1. Introduction
The World Health Organization (WHO) identified a cluster of respiratory illness cases with an unknown origin in Wuhan, China, in December 2019 [
1]. The respiratory illness, which was subsequently called ‘Coronavirus Disease 2019’ (COVID-19), spread rapidly to neighboring states and has turned into a worldwide pandemic [
2,
3]. The condition is caused by the RNA virus “severe acute respiratory syndrome coronavirus 2” (SARS-CoV-2) and is spread mostly via respiratory droplets and intimate contact with an infected person [
4,
5]. As of April 2022, WHO has verified around 500 million cases of COVID-19 globally, with more than 6 million fatalities [
6]. Patients with COVID-19 often have abnormal liver function, and some investigations have demonstrated that SARS-CoV-2 is related to liver dysfunction or damage [
7]. There are almost 400,000 chronic liver disease patients in Romania [
8], and doctors should be aware of the danger of liver involvement in COVID-19 patients, that several studies describe, particularly when the patient suffers from a pre-existent chronic liver condition [
9,
10,
11].
SARS is mostly a lung infection, thus being named ‘Severe Acute Respiratory Syndrome’ and ‘SARS atypical pneumonia’. However, various organ dysfunctions have been reported in individuals, including gastrointestinal complaints, poor liver function, lymphadenopathy, and splenic atrophy [
12]. However, most uncommon complications during and after COVID-19 have been reported in patients with multiple comorbidities, impaired immunity, and malignancies such as lung cancer [
13,
14]. These findings are indicative of extensive immunopathology or of SARS-coronavirus extrapulmonary dissemination and replication. Autopsies also suggest that the virus has infected various organs besides the liver [
15]. The pathogenic alterations may be explained by either the virus’s direct cytotoxic impact on the host or by an immunological response caused by the virus. Certain findings indicate that COVID-19 may have an effect on other organs, including variable degrees of liver damage in infected individuals [
16]. Recent research discovered that the SARS-CoV-2 virus binding to ACE 2 (ACE2) on cholangiocytes causes its malfunction, which may result in liver harm through an inflammatory response at the systemic level [
17,
18]. Several hospital-based investigations have documented liver damage in individuals with COVID-19 in the form of increased aspartate aminotransferase (AST) and alanine aminotransferase (ALT) values ranging from 14% to 53% above normal [
19,
20,
21]. Additionally, the presence of modest microvesicular steatosis and lobular and portal activity in the liver biopsy specimens of a deceased COVID-19 patient revealed that SARS-CoV-2 was involved in liver damage [
22].
Furthermore, it has been shown that older persons are more prone to get infected with the SARS-CoV-2 virus [
23]. Early reports from China indicated a rise in the severity of sickness and death among persons aged 65 and older, and similar trends were seen in Europe, with mortality rates as high as 10% among adults aged 70, compared to 1% among young adults [
24,
25]. Compared to younger persons, elderly patients have a greater requirement for intensive care unit (ICU) admission and mechanical ventilation; hence, the SARS-CoV-2 infection increases the death risk among those aged 65 and older [
26,
27]. A subsequent meta-analysis supported these results, observing that almost half of older patients with COVID-19 have a severe infection, one in five are seriously sick, and one in ten will eventually die [
28]. Moreover, older patients with a respiratory infection may appear with exhaustion, anorexia, and delirium in the absence of fever and productive cough, which may result in a delayed diagnosis and contribute to increased mortality in the elderly [
29]. Among other clinical features and complications in this population, it was reported that their recovery from SARS-CoV-2 infection is prolonged, leading to the so-called “long COVID”, irreversible lung damage, and psychological complications from maladaptive stress [
30,
31,
32].
With time passing, COVID-19 becomes a “mature” infection that becomes better understood by its molecular mechanisms and effects on the human body. The development of an efficient vaccine and a strong vaccination campaign among developed countries [
33,
34,
35], with over 4.5 billion people vaccinated worldwide at the time of the study [
36], allowed the populations at risk, such as the elderly, to become less vulnerable to COVID-19 [
37,
38]. However, the multitude of SARS-CoV-2 mutations that occurred allowed the virus to escape some immune mechanisms, determining infection with complications even in triple-vaccinated patients [
39]. Therefore, this research aims to describe the differences in liver test parameters in elderly individuals with COVID-19 in relation to other inflammatory markers and outcomes.
