Potential Values of Circulating microRNA-21 to Predict Early Recurrence in Patients with Colorectal Cancer after Treatments
Abstract
:1. Introduction
2. Materials and Methods
2.1. Enrolment of Clinical Patients and Healthy Volunteers
2.2. Plasma Preparation, CTC/CTM Detection, Exosome Isolation and Total microRNAs Extraction from Plasma and Exosome
2.2.1. Plasma Preparation and CTC/CTM Detection
2.2.2. Exosome Extraction and Identification
2.2.3. miRNAs Extraction and miR-21 Gene Expression
2.3. Statistical Analysis
3. Results
3.1. Demographics of Patients with CRC
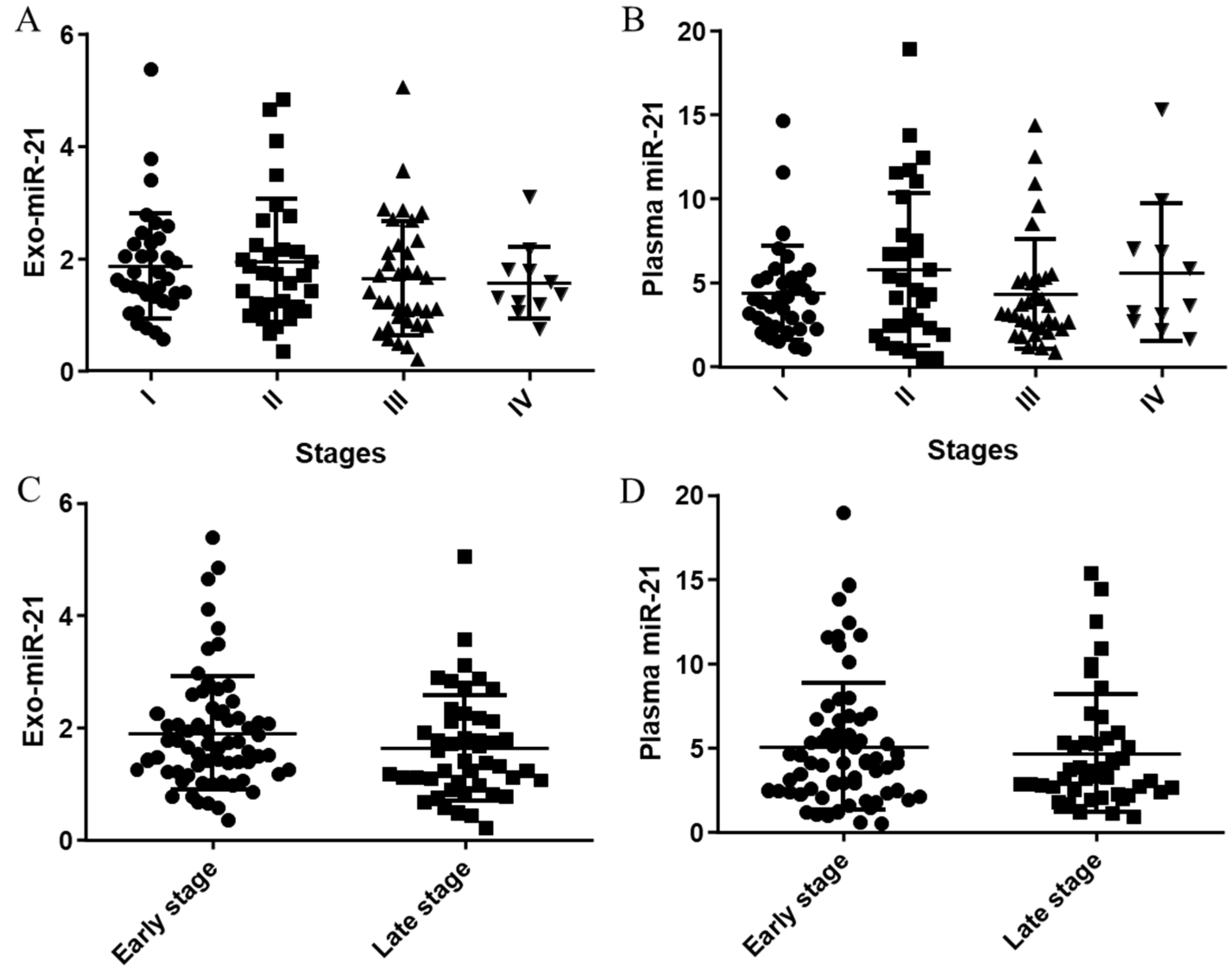
3.2. Distributions and Correlations of Plasma/Exosomal miR-21 Expression in Stages
3.2.1. Distributions of Plasma/Exosomal miR-21 Expressions
3.2.2. Correlations between Plasma miR-21 and exo-miR-21 Expressions
3.3. Correlations between Plasma/Exosomal miR-21 Expressions and Other Biomarkers
3.3.1. Correlations between Plasma/Exosomal miR-21 Expressions and Enumerations of EpCAM Positive CTCs (EpCTCs)
3.3.2. Correlations between Plasma/Exosomal miR-21 Expressions and Other Biomarkers
3.4. Prediction of CRC Recurrence in Patients Stratified by Biomarkers Individually and Combined
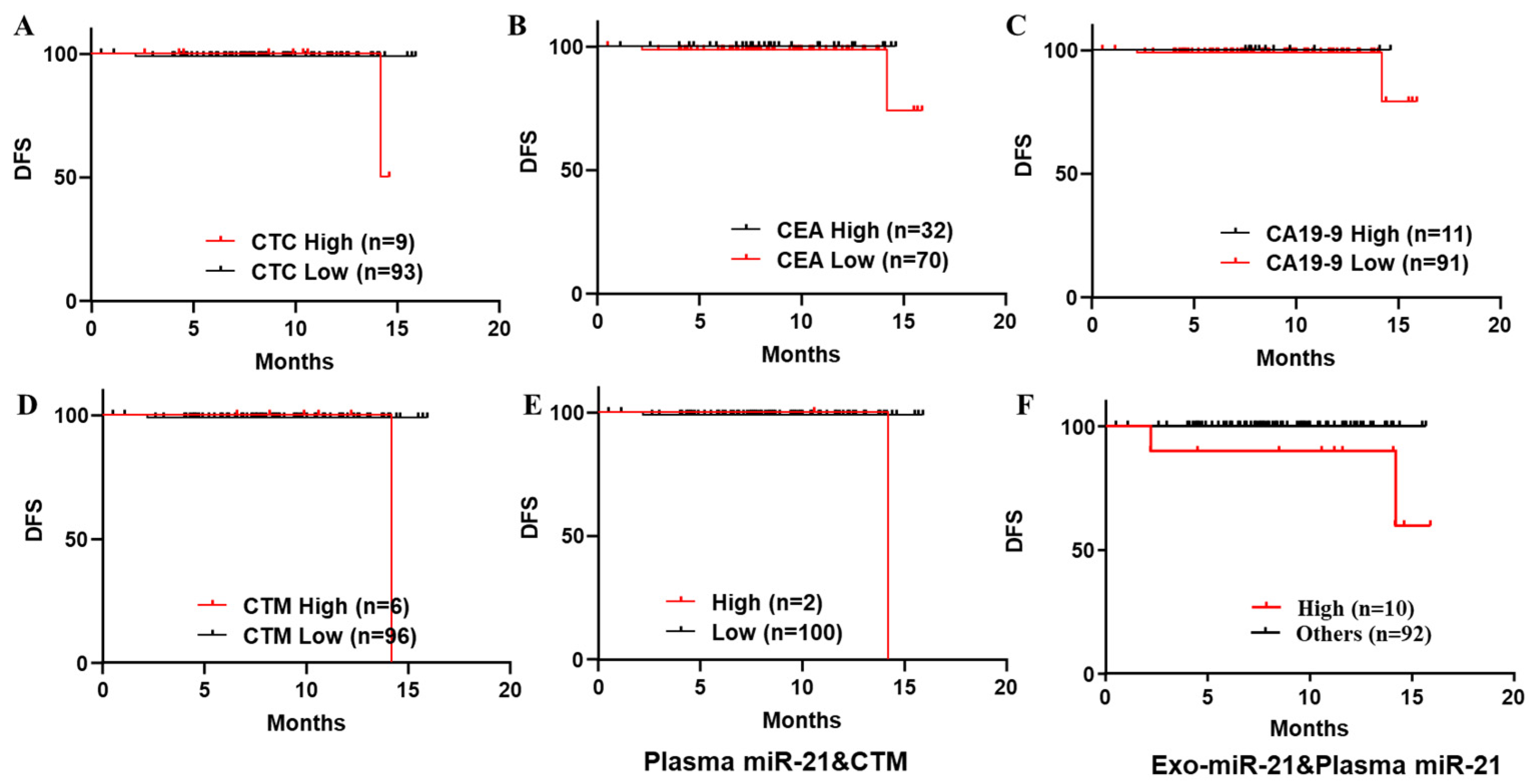
3.4.1. Receiver Operating Characteristic (ROC) Curves and Kaplan–Meier Survival Analyses of Plasma/exo-miR-21 Individually to Predict CRC Recurrence in Patients
3.4.2. Kaplan–Meier Survival Analyses on DFS of a Series of Biomarkers Individually and Combined to Predict Recurrence in Patients with Stage I to III CRC
3.4.3. Kaplan–Meier Survival Analyses on PFS of a Series of Biomarkers Individually and Combined to Predict Recurrence in Patients with Stage IV CRC
3.4.4. Recurrence Rate and Odds Ratio in Predicting CRC Recurrence in Patients in All Stages and Late Stages
Recurrence Rate and Odds Ratio in CRC Patients in ALL Stages
Recurrence Rate and Odds Ratio in CRC Patients in Late Stages
4. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Ladabaum, U.; Dominitz, J.A.; Kahi, C.; Schoen, R.E. Strategies for colorectal cancer screening. Gastroenterology 2020, 158, 418–432. [Google Scholar] [CrossRef]
- Vuik, F.E.R.; Nieuwenburg, S.A.V.; Moen, S.; Spada, C.; Senore, C.; Hassan, C.; Pennazio, M.; Rondonotti, E.; Pecere, S.; Kuipers, E.J.; et al. Colon capsule endoscopy in colorectal cancer screening: A systematic review. Endoscopy 2021, 53, 815–824. [Google Scholar] [CrossRef]
