How to Optimize ECLS Results beyond Ventricular Unloading: From ECMO to CentriMag® eVAD
Abstract
:1. Introduction
2. Materials and Methods
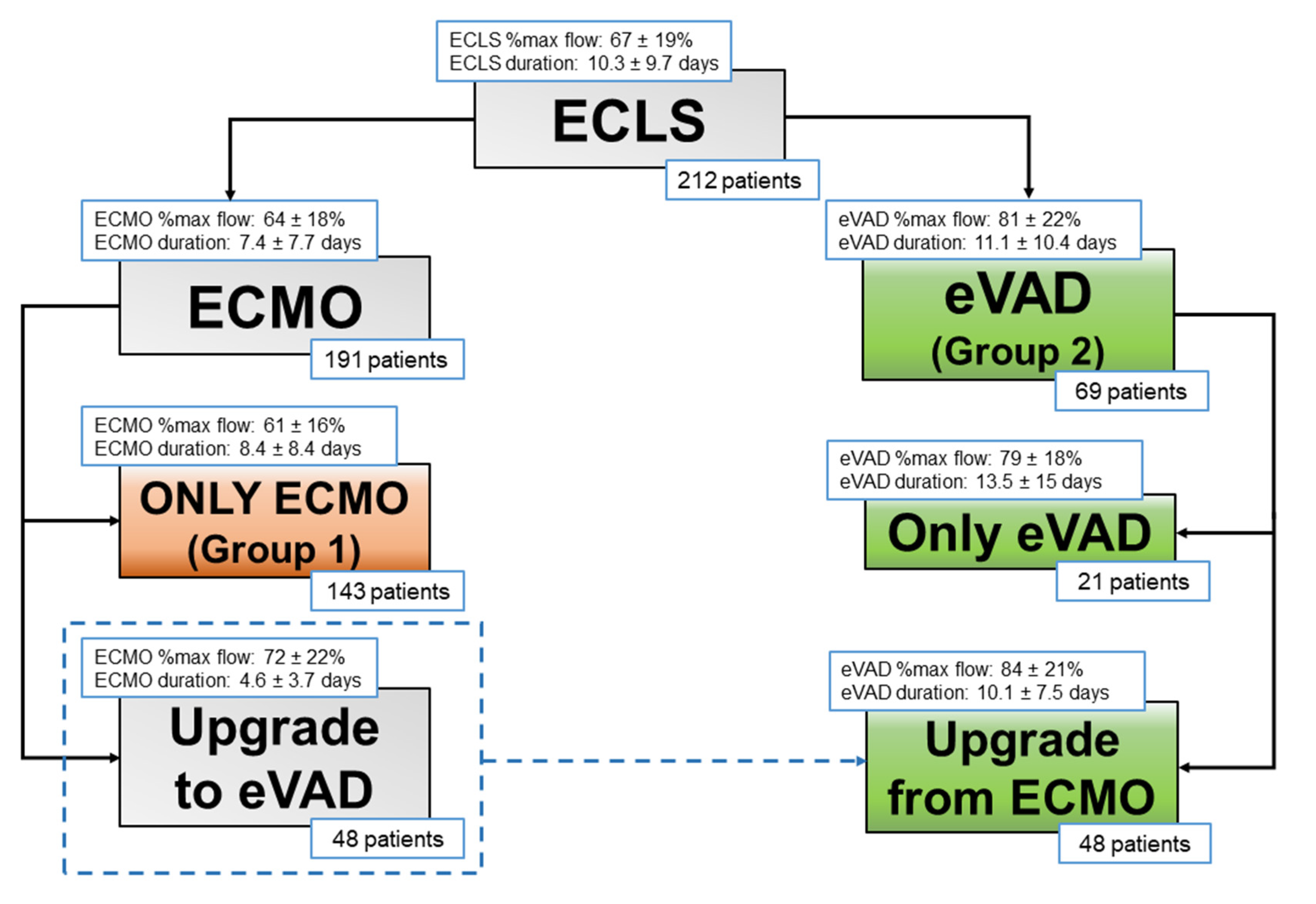
2.1. Study Population
2.2. ECLS Devices
2.3. ECLS Placement
2.4. Management of ECLS
2.5. Anticoagulation Management
2.6. Data Analysis
3. Results
3.1. Patients
3.2. Flow Patterns
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ECMO | extracorporeal membrane oxygenation |
| VA-ECMO | venous-arterial ECMO |
| eVAD | extracorporeal ventricular assistance device |
| ECLS | extracorporeal life support |
| LV | left ventricle |
| IABP | intra-aortic balloon pump |
| BSA | body surface area |
| aPTT | activated partial thromboplastin time |
| TAH | total artificial heart |
References
- ELSO Guidelines for Cardiopulmonary Extracorporeal Life Support Organization, Version 1.4, August 2017, Ann Arbor, MI, USA. Available online: www.elso.org/Resources/Guidelines.aspx (accessed on 30 September 2021).
