LASSO Regression-Based Diagnosis of Acute ST-Segment Elevation Myocardial Infarction (STEMI) on Electrocardiogram (ECG)
Abstract
:1. Introduction
2. Methods
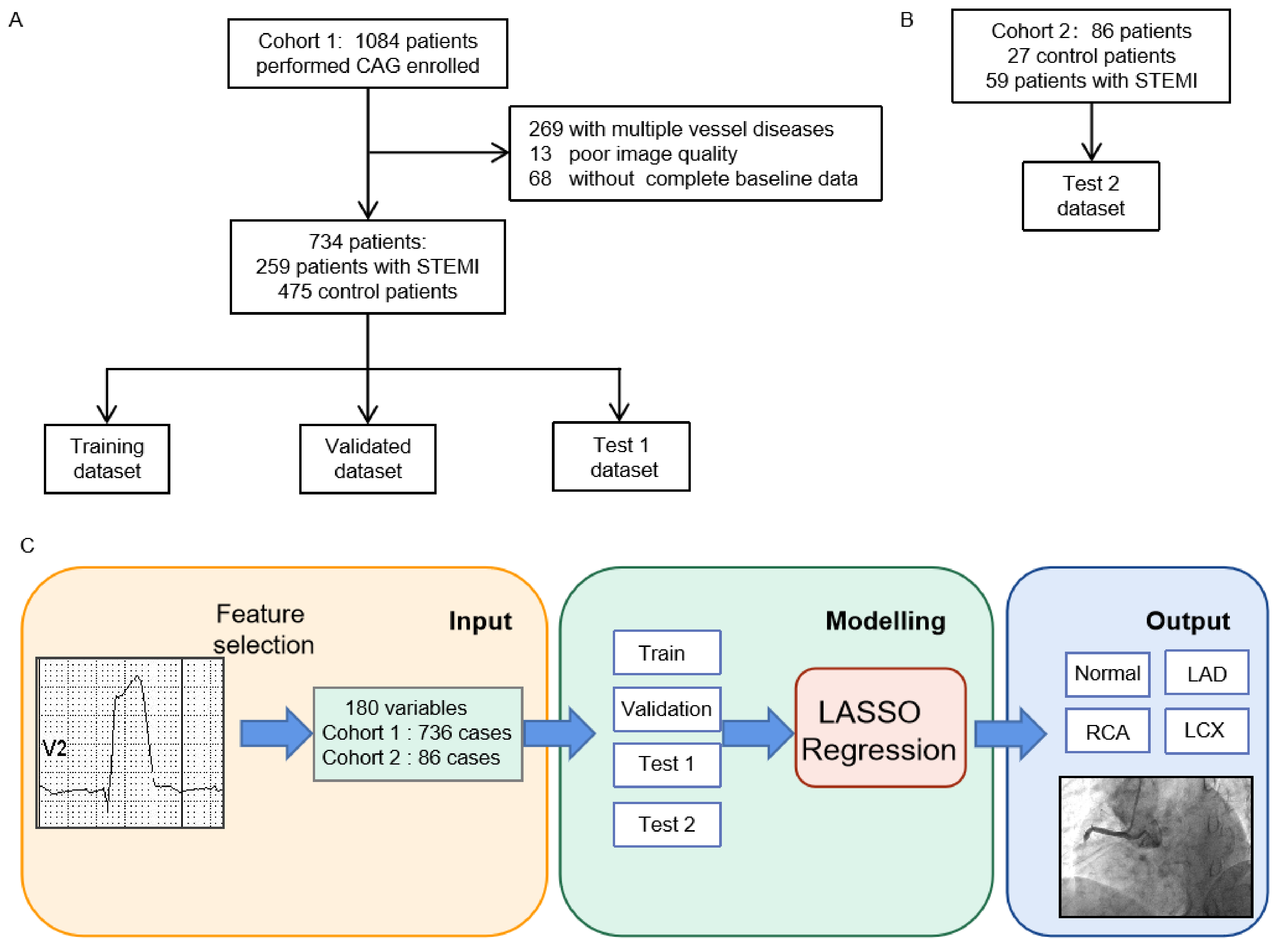
2.1. Study Design
2.2. Study Setting and Data Collection
2.3. Ecg Data
2.4. Lasso Regression
2.5. Comparative Test
2.6. Statistical Analysis
3. Results
3.1. Clinical Characteristics
3.2. Model Performance
3.3. The Interpretation of ECG Features
3.4. Comparative Test
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| STEMI | ST-segment elevation myocardial infarction |
| ECG | electrocardiogram |
| LAD | left anterior descending |
| LCx | left circumflex |
| RCA | right coronary artery |
| RCA | right coronary artery |
| RCA | right coronary artery |
References
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart disease and stroke statistics—2018 update: A report from the American Heart Association. Circulation 2018, 137, e67–e492. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.L.; Barber, R.M.; Foreman, K.J.; Ozgoren, A.A.; Abd-Allah, F.; Abera, S.F.; Aboyans, V.; Abraham, J.P.; Abubakar, I.; Abu-Raddad, L.J.; et al. Global, regional, and national disability-adjusted life years (dalys) for 306 diseases and injuries and healthy life expectancy (hale) for 188 countries, 1990–2013: Quantifying the epidemiological transition. Lancet 2015, 386, 2145–2191. [Google Scholar] [CrossRef]
- Andersen, H.R.; Nielsen, T.T.; Rasmussen, K.; Thuesen, L.; Kelbaek, H.; Thayssen, P.; Abildgaard, U.; Pedersen, F.; Madsen, J.K.; Grande, P.; et al. A comparison of coronary angioplasty with fibrinolytic therapy in acute myocardial infarction. N. Engl. J. Med. 2003, 349, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Simoons, M.L.; Chaitman, B.R.; White, H.D.; Joint ESC/ACCF/AHA/WHF Task Force for Universal Definition of Myocardial Infarction. Task force for the universal definition of myocardial infarction. Third universal definition of myocardial infarction. J. Am. Coll. Cardiol. 2012, 60, 1581–1598. [Google Scholar] [CrossRef] [PubMed]
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D. Executive Group on behalf of the Joint European Society of Cardiology (ESC)/American College of Cardiology (ACC)/American Heart Association (AHA)/World Heart Federation (WHF) Task Force for the Universal Definition of Myocardial Infarction. Fourth universal definition of myocardial infarction (2018). J. Am. Coll. Cardiol. 2018, 72, 2231–2264. [Google Scholar]