- Shaukat, A.; Kahi, C.J.; Burke, C.A.; Rabeneck, L.; Sauer, B.G.; Rex, D.K. ACG Clinical Guidelines: Colorectal Cancer Screening 2021. Am. J. Gastroenterol. 2021, 116, 458–479. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Goding Sauer, A.; Fedewa, S.A.; Butterly, L.F.; Anderson, J.C.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 145–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melli, F.; Bartolini, I.; Risaliti, M.; Tucci, R.; Ringressi, M.N.; Muiesan, P.; Taddei, A.; Amedei, A. Evaluation of prognostic factors and clinicopathological patterns of recurrence after curative surgery for colorectal cancer. World J. Gastrointest. Surg. 2021, 13, 50–75. [Google Scholar] [CrossRef]
- Howlader, N.; Noone, A.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M.E.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.R.; et al. SEER Cancer Statistics Review, 1975–2016; National Cancer Institute: Bethesda, MD, USA, 2019; Volume 1. [Google Scholar]
- Nassar, F.J.; Msheik, Z.S.; Itani, M.M.; Helou, R.E.; Hadla, R.; Kreidieh, F.; Bejjany, R.; Mukherji, D.; Shamseddine, A.; Nasr, R.R.; et al. Circulating miRNA as Biomarkers for Colorectal Cancer Diagnosis and Liver Metastasis. Diagnostics 2021, 11, 341. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, C.; Hayes, D.F. Circulating tumor cells. In Novel Biomarkers in the Continuum of Breast Cancer; Springer: Berlin/Heidelberg, Germany, 2016; pp. 235–258. [Google Scholar]
- Ghai, V.; Lee, I.; Wang, K. Circulating miRNAs as tumor biomarkers. In Oncogenomics; Elsevier: Amsterdam, The Netherlands, 2019; pp. 191–206. [Google Scholar]
- Balakrishnan, A.; Koppaka, D.; Anand, A.; Deb, B.; Grenci, G.; Viasnoff, V.; Thompson, E.W.; Gowda, H.; Bhat, R.; Rangarajan, A.; et al. Circulating Tumor Cell cluster phenotype allows monitoring response to treatment and predicts survival. Sci. Rep. 2019, 9, 7933. [Google Scholar] [CrossRef]
- Amintas, S.; Bedel, A.; Moreau-Gaudry, F.; Boutin, J.; Buscail, L.; Merlio, J.P.; Vendrely, V.; Dabernat, S.; Buscail, E. Circulating tumor cell clusters: United we stand divided we fall. Int. J. Mol. Sci. 2020, 21, 2653. [Google Scholar] [CrossRef] [Green Version]
- de Miguel Pérez, D.; Martínez, A.R.; Palomo, A.O.; Delgado Ureña, M.; Garcia Puche, J.L.; Robles Remacho, A.; Exposito Hernandez, J.; Lorente Acosta, J.A.; Ortega Sánchez, F.G.; Serrano, M.J. Extracellular vesicle-miRNAs as liquid biopsy biomarkers for disease identification and prognosis in metastatic colorectal cancer patients. Sci. Rep. 2020, 10, 1–13. [Google Scholar]
- Chu, H.Y.; Yang, C.Y.; Yeh, P.H.; Hsu, C.J.; Chang, L.W.; Chan, W.J.; Lin, C.P.; Lyu, Y.Y.; Wu, W.C.; Lee, C.W.; et al. Highly Correlated Recurrence Prognosis in Patients with Metastatic Colorectal Cancer by Synergistic Consideration of Circulating Tumor Cells/Microemboli and Tumor Markers CEA/CA19-9. Cells 2021, 10, 1149. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, Y.; Iinuma, H.; Tsukamoto, M.; Matsuda, K.; Hashiguchi, Y. Clinical significance of microRNA-21 as a biomarker in each Dukes’ stage of colorectal cancer. Oncol. Rep. 2015, 33, 573–582. [Google Scholar] [CrossRef] [Green Version]
