Abstract
In recent years, a significant improvement in left ventricular assist device (LVAD) technology has occurred, and the continuous-flow devices currently used can last more than 10 years in a patient. Current studies report that the 5-year survival rate after LVAD implantation approaches that after a heart transplant. However, the outcome is influenced by the correct selection of the patients, as well as the choice of the optimal time for implantation. This review summarizes the indications, the red flags for prompt initiation of LVAD evaluation, and the principles for appropriate patient screening.
1. Introduction
Management options for patients with advanced heart failure with reduced ejection fraction (advHFrEF) include orthotopic heart transplantation [1,2,3], long-term mechanical circulatory support [4], and intravenous inotropes [5]. Durable mechanical circulatory support through a left ventricular assist device (LVAD) has recently experienced significant technical and scientific development with the advent of fully magnetically levitated centrifugal pump technology. Recent studies indicate that 5-year survival after LVAD implantation approaches that after a heart transplant (5-year survival of 72.5% after heart transplant vs. 58.4 after LVAD implantation) [6]. However, enthusiasm resulting from improved outcomes after LVAD implantation has been tempered by postimplant complications, including stroke [7], infection [8], device thrombosis [9], and right ventricular failure (RVF) [10]. Many of these risks may be mitigated or at least anticipated by the careful selection of candidate patients for LVAD implantation. To provide guidance for general cardiologists, not only advHF specialists, in the proper selection of patients for LVADs, in this review, we provide an overview of the evaluation of advHFrEF patients for LVAD implantation.
2. Indications for LVADs
Generally, consideration of LVAD implantation is indicated in all patients with persistent NYHA class III–IV despite guideline-directed medical therapy (GDMT) and poor medium-term prognoses (Table 1) [11]. However, the Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) has further classified NYHA III–IV patients with AdvHFrEF [12]. According to this classification, the best outcome after LVAD implantation is achieved in INTERMACS class 2–3 patients [13]. However, recent technological advances have reduced postoperative complications and have paved the way for “early” LVAD implantation, which is now also being considered for patients in INTERMACS class 4 [14] (Figure 1).

Table 1.
Indications to LVADs according to international guidelines. ISHLT: International Society Heart and Lung Transplantation; AdvHFrEF: advanced heart failure with reduced ejection fraction; NYHA: New York Heart Association; GDMT: guideline-directed medical therapy; ACC/AHA: American College of Cardiology/American Heart Association; LVEF: left ventricular ejection fraction; CI: cardiac index; PCWP: pulmonary capillary wedge pressure; EACTS: European Association for Cardiothoracic Surgery; INTERMACS: Interagency Registry for Mechanically Assisted Circulatory Support; CPET: cardiopulmonary exercise test, COR: Class of Recommendation, LOE: Level of Evidence, NA: Not Available.
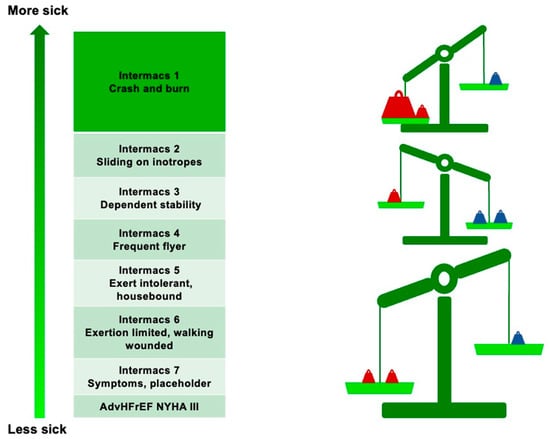
Figure 1.
Risk/benefit ratio of LVAD implantation according to INTERMACS class. Red weights represent the risks; blue weights represent the benefits.
There are three main indications for LVAD implantation:
- ►
- Bridge to Transplant (BTT): Indicated in patients on the orthotopic heart transplant list unable to maintain adequate end-organ perfusion with GDMT alone;
- ►
- Bridge to Candidacy (BTC): Suitable in selected patients with temporary contraindications to orthotopic heart transplantation that could be reversed after support with an LVAD (e.g., increased pulmonary vascular resistance, functional kidney disease);
- ►
- Destination Therapy (DT): Recommended in patients with an absolute and permanent contraindication to orthotopic heart transplantation, in whom an LVAD is used as a long-term therapy.