- Ayer, A.; Terkelsen, C.J. Difficult ECGs in STEMI: Lessons learned from serial sampling of pre-and in-hospital ECGs. J. Electrocardiol. 2014, 47, 448–458. [Google Scholar] [CrossRef]
- Gregg, R.E.; Babaeizadeh, S. Detection of culprit coronary lesion location in pre-hospital 12-lead ECG. J. Electrocardiol. 2014, 47, 890–894. [Google Scholar] [CrossRef]
- Zhao, Y.; Xiong, J.; Hou, Y.; Zhu, M.; Lu, Y.; Xu, Y.; Teliewubai, J.; Liu, W.; Xu, X.; Li, X.; et al. Early detection of st-segment elevated myocardial infarction by artificial intelligence with 12-lead electrocardiogram. Int. J. Cardiol. 2020, 317, 223–230. [Google Scholar] [CrossRef]
- Ko, W.; Siontis, K.C.; Attia, Z.I.; Carter, R.E.; Kapa, S.; Ommen, S.R.; Demuth, S.J.; Ackerman, M.J.; Gersh, B.J.; Arruda-Olson, A.M.; et al. Detection of hypertrophic cardiomyopathy using a convolutional neural network-enabled electrocardiogram. J. Am. Coll. Cardiol. 2020, 75, 722–733. [Google Scholar] [CrossRef]
- Ochoa, A.; Mena, L.J.; Felix, V.G. Noise-tolerant neural network approach for electrocardiogram signal classification. In Proceedings of the International Conference on Compute and Data Analysis, Lakeland, FL, USA, 19–23 May 2017; pp. 277–282. [Google Scholar]
- Pourbabaee, B.; Roshtkhari, M.J.; Khorasani, K. Deep convolutional neural networks and learning ecg features for screening paroxysmal atrial fibrillation patients. IEEE Trans. Syst. Man Cybern. Syst. 2018, 48, 2095–2104. [Google Scholar] [CrossRef]
- Hannun, A.Y.; Rajpurkar, P.; Haghpanahi, M.; Tison, G.H.; Bourn, C.; Turakhia, M.P.; Ng, A.Y. Cardiologist-level arrhythmia detection and classification in ambulatory electrocardiograms using a deep neural network. Nat. Med. 2019, 25, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Acharya, U.R.; Fujita, H.; Oh, S.L.; Hagiwara, Y.; Tan, J.H.; Adam, M. Application of deep convolutional neural network for automated detection of myocardial infarction using ecg signals. Inf. Sci. 2017, 415, 190–198. [Google Scholar] [CrossRef]
- Göksu, B.B.; Yildiz, O.; Özcan, İ. Diagnosis of acute coronary syndrome with a support vector machine. J. Med. Syst. 2016, 40, 84. [Google Scholar]