- Sazanov, A.A.; Kiselyova, E.V.; Zakharenko, A.A.; Romanov, M.N.; Zaraysky, M.I. Plasma and saliva miR-21 expression in colorectal cancer patients. J. Appl. Genet. 2017, 58, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Uratani, R.; Toiyama, Y.; Kitajima, T.; Kawamura, M.; Hiro, J.; Kobayashi, M.; Tanaka, K.; Inoue, Y.; Mohri, Y.; Mori, T.; et al. Diagnostic Potential of Cell-Free and Exosomal MicroRNAs in the Identification of Patients with High-Risk Colorectal Adenomas. PLoS ONE 2016, 11, e0160722. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, M.; Iinuma, H.; Yagi, T.; Matsuda, K.; Hashiguchi, Y. Circulating exosomal microRNA-21 as a biomarker in each tumor stage of colorectal cancer. Oncology 2017, 92, 360–370. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Zhu, J.; Mao, Y.; Li, X.; Hu, B.; Zhang, D. Associations between the epithelial-mesenchymal transition phenotypes of circulating tumor cells and the clinicopathological features of patients with colorectal cancer. Dis. Markers 2017, 2017, 9474532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, R.-J.; Hong, H.-J.; Sun, J.; Yu, C.R.; Liu, H.S.; Li, P.Y.; Zheng, M.H. Detection and Clinical Value of Circulating Tumor Cells as an Assisted Prognostic Marker in Colorectal Cancer Patients. Cancer Manag. Res. 2021, 13, 4567. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, E.A.; Silva, V.S.; Braun, A.C.; Gasparini, V.A.; Kupper, B.E.C.; Tariki, M.S.; Tarazona, J.G.R.; Takahashi, R.M.; Júnior, S.A.; Chinen, L.T.D. A higher platelet-to-lymphocyte ratio is prevalent in the presence of circulating tumor microemboli and is a potential prognostic factor for non-metastatic colon cancer. Transl. Oncol. 2021, 14, 100932. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhao, L.; Zhou, P.; Ma, H.; Huang, F.; Jin, M.; Dai, X.; Zheng, X.; Huang, S.; Zhang, T. Circulating tumor microemboli (CTM) and vimentin+ circulating tumor cells (CTCs) detected by a size-based platform predict worse prognosis in advanced colorectal cancer patients during chemotherapy. Cancer Cell Int. 2017, 17, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bu, H.; He, D.; He, X.; Wang, K. Exosomes: Isolation, Analysis, and Applications in Cancer Detection and Therapy. Chembiochem 2019, 20, 451–461. [Google Scholar] [CrossRef] [PubMed]