3. Clues to Prompt Initiation of LVAD Evaluation
Deriving the maximal mortality and morbidity reduction from LVAD implantation depends on the timing of the implant [15]. Proceeding with LVAD implantation too early in a patient’s heart failure course risks obviating the known benefit of pharmacologic and device guideline-directed therapy while exposing the patient to unnecessary perioperative risk. In contrast, if the LVAD is implanted too late in the course of the disease, the postoperative prognosis may be compromised by multiorgan dysfunction due to prolonged advHFrEF status [16]. Some degree of progressive multiorgan dysfunction is nearly ubiquitous in patients with end-stage heart failure and must be carefully evaluated when selecting candidate patients for LVADs. Therefore, identifying and appropriately referring patients with advHFrEF to centers capable of advanced therapies is critical to performing LVAD implantation in the “golden window” and, thus, to maximizing benefits and minimizing surgical procedure risks [17]. To help identify patients who may benefit from an LVAD at an appropriate time, several clinical, laboratory, and echocardiographic markers have been identified as “red flags”, indicating that the conventional approach is not enough for the patient and that LVAD implantation should be considered (Table 2).

Table 2.
Red flags that indicate the need to consider an LVAD implant.
3.1. Clinical and Laboratory Red Flags
Two or more episodes of worsening heart failure in the last 12 months indicates a poor prognosis in patients with advHFrEF [18]. A retrospective analysis involving 14,374 patients hospitalized for HF showed that patients with two or more hospitalizations for HF had a 1-year mortality rate of >40% [19]. More recently, a Swedish registry that enrolled 1801 patients with HF showed that worsening HF (defined as hospitalization for HF or an urgent need for intravenous diuretics) is associated with a reduction in the 5-year survival rate, particularly in patients with a left ventricular ejection fraction of ≤25% (log-rank p < 0.0001) [20].
The onset of intolerance to disease-modifier drugs due to symptomatic hypotension, resulting in the reduction or discontinuation of guideline-directed medical therapy (GDMT), is associated with a poor prognosis [21] unless it is due to hypovolemia [22]. In a retrospective analysis including 259 adult patients discharged from hospital after an episode of worsening heart failure, it was shown that those without an angiotensin-converting enzyme inhibitor because of hypotension or renal intolerance had a mortality rate higher than that of patients on angiotensin-converting enzyme inhibitors (57% vs. 22%; p = 0.0001) [23].
Inotrope dependence (the onset of symptomatic hypotension, recurrent congestive symptoms, or worsening renal function when inotropes are withdrawn) is a clear negative prognostic marker in patients with worsening advHFrEF [24] and should prompt the consideration of advanced therapies, including LVADs and/or heart transplant.
A high diuretic dose or the need to escalate the diuretic dose is also associated with poor prognosis. A hallmark study that enrolled 1354 patients with advHFrEF showed that patients in the highest quartile for diuretic dose (i.e., ≥160 mg of furosemide/daily) had the worst prognosis [25].
Arrhythmic storms (>3 episodes of sustained ventricular tachycardia or ventricular fibrillation in 24 h) are associated with a poor prognosis, mainly if they occur in patients in New York Heart Association (NYHA) functional class III–IV [26] or in a patient with prior ablation of the arrhythmic substrate [27]. Careful consideration of the arrhythmic substrate is required during evaluation, as ventricular tachyarrhythmias after LVAD implantation may also pose considerable risk for poor outcomes.
Numerous studies have shown that hyponatremia is a marker of poor prognosis in patients with acute heart failure [28,29]. More recently, the prognostic importance of persistent hyponatremia was documented in patients with advHFrEF in the ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial. In fact, in this study, the persistence of hyponatremia was associated with increased all-cause mortality and hospitalizations (73% vs. 50%; HR, 1.54) when compared with normonatremic patients [30].