- Jambukia, S.H.; Dabhi, V.K.; Prajapati, H.B. Classification of ecg signals using machine learning techniques: A survey. In Proceedings of the 2015 International Conference on Advances in Computer Engineering and Applications, Zhengzhou, China, 18–19 July 2015; pp. 714–721. [Google Scholar]
- Al-Zaiti, S.; Besomi, L.; Bouzid, Z.; Faramand, Z.; Frisch, S.; Martin-Gill, C.; Gregg, R.; Saba, S.; Callaway, C.; Sejdić, E. Machine learning-based prediction of acute coronary syndrome using only the pre-hospital 12-lead electrocardiogram. Nat. Commun. 2020, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.S.; Dodd; Henry, K.W.; Dvorak, T.D.; Pearce, D.M.; Lesly, A. Diagnosis of ST-elevation myocardial infarction in the presence of left bundle branch block with the ST-elevation to S-wave ratio in a modified Sgarbossa rule. Ann. Emerg. Med. 2012, 60, 766–776. [Google Scholar] [CrossRef]
- Siontis, K.C.; Yao, X.; Pirruccello, J.P.; Philippakis, A.A.; Noseworthy, P.A. How will machine learning inform the clinical care of atrial fibrillation? Circ. Res. 2020, 127, 155–169. [Google Scholar] [CrossRef]
- Celin, S.; Vasanth, K. Ecg signal classification using various machine learning techniques. J. Med. Syst. 2018, 42, 1–11. [Google Scholar] [CrossRef]
- Kung, B.; Hu, P.; Huang, C.; Lee, C.; Yao, C.; Kuan, C. An efficient ecg classification system using resource-saving architecture and random forest. IEEE J. Biomed. Health Inform. 2020, 25, 1904–1914. [Google Scholar] [CrossRef]
- Xie, T.; Li, R.; Shen, S.; Zhang, X.; Zhou, B.; Wang, Z. Intelligent analysis of premature ventricular contraction based on features and random forest. J. Healthc. Eng. 2019, 2019, 5787582. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, A.; Gao, M.; Chen, X.; Zhang, X.; Chen, X. Ecg-based multi-class arrhythmia detection using spatio-temporal attention-based convolutional recurrent neural network. Artif. Intell. Med. 2020, 106, 101856. [Google Scholar] [CrossRef]
- Han, C.; Shi, L. Automated interpretable detection of myocardial infarction fusing energy entropy and morphological features. Comput. Methods Programs Biomed. 2019, 175, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Nestelberger, T.; Cullen, L.; Lindahl, B.; Reichlin, T.; Greenslade, J.H.; Giannitsis, E.; Christ, M.; Morawiec, B.; Miro, O.; Martín-Sánchez, F.J.; et al. Diagnosis of acute myocardial infarction in the presence of left bundle branch block. Heart 2019, 105, 1559–1567. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.M.; Shofer, F.S.; Tabas, J.A.; Magid, D.J.; McCusker, C.M.; Hollander, J.E. Lack of association between left bundle-branch block and acute myocardial infarction in symptomatic ed patients. Am. J. Emerg. Med. 2009, 27, 916–921. [Google Scholar] [CrossRef] [PubMed]