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Ouweneel, D.M.; Schotborgh, J.V.; Limpens, J.; Sjauw, K.D.; Engström, A.E.; Lagrand, W.K.; Cherpanath, T.G.V.; Driessen, A.H.G.; De Mol, B.A.J.M.; Henriques, J.P.S. Extracorporeal life support during cardiac arrest and cardiogenic shock: A systematic review and meta-analysis. Intensive Care Med. 2016, 42, 1922–1934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donker, D.W.; Brodie, D.; Henriques, J.P.S.; Broomé, M. Left Ventricular Unloading during Veno-Arterial ECMO: A Simulation Study. ASAIO J. 2019, 65, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Briceno, N.; Annamalai, S.K.; Reyelt, L.; Crowley, P.; Qiao, X.; Swain, L.; Pedicini, R.; Foroutanjazi, S.; Jorde, L.; Yesodharan, G.; et al. Left Ventricular Unloading Increases the Coronary Collateral Flow Index Before Reperfusion and Reduces Infarct Size in a Swine Model of Acute Myocardial Infarction. J. Am. Heart Assoc. 2019, 8, e013586. [Google Scholar] [CrossRef]
- Russo, J.J.; Aleksova, N.; Pitcher, I.; Couture, E.; Parlow, S.; Faraz, M.; Visintini, S.; Simard, T.; Di Santo, P.; Mathew, R.; et al. Left Ventricular Unloading During Extracorporeal Membrane Oxygenation in Patients With Cardiogenic Shock. J. Am. Coll. Cardiol. 2019, 73, 654–662. [Google Scholar] [CrossRef]
- Meani, P.; Gelsomino, S.; Natour, E.; Johnson, D.M.; Rocca, H.B.; Pappalardo, F.; Bidar, E.; Makhoul, M.; Raffa, G.; Heuts, S.; et al. Modalities and Effects of Left Ventricle Unloading on Extracorporeal Life support: A Review of the Current Literature. Eur J. Heart Fail. 2017, 19 (Suppl. 2), 84–91. [Google Scholar] [CrossRef] [Green Version]
- Zeriouh, M.; Mohite, P.; Raj, B.; Sabashnikov, A.; Fatullayev, J.; Saez, D.G.; Zych, B.; Ghodsizad, A.; Rahmanian, P.; Choi, Y.-H.; et al. Short-term ventricular assist device as a bridge to decision in cardiogenic shock: Is it a justified strategy? Int. J. Artif. Organs 2016, 39, 114–120. [Google Scholar] [CrossRef]
- Conrad, S.A.; Broman, L.M.; Taccone, F.S.; Lorusso, R.; Malfertheiner, M.V.; Pappalardo, F.; Di Nardo, M.; Belliato, M.; Grazioli, L.; Barbaro, R.P.; et al. The Extracorporeal Life Support Organization Maastricht Treaty for Nomenclature in Extracorporeal Life Support. A Position Paper of the Extracorporeal Life Support Organization. Am. J. Respir. Crit. Care Med. 2018, 198, 447–451. [Google Scholar] [CrossRef]
- Burkhoff, D.; Sayer, G.; Doshi, D.; Uriel, N. Hemodynamics of Mechanical Circulatory Support. J. Am. Coll. Cardiol. 2015, 66, 2663–2674. [Google Scholar] [CrossRef] [Green Version]
- Truby, L.K.; Takeda, K.; Mauro, C.; Yuzefpolskaya, M.; Garan, A.R.; Kirtane, A.J.; Topkara, V.K.; Abrams, D.; Brodie, D.; Colombo, P.C.; et al. Incidence and Implications of Left Ventricular Distention During Venoarterial Extracorporeal Membrane Oxygenation Support. ASAIO J. 2017, 63, 257–265. [Google Scholar] [CrossRef]
