Periacetabular Tumour Resection under Anterosuperior Iliac Spine Allows Better Alloprosthetic Reconstruction than Above: Bone Contact Matters
Abstract
1. Introduction
2. Materials and Methods
2.1. Criteria of Data Collection
2.2. Classification System of Pelvic Resections Used
2.3. Data Collection
2.4. Statistical Analysis
3. Results
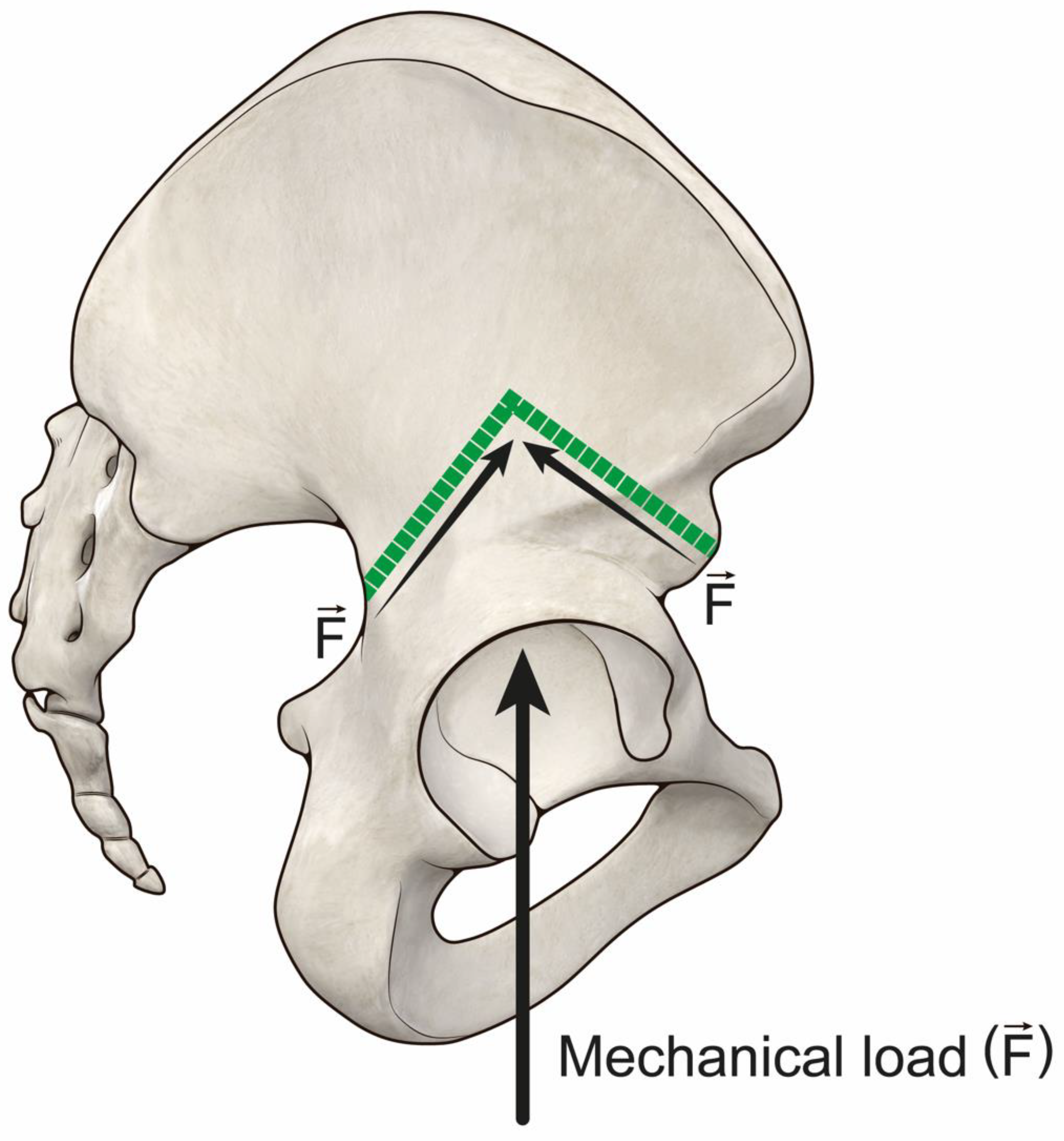
3.1. Impact of Bone-Allograft Contact on the Reconstruction
3.2. Impact of Age on the Reconstruction
3.3. Impact of Artificial Ligament on the Reconstruction
4. Discussion
4.1. Impact of Bone-Allograft Contact on the Reconstruction
4.2. Impact of Age on the Reconstruction
4.3. Impact of Artificial Ligament on the Reconstruction
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mayerson, J.L.; Wooldridge, A.N.; Scharschmidt, T.J. Pelvic Resection: Current Concepts. J. Am. Acad. Orthop. Surg. 2014, 22, 214–222. [Google Scholar] [CrossRef]
- Delloye, C.; Banse, X.; Brichard, B.; Docquier, P.-L.; Cornu, O. Pelvic Reconstruction with a Structural Pelvic Allograft After Resection of a Malignant Bone Tumor. J. Bone Jt. Surg. 2007, 89, 579–587. [Google Scholar] [CrossRef]
- Brown, T.S.; Salib, C.G.; Rose, P.S.; Sim, F.H.; Lewallen, D.G.; Abdel, M.P. Reconstruction of the Hip after Resection of Periacetabular Oncological Lesions: A Systematic Review. Bone Jt. J. 2018, 100-B, 22–30. [Google Scholar] [CrossRef]
- Campanacci, D.; Chacon, S.; Mondanelli, N.; Beltrami, G.; Scoccianti, G.; Caff, G.; Frenos, F.; Capanna, R. Pelvic Massive Allograft Reconstruction after Bone Tumour Resection. Int. Orthop. 2012, 36, 2529–2536. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, P.K.; Aston, W.J.S.; Grimer, R.J.; Abudu, A.; Carter, S.; Blunn, G.; Briggs, T.W.R.; Cannon, S. Peri-Acetabular Resection and Endoprosthetic Reconstruction for Tumours of the Acetabulum. J. Bone Jt. Surg. Br. 2008, 90-B, 1222–1227. [Google Scholar] [CrossRef]
- Donati, D.; Di Bella, C.; Frisoni, T.; Cevolani, L.; DeGroot, H. Alloprosthetic Composite Is a Suitable Reconstruction After Periacetabular Tumor Resection. Clin. Orthop. 2011, 469, 1450–1458. [Google Scholar] [CrossRef]