- Butt, M.U.; Patel, R.; Darrat, Y.H.; Morales, G.X.; Elayi, C.S. When a pseudo-infarct electrocardiogram (ECG) pattern in a posterior accessory (wolff-parkinson-white) pathway masks a true inferior infarct. Am. J. Case Rep. 2018, 19, 685. [Google Scholar] [CrossRef]
- de Castro, R.L., Jr.; de Lima, N.; de Costa Lino, D.O.; Bannon, S.F. Concealed wolff–parkinson–white syndrome revealed by acute coronary syndrome. Ann. Noninvasive Electrocardiol. 2020, 25, e12735. [Google Scholar] [CrossRef]
- Okin, P.M.; Hille, D.A.; Kjeldsen, S.E.; Devereux, R.B. Combining ecg criteria for left ventricular hypertrophy improves risk prediction in patients with hypertension. J. Am. Heart Assoc. 2017, 6, e007564. [Google Scholar] [CrossRef]
- Stiermaier, T.; Pöss, J.; Eitel, C.; de Waha, S.; Fuernau, G.; Desch, S.; Thiele, H.; Eitel, I. Impact of left ventricular hypertrophy on myocardial injury in patients with st-segment elevation myocardial infarction. Clin. Res. Cardiol. 2018, 107, 1013–1020. [Google Scholar] [CrossRef]
- ESC Committee for Practice Guidelines (CPG); Bax, J.J.; Baumgartner, H.; Ceconi, C.; Dean, V.; Deaton, C.; Fagard, R.; Funck-Brentano, C.; Hasdai, D.; Hoes, A.; et al. Third universal definition of myocardial infarction. J. Am. Coll. Cardiol. 2012, 60, 1581–1598. [Google Scholar]
- Pimor, A.; Auffret, V.; Didier, R.; Delaunay, R.; Filippi, E.; Hacot, J.P.; Saouli, D.; Rouault, G.; Druelles, P.; Bot, E.; et al. Immediate complete revascularization in patients with ST-segment elevation myocardial infarction and multivessel disease treated by primary percutaneous coronary intervention: Insights from the ORBI registry. Arch. Cardiovasc. Dis. 2018, 111, 656–665. [Google Scholar] [CrossRef]




| Cohort 1 | Cohort 2 | p | |
|---|---|---|---|
| n | 734 | 86 | |
| Age (years) | 57.0 ± 13.0 | 59.2 ± 10.6 | 0.080 |
| Sex (female) | 258 (35.1%) | 24 (27.9%) | 0.030 |
| Diabetes mellitus | 147 (20.0%) | 9(10.5%) | 0.033 |
| Hypertension | 321 (43.7%) | 16(18.6%) | 0.000 |
| Chronic kidney disease | 15 (2.0%) | 4(4.7%) | 0.129 |
