Clinical Utility of the Portable Pressure-Measuring Device for Compression Garment Pressure Measurement on Hypertrophic Scars by Burn Injury during Compression Therapy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Clinical Participants
2.2. Intervention
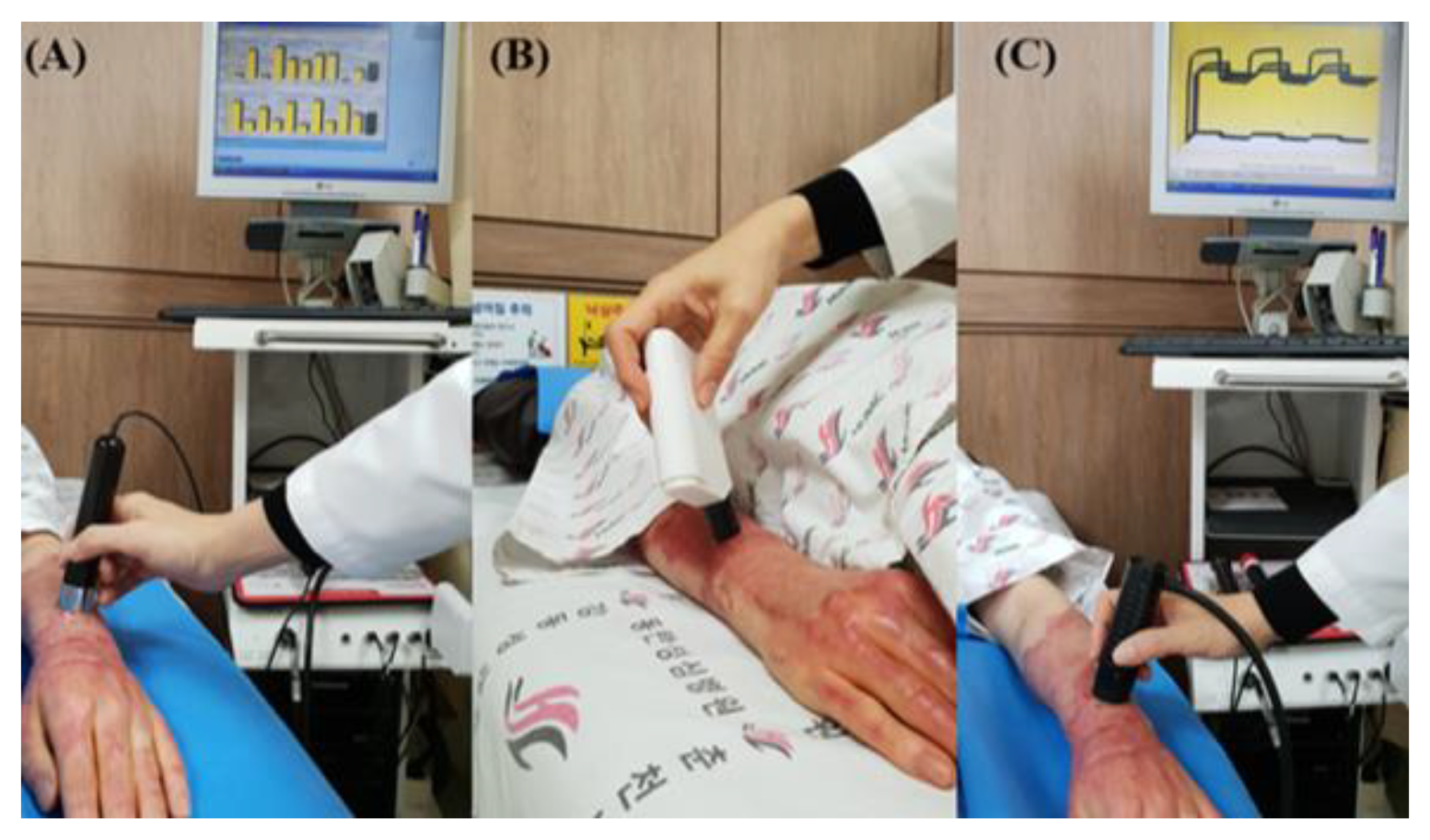
2.3. Outcome Measurements
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Prasarn, M.L.; Horodyski, M.; Schneider, P.S.; Pernik, M.N.; Gary, J.L.; Rechtine, G.R. Comparison of skin pressure measurements with the use of pelvic circumferential compression devices on pelvic ring injuries. Injury 2016, 47, 717–720. [Google Scholar] [CrossRef] [PubMed]
- Rimaud, D.; Convert, R.; Calmels, P. In vivo measurement of compression bandage interface pressures: The first study. Ann. Phys. Rehabil. Med. 2014, 57, 394–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crofton, E.; Meredith, P.; Gray, P.; O’Reilly, S.; Strong, J. Non-adherence with compression garment wear in adult burns patients: A systematic review and meta-ethnography. Burns 2020, 46, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Engrav, L.; Heimbach, D.; Rivara, F.; Moore, M.; Wang, J.; Carrougher, G.; Costa, B.; Numhom, S.; Calderon, J.; Gibran, N. 12-Year within-wound study of the effectiveness of custom pressure garment therapy. Burns 2010, 36, 975–983. [Google Scholar] [CrossRef]
- Macintyre, L.; Baird, M. Pressure garments for use in the treatment of hypertrophic scars—A review of the problems associated with their use. Burns 2006, 32, 10–15. [Google Scholar] [CrossRef]
- Van den Kerckhove, E.; Stappaerts, K.; Fieuws, S.; Laperre, J.; Massage, P.; Flour, M.; Boeckx, W. The assessment of erythema and thickness on burn related scars during pressure garment therapy as a preventive measure for hypertrophic scarring. Burns 2005, 31, 696–702. [Google Scholar] [CrossRef]
- Renò, F.; Sabbatini, M.; Lombardi, F.; Stella, M.; Pezzuto, C.; Magliacani, G.; Cannas, M. In vitro mechanical compression induces apoptosis and regulates cytokines release in hypertrophic scars. Wound Repair Regen. 2003, 11, 331–336. [Google Scholar] [CrossRef]
- Bosman, J. Lymphtaping for lymphoedema: An overview of the treatment and its uses. Br. J. Community Nurs. 2014, 19 (Suppl. 4), S12–S18. [Google Scholar] [CrossRef] [Green Version]
- Macintyre, L. Designing pressure garments capable of exerting specific pressures on limbs. Burns 2007, 33, 579–586. [Google Scholar] [CrossRef]
- Li, P.; Li-Tsang, C.W.P.; Deng, X.; Wang, X.; Wang, H.; Zhang, Y.; Tan, Z.; He, C. The recovery of post-burn hypertrophic scar in a monitored pressure therapy intervention programme and the timing of intervention. Burns 2018, 44, 1451–1467. [Google Scholar] [CrossRef]
