Evaluation of FIB-4, NFS, APRI and Liver Function Tests as Predictors for SARS-CoV-2 Infection in the Elderly Population: A Matched Case-Control Analysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Background, Design, and Ethics
2.2. Inclusion Criteria, Patient Characteristics, and Variables
2.3. Statistical Analysis
3. Results
3.1. Comparison of Baseline Characteristics
3.2. Laboratory Profile Analysis
3.3. Risk Factor Analysis
4. Discussion
4.1. Important Findings
4.2. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Timircan, M.; Bratosin, F.; Vidican, I.; Suciu, O.; Turaiche, M.; Bota, A.V.; Mitrescu, S.; Marincu, I. Coping Strategies and Health-Related Quality of Life in Pregnant Women with SARS-CoV-2 Infection. Medicina 2021, 57, 1113. [Google Scholar] [CrossRef] [PubMed]
- Popescu, A.; Craina, M.; Pantea, S.; Pirvu, C.; Radu, D.; Marincu, I.; Bratosin, F.; Bogdan, I.; Hosin, S.; Citu, C.; et al. COVID-19 Pandemic Impact on Surgical Treatment Methods for Early-Stage Cervical Cancer: A Population-Based Study in Romania. Healthcare 2022, 10, 639. [Google Scholar] [CrossRef] [PubMed]
- Timircan, M.; Bratosin, F.; Vidican, I.; Suciu, O.; Tirnea, L.; Avram, V.; Marincu, I. Exploring Pregnancy Outcomes Associated with SARS-CoV-2 Infection. Medicina 2021, 57, 796. [Google Scholar] [CrossRef]
- Popescu, A.; Craina, M.; Pantea, S.; Pirvu, C.; Chiriac, V.D.; Marincu, I.; Bratosin, F.; Bogdan, I.; Hosin, S.; Citu, C.; et al. COVID-19 Pandemic Effects on Cervical Cancer Diagnosis and Management: A Population-Based Study in Romania. Diagnostics 2022, 12, 907. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Xiang, R.; Huo, S.; Zhou, Y.; Jiang, S.; Wang, Q.; Yu, F. Molecular mechanism of interaction between SARS-CoV-2 and host cells and in-terventional therapy. Signal Transduct. Target. Ther. 2021, 6, 233. [Google Scholar] [CrossRef]
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int (accessed on 21 April 2022).
- Yadav, D.K.; Singh, A.; Zhang, Q.; Bai, X.; Zhang, W.; Yadav, R.K.; Singh, A.; Zhiwei, L.; Adhikari, V.P.; Liang, T. Involvement of liver in COVID-19: Systematic review and meta-analysis. Gut 2021, 70, 807–809. [Google Scholar] [CrossRef]
- Marincu, I.; Bratosin, F.; Curescu, M.; Suciu, O.; Turaiche, M.; Cerbu, B.; Vidican, I. Direct-Acting Antiviral Use for Genotype 1b Hepatitis C Patients with Associated Hematological Disorders from Romania. Medicina 2021, 57, 986. [Google Scholar] [CrossRef]
- Cabibbo, G.; Rizzo, G.E.M.; Stornello, C.; Craxì, A. SARS-CoV-2 infection in patients with a normal or abnormal liver. J. Viral Hepat. 2021, 28, 4–11. [Google Scholar] [CrossRef]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef]
- Docherty, A.B.; Harrison, E.M.; Green, C.A.; Hardwick, H.E.; Pius, R.; Norman, L.; Holden, K.A.; Read, J.M.; Dondelinger, F.; Carson, G.; et al. Features of 20 133 UK patients in hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: Prospective observational cohort study. BMJ 2020, 369, m1985. [Google Scholar] [CrossRef]
- Zhao, W.; Li, H.; Li, J.; Xu, B.; Xu, J. The mechanism of multiple organ dysfunction syndrome in patients with COVID-19. J. Med. Virol. 2022, 94, 1886–1892. [Google Scholar] [CrossRef]