- Donker, D.W.; Brodie, D.; Henriques, J.P.S.; Broomé, M. Left ventricular unloading during veno-arterial ECMO: A review of percutaneous and surgical unloading interventions. Perfusion 2019, 34, 98–105. [Google Scholar] [CrossRef] [Green Version]
- Caruso, M.V.; Gramigna, V.; Renzulli, A.; Fragomeni, G. Computational analysis of aortic hemodynamics during total and partial extracorporeal membrane oxygenation and intra-aortic balloon pump support. Acta Bioeng. Biomech. 2016, 18, 3–9. [Google Scholar]
- Lorusso, R. Are two crutches better than one? The ongoing dilemma on the effects and need for left ventricular unloading during veno-arterial extracorporeal membrane oxygenation. Eur. J. Heart Fail. 2017, 19, 413–415. [Google Scholar] [CrossRef]
- Mirabel, M.; Luyt, C.E.; Leprince, P.; Trouillet, J.L.; Léger, P.; Pavie, A.; Chastre, J.; Combes, A. Outcomes, long-term quality of life, and psychologic assessment of fulminant myocarditis patients rescued by mechanical circulatory support. Crit. Care Med. 2011, 39, 1029–1035. [Google Scholar] [CrossRef]
- Kapur, N.K.; Annamalai, S.; Reyelt, L.; Karmiy, S.J.; Razavi, A.A.; Foroutanjazi, S.; Chennojwala, A.; Ishikawa, K. From bedside to bench and back again: Translational studies of mechanical unloading of the left ventricle to promote recovery after acute myocardial infarction. F1000Research 2018, 7, 1852. [Google Scholar] [CrossRef] [Green Version]
- Tarzia, V.; Bortolussi, G.; Bianco, R.; Buratto, E.; Bejko, J.; Carrozzini, M.; De Franceschi, M.; Gregori, D.; Fichera, D.; Zanella, F.; et al. Extracorporeal life support in cardiogenic shock: Impact of acute versus chronic etiology on outcome. J. Thorac. Cardiovasc. Surg. 2015, 150, 333–340. [Google Scholar] [CrossRef] [Green Version]
- Merkle-Storms, J.; Djordjevic, I.; Sabashnikov, A.; Eghbalzadeh, K.; Gkouziouta, A.; Fiane, A.; Stockman, B.; Montalto, A.; Bernhardt, A.; Meyns, B.; et al. Comparative analysis of LVAD patients in regard of ischaemic or idiopathic cardiomyopathy: A propensity-score analysis of EUROMACS data. Int. J. Artif. Organs 2022, 45, 284–291. [Google Scholar] [CrossRef]
- Cevasco, M.; Takayama, H.; Ando, M.; Garan, A.R.; Naka, Y.; Takeda, K. Left ventricular distension and venting strategies for patients on venoarterial extracorporeal membrane oxygenation. J. Thorac. Dis. 2019, 11, 1676–1683. [Google Scholar] [CrossRef]
- Aso, S.; Matsui, H.; Fushimi, K.; Yasunaga, H. The Effect of Intraaortic Balloon Pumping Under Venoarterial Extracorporeal Membrane Oxygenation on Mortality of Cardiogenic Patients: An Analysis Using a Nationwide Inpatient Database. Crit. Care Med. 2016, 44, 1974–1979. [Google Scholar] [CrossRef]
