Associations of the Alpha-Actinin Three Genotype with Bone and Muscle Mass Loss among Middle-Aged and Older Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. ACTN3 Genotyping
2.3. Measurement of Bone Mass
2.4. Measurement of Muscle Mass
2.5. Physical Performance
2.6. Sociodemographic Characteristics
2.7. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Aging Overview. Available online: https://www.who.int/news-room/fact-sheets/detail/ageing-and-health (accessed on 1 February 2022).
- Peeters, G.; van Schoor, N.M.; Lips, P. Fall risk: The clinical relevance of falls and how to integrate fall risk with fracture risk. Best Pract. Res. Clin. Rheumatol. 2009, 23, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Tinetti, M.E. Clinical practice. Preventing falls in elderly persons. N. Engl. J. Med. 2003, 348, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Leibson, C.L.; Tosteson, A.N.; Gabriel, S.E.; Ransom, J.E.; Melton, L.J. Mortality, disability, and nursing home use for persons with and without hip fracture: A population-based study. J. Am. Geriatr. Soc. 2002, 50, 1644–1650. [Google Scholar] [CrossRef]
- Miyakoshi, N.; Hongo, M.; Mizutani, Y.; Shimada, Y. Prevalence of sarcopenia in Japanese women with osteopenia and osteoporosis. J. Bone Miner. Metab. 2013, 31, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Di Monaco, M.; Vallero, F.; Di Monaco, R.; Tappero, R. Prevalence of sarcopenia and its association with osteoporosis in 313 older women following a hip fracture. Arch. Gerontol. Geriatr. 2011, 52, 71–74. [Google Scholar] [CrossRef]
- He, H.; Liu, Y.; Tian, Q.; Papasian, C.J.; Hu, T.; Deng, H.W. Relationship of sarcopenia and body composition with osteoporosis. Osteoporos. Int. 2016, 27, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Cariati, I.; Bonanni, R.; Onorato, F.; Mastrogregori, A.; Rossi, D.; Iundusi, R.; Gasbarra, E.; Tancredi, V.; Tarantino, U. Role of physical activity in bone-muscle crosstalk: Biological aspects and clinical implications. J. Funct. Morphol. Kinesiol. 2021, 6, 55. [Google Scholar] [CrossRef]
- Sapir-Koren, R.; Livshits, G. Osteocyte control of bone remodeling: Is sclerostin a key molecular coordinator of the balanced bone resorption-formation cycles? Osteoporos. Int. 2014, 25, 2685–2700. [Google Scholar] [CrossRef]
- Goodman, C.A.; Hornberger, T.A.; Robling, A.G. Bone and skeletal muscle: Key players in mechanotransduction and potential overlapping mechanisms. Bone 2015, 80, 24–36. [Google Scholar] [CrossRef]