- Yildiz, N. A novel technique to determine pressure in pressure garments for hypertrophic burn scars and comfort properties. Burns 2007, 33, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.H.; Li-Tsang, C.W. Validation of the Pliance X System in measuring interface pressure generated by pressure garment. Burns 2009, 35, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Mann, R.; Yeong, E.K.; Moore, M.L.; Engrav, L.H. A New Tool to Measure Pressure Under Burn Garments. J. Burn. Care Rehabil. 1997, 18, 160–163; discussion 159. [Google Scholar] [CrossRef] [PubMed]
- Chi, Y.-W.; Tseng, K.-H.; Li, R.; Pan, T. Comparison of piezoresistive sensor to PicoPress® in in-vitro interface pressure measurement. Phlebology 2018, 33, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Alkhalil, A.; Tejiram, S.; Travis, T.E.; Prindeze, N.J.; Carney, B.C.; Moffatt, L.T.; Johnson, L.S.; Ramella-Roman, J.; Shupp, J.W. A Translational Animal Model for Scar Compression Therapy Using an Automated Pressure Delivery System. Eplasty 2015, 15, e29. [Google Scholar]
- Brorson, H.; Hansson, E.; Jense, E.; Freccero, C. Development of a Pressure-Measuring Device to Optimize Compression Treatment of Lymphedema and Evaluation of Change in Garment Pressure with Simulated Wear and Tear. Lymphat. Res. Biol. 2012, 10, 74–80. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.Y.; Cho, Y.S.; Joo, S.Y.; Seo, C.H. Comparison between the portable pressure measuring device and PicoPress® for garment pressure measurement on hypertrophic burn scar during compression therapy. Burns 2021, 47, 1621–1626. [Google Scholar] [CrossRef]
- Broughton, G., 2nd; Janis, J.E.; Attinger, C.E. Wound Healing: An Overview. Plast. Reconstr. Surg. 2006, 117, 1e-S–32e-S. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.Y.; Joo, S.Y.; Cho, Y.S.; Hur, G.Y.; Seo, C.H. Effect of extracorporeal shock wave therapy for burn scar regeneration: A prospective, randomized, double-blinded study. Burns 2021, 47, 821–827. [Google Scholar] [CrossRef]
- Tejiram, S.; Zhang, J.; Travis, T.E.; Carney, B.C.; Alkhalil, A.; Moffatt, L.T.; Johnson, L.S.; Shupp, J.W. Compression therapy affects collagen type balance in hypertrophic scar. J. Surg. Res. 2016, 201, 299–305. [Google Scholar] [CrossRef] [Green Version]
- Li-Tsang, C.W.P.; Zheng, Y.P.; Lau, J.C.M. A Randomized Clinical Trial to Study the Effect of Silicone Gel Dressing and Pressure Therapy on Posttraumatic Hypertrophic Scars. J. Burn Care Res. 2010, 31, 448–457. [Google Scholar] [CrossRef] [PubMed]
- Li-Tsang, C.W.; Feng, B.; Huang, L.; Liu, X.; Shu, B.; Chan, Y.T.; Cheung, K.-K. A histological study on the effect of pressure therapy on the activities of myofibroblasts and keratinocytes in hypertrophic scar tissues after burn. Burns 2015, 41, 1008–1016. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.-W.; Deng, W.-P.; Yeong, E.-K.; Wu, C.-Y.; Yeh, S.-W. Pressure Effects on the Growth of Human Scar Fibroblasts. J. Burn Care Res. 2008, 29, 835–841. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghassemi, P.; Shupp, J.W.; Travis, T.E.; Gravunder, A.J.; Moffatt, L.T.; Ramella-Roman, J.C. A portable automatic pressure delivery system for scar compression therapy in large animals. Rev. Sci. Instrum. 2015, 86, 015101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, F.; Knapp, D.; Wallen, M. Comparison of the characteristics and features of pressure garments used in the management of burn scars. Burns 1998, 24, 329–335. [Google Scholar] [CrossRef]