- Bréchot, N.; Demondion, P.; Santi, F.; Lebreton, G.; Pham, T.; Dalakidis, A.; Gambotti, L.; Luyt, C.E.; Schmidt, M.; Hekimian, G.; et al. Intra-aortic balloon pump protects against hydrostatic pulmonary oedema during peripheral venoarterial-extracorporeal membrane oxygenation. Eur. Heart J. Acute Cardiovasc. Care 2018, 7, 62–69. [Google Scholar] [CrossRef]
- Li, Y.; Yan, S.; Gao, S.; Liu, M.; Lou, S.; Liu, G.; Ji, G.; Bao, G. Effect of an intra-aortic balloon pump with venoarterial extracorporeal membrane oxygenation on mortality of patients with cardiogenic shock: A systematic review and meta-analysis. Eur J. Cardiothorac. Surg. 2019, 55, 395–404. [Google Scholar] [CrossRef]
- Vallabhajosyula, S.; O’Horo, J.C.; Antharam, P.; Ananthaneni, S.; Vallabhajosyula, S.; Stulak, J.M.; Eleid, M.F.; Dunlay, S.M.; Gersh, B.J.; Rihal, C.S.; et al. Concomitant Intra-Aortic Balloon Pump Use in Cardiogenic Shock Requiring Veno-Arterial Extracorporeal Membrane Oxygenation. Circ. Cardiovasc. Interv. 2018, 11, e006930. [Google Scholar] [CrossRef]
- Centofanti, P.; Attisani, M.; La Torre, M.; Ricci, D.; Boffini, M.; Baronetto, A.; Simonato, E.; Clerici, A.; Rinaldi, M. Left Ventricular Unloading during Peripheral Extracorporeal Membrane Oxygenator Support: A Bridge To Life In Profound Cardiogenic Shock. J. Extra Corpor. Technol. 2017, 49, 201–205. [Google Scholar]
- Fiedler, A.G.; Dalia, A.; Axtell, A.L.; Ortoleva, J.; Thomas, S.M.; Roy, N.; Villavicencio, M.A.; D’Alessandro, D.A.; Cudemus, G. Impella Placement Guided by Echocardiography Can Be Used as a Strategy to Unload the Left Ventricle During Peripheral Venoarterial Extracorporeal Membrane Oxygenation. J. Cardiothorac. Vasc. Anesth. 2018, 32, 2585–2591. [Google Scholar] [CrossRef]
- Pappalardo, F.; Schulte, C.; Pieri, M.; Schrage, B.; Contri, R.; Soeffker, G.; Greco, T.; Lembo, R.; Müllerleile, K.; Colombo, A.; et al. Concomitant implantation of Impella® on top of veno-arterial extracorporeal membrane oxygenation may improve survival of patients with cardiogenic shock. Eur. J. Heart Fail. 2017, 19, 404–412. [Google Scholar] [CrossRef]
- Pappalardo, F.; Scandroglio, A.M.; Latib, A. Full percutaneous biventricular support with two Impella pumps: The Bi-Pella approach. ESC Heart Fail. 2018, 5, 368–371. [Google Scholar] [CrossRef] [Green Version]
- John, R.; Long, J.W.; Massey, H.T.; Griffith, B.P.; Sun, B.C.; Tector, A.J.; Frazier, O.H.; Joyce, L.D. Outcomes of a multicenter trial of the Levitronix CentriMag ventricular assist system for short-term circulatory support. J. Thorac. Cardiovasc. Surg. 2011, 141, 932–939. [Google Scholar] [CrossRef] [Green Version]
- Takayama, H.; Soni, L.; Kalesan, B.; Truby, L.K.; Ota, T.; Cedola, S.; Khalpey, Z.; Uriel, N.; Colombo, P.; Mancini, D.M.; et al. Bridge-to-decision therapy with a continuous-flow external ventricular assist device in refractory cardiogenic shock of various causes. Circ. Heart Fail. 2015, 7, 799–806. [Google Scholar] [CrossRef] [Green Version]