- Junqueira-Neto, S.; Batista, I.A.; Costa, J.L.; Melo, S.A. Liquid Biopsy beyond Circulating Tumor Cells and Cell-Free DNA. Acta Cytol. 2019, 63, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Allard, W.J.; Matera, J.; Miller, M.C.; Repollet, M.; Connelly, M.C.; Rao, C.; Tibbe, A.G.; Uhr, J.W.; Terstappen, L.W. Tumor cells circulate in the peripheral blood of all major carcinomas but not in healthy subjects or patients with nonmalignant diseases. Clin. Cancer Res. 2004, 10, 6897–6904. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lianidou, E.S.; Strati, A.; Markou, A. Circulating tumor cells as promising novel biomarkers in solid cancers. Crit. Rev. Clin. Lab. Sci. 2014, 51, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.Y.; Lu, L.S.; Cho, W.; Wu, S.Y.; Chang, Y.C.; Lin, C.P.; Yang, C.Y.; Lin, C.H.; Jiang, J.K.; Tseng, F.G. Enumerating Circulating Tumor Cells with a Self-Assembled Cell Array (SACA) Chip: A Feasibility Study in Patients with Colorectal Cancer. Cancers 2019, 11, 56. [Google Scholar] [CrossRef] [Green Version]
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.; et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef] [Green Version]
- Krebs, M.G.; Renehan, A.G.; Backen, A.; Gollins, S.; Chau, I.; Hasan, J.; Valle, J.W.; Morris, K.; Beech, J.; Ashcroft, L.; et al. Circulating Tumor Cell Enumeration in a Phase II Trial of a Four-Drug Regimen in Advanced Colorectal Cancer. Clin. Colorectal Cancer 2015, 14, 115–122.e1–2. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, C.; Meropol, N.; Punt, C.; Iannotti, N.; Saidman, B.H.; Sabbath, K.D.; Gabrail, N.Y.; Picus, J.; Morse, M.A.; Mitchell, E.J.A.O.O.; et al. Relationship among circulating tumor cells, CEA and overall survival in patients with metastatic colorectal cancer. Ann. Oncol. 2013, 24, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Ghossein, R.A.; Bhattacharya, S.; Rosai, J. Molecular detection of micrometastases and circulating tumor cells in solid tumors. Clin. Cancer Res. 1999, 5, 1950–1960. [Google Scholar]
- Umer, M.; Vaidyanathan, R.; Nguyen, N.T.; Shiddiky, M.J.A. Circulating tumor microemboli: Progress in molecular understanding and enrichment technologies. Biotechnol. Adv. 2018, 36, 1367–1389. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ying, S.-Y.; Chang, D.C.; Lin, S.-L. The microRNA (miRNA): Overview of the RNA genes that modulate gene function. Mol. Biotechnol. 2008, 38, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.C.; Feinbaum, R.L.; Ambros, V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 1993, 75, 843–854. [Google Scholar] [CrossRef]
- Lau, N.C.; Lim, L.P.; Weinstein, E.G.; Bartel, D.P. An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditis elegans. Science 2001, 294, 858–862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ying, S.-Y.; Chang, D.C.; Lin, S.-L. The MicroRNA. In MicroRNA Protocols; Springer: Berlin/Heidelberg, Germany, 2018; pp. 1–25. [Google Scholar]