The persistence of elevated plasma levels of NT-proBNP despite GDMT is associated with poor prognosis in patients with advHFrEF. In a prospective study that enrolled 550 patients with heart failure of ischemic and non-ischemic etiology, NT-proBNP levels of >5000 pg/mL were associated with increased mortality (mortality rate of 28.4% per year), which markedly discriminated advHFrEF candidates for LVAD implantation or urgent heart transplantation [31]. Furthermore, in the BioSHiFT (Role of Biomarkers and Echocardiography in Prediction of Prognosis of Chronic HF Patients) study, an increase in serial assessments of plasma NT-proBNP levels was associated with cardiac death, heart transplantation, or LVAD implantation [32].
3.2. Echocardiographic Red Flags
Certain echocardiographic features may suggest the presence of an end-stage form of heart failure, such as the following [33]:
- ►
- Marked left ventricular dilatation (left ventricular end-diastolic diameter of >80 mm);
- ►
- Left ventricular ejection fraction of ≤25%;
- ►
- Persistent restrictive mitral filling patterns or echocardiographic findings suggesting pulmonary hypertension despite aggressive diuretic therapy and therapy with inodilators (i.e., milrinone, levosimendan) [34].
Despite the importance of echocardiographic red flags, they should always be contextualized in the clinical scenario to best determine whether a patient warrants referral for the consideration of LVADs or other advanced therapies.
3.3. Invasive Hemodynamics Red Flags
Right heart catheterization is often used in patients with suspected advHFrEF to quantify the degree of hemodynamic dysfunction [35].
The persistence of a reduced cardiac index or increased left ventricular filling pressures despite the aggressive use of intravenous diuretics and inodilators is an important predictor of poor outcomes [36] and warrants evaluation for an LVAD.
4. Assessment of Patients
Although the evaluation of patients for LVADs should be performed in specialized centers for advHFrEF treatment, we consider it useful for the clinical cardiologist to be familiar with the basic principles of LVAD preimplant evaluation. Therefore, in this session, we practically cover the rationale for the primary investigations that a patient candidate for LVAD implantation undergoes in the preoperative phase. The class of recommendations (COR) and level of evidence (LOE) reported for each single indication are provided according to the latest recommendations by the International Society for Heart and Lung Transplantation [15].
4.1. Blood Investigation
Blood analysis represents an important step for the evaluation of patients with advHFrEF as potential candidates for LVAD implantation; mainly, the following parameters should be evaluated:
- ►
- Kidney function: Because the presence of advanced chronic kidney disease (CKD) strongly affects the LVAD post implantation prognosis [37], evaluation of serum creatinine blood urea nitrogen, creatinine clearance, and proteinuria is recommended in all patients (COR I, LOE C). Such assessments should be done in hemodynamically stable patients or after an adequate period of stabilization with diuretics, inotropes, and/or temporary mechanical circulatory support, if clinically indicated (COR I, LOE C). In patients with CKD status >IV–V (i.e., an estimated glomerular filtration rate of <30 mL/min/1.73 m2), the implantation of an LVAD should also be carefully evaluated by a multidisciplinary team (COR IIa, LOE C) as the anticipation of permanent dialysis is a contraindication to LVAD implantation (COR III, LOE C).
- ►
- Hepatic function: Severe hepatic dysfunction is strongly related to an adverse prognosis post-LVAD implantation [38], so a liver enzyme and hepatic protein synthesis evaluation is mandatory in all patients.
- ►
- Blood glucose and glycated hemoglobin: Diabetes has been associated with infection and late mortality in LVAD patients [39], so in all LVAD candidates, the presence of diabetes should be carefully evaluated (COR I, LOE C). Patients with uncontrolled diabetes should undergo optimization of their glycemic control (endocrine expertise recommended; COR I, LOE C), and patients with diabetes-related organ damage (proliferative retinopathy, vasculopathy, nephropathy) have a relative contraindication for LVAD implantation (COR IIb, LOE C).
- ►
- Platelet count and coagulation parameters: Because major post-LVAD complications include both thrombotic (particularly pump thrombosis) and hemorrhagic (mainly gastrointestinal) events [40,41], all patients should undergo preoperative evaluation of their international normalized ratio of prothrombin time, activated prothrombin time, and platelet count (COR I, LOE C). Baseline abnormalities in coagulation parameters not due to drug therapy or an altered platelet count (thrombocythemia and thrombocytosis) should be promptly evaluated to determine their etiology before LVAD implantation (COR I, LOE C).