- Keller, K.; Krenzer-Scheidemantel, G.; Meyer, T. A systematic analysis of the compression pressure in the compression therapy for scars in childhood with the help of the Kikuhime® pressure device. Zentralbl. Chir. 2014, 139, 638–642. [Google Scholar] [CrossRef]
- Kerckhove, E.V.D.; Fieuws, S.; Massagé, P.; Hierner, R.; Boeckx, W.; Deleuze, J.-P.; Laperre, J.; Anthonissen, M. Reproducibility of repeated measurements with the Kikuhime pressure sensor under pressure garments in burn scar treatment. Burns 2007, 33, 572–578. [Google Scholar] [CrossRef]
- Harries, C.; Pegg, S. Measuring pressure under burns pressure garments using the Oxford Pressure Monitor. Burns 1989, 15, 187–189. [Google Scholar] [CrossRef]
- Catarino, C.M.; Kaiser, K.; Baltazar, T.; Catarino, L.M.; Brewer, J.R.; Karande, P. Evaluation of native and non-native biomaterials for engineering human skin tissue. Bioeng. Transl. Med. 2022, 7, e10297. [Google Scholar] [CrossRef]
- Zhang, C.-P.; Fu, X.-B. Therapeutic potential of stem cells in skin repair and regeneration. Chin. J. Traumatol. 2008, 11, 209–221. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Peng, Y.; Fan, H.; Zhang, Y.; Min, P. Microneedle delivery of botulinum toxin type A combined with hyaluronic acid for the synergetic management of multiple sternal keloids with oily skin: A retrospective clinical investigation. J. Cosmet. Dermatol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Daly, C.H. Biomechanical Properties of Dermis. J. Investig. Dermatol. 1982, 79 (Suppl. 1), 17s–20s. [Google Scholar] [CrossRef] [PubMed]
- DeBruler, D.M.; Baumann, M.E.; Zbinden, J.C.; Blackstone, M.B.N.; Bailey, J.K.; Supp, D.M.; Powell, H.M. Improved Scar Outcomes with Increased Daily Duration of Pressure Garment Therapy. Adv. Wound Care 2020, 9, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Kischer, C.W.; Shetlar, M.R.; Shetlar, C.L. Alteration of Hypertrophic Scars Induced by Mechanical Pressure. Arch. Dermatol. 1975, 111, 60–64. [Google Scholar] [CrossRef] [PubMed]
- Reno, F.; Sabbatini, M.; Stella, M.; Magliacani, G.; Cannas, M. Effect of in vitro mechanical compression on Epilysin (matrix metalloproteinase-28) expression in hypertrophic scars. Wound Repair Regen. 2005, 13, 255–261. [Google Scholar] [CrossRef] [PubMed]


| Pressure Monitoring (n = 30) | Control (n = 30) | p | |
|---|---|---|---|
| Male:Female, n | 28:2 | 25:5 | 0.62 |
| Age (years) | 44.13 ± 14.51 | 46.03 ± 11.78 | 0.58 |
| TBSA (%) | 31.93 ± 16.82 | 35.57 ± 14.57 | 0.60 |
| Site of burn 0.70 | |||
| Arms, thigh | 3 (10) | 2 (7) | |
