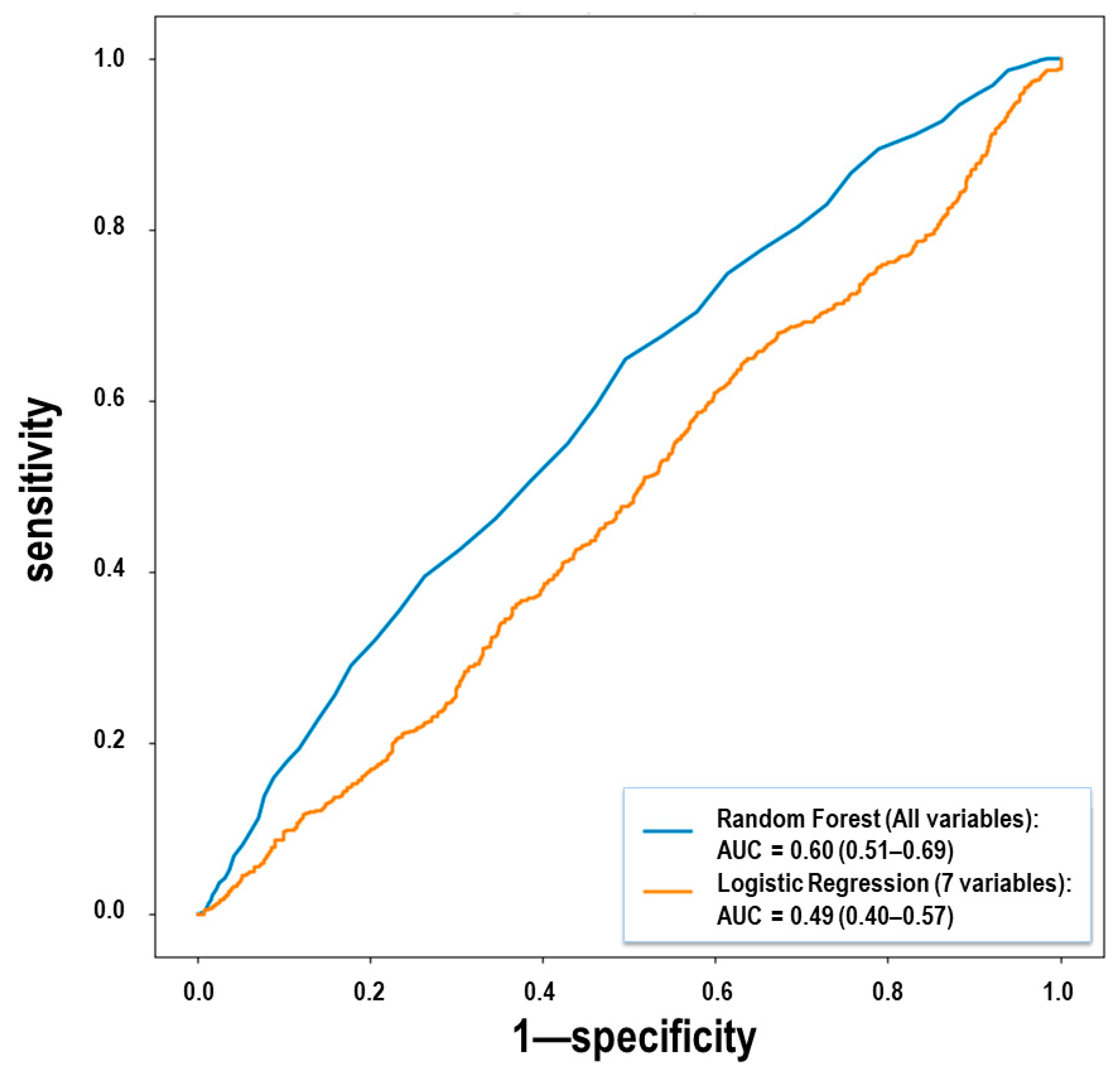
| Variable | Relative Importance |
|---|---|
| Mean arterial pressure | 1.000 |
| Magnesium levels | 0.822 |
| Protein levels | 0.759 |
| Lactate dehydrogenase (ldh) | 0.651 |
| Sodium levels | 0.434 |
| Hemoptysis | 0.429 |
| D-dimer levels | 0.404 |
| Neutrophil count | 0.380 |
| Aspartate aminotransferase (ast) levels | 0.370 |
| Lymphocyte count | 0.369 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Belhassen-García, M.; Sánchez-Puente, A.; Dorado-Díaz, P.-I.; López-Bernús, A.; Sampedro-Gómez, J.; Azibeiro-Melchor, R.; Marcano-Millán, E.; Rodríguez-Alonso, B.; Sánchez-Barrado, M.-E.; Hernández-García, I.; et al. Date of Admission during COVID-19 Pandemic Impacted Patient Outcomes in Addition to the Higher Efficacy of Tocilizumab Plus High-Dose Corticosteroid Therapy Compared to Tocilizumab Alone. J. Clin. Med. 2022, 11, 198. https://doi.org/10.3390/jcm11010198
Belhassen-García M, Sánchez-Puente A, Dorado-Díaz P-I, López-Bernús A, Sampedro-Gómez J, Azibeiro-Melchor R, Marcano-Millán E, Rodríguez-Alonso B, Sánchez-Barrado M-E, Hernández-García I, et al. Date of Admission during COVID-19 Pandemic Impacted Patient Outcomes in Addition to the Higher Efficacy of Tocilizumab Plus High-Dose Corticosteroid Therapy Compared to Tocilizumab Alone. Journal of Clinical Medicine. 2022; 11(1):198. https://doi.org/10.3390/jcm11010198
Chicago/Turabian StyleBelhassen-García, Moncef, Antonio Sánchez-Puente, Pedro-Ignacio Dorado-Díaz, Amparo López-Bernús, Jesús Sampedro-Gómez, Raúl Azibeiro-Melchor, Edgard Marcano-Millán, Beatriz Rodríguez-Alonso, María-Elisa Sánchez-Barrado, Ignacio Hernández-García, and et al. 2022. "Date of Admission during COVID-19 Pandemic Impacted Patient Outcomes in Addition to the Higher Efficacy of Tocilizumab Plus High-Dose Corticosteroid Therapy Compared to Tocilizumab Alone" Journal of Clinical Medicine 11, no. 1: 198. https://doi.org/10.3390/jcm11010198