- Folescu, R.; Levai, C.M.; Grigoras, M.L.; Arghirescu, T.S.; Talpos, I.C.; Gindac, C.M.; Zamfir, C.L.; Poroch, V.; Anghel, M.D. Expression and significance of Ki-67 in lung cancer. Rom. J. Morphol. Embryol. 2018, 59, 227–233. [Google Scholar]
- Grigoras, M.L.; Arghirescu, T.S.; Folescu, R.; Talpoş, I.C.; Gîndac, C.M.; Zamfir, C.L.; Cornianu, M.; Anghel, M.D.; Levai, C.M. Expression of E-cadherin in lung carcinoma, other than those with small cells (NSCLC). Rom. J. Morphol. Embryol. 2017, 58, 1317–1325. [Google Scholar] [PubMed]
- Wong, D.W.L.; Klinkhammer, B.M.; Djudjaj, S.; Villwock, S.; Timm, M.; Buhl, E.; Wucherpfennig, S.; Cacchi, C.; Braunschweig, T.; Knüchel-Clarke, R.; et al. Multisystemic Cellular Tropism of SARS-CoV-2 in Autopsies of COVID-19 Patients. Cells 2021, 10, 1900. [Google Scholar] [CrossRef]
- Yu, D.; Du, Q.; Yan, S.; Guo, X.-G.; He, Y.; Zhu, G.; Zhao, K.; Ouyang, S. Liver injury in COVID-19: Clinical features and treatment management. Virol. J. 2021, 18, 121. [Google Scholar] [CrossRef] [PubMed]
- Citu, I.M.; Citu, C.; Gorun, F.; Neamtu, R.; Motoc, A.; Burlea, B.; Rosca, O.; Bratosin, F.; Hosin, S.; Manolescu, D.; et al. Using the NYHA Classification as Forecasting Tool for Hospital Readmission and Mortality in Heart Failure Patients with COVID-19. J. Clin. Med. 2022, 11, 1382. [Google Scholar] [CrossRef]
- Pilut, C.N.; Citu, C.; Gorun, F.; Bratosin, F.; Gorun, O.M.; Burlea, B.; Citu, I.M.; Grigoras, M.L.; Manolescu, D.; Gluhovschi, A. The Utility of Laboratory Parameters for Cardiac Inflammation in Heart Failure Patients Hospitalized with SARS-CoV-2 Infection. Diagnostics 2022, 12, 824. [Google Scholar] [CrossRef]
- Cerbu, B.; Pantea, S.; Bratosin, F.; Vidican, I.; Turaiche, M.; Frent, S.; Borsi, E.; Marincu, I. Liver Impairment and Hematological Changes in Patients with Chronic Hepatitis C and COVID-19: A Retrospective Study after One Year of Pandemic. Medicina 2021, 57, 597. [Google Scholar] [CrossRef]
- Pazgan-Simon, M.; Ser-afińska, S.; Kukla, M.; Kucharska, M.; Zuwała-Jagiełło, J.; Buczyńska, I.; Zielińska, K.; Simon, K. Liver Injury in Patients with COVID-19 without Underlying Liver Disease. J. Clin. Med. 2022, 11, 308. [Google Scholar] [CrossRef] [PubMed]
- Cerbu, B.; Grigoras, M.L.; Bratosin, F.; Bogdan, I.; Citu, C.; Bota, A.V.; Timircan, M.; Bratu, M.L.; Levai, M.C.; Marincu, I. Laboratory Profile of COVID-19 Patients with Hepatitis C-Related Liver Cirrhosis. J. Clin. Med. 2022, 11, 652. [Google Scholar] [CrossRef]
- Zghal, M.; Bouhamed, M.; Mellouli, M.; Triki, M.; Kallel, R.; Ayedi, L.; Boudawara, T.S.; Makni, S. Liver injury in COVID-19: Pathological findings. Pan Afr. Med. J. 2022, 41, 56. [Google Scholar] [CrossRef] [PubMed]
- Marincu, I.; Bratosin, F.; Vidican, I.; Bostanaru, A.-C.; Frent, S.; Cerbu, B.; Turaiche, M.; Tirnea, L.; Timircan, M. Predictive Value of Comorbid Conditions for COVID-19 Mortality. J. Clin. Med. 2021, 10, 2652. [Google Scholar] [CrossRef] [PubMed]