- Kirk, B.; Feehan, J.; Lombardi, G.; Duque, G. Muscle, Bone, and Fat Crosstalk: The Biological Role of Myokines, Osteokines, and Adipokines. Curr. Osteoporos. Rep. 2020, 18, 388–400. [Google Scholar] [CrossRef]
- Kirk, B.; Zanker, J.; Duque, G. Osteosarcopenia: Epidemiology, diagnosis, and treatment-facts and numbers. J. Cachexia Sarcopenia Muscle 2020, 11, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Sepúlveda-Loyola, W.; Phu, S.; Bani Hassan, E.; Brennan-Olsen, S.L.; Zanker, J.; Vogrin, S.; Conzade, R.; Kirk, B.; Al Saedi, A.; Probst, V.; et al. The joint occurrence of osteoporosis and sarcopenia (osteosarcopenia): Definitions and characteristics. J. Am. Med. Dir. Assoc. 2020, 21, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Atlihan, R.; Kirk, B.; Duque, G. Non-pharmacological interventions in osteosarcopenia: A systematic review. J. Nutr. Health Aging 2021, 25, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Huo, Y.R.; Suriyaarachchi, P.; Gomez, F.; Curcio, C.L.; Boersma, D.; Muir, S.W.; Montero-Odasso, M.; Gunawardene, P.; Demontiero, O.; Duque, G. Phenotype of osteosarcopenia in older individuals with a history of falling. J. Am. Med. Dir. Assoc. 2015, 16, 290–295. [Google Scholar] [CrossRef]
- Wang, Y.J.; Wang, Y.; Zhan, J.K.; Tang, Z.Y.; He, J.Y.; Tan, P.; Deng, H.Q.; Huang, W.; Liu, Y.S. Sarco-osteoporosis: Prevalence and association with frailty in chinese community-dwelling older adults. Int. J. Endocrinol. 2015, 2015, 482940. [Google Scholar] [CrossRef]
- Drey, M.; Sieber, C.C.; Bertsch, T.; Bauer, J.M.; Schmidmaier, R.; FiAT intervention group. Osteosarcopenia is more than sarcopenia and osteopenia alone. Aging Clin. Exp. Res. 2016, 28, 895–899. [Google Scholar] [CrossRef]
- Teng, Z.; Zhu, Y.; Teng, Y.; Long, Q.; Hao, Q.; Yu, X.; Yang, L.; Lv, Y.; Liu, J.; Zeng, Y.; et al. The analysis of osteosarcopenia as a risk factor for fractures, mortality, and falls. Osteoporos. Int. 2021, 32, 2173–2183. [Google Scholar] [CrossRef]
- Kirk, B.; Al Saedi, A.; Duque, G. Osteosarcopenia: A case of geroscience. Aging Med. 2019, 2, 147–156. [Google Scholar] [CrossRef]
- Karasik, D.; Kiel, D.P. Genetics of the musculoskeletal system: A pleiotropic approach. J. Bone Miner. Res. 2008, 23, 788–802. [Google Scholar] [CrossRef]
- Pickering, C.; Kiely, J. ACTN3, morbidity, and healthy aging. Front. Genet. 2018, 9, 15. [Google Scholar] [CrossRef]
- Houweling, P.J.; Papadimitriou, I.D.; Seto, J.T.; Pérez, L.M.; Coso, J.D.; North, K.N.; Lucia, A.; Eynon, N. Is evolutionary loss our gain? The role of ACTN3 p.Arg577Ter (R577X) genotype in athletic performance, ageing, and disease. Hum. Mutat. 2018, 39, 1774–1787. [Google Scholar] [CrossRef] [PubMed]