2. Materials and Methods
2.1. Background, Design, and Ethics
This current observational single-center research followed a case-control design of elderly patients hospitalized for SARS-CoV-2 infection. The research was conducted at a tertiary emergency hospital in western Romania, where patients were hospitalized in the COVID-19 unit of the Timisoara Municipal Hospital. Data were collected between 1 March 2020 and 1 March 2022. The sample size and key features were identified via the use of a population-based administrative database of patients who attended the same clinic’s inpatient setting over the study period. Our comprehensive database included patient medical records that were protected by privacy laws and gathered with the patient’s permission. The patient’s demographics, medical history, laboratory profile, and in-hospital treatment were all included in this data. All patients’ baseline characteristics and procedures were recorded in the hospital database and in paper patient records inspected by certified clinicians participating in the current inquiry.
The Ethics Committee of the “Victor Babes” University of Medicine and Pharmacy in Timisoara, Romania, as well as the Ethics Committee of the Timisoara Municipal Hospital, accepted the study protocol. The institutions are governed by the provisions of Article 167 of Law No. 95/2006, Article 28 of Order 904/2006, and the EU Good Clinical Practice Directives 2005/28/EC, the International Conference on Harmonization of Technical Requirements for the Registration of Pharmaceuticals for Human Use (ICH), and the Declaration of Helsinki—Recommendations Guiding Mediation. On 23 December 2021, the present research was accepted with approval number I-32467.
2.2. Inclusion Criteria, Patient Characteristics, and Variables
The inclusion criteria were established for all patients over the age of 65, as a threshold for the age of retirement in Romania, as well as a threshold for old age as considered by multiple medical research studies [
40,
41]. Patients were included if they had a history of hospitalization in our department for SARS-CoV-2 infection, as determined by real-time polymerase chain reaction (RT-PCR). Exclusion criteria accounted for patients who suffered from cirrhosis, insufficient patient profiles in terms of imaging exams and laboratory data, as well as records without patient consent. The collection of data was performed by qualified doctors who volunteered to participate in this study, and the database information was validated against existing patient paper records. From the elderly population hospitalized in the COVID-19 department, we included a total of 316 cases of SARS-CoV-2 infection that were eligible for inclusion. The cohort of elderly patients with COVID-19 validated for study inclusion was compared with 316 adult patients younger than 65 years and a history of SARS-CoV-2 infection matched by gender and body mass index (BMI).
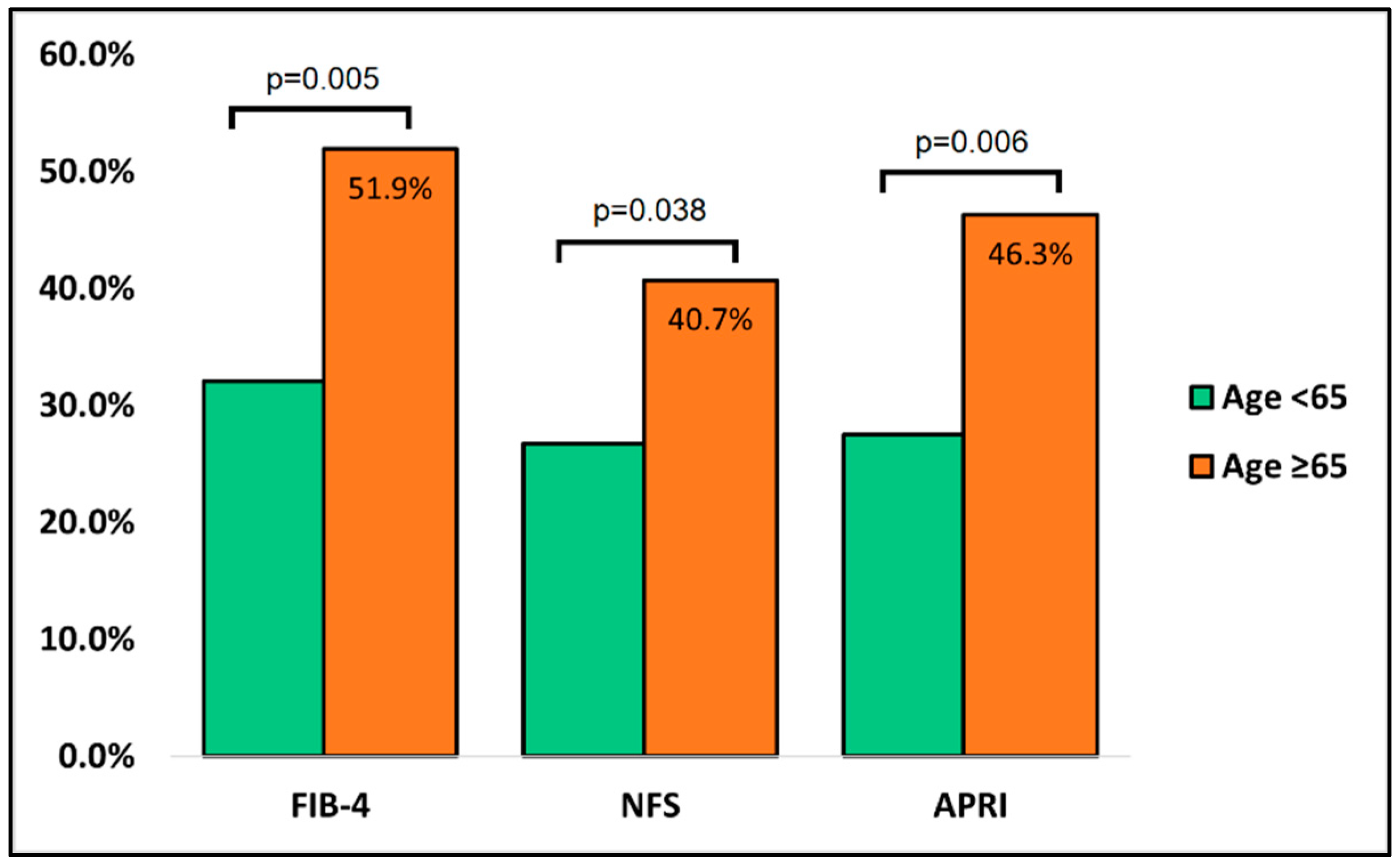