- Yanez, N.D.; Weiss, N.S.; Romand, J.A.; Treggiari, M.M. COVID-19 mortality risk for older men and women. BMC Public Health 2020, 20, 1742. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Yuan, W.; Shao, J.; Huang, K.; Wang, Q.; Yao, S.; Lu, W.; Liu, L.; Fu, T. Risk factors for mortality in patients over 70 years old with COVID-19 in Wuhan at the early break: Retrospective case series. BMC Infect. Dis. 2021, 21, 821. [Google Scholar] [CrossRef] [PubMed]
- Gkoufa, A.; Maneta, E.; Ntoumas, G.N.; Georgakopoulou, V.E.; Mantelou, A.; Kokkoris, S.; Routsi, C. Elderly adults with COVID-19 admitted to intensive care unit: A narrative review. World J. Crit. Care Med. 2021, 10, 278–289. [Google Scholar] [CrossRef]
- Citu, C.; Gorun, F.; Motoc, A.; Ratiu, A.; Gorun, O.M.; Burlea, B.; Neagoe, O.; Citu, I.M.; Rosca, O.; Bratosin, F.; et al. Evaluation and Comparison of the Predictive Value of 4C Mortality Score, NEWS, and CURB-65 in Poor Outcomes in COVID-19 Patients: A Retrospective Study from a Single Center in Romania. Diagnostics 2022, 12, 703. [Google Scholar] [CrossRef]
- Singhal, S.; Kumar, P.; Singh, S.; Saha, S.; Dey, A.B. Clinical features and outcomes of COVID-19 in older adults: A systematic review and meta-analysis. BMC Geriatr. 2021, 21, 321. [Google Scholar] [CrossRef]
- Limpawattana, P.; Phungoen, P.; Mitsungnern, T.; Laosuangkoon, W.; Tansangworn, N. Atypical presentations of older adults at the emergency department and associated factors. Arch. Gerontol. Geriatr. 2016, 62, 97–102. [Google Scholar] [CrossRef]
- Dehelean, L.; Romosan, A.M.; Papava, I.; Bredicean, C.A.; Dumitrascu, V.; Ursoniu, S.; Romosan, R.-S. Prolactin response to antipsychotics: An inpatient study. PLoS ONE 2020, 15, e0228648. [Google Scholar] [CrossRef]
- Dehelean, L.; Marinescu, I.; Stovicek, P.O.; Andor, M. Cardiovascular anomalies and evolutionary risk factors in schizophrenia—Multifactorial approach. Rom. J. Morphol. Embryol. 2019, 60, 1105–1113. [Google Scholar]
- Dehelean, L.; Papava, I.; Musat, M.I.; Bondrescu, M.; Bratosin, F.; Bucatos, B.O.; Bortun, A.-M.C.; Mager, D.V.; Romosan, R.S.; Romosan, A.-M.; et al. Coping Strategies and Stress Related Disorders in Patients with COVID-19. Brain Sci. 2021, 11, 1287. [Google Scholar] [CrossRef] [PubMed]
- Citu, I.M.; Citu, C.; Gorun, F.; Motoc, A.; Gorun, O.M.; Burlea, B.; Bratosin, F.; Tudorache, E.; Margan, M.-M.; Hosin, S.; et al. Determinants of COVID-19 Vaccination Hesitancy among Romanian Pregnant Women. Vaccines 2022, 10, 275. [Google Scholar] [CrossRef] [PubMed]
- Götz, G.; Herold, D.; Klotz, P.-A.; Schäfer, J.T. Efficiency in COVID-19 Vaccination Campaigns—A Comparison across Germany’s Federal States. Vaccines 2021, 9, 788. [Google Scholar] [CrossRef]
- Citu, I.M.; Citu, C.; Gorun, F.; Sas, I.; Bratosin, F.; Motoc, A.; Burlea, B.; Rosca, O.; Malita, D.; Gorun, O.M. The Risk of Spontaneous Abortion Does Not Increase Following First Trimester mRNA COVID-19 Vaccination. J. Clin. Med. 2022, 11, 1698. [Google Scholar] [CrossRef]
- Ritchie, H.; Mathieu, E.; Rodés-Guirao, L.; Appel, C.; Giattino, C.; Ortiz-Ospina, E.; Hasell, J.; Macdonald, B.; Beltekian, D.; Roser, M. Coronavirus Pandemic (COVID-19). 2020. Available online: https://ourworldindata.org/coronavirus (accessed on 12 April 2022).