- North, K.N.; Yang, N.; Wattanasirichaigoon, D.; Mills, M.; Easteal, S.; Beggs, A.H. A common nonsense mutation results in alpha-actinin-3 deficiency in the general population. Nat. Genet. 1999, 21, 353–354. [Google Scholar] [CrossRef] [PubMed]
- Vincent, B.; De Bock, K.; Ramaekers, M.; Van den Eede, E.; Van Leemputte, M.; Hespel, P.; Thomis, M.A. ACTN3 (R577X) genotype is associated with fiber type distribution. Physiol. Genomics 2007, 32, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Schindeler, A.; McDonald, M.M.; Seto, J.T.; Houweling, P.J.; Lek, M.; Hogarth, M.; Morse, A.R.; Raftery, J.M.; Balasuriya, D.; et al. alpha-Actinin-3 deficiency is associated with reduced bone mass in human and mouse. Bone 2011, 49, 790–798. [Google Scholar] [CrossRef]
- Kiuchi, Y.; Makizako, H.; Nakai, Y.; Taniguchi, Y.; Tomioka, K.; Sato, N.; Wada, A.; Doi, T.; Kiyama, R.; Takenaka, T. Associations of alpha-actinin-3 genotype with thigh muscle volume and physical performance in older adults with sarcopenia or pre-sarcopenia. Exp. Gerontol. 2021, 154, 111525. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.; Lee, I.; Kang, H. ACTN3 gene and susceptibility to sarcopenia and osteoporotic status in older Korean adults. BioMed Res. Int. 2017, 2017, 4239648. [Google Scholar] [CrossRef] [PubMed]
- Min, S.K.; Lim, S.T.; Kim, C.S. Association of ACTN3 polymorphisms with BMD, and physical fitness of elderly women. J. Phys. Ther. Sci. 2016, 28, 2731–2736. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.K.; Woo, J.; Assantachai, P.; Auyeung, T.W.; Chou, M.Y.; Iijima, K.; Jang, H.C.; Kang, L.; Kim, M.; Kim, S.; et al. Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J. Am. Med. Dir. Assoc. 2020, 21, 300–307. [Google Scholar] [CrossRef]
- Nelson, H.D. Menopause. Lancet 2008, 371, 760–770. [Google Scholar] [CrossRef]
- Subramaniam, S.; Chan, C.Y.; Soelaiman, I.N.; Mohamed, N.; Muhammad, N.; Ahmad, F.; Ng, P.Y.; Jamil, N.A.; Aziz, N.A.; Chin, K.Y. The performance of a calcaneal quantitative ultrasound device, CM-200, in stratifying osteoporosis risk among Malaysian population aged 40 years and above. Diagnostics 2020, 10, 178. [Google Scholar] [CrossRef]
- Chin, K.Y.; Soelaiman, I.N.; Mohamed, I.N.; Ibrahim, S.; Wan Ngah, W.Z. The effects of age, physical activity level, and body anthropometry on calcaneal speed of sound value in men. Arch. Osteoporos. 2012, 7, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Kurose, S.; Nishikawa, S.; Nagaoka, T.; Kusaka, M.; Kawamura, J.; Nishioka, Y.; Sato, S.; Tsutsumi, H.; Kimura, Y. Prevalence and risk factors of sarcopenia in community-dwelling older adults visiting regional medical institutions from the Kadoma sarcopenia Study. Sci. Rep. 2020, 10, 19129. [Google Scholar] [CrossRef] [PubMed]