- De Robertis, F.; Birks, E.J.; Rogers, P.; Dreyfus, G.; Pepper, J.R.; Khaghani, A. Clinical performance with the Levitronix Centrimag short-term ventricular assist device. J. Heart Lung Transplant. 2006, 25, 181–186. [Google Scholar] [CrossRef]
- Chan, C.H.; Pieper, I.L.; Hambly, R.; Radley, G.; Jones, A.; Friedmann, Y.; Hawkins, K.M.; Westaby, S.; Foster, G.; Thornton, C.A. The CentriMag centrifugal blood pump as a benchmark for in vitro testing of hemocompatibility in implantable ventricular assist devices. Artif. Organs 2015, 39, 93–101. [Google Scholar] [CrossRef]


| Overall | Group 1 | Group 2 | p-Value | |
|---|---|---|---|---|
| (n = 212) | (n = 143) | (n = 69) | ||
| Age (years) | 53.75 ± 14.61 | 54.87 ± 15.02 | 51.42 ± 13.54 | 0.108 |
| Male | 162 (76%) | 115 (80%) | 47 (68%) | 0.058 |
| BSA (m2) | 1.88 ± 0.28 | 1.89 ± 0.31 | 1.85 ± 0.21 | 0.258 |
| Etiology | ||||
| Acute coronary syndrome | 92 (43%) | 71 (50%) | 21 (30%) | 0.012 |
| Myocarditis | 12 (6%) | 4 (3%) | 8 (12%) | 0.021 |
| Pulmonary embolism | 5 (2%) | 4 (3%) | 1 (1%) | 0.475 |
| Ischemic cardiomyopathy | 34 (16%) | 20 (14%) | 14 (20%) | 0.318 |
| Dilated cardiomyopathy | 52 (25%) | 27 (19%) | 25 (36%) | 0.010 |
| Congenital heart disease | 2 (1%) | 2 (1%) | 0 (0%) | 0.454 |
| Other | 17 (8%) | 15 (10%) | 2 (3%) | 0.063 |
| Isolated LV failure | 126 (59%) | 91 (64%) | 35 (51%) | 0.073 |
| Biventricular failure | 86 (41%) | 52 (36%) | 34 (49%) | 0.073 |
| Mechanical ventilation | 180 (85%) | 118 (83%) | 62 (90%) | 0.219 |
| Renal failure | 89 (42%) | 62 (43%) | 27 (39%) | 0.656 |
| Continuous hemofiltration | 19 (9%) | 12 (8%) | 7 (10%) | 0.797 |
| Hepatic failure | 36 (17%) | 25 (17%) | 11 (16%) | 0.847 |
| MELD score | 13.29 ± 7.60 | 13.56 ± 7.48 | 12.78 ± 7.84 | 0.490 |
| Number of inotropes | 1.45 ± 1.15 | 1.49 ± 1.12 | 1.37 ± 1.23 | 0.513 |
| Intra-aortic balloon pump | 63 (30%) | 46 (32%) | 17 (25%) | 0.336 |
| Cardio-pulmonary resuscitation | 47 (22%) | 33 (23%) | 14 (20%) | 0.647 |
| Overall | Group 1 | Group 2 | p-Value | |
|---|---|---|---|---|
| (n = 212) | (n = 143) | (n = 69) | ||
| ECMO duration (days) | 7.43 ± 7.72 | 8.37 ± 8.43 | 4.55 ± 3.73 | 0.003 |
| ECMO mean flow/theoretic flow (%) | 64.02 ± 18.33 | 61.21 ± 16.01 | 72.21 ± 22.05 | 0.002 |
| Left eVAD implantation | 35 (17%) | - | 35 (51%) | - |
| Biventricular eVAD implantation | 34 (16%) | - | 34 (49%) | - |
| eVAD duration (days) | 11.12 ± 10.35 | - | 11.12 ± 10.35 | - |
| eVAD mean flow/theoretic flow (%) | 79.49 ± 18.42 | - | 79.49 ± 18.42 | - |
| Total ECLS duration (days) | 10.29 ± 9.66 | 8.37 ± 8.43 | 14.25 ± 10.84 | 0.001 |
| Total ECLS mean flow/theoretic flow (%) | 67.19 ± 18.87 | 61.21 ± 16.01 | 79.49 ± 18.42 | 0.001 |
| Overall | Group 1 | Group 2 | p-Value | |
|---|---|---|---|---|
| (n = 212) | (n = 143) | (n = 69) | ||
| Neurological event | 35 (17%) | 27 (19%) | 9 (13%) | 0.114 |