- Zhang, B.; Pan, X.; Cobb, G.P.; Anderson, T.A. microRNAs as oncogenes and tumor suppressors. Dev. Biol. 2007, 302, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Schee, K.; Fodstad, Ø.; Flatmark, K. MicroRNAs as biomarkers in colorectal cancer. Am. J. Pathol. 2010, 177, 1592–1599. [Google Scholar] [CrossRef]
- Chen, B.; Xia, Z.; Deng, Y.N.; Yang, Y.; Zhang, P.; Zhu, H.; Xu, N.; Liang, S. Emerging microRNA biomarkers for colorectal cancer diagnosis and prognosis. Open Biol. 2019, 9, 180212. [Google Scholar] [CrossRef] [Green Version]
- Allegra, A.; Alonci, A.; Campo, S.; Penna, G.; Petrungaro, A.; Gerace, D.; Musolino, C. Circulating microRNAs: New biomarkers in diagnosis, prognosis and treatment of cancer (review). Int. J. Oncol. 2012, 41, 1897–1912. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, T.; Leong, M.H.; Harms, B.; Kennedy, G.; Chen, L. MicroRNA-21 as a potential colon and rectal cancer biomarker. World J. Gastroenterol. 2013, 19, 5615. [Google Scholar] [CrossRef]
- Ogata-Kawata, H.; Izumiya, M.; Kurioka, D.; Honma, Y.; Yamada, Y.; Furuta, K.; Gunji, T.; Ohta, H.; Okamoto, H.; Sonoda, H.; et al. Circulating exosomal microRNAs as biomarkers of colon cancer. PLoS ONE 2014, 9, e92921. [Google Scholar] [CrossRef]
- Clancy, C.; Joyce, M.R.; Kerin, M.J. The use of circulating microRNAs as diagnostic biomarkers in colorectal cancer. Cancer Biomark. 2015, 15, 103–113. [Google Scholar] [CrossRef]
- Fukada, M.; Matsuhashi, N.; Takahashi, T.; Sugito, N.; Heishima, K.; Yoshida, K.; Akao, Y. Postoperative changes in plasma miR21-5p as a novel biomarker for colorectal cancer recurrence: A prospective study. Cancer Sci. 2021, 112, 4270. [Google Scholar] [CrossRef]
- Schetter, A.J.; Leung, S.Y.; Sohn, J.J.; Zanetti, K.A.; Bowman, E.D.; Yanaihara, N.; Yuen, S.T.; Chan, T.L.; Kwong, D.L.; Au, G.K.; et al. MicroRNA expression profiles associated with prognosis and therapeutic outcome in colon adenocarcinoma. JAMA 2008, 299, 425–436. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srivastava, A.; Filant, J.; Moxley, K.M.; Sood, A.; McMeekin, S.; Ramesh, R. Exosomes: A role for naturally occurring nanovesicles in cancer growth, diagnosis and treatment. Curr. Gene Ther. 2015, 15, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Hannafon, B.N.; Ding, W.Q. Intercellular communication by exosome-derived microRNAs in cancer. Int. J. Mol. Sci. 2013, 14, 14240–14269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tovar-Camargo, O.A.; Toden, S.; Goel, A. Exosomal microRNA Biomarkers: Emerging Frontiers in Colorectal and Other Human Cancers. Expert Rev. Mol. Diagn. 2016, 16, 553–567. [Google Scholar] [CrossRef] [Green Version]
- Whiteside, T.L. The potential of tumor-derived exosomes for noninvasive cancer monitoring. Expert Rev. Mol. Diagn. 2015, 15, 1293–1310. [Google Scholar] [CrossRef] [Green Version]