- Makizako, H.; Kubozono, T.; Kiyama, R.; Takenaka, T.; Kuwahata, S.; Tabira, T.; Kanoya, T.; Horinouchi, K.; Shimada, H.; Ohishi, M. Associations of social frailty with loss of muscle mass and muscle weakness among community-dwelling older adults. Geriatr. Gerontol. Int. 2019, 19, 76–80. [Google Scholar] [CrossRef]
- Kiuchi, Y.; Makizako, H.; Nakai, Y.; Tomioka, K.; Taniguchi, Y.; Kimura, M.; Kanouchi, H.; Takenaka, T.; Kubozono, T.; Ohishi, M. The association between dietary variety and physical frailty in community-dwelling older adults. Healthcare 2021, 9, 32. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, N.; Miyamoto-Mikami, E.; Hirata, K.; Kimura, N.; Fuku, N. Association analysis of the ACTN3 R577X polymorphism with passive muscle stiffness and muscle strain injury. Scand. J. Med. Sci. Sport. 2018, 28, 1209–1214. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, H.; Tobina, T.; Ichinoseki-Sekine, N.; Kakigi, R.; Tsuzuki, T.; Zempo, H.; Shiose, K.; Yoshimura, E.; Kumahara, H.; Ayabe, M.; et al. Role of selected polymorphisms in determining muscle fiber composition in Japanese men and women. J. Appl. Physiol. 2018, 124, 1377–1384. [Google Scholar] [CrossRef]
- Kikuchi, N.; Tsuchiya, Y.; Nakazato, K.; Ishii, N.; Ochi, E. Effects of the ACTN3 R577X genotype on the muscular strength and range of motion before and after eccentric contractions of the elbow flexors. Int. J. Sports Med. 2018, 39, 148–153. [Google Scholar] [CrossRef]
- Mikami, E.; Fuku, N.; Murakami, H.; Tsuchie, H.; Takahashi, H.; Ohiwa, N.; Tanaka, H.; Pitsiladis, Y.P.; Higuchi, M.; Miyachi, M.; et al. ACTN3 R577X genotype is associated with sprinting in elite Japanese athletes. Int. J. Sports Med. 2014, 35, 172–177. [Google Scholar] [CrossRef]
- Genaro, P.S.; Pereira, G.A.; Pinheiro, M.M.; Szejnfeld, V.L.; Martini, L.A. Influence of body composition on bone mass in postmenopausal osteoporotic women. Arch. Gerontol. Geriatr. 2010, 51, 295–298. [Google Scholar] [CrossRef]
- Alfred, T.; Ben-Shlomo, Y.; Cooper, R.; Hardy, R.; Cooper, C.; Deary, I.J.; Gunnell, D.; Harris, S.E.; Kumari, M.; Martin, R.M.; et al. ACTN3 genotype, athletic status, and life course physical capability: Meta-analysis of the published literature and findings from nine studies. Hum. Mutat. 2011, 32, 1008–1018. [Google Scholar] [CrossRef]