- Citu, I.M.; Citu, C.; Gorun, F.; Sas, I.; Tomescu, L.; Neamtu, R.; Motoc, A.; Gorun, O.M.; Burlea, B.; Bratosin, F.; et al. Immunogenicity Following Administration of BNT162b2 and Ad26.COV2.S COVID-19 Vaccines in the Pregnant Population during the Third Trimester. Viruses 2022, 14, 307. [Google Scholar] [CrossRef] [PubMed]
- Marincu, I.; Citu, C.; Bratosin, F.; Bogdan, I.; Timircan, M.; Gurban, C.V.; Bota, A.V.; Braescu, L.; Grigoras, M.L. Clinical Characteristics and Outcomes of COVID-19 Hospitalized Patients: A Comparison between Complete mRNA Vaccination Profile and Natural Immunity. J. Pers. Med. 2022, 12, 259. [Google Scholar] [CrossRef]
- Accorsi, E.K.; Britton, A.; Fleming-Dutra, K.E.; Smith, Z.R.; Shang, N.; Derado, G.; Miller, J.; Schrag, S.J.; Verani, J.R. Association between 3 Doses of mRNA COVID-19 Vaccine and Symptomatic Infection Caused by the SARS-CoV-2 Omicron and Delta Variants. JAMA 2022, 327, 639–651. [Google Scholar] [CrossRef]
- Franssen, T.; Stijnen, M.; Hamers, F.; Schneider, F. Age differences in demographic, social and health-related factors associated with loneliness across the adult life span (19–65 years): A cross-sectional study in the Netherlands. BMC Public Health 2020, 20, 1118. [Google Scholar] [CrossRef]
- Shinan-Altman, S.; Werner, P. Subjective Age and Its Correlates Among Middle-Aged and Older Adults. Int. J. Aging Hum. Dev. 2019, 88, 3–21. [Google Scholar] [CrossRef]
- Vallet-Pichard, A.; Mallet, V.; Nalpas, B.; Verkarre, V.; Nalpas, A.; Dhalluin-Venier, V.; Fontaine, H.; Pol, S. FIB-4: An inexpensive and accurate marker of fibrosis in HCV infection. comparison with liver biopsy and fibrotest. Hepatology 2007, 46, 32–36. [Google Scholar] [CrossRef]
- Treeprasertsuk, S.; Björnsson, E.; Enders, F.; Suwanwalaikorn, S.; Lindor, K.D. NAFLD fibrosis score: A prognostic predictor for mortality and liver complications among NAFLD patients. World J. Gastroenterol. 2013, 19, 1219–1229. [Google Scholar] [CrossRef]
- De Matteis, C.; Cariello, M.; Graziano, G.; Battaglia, S.; Suppressa, P.; Piazzolla, G.; Sabbà, C.; Moschetta, A. AST to Platelet Ratio Index (APRI) is an easy-to-use predictor score for cardiovascular risk in metabolic subjects. Sci. Rep. 2021, 11, 14834. [Google Scholar] [CrossRef] [PubMed]
- Statista. Romania: Age Structure from 2011 to 2021. Available online: https://www.statista.com/statistics/373125/age-structure-in-romania/ (accessed on 8 August 2022).