| Sepsis | 23 (11%) | 14 (10%) | 9 (13%) | 0.471 |
| Renal failure | 150 (71%) | 97 (68%) | 53 (77%) | 0.178 |
| Continuous hemofiltration | 77 (36%) | 45 (31%) | 28 (41%) | 0.057 |
| Hepatic failure | 91 (43%) | 59 (41%) | 32 (46%) | 0.481 |
| ARDS | 23 (11%) | 11 (8%) | 6 (9%) | 0.367 |
| Hemolysis | 52 (25%) | 32 (22%) | 20 (29%) | 0.234 |
| Major bleeding | 31 (15%) | 20 (14%) | 11 (16%) | 0.532 |
| Overall (n = 212) | Partial-Flow ECMO (n = 50) | Medium-Flow ECMO (n = 44) | High-Flow ECMO (n = 49) | High-Flow eVAD (n = 69) | p-Value (All Subgroups Comparisons) | p-Value (High-Flow ECMO vs. High-Flow eVAD) | |
|---|---|---|---|---|---|---|---|
| Mortality during ECLS | 51 (24%) | 4 (8%) | 12 (27%) | 20 (41%) | 15 (22%) | 0.002 | 0.021 |
| In-hospital mortality | 99 (47%) | 16 (32%) | 21 (48%) | 31 (63%) | 31 (45%) | 0.020 | 0.037 |
| Myocardial recovery | 59 (28%) | 31 (62%) | 13 (30%) | 7 (14%) | 8 (12%) | 0.001 | 0.435 |
| Heart transplantation after ECLS | 45 (21%) | 8 (16%) | 11 (25%) | 5 (10%) | 21 (30%) | 0.041 | 0.007 |
| LVAD implantation after ECLS | 36 (17%) | 7 (14%) | 5 (11%) | 5 (10%) | 19 (28%) | 0.039 | 0.017 |
| TAH implantation after ECLS | 4 (2%) | 0 (0%) | 0 (0%) | 4 (8%) | 0 (0%) | 0.004 | 0.028 |
| Conventional surgery after ECLS | 10 (5%) | 3 (6%) | 1 (2%) | 6 (12%) | 0 (0%) | 0.016 | 0.004 |
| Device | LV Unloading | High-Flow Support | Physiological Flow | Long-Term Support |
|---|---|---|---|---|
| ECMO + IABP | ↑ | - | - | - |
| ECMELLA | ↑↑ | ↑ | - | - |
| ECMO + Apical Vent | ↑↑↑ | ↑↑ | - | ↑ |
| BiPELLA | ↑↑ | ↑↑ | ↑↑ | - |
| CentriMag® eVAD | ↑↑↑ | ↑↑↑ | ↑↑↑ | ↑↑↑ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tarzia, V.; Bagozzi, L.; Ponzoni, M.; Bortolussi, G.; Folino, G.; Bianco, R.; Zanella, F.; Bottio, T.; Gerosa, G. How to Optimize ECLS Results beyond Ventricular Unloading: From ECMO to CentriMag® eVAD. J. Clin. Med. 2022, 11, 4605. https://doi.org/10.3390/jcm11154605
Tarzia V, Bagozzi L, Ponzoni M, Bortolussi G, Folino G, Bianco R, Zanella F, Bottio T, Gerosa G. How to Optimize ECLS Results beyond Ventricular Unloading: From ECMO to CentriMag® eVAD. Journal of Clinical Medicine. 2022; 11(15):4605. https://doi.org/10.3390/jcm11154605
Chicago/Turabian StyleTarzia, Vincenzo, Lorenzo Bagozzi, Matteo Ponzoni, Giacomo Bortolussi, Giulio Folino, Roberto Bianco, Fabio Zanella, Tomaso Bottio, and Gino Gerosa. 2022. "How to Optimize ECLS Results beyond Ventricular Unloading: From ECMO to CentriMag® eVAD" Journal of Clinical Medicine 11, no. 15: 4605. https://doi.org/10.3390/jcm11154605
APA StyleTarzia, V., Bagozzi, L., Ponzoni, M., Bortolussi, G., Folino, G., Bianco, R., Zanella, F., Bottio, T., & Gerosa, G. (2022). How to Optimize ECLS Results beyond Ventricular Unloading: From ECMO to CentriMag® eVAD. Journal of Clinical Medicine, 11(15), 4605. https://doi.org/10.3390/jcm11154605