- Wubbolts, R.; Leckie, R.S.; Veenhuizen, P.T.; Schwarzmann, G.; Möbius, W.; Hoernschemeyer, J.; Slot, J.W.; Geuze, H.J.; Stoorvogel, W. Proteomic and biochemical analyses of human B cell-derived exosomes. Potential implications for their function and multivesicular body formation. J. Biol. Chem. 2003, 278, 10963–10972. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karlsen, T.A.; Aae, T.F.; Brinchmann, J.E. Robust profiling of microRNAs and isomiRs in human plasma exosomes across 46 individuals. Sci. Rep. 2019, 9, 19999. [Google Scholar] [CrossRef] [PubMed]
- Hur, J.Y.; Lee, K.Y. Characteristics and Clinical Application of Extracellular Vesicle-Derived DNA. Cancers 2021, 13, 3827. [Google Scholar] [CrossRef] [PubMed]
- Sabry, D.; El-Deek, S.E.; Maher, M.; El-Baz, M.A.; El-Bader, H.M.; Amer, E.; Hassan, E.A.; Fathy, W. and El-Deek, H.E. Role of miRNA-210, miRNA-21 and miRNA-126 as diagnostic biomarkers in colorectal carcinoma: Impact of HIF-1α-VEGF signaling pathway. Mol. Cell. Biochem. 2019, 454, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Shibutani, M.; Maeda, K.; Nagahara, H.; Ohtani, H.; Sakurai, K.; Toyokawa, T.; Kubo, N.; Tanaka, H.; Muguruma, K.; Ohira, M.; et al. Significance of CEA and CA19-9 combination as a prognostic indicator and for recurrence monitoring in patients with stage II colorectal cancer. Anticancer. Res. 2014, 34, 3753–3758. [Google Scholar] [PubMed]
- Wang, J.; Wang, X.; Yu, F.; Chen, J.; Zhao, S.; Zhang, D.; Yu, Y.; Liu, X.; Tang, H. and Peng, Z. Combined detection of preoperative serum CEA, CA19-9 and CA242 improve prognostic prediction of surgically treated colorectal cancer patients. Int. J. Clin. Exp. Pathol. 2015, 8, 14853–14863. [Google Scholar] [PubMed]
- Chiu, S.Y.; Hsieh, C.H.; You, J.F.; Chu, P.Y.; Hung, H.Y.; Chu, P.H. and Wu, M.H. Enhancing Prediction Performance by Add-On Combining Circulating Tumor Cell Count, CD45(neg) EpCAM(neg) Cell Count on Colorectal Cancer, Advance, and Metastasis. Cancers 2021, 13, 2521. [Google Scholar] [CrossRef]
- Pesta, M.; Kucera, R.; Topolcan, O.; Karlikova, M.; Houfkova, K.; Polivka, J.; Macanova, T.; Machova, I.; Slouka, D. and Kulda, V.Plasma microRNA Levels Combined with CEA and CA19-9 in the Follow-Up of Colorectal Cancer Patients. Cancers 2019, 11, 864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-C.; Yang, S.-H.; Lin, J.-K.; Lin, T.C.; Chen, W.S.; Jiang, J.K.; Wang, H.S. and Chang, S.C.Is it reasonable to add preoperative serum level of CEA and CA19-9 to staging for colorectal cancer? J. Surg. Res. 2005, 124, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Takakura, Y.; Ikeda, S.; Imaoka, Y.; Urushihara, T.; Itamoto, T. An elevated preoperative serum carbohydrate antigen 19-9 level is a significant predictor for peritoneal dissemination and poor survival in colorectal cancer. Colorectal Dis. 2015, 17, 417–425. [Google Scholar] [CrossRef] [PubMed]