- Peckham, H.; de Gruijter, N.M.; Raine, C.; Radziszewska, A.; Ciurtin, C.; Wedderburn, L.R.; Rosser, E.C.; Webb, K.; Deakin, C.T. Male sex identified by global COVID-19 meta-analysis as a risk factor for death and ITU admission. Nat. Commun. 2020, 11, 6317. [Google Scholar] [CrossRef]
- Liu, M.; Mei, K.; Tan, Z.; Huang, S.; Liu, F.; Deng, C.; Ma, J.; Yu, P.; Liu, X. Liver Fibrosis Scores and Hospitalization, Mechanical Ventilation, Severity, and Death in Patients with COVID-19: A Systematic Review and Dose-Response Meta-Analysis. Can. J. Gastroenterol. Hepatol. 2022, 2022, 7235860. [Google Scholar] [CrossRef] [PubMed]
- Yao, R.; Zhu, L.; Wang, J.; Liu, J.; Xue, R.; Xue, L.; Liu, L.; Li, C.; Zhao, H.; Cheng, J.; et al. Risk of severe illness of COVID-19 patients with NAFLD and increased NAFLD fibrosis scores. J. Clin. Lab. Anal. 2021, 35, e23880. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.W.; Jin, H.Y.; Yon, D.K.; Effenberger, M.; Shin, Y.H.; Kim, S.Y.; Yang, J.M.; Kim, M.S.; Koyanagi, A.; Jacob, L.; et al. Non-alcoholic Fatty Liver Disease and COVID-19 Susceptibility and Outcomes: A Korean Nationwide Cohort. J. Korean Med. Sci. 2021, 36, e291. [Google Scholar] [CrossRef]
- Yuki, K.; Fujiogi, M.; Koutsogiannaki, S. COVID-19 pathophysiology: A review. Clin. Immunol. 2020, 215, 108427. [Google Scholar] [CrossRef]
- Ali, F.E.M.; Mohammedsaleh, Z.M.; Ali, M.M.; Ghogar, O.M. Impact of cytokine storm and systemic inflammation on liver impairment patients infected by SARS-CoV-2: Prospective therapeutic challenges. World J. Gastroenterol. 2021, 27, 1531–1552. [Google Scholar] [CrossRef]
- Kang, S.J.; Jung, S.I. Age-Related Morbidity and Mortality among Patients with COVID-19. Infect. Chemother. 2020, 52, 154–164. [Google Scholar] [CrossRef]
- Fruh, S.M. Obesity: Risk factors, complications, and strategies for sustainable long-term weight management. J. Am. Assoc. Nurse Pract. 2017, 29 (Suppl. S1), S3–S14. [Google Scholar] [CrossRef]
- Fan, G.; Song, H.; Yip, S.; Zhang, T.; He, D. Impact of low vaccine coverage on the resurgence of COVID-19 in Central and Eastern Europe. One Health 2022, 14, 100402. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, J.V.; Wyka, K.; White, T.M.; Picchio, C.A.; Rabin, K.; Ratzan, S.C.; Leigh, J.P.; Hu, J.; El-Mohandes, A. Revisiting COVID-19 vaccine hesitancy around the world using data from 23 countries in 2021. Nat. Commun. 2022, 13, 3801. [Google Scholar] [CrossRef] [PubMed]