| CVD family history | 38 (5.2%) | 5(5.8%) | 0.797 |
| BUN (mmol/L) | 5.17 ± 2.6 | 7.13 ± 3.96 | 0.000 |
| Cr (mmol/L) | 78.31 ± 53.68 | 91.93 ± 42.92 | 0.024 |
| CHOL (mmol/L) | 4.55 ± 1.24 | 4.27 ± 1.33 | 0.047 |
| TG (mmol/L) | 1.63 ± 1.1 | 1.42 ± 1.15 | 0.086 |
| HDL-C (mmol/L) | 1.09 ± 0.34 | 1.08 ± 0.35 | 0.909 |
| LDL-C (mmol/L) | 2.85 ± 1.03 | 2.65 ± 1.11 | 0.086 |
| CK-MB (U/L) | 17.13 ± 40.49 | 11.84 ± 13.05 | 0.010 |
| STEMI | 259 (35.2%) | 59 (68.6%) | 0.022 |
| Localization of infarct-related arteries, n | |||
| LAD | 128 (49.4%) | 30 (50.8%) | 0.912 |
| RCA | 95 (36.6%) | 20 (33.9%) | 0.862 |
| LCX | 36 (13.9%) | 9 (15.3%) | 0.831 |
| ECG abnormal phenomenon | |||
| Complete left bundle branch block | 2 (0.3%) | 1 (1.2%) | 0.034 |
| Ventricular premature beat | 28 (3.8%) | 7 (7.8%) | 0.077 |
| Preexcitation syndrome | 3 (0.4%) | 1 (1.2%) | 0.010 |
| Complete right bundle branch block | 15 (2.0%) | 6 (7.0%) | 0.008 |
| Left ventricular hypertrophy | 16 (2.2%) | 10 (11.6%) | 0.000 |
| Atrial fibrillation | 5 (0.7%) | 1 (1.2%) | 0.501 |
| Pacing | 1 (0.1%) | 0 (0) | 1.000 |
| Training Dataset | Validation Dataset | Internal Testing Dataset | External Testing Dataset | p | |
|---|---|---|---|---|---|
| n | 445 | 144 | 145 | 86 | |
| Age (years) | 56.8 ± 13.5 | 57.8 ± 12.3 | 57.1 ± 12.5 | 59.2 ± 10.6 | 0.409 |
| Sex (female) | 148 (33.3%) | 51 (35.4%) | 59 (40.7%) | 24 (27.9%) | 0.215 |
| Diabetes | 86 (19.3%) | 23 (16.0%) | 38 (26.2%) | 9 (10.5%) | 0.020 |
| Hypertension | 184 (54.3%) | 68 (47.2%) | 69 (47.6%) | 16 (18.6%) | 0.000 |
| CKD | 7 (1.6%) | 3 (2.1%) | 5 (3.4%) | 4 (4.7%) | 0.260 |
| CVD family history | 21 (4.7%) | 10 (6.9%) | 7 (4.8%) | 5 (5.8%) | 0.758 |
| WBC (×10/L) | 8.0 ± 3.3 | 8.2 ± 3.0 | 8.3 ± 3.3 | 7.2 ± 2.4 | 0.070 |
| RBC (×10/L) | 4.4 ± 0.9 | 4.2 ± 0.8 | 4.3 ± 1.0 | 4.3 ± 0.8 | 0.376 |
| HGB (g/mL) | 130.6 ± 17.1 | 128.1 ± 17.8 | 128.4 ± 18.5 | 127.0 ± 22.2 | 0.197 |
| PLT (×10/L) | 210.8 ± 60.5 | 206.9 ± 58.5 | 205.9 ± 54.7 | 227.3 ± 66.2 | 0.045 |
| ALB (g/L) | 39.2 ± 4.2 | 38.9 5.0 | 38.8 ± 4.5 | 39.5 ± 4.9 | 0.676 |
| GLB (g/L) | 24.8 ± 4.2 | 24.3 ± 4.2 | 24.5 ± 4.6 | 26.6 ± 4.7 | 0.001 |
| K (mmol/L) | 3.59 ± 0.54 | 3.6 ± 0.56 | 3.62 ± 0.54 | 4.2 ± 0.65 | 0.000 |
| Na (mmol/L) | 137.44 ± 4.4 | 136.8 ± 4.04 | 136.58 ± 5.03 | 141.7± 3.07 | 0.000 |