- Kiatisevi, P.; Sukunthanak, B.; Pakpianpairoj, C.; Liupolvanish, P. Functional Outcome and Complications Following Reconstruction for Harrington Class II and III Periacetabular Metastasis. World J. Surg. Oncol. 2015, 13, 4. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.A.; van de Sande, M.A.J.; Dijkstra, P.D.S. Poor Long-Term Clinical Results of Saddle Prosthesis After Resection of Periacetabular Tumors. Clin. Orthop. 2013, 471, 324–331. [Google Scholar] [CrossRef]
- Takami, M.; Ieguchi, M.; Aono, M.; Hoshi, M.; Takada, J.; Oebisu, N.; Iwai, T. Flail Hip Joint Following Periacetabular Tumor Resection of the Pelvis Using Upper Surface of the Femoral Neck as a Saddle: A Case Report. Oncol. Lett. 2015, 10, 3529–3531. [Google Scholar] [CrossRef][Green Version]
- Langlais, F.; Lambotte, J.C.; Thomazeau, H. Long-Term Results of Hemipelvis Reconstruction with Allografts. Clin. Orthop. 2001, 388, 178–186. [Google Scholar] [CrossRef]
- Ozaki, T.; Hoffmann, C.; Hillmann, A.; Gosheger, G.; Lindner, N.; Winkelmann, W. Implantation of Hemipelvic Prosthesis After Resection of Sarcoma. Clin. Orthop. 2002, 396, 197–205. [Google Scholar] [CrossRef]
- Hillmann, A.; Hoffmann, C.; Gosheger, G.; Rödl, R.; Winkelmann, W.; Ozaki, T. Tumors of the Pelvis: Complications after Reconstruction. Arch. Orthop. Trauma Surg. 2003, 123, 340–344. [Google Scholar] [CrossRef]
- Angelini, A.; Kotrych, D.; Trovarelli, G.; Szafrański, A.; Bohatyrewicz, A.; Ruggieri, P. Analysis of Principles Inspiring Design of Three-Dimensional-Printed Custom-Made Prostheses in Two Referral Centres. Int. Orthop. 2020, 44, 829–837. [Google Scholar] [CrossRef]
- De Paolis, M.; Sambri, A.; Zucchini, R.; Frisoni, T.; Spazzoli, B.; Taddei, F.; Donati, D.M. Custom-Made 3D-Printed Prosthesis in Periacetabular Resections Through a Novel Ileo-Adductor Approach. Orthopedics 2022, 45, e110–e114. [Google Scholar] [CrossRef]
- Enneking, W.F.; Dunham, W.K. Resection and Reconstruction for Primary Neoplasms Involving the Innominate Bone. J. Bone Jt. Surg. 1978, 60, 731–746. [Google Scholar] [CrossRef]