| Variables * | Age < 65 (n = 316) | Age ≥ 65 (n = 316) | p-Value |
|---|---|---|---|
| Background data | |||
| Age, years (mean ± SD) | 58.0 ± 11.8 | 71.4 ± 9.2 | <0.001 |
| BMI, kg/m2 (mean ± SD) | 24.4 ± 4.1 | 24.8 ± 4.0 | 0.214 |
| Gender (men) | 172 (54.4%) | 172 (54.4%) | 1 |
| Area of residence (urban) | 187 (59.2%) | 169 (53.5%) | 0.148 |
| Smoking | 114 (36.1%) | 95 (30.1%) | 0.108 |
| Chronic alcohol use | 24 (7.6%) | 29 (9.2%) | 0.473 |
| Complete COVID-19 Vaccination | 7 (2.2%) | 13 (4.1%) | 0.172 |
| Hepatitis B vaccine | 11 (3.5%) | 6 (1.9%) | 0.218 |
| Comorbidities | |||
| Malignancy | 18 (5.7%) | 25 (7.9%) | 0.268 |
| Chronic lung disease | 28 (8.9%) | 39 (12.3%) | 0.155 |
| Cardiovascular disease | 107 (33.9%) | 132 (41.8%) | 0.040 |
| Cerebrovascular disease | 24 (7.6%) | 49 (15.5%) | 0.001 |
| Diabetes mellitus | 41 (13.0%) | 48 (15.2%) | 0.423 |
| Autoimmune disease | 13 (4.1%) | 15 (4.7%) | 0.699 |
| Chronic kidney disease | 16 (5.1%) | 21 (6.6%) | 0.396 |
| Digestive and liver disease ** | 23 (7.3%) | 30 (9.5%) | 0.315 |
| CCI score (≥2) | 76 (24.1%) | 107 (33.9%) | 0.006 |
| Oxygen supplementation | <0.001 | ||
| No supplementation | 35 (11.1%) | 12 (3.8%) | |
| Non-invasive ventilation | 241 (76.3%) | 248 (78.5%) | |
| Invasive ventilation | 40 (12.7%) | 56 (17.7%) | |
| COVID-19 severity | 0.016 | ||
| Mild | 106 (33.5%) | 82 (25.9%) | |
| Moderate | 121 (38.3%) | 113 (35.8%) | |
| Severe | 89 (28.2%) | 121 (38.3%) | |
| Disease outcomes | |||
| Days of hospitalization (mean ± SD) | 11 ± 6.6 | 19 ± 8.3 | <0.001 |
| ICU admission | 40 (12.7%) | 72 (22.8%) | <0.001 |
| In-hospital mortality | 28 (8.9%) | 54 (17.1%) | 0.002 |
| Variables * | Normal Range | Age < 65 (n = 316) | Age ≥ 65 (n = 316) | p-Value |
|---|---|---|---|---|
| RBC (millions/mm3) | 4.35–5.65 | 61 (19.3%) | 108 (34.2%) | <0.001 |
| WBC (thousands/mm3) | 4.5–11.0 | 72 (22.8%) | 121 (38.3%) | <0.001 |
| Hemoglobin (g/dL) | 13.0–17.0 | 54 (17.1%) | 83 (26.3%) | 0.005 |
| Hematocrit (%) | 36–48 | 39 (12.3%) | 67 (21.2%) | 0.002 |
| Platelets (thousands/mm3) | 150–450 | 41 (13.0%) | 53 (16.8%) | 0.179 |
| Ferritin (ng/mL) | 20–250 | 48 (15.2%) | 66 (20.9%) | 0.062 |
| ESR (mm/h) | 0–22 | 124 (39.2%) | 149 (47.2%) | 0.044 |
| CRP (mg/L) | 0–10 | 145 (45.9%) | 166 (52.5%) | 0.094 |
| Fibrinogen (g/L) | 2–4 | 169 (53.5%) | 180 (57.0%) | 0.378 |