| Exosome miR21 in PB | Plasma miR21 in PB | CTCs in PB | CTM in PB | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Total | High | Low | p | High | Low | p | High | Low | p | High | Low | p | |
| N = 113 | N = 37 | N = 76 | N =20 | N = 93 | N = 11 | N = 102 | N = 9 | N = 104 | |||||
| Age | 65.19 | 67.08 | 64.28 | 66.5 | 64.91 | 65.19 | 69.45 | 61.22 | 65.54 | ||||
| (39–93) | (42–93) | (39–90) | (42–93) | (39–92) | (49–85) | (39–92) | (40–83) | (39–93) | |||||
| Gender | |||||||||||||
| Male | 77 (68%) | 26 | 51 | 0.8311 | 13 | 64 | 0.7936 | 6 | 71 | 0.3231 | 6 | 71 | >0.9999 |
| Female | 36 (32%) | 11 | 25 | 7 | 29 | 5 | 31 | 3 | 33 | ||||
| TNM Stage | |||||||||||||
| I | 36 (32%) | 10 | 26 | 0.7189 | 3 | 33 | 0.0003 *** | 3 | 33 | 3 | 33 | 0.0788 | |
| II | 31 (27%) | 11 | 20 | 6 | 25 | 2 | 29 | 0.6896 | 1 | 30 | |||
| III | 35 (31%) | 11 | 24 | 4 | 31 | 4 | 31 | 2 | 33 | ||||
| IV | 11 (10%) | 5 | 6 | 7 | 4 | 2 | 9 | 3 | 8 | ||||
| T stage | |||||||||||||
| T1–T2 | 39 (35%) | 10 | 29 | 0.2947 | 3 | 36 | 0.0674 | 3 | 36 | 0.7454 | 3 | 36 | >0.9999 |
| T3–T4 | 74 (65%) | 27 | 47 | 17 | 57 | 8 | 66 | 6 | 68 | ||||
| N stage | |||||||||||||
| N0 | 70 (62%) | 23 | 47 | 0.4572 | 11 | 59 | 0.0133 * | 6 | 64 | 0.2162 | 6 | 64 | 0.9487 |
| N1 | 30 (27%) | 8 | 22 | 3 | 27 | 2 | 28 | 2 | 28 | ||||
| N2 | 13 (12%) | 6 | 7 | 6 | 7 | 3 | 10 | 1 | 12 | ||||
| M stage | |||||||||||||
| M0 | 102 (90%) | 32 | 70 | 0.4996 | 13 | 89 | 0.0004 *** | 9 | 93 | 0.2905 | 6 | 96 | 0.0422 * |
| M1 | 11 (10%) | 5 | 6 | 7 | 4 | 2 | 9 | 3 | 8 | ||||
| Tumor size (cm2) | |||||||||||||
| ≥5 | 24 (21%) | 12 | 12 | 0.0521 | 7 | 17 | 0.1302 | 5 | 19 | 0.0537 | 2 | 22 | >0.9999 |
| <5 | 89 (79%) | 25 | 64 | 13 | 76 | 6 | 83 | 7 | 82 | ||||
| Differentiation | |||||||||||||
| Poor | 5 (4%) | 1 | 4 | 0.8246 | 1 | 4 | 0.7619 | 1 | 4 | 0.2641 | 1 | 4 | 0.1449 |
| Moderate | 105 (93%) | 35 | 70 | 18 | 87 | 9 | 96 | 7 | 98 | ||||
| Well | 3 (3%) | 1 | 2 | 1 | 2 | 1 | 2 | 1 | 2 | ||||
| Location | |||||||||||||
| Right colon | 26 (23%) | 10 | 16 | 0.7366 | 4 | 22 | 0.3203 | 4 | 22 | 0.0781 | 2 | 24 | 0.8861 |
| Left colon | 57 (50%) | 17 | 40 | 8 | 49 | 2 | 55 | 4 | 53 | ||||
| Rectal | 30 (27%) | 10 | 20 | 8 | 22 | 5 | 25 | 3 | 27 | ||||
| CEA (5 ng/mL) | |||||||||||||
| >5 | 38 (34%) | 15 | 23 | 0.2958 | 9 | 29 | 0.2977 | 5 | 33 | 0.5032 | 8 | 30 | 0.0006 *** |
| ≤5 | 75 (66%) | 22 | 53 | 11 | 64 | 6 | 69 | 1 | 74 | ||||
| CA19-9 (U/mL) | |||||||||||||
| >37 | 15 (13%) | 10 | 5 | 0.0058 ** | 6 | 9 | 0.0257 * | 3 | 11 | 0.1371 | 1 | 13 | >0.9999 |