- Enneking, W.F.; Dunham, W.; Gebhardt, M.C.; Malawar, M.; Pritchard, D.J. A System for the Functional Evaluation of Reconstructive Procedures after Surgical Treatment of Tumors of the Musculoskeletal System. In Classic Papers in Orthopaedics; Springer: London, UK, 1993; pp. 241–246. [Google Scholar]
- Kaplan, E.L.; Meier, P. Nonparametric Estimation from Incomplete Observations. J. Am. Stat. Assoc. 1958, 53, 457–481. [Google Scholar] [CrossRef]
- Beadel, G.P.; McLaughlin, C.E.; Wunder, J.S.; Griffin, A.M.; Ferguson, P.C.; Bell, R.S. Outcome in Two Groups of Patients with Allograft-Prosthetic Reconstruction of Pelvic Tumor Defects. Clin. Orthop. 2005, 438, 30–35. [Google Scholar] [CrossRef]
- Guo, W.; Li, D.; Tang, X.; Yang, Y.; Ji, T. Reconstruction with Modular Hemipelvic Prostheses for Periacetabular Tumor. Clin. Orthop. 2007, 461, 180–188. [Google Scholar] [CrossRef]
- Avedian, R.S.; Haydon, R.C.; Peabody, T.D. Multiplanar Osteotomy with Limited Wide Margins: A Tissue Preserving Surgical Technique for High-Grade Bone Sarcomas. Clin. Orthop. 2010, 468, 2754–2764. [Google Scholar] [CrossRef]
- Coccolini, F.; Stahel, P.F.; Montori, G.; Biffl, W.; Horer, T.M.; Catena, F.; Kluger, Y.; Moore, E.E.; Peitzman, A.B.; Ivatury, R.; et al. Pelvic Trauma: WSES Classification and Guidelines. World J. Emerg. Surg. 2017, 12, 5. [Google Scholar] [CrossRef]
- Pool-Goudzwaard, A.; Hoek van Dijke, G.; van Gurp, M.; Mulder, P.; Snijders, C.; Stoeckart, R. Contribution of Pelvic Floor Muscles to Stiffness of the Pelvic Ring. Clin. Biomech. 2004, 19, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Gerbers, J.G.; Jutte, P.C. Hip-Sparing Approach Using Computer Navigation in Periacetabular Chondrosarcoma. Comput. Aided Surg. 2013, 18, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Yi, C.; Zheng, J.; Li, R.; Lan, Y.; He, M.; Lai, J.; Guan, T.; Pang, F.; Mo, Z.; Chen, P.; et al. Preliminary Proposal: A Classification System for Reconstruction with Autologous Femoral Head after Periacetabular Tumors Resection. J. Orthop. Surg. 2021, 16, 119. [Google Scholar] [CrossRef] [PubMed]
- Xiao, C.; Zhou, Y.; Zhang, W.; Luo, Y.; Tu, C. An Acetabular-Preserving Procedure for Pelvic Giant Cell Tumor Involving Partial Acetabulum. World J. Surg. Oncol. 2017, 15, 200. [Google Scholar] [CrossRef] [PubMed]