| Procalcitonin (ug/L) | 0–0.25 | 53 (16.8%) | 92 (29.1%) | <0.001 |
| D-dimers (ng/mL) | <250 | 27 (8.5%) | 44 (13.9%) | 0.032 |
| IL-6 (pg/mL) | 0–16 | 52 (16.5%) | 65 (20.6%) | 0.183 |
| Creatinine (µmol/L) | 0.74–1.35 | 30 (9.5%) | 68 (21.5%) | <0.001 |
| Variables * | Normal Range | Age < 65 (n = 316) | Age ≥ 65 (n = 316) | p-Value |
|---|---|---|---|---|
| Fasting glucose (mmol/L) | 60–125 | 39.2% | 48.4% | 0.020 |
| ALT (U/L) | 7–35 | 41.5% | 56.3% | <0.001 |
| AST (U/L) | 10–40 | 40.2% | 57.6% | <0.001 |
| ALP (U/L) | 40–130 | 34.5% | 52.8% | <0.001 |
| Serum albumin (g/dL) | 3.4–5.4 | 31.3% | 36.1% | 0.206 |
| Total proteins (g/dL) | 6.0–8.3 | 26.3% | 30.4% | 0.251 |
| Total bilirubin (g/dL) | 0.3–1.2 | 20.6% | 25.6% | 0.131 |
| GGT (U/L) | 0–30 | 22.8% | 27.2% | 0.198 |
| LDH (U/L) | 140–280 | 27.8% | 38.0% | 0.006 |
| PT (seconds) | 11.0–13.5 | 28.8% | 34.5% | 0.123 |
| APTT (seconds) | 30–40 | 27.2% | 32.3% | 0.163 |
| FIB-4 | 1.45–3.25 | 26.3% | 40.5% | <0.001 |
| NFS | <−1.5 | 15.2% | 31.0% | <0.001 |
| APRI | 0.5–1.5 | 21.2% | 32.9% | <0.001 |
| Variables * | Normal Range | Age < 65 (n = 288) | Age ≥ 65 (n = 262) | p-Value |
|---|---|---|---|---|
| Fasting glucose (mmol/L) | 60–125 | 31.3% | 36.7% | 0.153 |
| ALT (U/L) | 7–35 | 27.8% | 35.1% | 0.048 |
| AST (U/L) | 10–40 | 29.1% | 37.7% | 0.022 |
| ALP (U/L) | 40–130 | 22.8% | 30.4% | 0.030 |
| Serum albumin (g/dL) | 3.4–5.4 | 21.2% | 25.9% | 0.159 |
| Total proteins (g/dL) | 6.0–8.3 | 15.2% | 22.5% | 0.019 |
| Total bilirubin (g/dL) | 0.3–1.2 | 12.3% | 16.8% | 0.363 |
| GGT (U/L) | 0–30 | 15.2% | 20.9% | 0.062 |
| LDH (U/L) | 140–280 | 13.0% | 16.8% | 0.179 |
| PT (seconds) | 11.0–13.5 | 17.1% | 21.8% | 0.131 |
| APTT (seconds) | 30–40 | 16.8% | 20.6% | 0.220 |
| FIB-4 | 1.45–3.25 | 20.9% | 28.5% | 0.026 |
| NFS | <−1.5 | 9.5% | 15.2% | 0.029 |
| APRI | 0.5–1.5 | 10.4% | 17.4% | 0.011 |
| Variables * | Normal Range | Survivors (n = 262) | Deceased (n = 54) | p-Value |
|---|---|---|---|---|
| Fasting glucose (mmol/L) | 60–125 | 96 (36.6%) | 29 (53.7%) | 0.019 |
| ALT (U/L) | 7–35 | 68 (32.4%) | 31 (57.4%) | <0.001 |
| AST (U/L) | 10–40 | 97 (37.0%) | 36 (66.7%) | <0.001 |
| ALP (U/L) | 40–130 | 101 (38.5%) | 30 (55.6%) | 0.020 |
| Serum albumin (g/dL) | 3.4–5.4 | 64 (24.4%) | 25 (46.3%) | 0.001 |
| Total proteins (g/dL) | 6.0–8.3 | 67 (25.6%) | 23 (42.6%) | 0.011 |
| Total bilirubin (g/dL) | 0.3–1.2 | 60 (22.9%) | 18 (33.3%) | 0.105 |