| Ca (mmol/L) | 1.98 ± 1.05 | 1.87 ± 0.34 | 1.94 ± 0.32 | 2.06 ± 0.24 | 0.312 |
| GLU (mmol/L) | 6.84 ± 3.07 | 7.34 ± 3.45 | 6.86 ± 2.92 | 6.77 ± 1.9 | 0.335 |
| BUN (mmol/L) | 5.14 ± 2.25 | 5.01 ± 2.93 | 5.43 ± 3.21 | 7.13 ± 3.96 | 0.000 |
| Cr (mmol/L) | 75.3 ± 45.3 | 83.8 ± 75.9 | 82.1 ± 50.7 | 91.9 ± 42.9 | 0.031 |
| CHO L (mmol/L) | 4.53 ± 1.19 | 4.6 ± 1.39 | 4.54 ± 1.23 | 4.27± 1.33 | 0.234 |
| TG (mmol/L) | 1.62 ± 1.08 | 1.69 ± 1.28 | 1.62 ± 0.94 | 1.42 ± 1.15 | 0.325 |
| HDL-C (mmol/L) | 1.07 ± 0.33 | 1.11 ± 0.38 | 1.11 ± 0.34 | 1.08 ± 0.35 | 0.460 |
| LDL-C (mmol/L) | 2.84 ± 0.99 | 2.92 ± 1.14 | 2.83 ± 1.05 | 2.65 ± 1.11 | 0.291 |
| CK-MB (U/L) | 16.89 ± 36.82 | 18.51 ± 51.09 | 16.51 ± 39.65 | 11.84 ± 13.05 | 0.640 |
| AUC | Accuracy | SEN | SPE | PPV | NPV | ||
|---|---|---|---|---|---|---|---|
| Model 1 | |||||||
| Train dataset | 0.98 (0.97–0.99) | 0.93 | 0.91 | 0.96 | 0.92 | 0.95 | |
| Validation dataset | 0.97 (0.94–0.99) | 0.89 | 0.86 | 0.92 | 0.85 | 0.92 | |
| Internal dataset | 0.94 (0.90–0.98) | 0.85 | 0.85 | 0.86 | 0.76 | 0.91 | |
| External dataset | 0.93 (0.88–0.98) | 0.84 | 0.85 | 0.82 | 0.89 | 0.78 | |
| Model 2 | |||||||
| Train dataset | 0.94 (0.91–0.98) | 0.92 | 0.95 | 0.84 | 0.86 | 0.94 | |
| Validation dataset | 0.94 (0.85–1.00) | 0.88 | 0.96 | 0.8 | 0.83 | 0.95 | |
| Internal dataset | 0.92 (0.83–0.99) | 0.84 | 0.88 | 0.79 | 0.81 | 0.80 | |
| External dataset | 0.98 (0.95–1.00) | 0.95 | 0.97 | 0.93 | 0.93 | 0.97 |
| Feature | se | z | p | ||
|---|---|---|---|---|---|
| Mode1 | |||||
| (Intercept) | 0.714 | 3.456 | 0.207 | 0.836 | |
| V4(PB) (mV) | −33.047 | 9.296 | −3.555 | <0.001 | |
| V6(Q) (mV) | −18.559 | 5.335 | −3.479 | 0.001 | |
| III(TE) (mV) | 29.427 | 5.941 | 4.953 | <0.001 | |
| AVL(TB) (mV) | 23.027 | 4.46 | 5.163 | <0.001 | |
| II(R) (mV) | −4.4 | 0.835 | −5.266 | <0.001 | |
| II(TB) (mV) | −17.124 | 4.374 | −3.915 | <0.001 | |
| V1(Q) (mV) | −3.264 | 0.749 | −4.358 | <0.001 | |
| III(ST80) (mV) | 12.8 | 4.369 | 2.93 | 0.003 | |
| V2(Q) (mV) | −2.083 | 0.768 | −2.714 | 0.007 | |
| V2(TB) (mV) | 3.271 | 1.162 | 2.816 | 0.005 | |
| V3(ST40) (mV) | 8.861 | 2.719 | 3.259 | 0.001 | |
| V5(TE) (mV) | −5.696 | 2.265 | −2.515 | 0.012 | |
| R-Q interval (ms) | −0.229 | 0.059 | −3.858 | <0.001 | |
| QTc interval (ms) | 0.025 | 0.008 | 3.33 | 0.001 | |
| Model 2 | |||||
| (Intercept) | 0.454 | 0.518 | 0.878 | 0.38 | |