- Lam, Y.; Yau, R.; Ho, K.W.Y.; Mak, K.; Fong, S.; So, T.Y.C. Is It Possible and Safe to Perform Acetabular-Preserving Resections for Malignant Neoplasms of the Periacetabular Region? Clin. Orthop. 2017, 475, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.; Cai, H.; Kuong, E.; Chui, E.; Siu, Y.C.; Ji, T.; Drstvenšek, I. Surgical Applications of Three-Dimensional Printing in the Pelvis and Acetabulum: From Models and Tools to Implants. Unfallchirurg 2019, 122, 278–285. [Google Scholar] [CrossRef]
- Wong, K.C.; Kumta, S.M.; Geel, N.V.; Demol, J. One-Step Reconstruction with a 3D-Printed, Biomechanically Evaluated Custom Implant after Complex Pelvic Tumor Resection. Comput. Aided Surg. 2015, 20, 14–23. [Google Scholar] [CrossRef]
- Syftestad, G.T.; Urist, M.R. Bone Aging. Clin. Orthop. 1982, 162, 288–297. [Google Scholar] [CrossRef]
- Heck, D.A.; Nakajima, I.; Kelly, P.J.; Chao, E.Y. The Effect of Load Alteration on the Biological and Biomechanical Performance of a Titanium Fiber-Metal Segmental Prosthesis. J. Bone Jt. Surg. 1986, 68, 118–126. [Google Scholar] [CrossRef]
- Eckhoff, D.G.; Turner, A.S.; Aberman, H.M. Effect of Age on Bone Formation around Orthopaedic Implants. Clin. Orthop. 1995, 312, 253–260. [Google Scholar]
- Fujishiro, T.; Nishikawa, T.; Takikawa, S.; Saegusa, Y.; Yoshiya, S.; Kurosaka, M. Reconstruction of the Iliofemoral Ligament with an Artificial Ligament for Recurrent Anterior Dislocation of Total Hip Arthroplasty. J. Arthroplast. 2003, 18, 524–527. [Google Scholar] [CrossRef]
- Aota, S.; Kikuchi, S.; Ohashi, H.; Kitano, N.; Hakozaki, M.; Konno, S. Soft Tissue Reinforcement with a Leeds-Keio Artificial Ligament in Revision Surgery for Dislocated Total Hip Arthroplasty. HIP Int. 2018, 28, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Klein, W.; Jensen, K.-U. Synovitis and Artificial Ligaments. Arthrosc. J. Arthrosc. Relat. Surg. 1992, 8, 116–124. [Google Scholar] [CrossRef]