- Pereira, A.; Costa, A.M.; Izquierdo, M.; Silva, A.J.; Bastos, E.; Marques, M.C. ACE I/D and ACTN3 R/X polymorphisms as potential factors in modulating exercise-related phenotypes in older women in response to a muscle power training stimuli. Age 2013, 35, 1949–1959. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, Y.; Makizako, H.; Kiyama, R.; Tomioka, K.; Nakai, Y.; Kubozono, T.; Takenaka, T.; Ohishi, M. The association between osteoporosis and grip strength and skeletal muscle mass in community-dwelling older women. Int. J. Environ. Res. Public Health 2019, 16, 1228. [Google Scholar] [CrossRef] [PubMed]
- Curtis, E.; Litwic, A.; Cooper, C.; Dennison, E. Determinants of Muscle and Bone Aging. J. Cell Physiol. 2015, 230, 2618–2625. [Google Scholar] [CrossRef] [PubMed]
- Seto, J.T.; Roeszler, K.N.; Meehan, L.R.; Wood, H.D.; Tiong, C.; Bek, L.; Lee, S.F.; Shah, M.; Quinlan, K.G.R.; Gregorevic, P.; et al. ACTN3 genotype influences skeletal muscle mass regulation and response to dexamethasone. Sci. Adv. 2021, 7, eabg0088. [Google Scholar] [CrossRef] [PubMed]
- Levinger, I.; Yan, X.; Bishop, D.; Houweling, P.J.; Papadimitriou, I.; Munson, F.; Byrnes, E.; Vicari, D.; Brennan-Speranza, T.C.; Eynon, N. The influence of alpha-actinin-3 deficiency on bone remodelling markers in young men. Bone 2017, 98, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Fatima, M.; Brennan-Olsen, S.L.; Duque, G. Therapeutic approaches to osteosarcopenia: Insights for the clinician. Ther. Adv. Musculoskelet. Dis. 2019, 11, 1759720X19867009. [Google Scholar] [CrossRef]
- Trajanoska, K.; Rivadeneira, F.; Kiel, D.P.; Karasik, D. Genetics of bone and muscle interactions in umans. Curr. Osteoporos. Rep. 2019, 17, 86–95. [Google Scholar] [CrossRef]
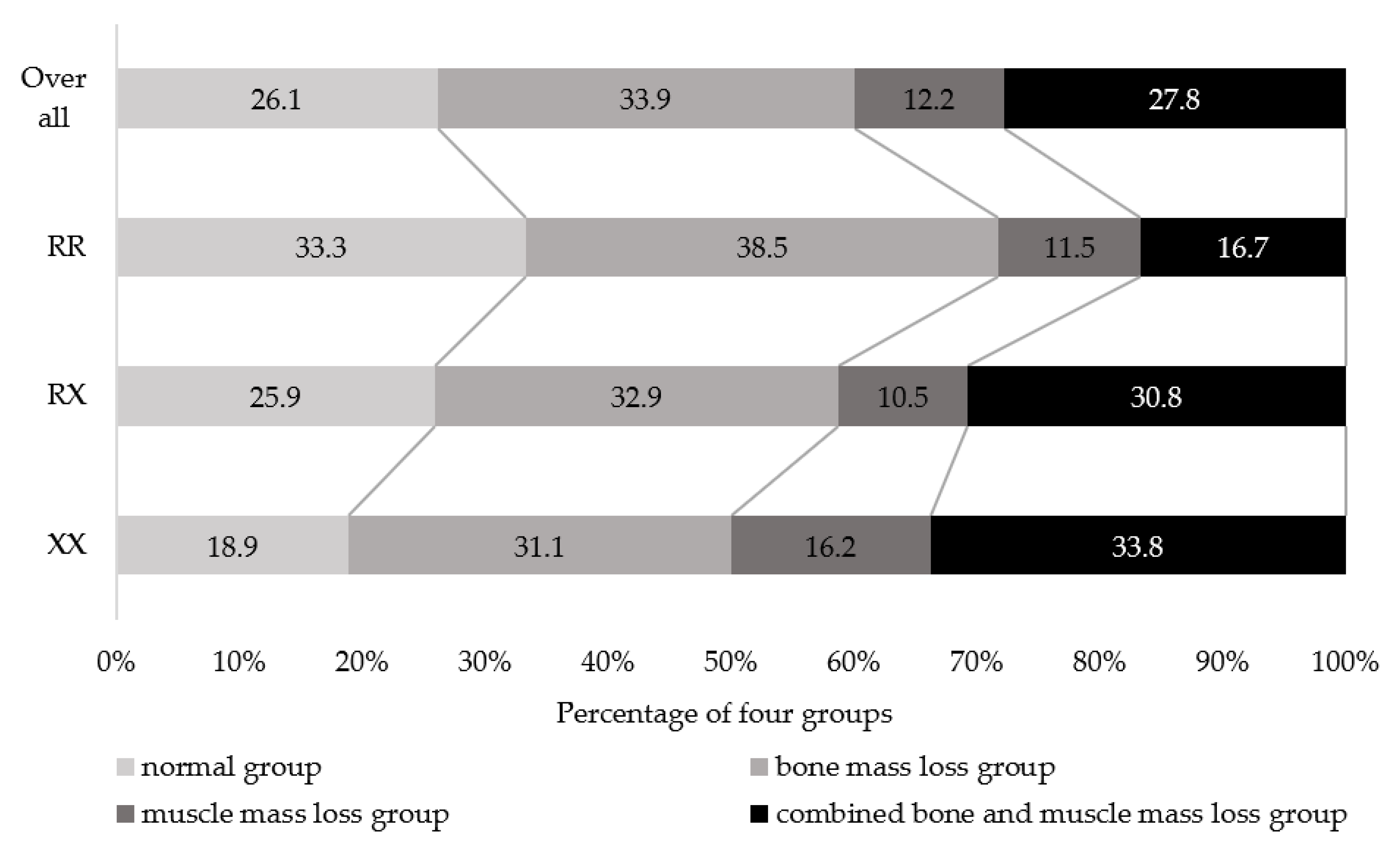
| Genotype | |||||
|---|---|---|---|---|---|
| Variable | Overall (n = 295) | XX (n = 74, 25.1%) | RX (n = 143, 48.5%) | RR (n = 78, 26.4%) | p |
| Age, years | 73.2 ± 6.5 | 73.9 ± 6.9 | 73.0 ± 6.9 | 72.8 ± 5.3 | 0.500 |
| Female, n (%) | 187 (63.4) | 46 (62.2) | 102 (71.3) | 39 (50.0) | 0.109 |
| BMI, kg/m2 | 22.8 ± 3.1 | 22.2 ± 3.2 | 22.9 ± 3.2 | 23.2 ± 2.8 | 0.122 |
| Medications, number/day | 3.0 ± 3.0 | 3.7 ± 3.3 | 2.9 ± 3.0 | 2.5 ± 2.5 | 0.054 |
| Osteoporosis medications, n (%) | 46 (15.6) | 11 (14.9) | 27 (18.9) | 8 (10.3) | 0.419 |
| Medical history, n (%) | |||||
| Heart disease | 23 (7.8) | 6 (7.7) | 11 (7.7) | 6 (8.1) | 0.925 |
| Diabetes mellitus | 36 (12.2) | 10 (13.5) | 16 (11.2) | 10 (12.8) | 0.904 |