| Forearms, leg | 15 (50) | 16 (53) | |
| Hands, foot | 12 (40) | 12 (40) | |
| Mechanism of burn | 0.65 | ||
| Flame | 20 (67) | 18 (60) | |
| Electrical | 7 (23) | 7 (23) | |
| Scalding | 3 (10) | 5 (17) | |
| Duration (days) between the burn injury and treatment | 69.00 ± 30.05 | 65.67 ± 20.00 | 0.90 |
| Pressure Monitoring (n = 30) | Control (n = 30) | p | |
|---|---|---|---|
| Thickness (cm) | 0.19 ± 0.06 | 0.18 ± 0.05 | 0.96 |
| Melanin (AU) | 185.93 ± 78.49 | 159.60 ± 73.18 | 0.84 |
| Erythema (AU) | 517.57 ± 114.30 | 491.43 ± 100.11 | 0.78 |
| TEWL (g/h/m2) | 17.31 ± 5.45 | 15.92 ± 5.86 | 0.89 |
| Sebum | 33.30 ± 54.93 | 30.57 ± 46.52 | 0.27 |
| Skin distensibility | 0.66 ± 0.61 | 0.61 ± 0.58 | 0.90 |
| Biologic skin elasticity | 0.43 ± 0.23 | 0.42 ± 0.24 | 0.71 |
| Gross skin elasticity | 0.60 ± 0.29 | 0.62 ± 0.21 | 0.69 |
| Skin viscoelasticity | 0.49 ± 0.44 | 0.45 ± 0.20 | 0.77 |
| Pressure Monitoring (n = 30) | Control (n = 30) | p | |
| Thickness (cm) | −0.01 ± 0.08 | 0.06 ± 0.10 | 0.03 * |
| Melanin (AU) | 8.87 ± 86.31 | 15.80 ± 82.07 | 0.42 |
| Erythema (AU) | −93. 73 ± 116.43 | −32.73 ± 117.97 | 0.03 * |
| TEWL (g/h/m2) | 0.89 ± 6.65 | 0.72 ± 7.81 | 0.56 |
| Sebum | 74.97 ± 80.62 | 36.20 ± 93.63 | 0.02 * |
| Skin distensibility | 0.07 ± 0.52 | −0.08 ± 0.64 | 0.66 |
| Biologic skin elasticity | 0.01 ± 0.15 | 0.00 ± 0.19 | 0.83 |
| Gross skin elasticity | −0.01 ± 0.19 | −0.10 ± 0.29 | 0.10 |
| Skin viscoelasticity | 0.02 ± 0.47 | 0.08 ± 0.16 | 0.18 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joo, S.Y.; Cho, Y.S.; Yoo, J.W.; Kim, Y.H.; Sabangan, R.; Lee, S.Y.; Seo, C.H. Clinical Utility of the Portable Pressure-Measuring Device for Compression Garment Pressure Measurement on Hypertrophic Scars by Burn Injury during Compression Therapy. J. Clin. Med. 2022, 11, 6743. https://doi.org/10.3390/jcm11226743
Joo SY, Cho YS, Yoo JW, Kim YH, Sabangan R, Lee SY, Seo CH. Clinical Utility of the Portable Pressure-Measuring Device for Compression Garment Pressure Measurement on Hypertrophic Scars by Burn Injury during Compression Therapy. Journal of Clinical Medicine. 2022; 11(22):6743. https://doi.org/10.3390/jcm11226743
Chicago/Turabian StyleJoo, So Young, Yoon Soo Cho, Ji Won Yoo, Yi Hyun Kim, Rachael Sabangan, Seung Yeol Lee, and Cheong Hoon Seo. 2022. "Clinical Utility of the Portable Pressure-Measuring Device for Compression Garment Pressure Measurement on Hypertrophic Scars by Burn Injury during Compression Therapy" Journal of Clinical Medicine 11, no. 22: 6743. https://doi.org/10.3390/jcm11226743
APA StyleJoo, S. Y., Cho, Y. S., Yoo, J. W., Kim, Y. H., Sabangan, R., Lee, S. Y., & Seo, C. H. (2022). Clinical Utility of the Portable Pressure-Measuring Device for Compression Garment Pressure Measurement on Hypertrophic Scars by Burn Injury during Compression Therapy. Journal of Clinical Medicine, 11(22), 6743. https://doi.org/10.3390/jcm11226743