All individuals got a SARS-CoV-2 outpatient examination, having the infection validated by nasopharyngeal RT-PCR for SARS-CoV-2 RNA, in accordance with criteria at the time of the research. The infections were classified as mild, moderate, or severe COVID-19 based on clinical findings and computed tomography (CT) data. Therefore, all patients with pulmonary lesions were classified as mild (30 percent pulmonary damage), moderate (30–60 percent pulmonary damage), or severe (more than 60 percent of lung area damaged). A complete medical history, clinical examination, and other additional studies were assessed at the initial presentation. Representatives of the clinical teams collected anonymized laboratory data on all cases of COVID-19 diagnosed throughout the research period, including the following biological parameters: the red blood cell count, white blood cell count, hemoglobin, hematocrit, platelets, ferritin, erythrocyte sedimentation rate, c-reactive protein, fibrinogen, procalcitonin, d-dimers, interleukin-6, and creatinine. The liver studies comprised a fasting glucose check, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, serum albumin, total proteins, total bilirubin, gamma glutamate transpeptidase, lactate dehydrogenase, prothrombin time, partial thromboplastin time, FIB-4, NFS, and APRI scores.
Other variables analyzed included the background characteristics of patients (age, body mass index, gender), area of residence, smoking status, history of an alcohol use disorder, the status of complete COVID-19 vaccination with three doses, comorbidities (malignancy, chronic lung disease, cardiovascular disease, cerebrovascular disease, diabetes mellitus, autoimmune disease, chronic kidney disease, digestive and liver disease), Charlson Comorbidity Index (CCI), oxygen supplementation, COVID-19 severity, intensive care unit admission, and in-hospital mortality. The Fibrosis-4 (FIB-4), nonalcoholic fatty liver disease (NAFLD), non-invasive fibrosis score (NFS), and AST to platelet ratio index (APRI) scores were reported as markers of liver fibrosis and clinical outcomes for the elderly admitted for SARS-CoV-2 infection and calculated as indicated by their validation studies [
42,
43,
44] using the following formulas:
2.3. Statistical Analysis
The sample size was calculated for a confidence interval of 95%, margin of error 5% and a population proportion of approximately 20% of people older than 65 from the 19 million population size, according to the most recent statistics for age structure in Romania [
45]. The sample size was considered adequate for a calculated number of 246 patients. For statistical analysis, MS EXCEL and IBM SPSS were used. Continuous variables were presented as the mean, standard deviation (SD), or median with interquartile range (IQR). Descriptive statistical analyses were done to obtain the means and standard deviations, and the Student’s
t-test was used to estimate the
p-value. To examine proportional differences, the Chi-square test was performed. The liver markers were included in a multivariate regression analysis that was adjusted for confounding factors (age, comorbidities, and COVID-19 vaccination status), with results expressed as odds ratio (OR) and confidence interval (CI). A
p-value of 0.05 was determined to be statistically significant.