| GGT (U/L) | 0–30 | 63 (24.0%) | 20 (37.0%) | 0.048 |
| LDH (U/L) | 140–280 | 75 (28.6%) | 24 (44.4%) | 0.022 |
| PT (seconds) | 11.0–13.5 | 71 (27.1%) | 21 (38.9%) | 0.082 |
| APTT (seconds) | 30–40 | 63 (24.0%) | 19 (35.2%) | 0.089 |
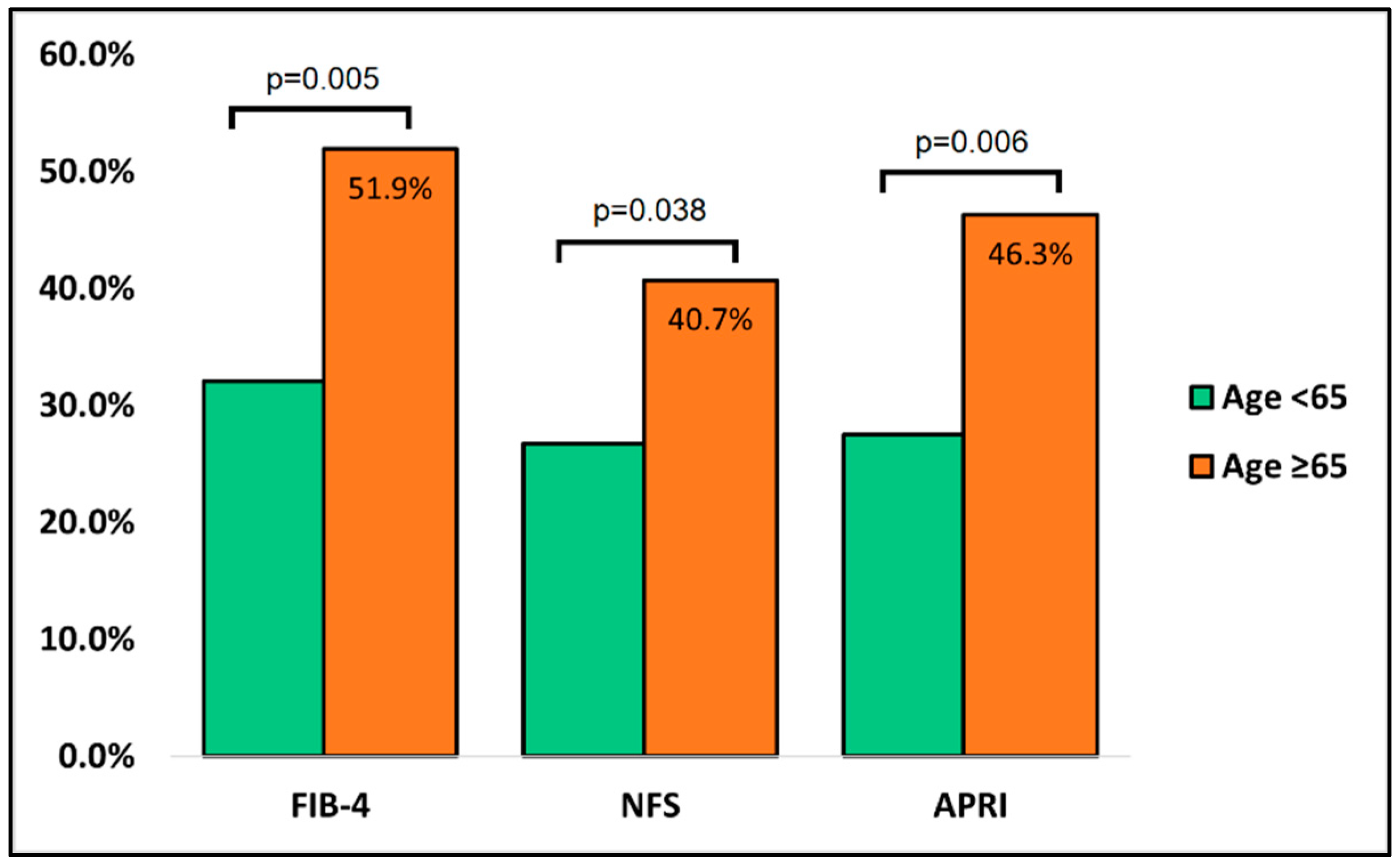
| FIB-4 | 1.45–3.25 | 84 (32.1%) | 28 (51.9%) | 0.005 |
| NFS | <−1.5 | 70 (26.7%) | 22 (40.7%) | 0.038 |
| APRI | 0.5–1.5 | 72 (27.5%) | 25 (46.3%) | 0.006 |
| Risk Factors | OR | 95% CI | p-Value |
|---|---|---|---|
| ALP (U/L) | 1.26 | 1.03–1.84 | 0.033 |
| LDH (U/L) | 1.68 | 1.22–2.97 | 0.001 |
| AST (U/L) | 1.98 | 1.49–3.15 | 0.001 |
| ALT (U/L) | 2.34 | 1.52–3.66 | <0.001 |
| APRI > 1.5 | 2.69 | 1.65–4.07 | <0.001 |
| NFS > 1.5 | 3.05 | 1.83–4.61 | <0.001 |
| FIB-4 > 3.25 | 3.13 | 1.95–4.86 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grigoras, M.L.; Citu, I.M.; Citu, C.; Chiriac, V.D.; Gorun, F.; Levai, M.C.; Manolescu, D.; Rosca, O.; Bratosin, F.; Gurumurthy, S.; et al. Evaluation of FIB-4, NFS, APRI and Liver Function Tests as Predictors for SARS-CoV-2 Infection in the Elderly Population: A Matched Case-Control Analysis. J. Clin. Med. 2022, 11, 5149. https://doi.org/10.3390/jcm11175149
Grigoras ML, Citu IM, Citu C, Chiriac VD, Gorun F, Levai MC, Manolescu D, Rosca O, Bratosin F, Gurumurthy S, et al. Evaluation of FIB-4, NFS, APRI and Liver Function Tests as Predictors for SARS-CoV-2 Infection in the Elderly Population: A Matched Case-Control Analysis. Journal of Clinical Medicine. 2022; 11(17):5149. https://doi.org/10.3390/jcm11175149
Chicago/Turabian StyleGrigoras, Mirela Loredana, Ioana Mihaela Citu, Cosmin Citu, Veronica Daniela Chiriac, Florin Gorun, Mihaela Codrina Levai, Diana Manolescu, Ovidiu Rosca, Felix Bratosin, Srivathsava Gurumurthy, and et al. 2022. "Evaluation of FIB-4, NFS, APRI and Liver Function Tests as Predictors for SARS-CoV-2 Infection in the Elderly Population: A Matched Case-Control Analysis" Journal of Clinical Medicine 11, no. 17: 5149. https://doi.org/10.3390/jcm11175149
APA StyleGrigoras, M. L., Citu, I. M., Citu, C., Chiriac, V. D., Gorun, F., Levai, M. C., Manolescu, D., Rosca, O., Bratosin, F., Gurumurthy, S., Wulandari, P. H., & Cretu, O. M. (2022). Evaluation of FIB-4, NFS, APRI and Liver Function Tests as Predictors for SARS-CoV-2 Infection in the Elderly Population: A Matched Case-Control Analysis. Journal of Clinical Medicine, 11(17), 5149. https://doi.org/10.3390/jcm11175149