- Ochen, Y.; Beks, R.B.; Emmink, B.L.; Wittich, P.; van der Velde, D.; Houwert, R.M.; Keizer, J. Surgical Treatment of Acute and Chronic AC Joint Dislocations: Five-Year Experience with Conventional and Modified LARS Fixation by a Single Surgeon. J. Orthop. 2020, 17, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Sinagra, Z.P.; Kop, A.; Pabbruwe, M.; Parry, J.; Clark, G. Foreign Body Reaction Associated with Artificial LARS Ligaments: A Retrieval Study. Orthop. J. Sports Med. 2018, 6, 232596711881160. [Google Scholar] [CrossRef] [PubMed]
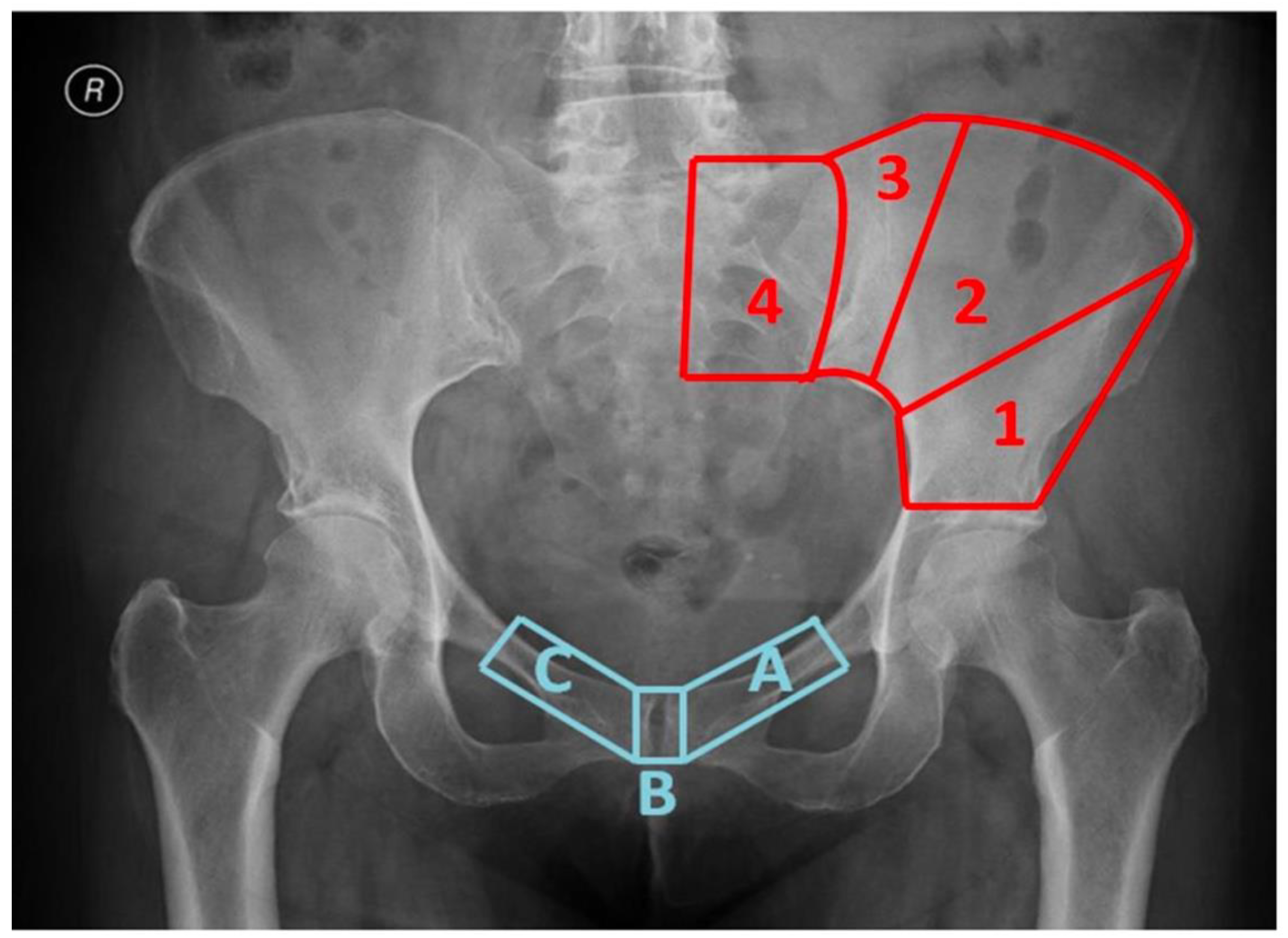





| Region of Proximal Osteotomy Line: | Region of Distal Osteotomy Line: |
|---|---|
| 1Osteotomy inside an area comprised between the roof of the acetabulum and a line that connects the apex of the ischiatic notch and the anterosuperior iliac spine | AOsteotomy involving the ipsilateral pubic ramus |
| 2Osteotomy inside an area comprised between the anterosuperior iliac spine and the lateral margin of posterosuperior iliac spine bone bulk | BOsteotomy involving the pubic symphysis |
| 3Osteotomy inside an area comprised between the lateral margin of posterosuperior iliac spine bone bulk and the sacro-iliac joint (excluded) | COsteotomy involving the contralateral pubic ramus |
| 4Osteotomy medial to the sacro-iliac joint (included) |
| Number | Gender | Age | Diagnosis | Stage | Resection Type | Postoperative Chemotherapy | Patient Follow Up (Months) | Status |
|---|---|---|---|---|---|---|---|---|
| 1 | M | 40 | Chs c | II B | 1A | No | 194 | Ned |
| 2 | M | 32 | Chs c | I B | 1A | No | 187 | Ned |
| 3 | F | 56 | Chs c | I A | 2A | No | 256 | Ned |
| 4 | F | 69 | Chs c | I B | 1A | No | 168 | Ned |
| 5 | M | 34 | GCT | 3 | 1A | No | 286 | Ned |
| 6 | F | 59 | Chs c | I B | 4A | No | 255 | Ned |
| 7 | M | 38 | Chs c | I A | 1A | No | 194 | Dod |
| 8 | M | 62 | Chs c | IB | 1A | No | 110 | Ned |
| 9 | F | 49 | Chs dediff | II B | 4B | Yes | 100 | Dod |
| 10 | F | 25 | Chs c | I B | 3B | No | 277 | Ned |
| 11 | M | 22 | Os L | I A | 2B | No | 258 | Ned |
| 12 | F | 17 | Os H | II B | 4A | Yes | 252 | Ned |
| 13 | F | 22 | Os H | II B | 4A | Yes | 252 | Ned |
| 14 | M | 21 | Chs c | IIB | 4A | No | 295 | Ned |
| 15 | F | 33 | GCT | 3 | 1A | No | 230 | Ned |
| 16 | M | 57 | Chs dediff | II B | 4B | No | 300 | Ned |
| 17 | M | 56 | Chs c | IB | 1A | No | 142 | Ned |
| 18 | M | 60 | Chs c | I B | 4B | No | 146 | Ned |