| Osteoporosis | 55 (18.6) | 15 (20.3) | 32 (22.4) | 8 (10.3) | 0.107 |
| Thyroid disease | 22 (7.5) | 1 (1.4) | 17 (11.9) | 4 (5.1) | 0.405 |
| Respiratory disease | 29 (9.8) | 10 (13.5) | 14 (9.8) | 5 (6.4) | 0.142 |
| Malignant tumor | 33 (11.2) | 6 (8.1) | 19 (13.3) | 8 (10.3) | 0.690 |
| GDS-15, score | 2.6 ± 2.4 | 2.9 ± 2.6 | 2.7 ± 2.5 | 2.4 ± 2.1 | 0.397 |
| Fall history, n (%) | 45 (15.3) | 13 (17.6) | 22 (15.4) | 10 (12.8) | 0.416 |
| Exercise habit, n (%) | 237 (80.3) | 61 (82.4) | 112 (78.3) | 64 (82.1) | 0.965 |
| Muscle mass loss, n (%) | 118 (40.0) | 37 (50.0) | 59 (41.3) | 22 (28.2) | 0.006 |
| Bone mass loss, n (%) | 182 (61.7) | 48 (64.9) | 91 (63.6) | 43 (55.1) | 0.213 |
| Grip strength, kg | 25.7 ± 7.9 | 25.3 ± 7.2 | 24.8 ± 7.8 | 27.9 ± 8.4 | 0.015 |
| Walking speed, m/s | 1.3 ± 0.2 | 1.3 ± 0.2 | 1.3 ± 0.2 | 1.3 ± 0.2 | 0.979 |
| Normal Group vs | |||
|---|---|---|---|
| Independent Variable | Bone Mass Loss Group OR (95% CI) | Muscle Mass Loss Group OR (95% CI) | Combined Bone and Muscle Mass Loss Group OR (95% CI) |
| RR | reference | reference | reference |
| RX | 0.75 (0.35–1.60) | 1.03 (0.37–2.88) | 1.62 (0.66–3.98) |
| XX | 1.14 (0.45–2.89) | 2.30 (0.73–7.30) | 3.00 (1.05–8.54) * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taniguchi, Y.; Makizako, H.; Nakai, Y.; Kiuchi, Y.; Akaida, S.; Tateishi, M.; Takenaka, T.; Kubozono, T.; Ohishi, M. Associations of the Alpha-Actinin Three Genotype with Bone and Muscle Mass Loss among Middle-Aged and Older Adults. J. Clin. Med. 2022, 11, 6172. https://doi.org/10.3390/jcm11206172
Taniguchi Y, Makizako H, Nakai Y, Kiuchi Y, Akaida S, Tateishi M, Takenaka T, Kubozono T, Ohishi M. Associations of the Alpha-Actinin Three Genotype with Bone and Muscle Mass Loss among Middle-Aged and Older Adults. Journal of Clinical Medicine. 2022; 11(20):6172. https://doi.org/10.3390/jcm11206172
Chicago/Turabian StyleTaniguchi, Yoshiaki, Hyuma Makizako, Yuki Nakai, Yuto Kiuchi, Shoma Akaida, Mana Tateishi, Toshihiko Takenaka, Takuro Kubozono, and Mitsuru Ohishi. 2022. "Associations of the Alpha-Actinin Three Genotype with Bone and Muscle Mass Loss among Middle-Aged and Older Adults" Journal of Clinical Medicine 11, no. 20: 6172. https://doi.org/10.3390/jcm11206172
APA StyleTaniguchi, Y., Makizako, H., Nakai, Y., Kiuchi, Y., Akaida, S., Tateishi, M., Takenaka, T., Kubozono, T., & Ohishi, M. (2022). Associations of the Alpha-Actinin Three Genotype with Bone and Muscle Mass Loss among Middle-Aged and Older Adults. Journal of Clinical Medicine, 11(20), 6172. https://doi.org/10.3390/jcm11206172