| V1(Q) (mV) | 1.569 | 0.691 | 2.27 | 0.023 | |
| V2(Q) (mV) | 1.917 | 0.73 | 2.624 | 0.009 | |
| V2(TB) (mV) | 4.742 | 1.342 | 3.535 | <0.001 | |
| V3(ST40) (mV) | −7.373 | 2.274 | −3.241 | 0.001 |
| AUC | SEN | SPE | PPV | NPV | ||
|---|---|---|---|---|---|---|
| Model 1 | ||||||
| Experienced cardiologists | 0.92 (0.90–0.95) | 0.88 | 0.92 | 0.96 | 0.97 | |
| Emergency physicians | 0.86 (0.82–0.89) | 0.74 | 0.84 | 0.95 | 0.97 | |
| Internal medicine residents | 0.83 (0.80–0.86) | 0.69 | 0.82 | 0.96 | 0.98 | |
| Medical interns | 0.76 (0.72–0.80) | 0.63 | 0.77 | 0.8 | 0.89 | |
| Our model | 0.94 (0.90–0.98) | 0.85 | 0.86 | 0.76 | 0.91 | |
| Model 2 | ||||||
| Experienced cardiologists | 0.83 (0.78–0.89) | 0.72 | 0.93 | 0.75 | 0.94 | |
| Emergency physicians | 0.81 (0.76–0.87) | 0.70 | 0.93 | 0.71 | 0.93 | |
| Internal medicine residents | 0.79 (0.74–0.85) | 0.69 | 0.92 | 0.63 | 0.90 | |
| Medical interns | 0.68 (0.62–0.73) | 0.53 | 0.87 | 0.43 | 0.82 | |
| Our model | 0.98 (0.95–1.00) | 0.88 | 0.79 | 0.81 | 0.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, L.; Zhou, B.; Liu, D.; Wang, L.; Zhang, X.; Xu, L.; Yuan, L.; Zhang, H.; Ling, Y.; Shi, G.; et al. LASSO Regression-Based Diagnosis of Acute ST-Segment Elevation Myocardial Infarction (STEMI) on Electrocardiogram (ECG). J. Clin. Med. 2022, 11, 5408. https://doi.org/10.3390/jcm11185408
Wu L, Zhou B, Liu D, Wang L, Zhang X, Xu L, Yuan L, Zhang H, Ling Y, Shi G, et al. LASSO Regression-Based Diagnosis of Acute ST-Segment Elevation Myocardial Infarction (STEMI) on Electrocardiogram (ECG). Journal of Clinical Medicine. 2022; 11(18):5408. https://doi.org/10.3390/jcm11185408
Chicago/Turabian StyleWu, Lin, Bin Zhou, Dinghui Liu, Linli Wang, Ximei Zhang, Li Xu, Lianxiong Yuan, Hui Zhang, Yesheng Ling, Guangyao Shi, and et al. 2022. "LASSO Regression-Based Diagnosis of Acute ST-Segment Elevation Myocardial Infarction (STEMI) on Electrocardiogram (ECG)" Journal of Clinical Medicine 11, no. 18: 5408. https://doi.org/10.3390/jcm11185408
APA StyleWu, L., Zhou, B., Liu, D., Wang, L., Zhang, X., Xu, L., Yuan, L., Zhang, H., Ling, Y., Shi, G., Ke, S., He, X., Tian, B., Chen, Y., & Qian, X. (2022). LASSO Regression-Based Diagnosis of Acute ST-Segment Elevation Myocardial Infarction (STEMI) on Electrocardiogram (ECG). Journal of Clinical Medicine, 11(18), 5408. https://doi.org/10.3390/jcm11185408