| 19 | M | 65 | Chs c | II B | 2B | No | 266 | Dod |
| 20 | F | 37 | Chs c | II B | 4B | No | 305 | Ned |
| 21 | M | 51 | Chs c | I B | 1B | No | 138 | Ned |
| 22 | F | 63 | Angios | IIB | 4A | No | 38 | Dod |
| 23 | M | 29 | Tcg | III | 4A | No | 331 | Ned |
| 24 | F | 51 | Os H | II B | 1A | Yes | 65 | Dod |
| 25 | M | 48 | Chs c | I B | 1A | No | 169 | Ned |
| 26 | M | 66 | Chs c | I B | 1A | No | 123 | Ned |
| 27 | F | 24 | Chs dediff | II B | 1A | No | 276 | Ned |
| 28 | F | 43 | Os H | I B | 1A | No | 167 | Ned |
| 29 | M | 59 | GCT | 3 | 1A | No | 250 | Ned |
| 30 | F | 35 | Spindle cell s | II B | 4A | Yes | 184 | Ned |
| 31 | M | 53 | Chs c | I A | 1B | No | 173 | Ned |
| 21 | F | 59 | Chs c | I B | 1A | No | 51 | Ned |
| 33 | M | 18 | Ewing | II | 1A | Yes | 157 | Ned |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruschi, A.; Cevolani, L.; Spazzoli, B.; Focaccia, M.; Pasini, S.; Frisoni, T.; Donati, D.M. Periacetabular Tumour Resection under Anterosuperior Iliac Spine Allows Better Alloprosthetic Reconstruction than Above: Bone Contact Matters. J. Clin. Med. 2022, 11, 4499. https://doi.org/10.3390/jcm11154499
Bruschi A, Cevolani L, Spazzoli B, Focaccia M, Pasini S, Frisoni T, Donati DM. Periacetabular Tumour Resection under Anterosuperior Iliac Spine Allows Better Alloprosthetic Reconstruction than Above: Bone Contact Matters. Journal of Clinical Medicine. 2022; 11(15):4499. https://doi.org/10.3390/jcm11154499
Chicago/Turabian StyleBruschi, Alessandro, Luca Cevolani, Benedetta Spazzoli, Marco Focaccia, Stefano Pasini, Tommaso Frisoni, and Davide Maria Donati. 2022. "Periacetabular Tumour Resection under Anterosuperior Iliac Spine Allows Better Alloprosthetic Reconstruction than Above: Bone Contact Matters" Journal of Clinical Medicine 11, no. 15: 4499. https://doi.org/10.3390/jcm11154499
APA StyleBruschi, A., Cevolani, L., Spazzoli, B., Focaccia, M., Pasini, S., Frisoni, T., & Donati, D. M. (2022). Periacetabular Tumour Resection under Anterosuperior Iliac Spine Allows Better Alloprosthetic Reconstruction than Above: Bone Contact Matters. Journal of Clinical Medicine, 11(15), 4499. https://doi.org/10.3390/jcm11154499