| ≤37 | 98 (87%) | 27 | 71 | 14 | 84 | 8 | 91 | 8 | 91 | ||||
| Treatments | |||||||||||||
| pre-operation | 18 (16%) | 6 | 12 | >0.9999 | 5 | 13 | 0.3086 | 2 | 16 | 0.6871 | 2 | 16 | 0.6335 |
| non-pre operation | 95 (84%) | 31 | 64 | 15 | 80 | 9 | 86 | 7 | 88 | ||||
| All Stages | Number of Cases | Recurrence Rate (%) | Odds Ratio | |
|---|---|---|---|---|
| 113 Cases | Recurrence (+) | Recurrence (−) | ||
| High Exosome miR-21 | 7 | 30 | 18.9 | 17.5 |
| Low Exosome miR-21 | 1 | 75 | 1.3 | p value = 0.0016 ** |
| High Plasma miR-21 | 7 | 12 | 37 | 54.3 |
| Low Plasma miR-21 | 1 | 93 | 1.1 | p value < 0.0001 **** |
| High CTC | 3 | 8 | 27.3 | 7.3 |
| Low CTC | 5 | 97 | 4.9 | p value = 0.0298 * |
| Presence of CTM | 2 | 7 | 22.2 | 4.7 |
| Absence of CTM | 6 | 98 | 5.8 | p value = 0.123 |
| CEA > 5 ng/mL | 4 | 34 | 10.5 | 2.1 |
| CEA ≤ 5 ng/mL | 4 | 71 | 5.3 | p value = 0.4388 |
| CA19-9 > 37 U/mL | 3 | 12 | 20 | 4.7 |
| CA19-9 ≤ 37 U/mL | 5 | 93 | 5.1 | p value = 0.071 |
| High Exosome miR-21 | 6 | 8 | 42.9 | 36.4 p value < 0.0001 **** |
| High Plasma miR-21 | ||||
| Others | 2 | 97 | 2 | |
| High Exosome miR-21 | 3 | 3 | 50 | 20.4 p value = 0.0043 ** |
| High Plasma miR-21 | ||||
| High CTC | ||||
| Others | 5 | 102 | 4.7 | |
| High Exosome miR-21 | 1 | 1 | 50 | 14.9 p value = 0.1372 |
| High Plasma miR-21 | ||||
| With CTM | ||||
| Others | 7 | 104 | 6.3 | |
| High Exosome miR-21 | 4 | 5 | 44.4 | 20 p value = 0.0012 ** |
| High Plasma miR-21 | ||||
| CEA > 5 ng/mL | ||||
| Others | 4 | 100 | 3.8 | |
| High Exosome miR-21 | 3 | 3 | 50 | 20.4 p value = 0.0043 ** |
| High Plasma miR-21 | ||||
| CA19-9 > 37 U/mL | ||||
| Others | 5 | 102 | 4.7 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hao, Y.-J.; Yang, C.-Y.; Chen, M.-H.; Chang, L.-W.; Lin, C.-P.; Lo, L.-C.; Huang, S.-C.; Lyu, Y.-Y.; Jiang, J.-K.; Tseng, F.-G. Potential Values of Circulating microRNA-21 to Predict Early Recurrence in Patients with Colorectal Cancer after Treatments. J. Clin. Med. 2022, 11, 2400. https://doi.org/10.3390/jcm11092400
Hao Y-J, Yang C-Y, Chen M-H, Chang L-W, Lin C-P, Lo L-C, Huang S-C, Lyu Y-Y, Jiang J-K, Tseng F-G. Potential Values of Circulating microRNA-21 to Predict Early Recurrence in Patients with Colorectal Cancer after Treatments. Journal of Clinical Medicine. 2022; 11(9):2400. https://doi.org/10.3390/jcm11092400
Chicago/Turabian StyleHao, Yun-Jie, Chih-Yung Yang, Ming-Hsien Chen, Lu-Wey Chang, Chien-Ping Lin, Liang-Chuan Lo, Sheng-Chieh Huang, You-You Lyu, Jeng-Kai Jiang, and Fan-Gang Tseng. 2022. "Potential Values of Circulating microRNA-21 to Predict Early Recurrence in Patients with Colorectal Cancer after Treatments" Journal of Clinical Medicine 11, no. 9: 2400. https://doi.org/10.3390/jcm11092400