4.2. Echocardiography
All patients should undergo echocardiography before LVAD implantation to assess their right ventricular size and function and to identify and quantify any coexisting valvulopathies (COR I, LOE C).
- ►
- Right ventricle evaluation: Most echocardiography-based predictors of right ventricular failure (RVF) post-LVAD implantation are based on assessing RV systolic–diastolic function. However, it was recently demonstrated that evaluating the RV–pulmonary artery (PA) parameters may increase the possibility of predicting postimplant RVF [42]. A systolic tricuspid annulus plane excursion (TAPSE) of <8 mm has been associated with RVF [43,44]; similarly, a right ventricular free wall s′ value on tissue Doppler of <5–8 cm/s [45] and an RV fractional area change (FAC) value of <25–30% [45] are associated with postimplant RVF. RV diastolic dysfunction also provides essential information on the occurrence of RVF after LVAD implantation. Indeed, a preimplant tricuspid E/e′ ratio of >10 predicts RVF [46]. Finally, recently, measures of the RV–PA coupling, which are less affected by afterload changes [47], have been shown to predict the onset of post-LVAD RHF. Specifically, a value of <24 obtained from the product of the peak systolic longitudinal strain rate of RV and the mean RV–PA gradient [48] or an RV load adaptation index of <14 [49] identifies patients with a failing RV unable to adapt to the increase in blood flow post-LVAD implantation.
- ►
- Valvular evaluation: Aortic valve regurgitation results in recirculation and reduced forward flow in patients with LVADs [50]. Therefore, the presence of more than mild aortic valve regurgitation is an indication for valve replacement with a biological valve (COR I, LOE C). Conversely, mild to moderate aortic stenosis does not impact LVAD function unless there is concomitant valve insufficiency [51]; therefore, it is common practice to replace the aortic valve only in patients with pre-existing severe aortic stenosis (COR I, LOE C). Significant mitral stenosis prevents inflow to the LVAD [52]; therefore, moderate or more significant mitral stenosis indicates the need for commissurotomy or valve replacement with a biological valve (COR I, LOE C). Mitral insufficiency, even of severe grade, is not a contraindication because left ventricular unloading almost always reduces the severity of mitral regurgitation [53,54]; on the other hand, since significant tricuspid regurgitation contributes to the occurrence of RVF [55,56], repair or replacement with a bioprosthetic valve should be performed in patients with a moderate or greater degree of tricuspid valve insufficiency that is anticipated to persist after LVAD support (COR IIb, LOE B).
Recommendations for patients with preexisting prosthetic valves are shown in Table 3.

Table 3.
Surgical recommendations for LVAD candidate patients with prosthetic valves. LVAD: left ventricular assist device.
Concomitant valve surgery with LVAD implantation increases the surgical risk due to the prolonged extracorporeal circulation time and increased incidence of postoperative RVF [57]. These factors must be considered during the patient selection process.
4.3. Other Imaging Techniques
In addition to transthoracic echocardiography, other imaging techniques should be used for the appropriate selection of candidate patients for LVAD implantation:
- ►
- Chest imaging: Evaluation of the intrathoracic anatomy is mandated in the preoperative assessment for LVAD surgery [58]. Therefore, chest radiography should be performed in all patients (COR I, LOE C). In patients with suspected intrathoracic abnormalities or previous cardiac surgery, computed tomography (CT) or magnetic resonance imaging should be used to better define the thoracic anatomy (COR I, LOE C).
- ►
- Liver imaging: In patients whose clinical history and/or laboratory investigations suggest hepatic dysfunction, screening for fibrosis or cirrhosis via ultrasonography or CT scan (COR I, LOE C) is necessary. Since fibrosis in the absence of significant portal hypertension may not be a contraindication to LVAD implantation [59], especially in the case of a normal hepatic synthetic capacity, a consultation with a hepatologist is indicated in such patients (COR I, LOE C). Conversely, cirrhosis or advanced hepatic dysfunction represents a contraindication to LVADs. A biopsy may be required to assess for cirrhosis. An analysis of 524 advHFrEF patients undergoing LVAD implantation showed that patients who had a MELDIX (End-Stage Liver Disease eXcluding International Normalized Ratio model) score of >14 had poorer short- and long-term survival, as well as an increased risk of early right ventricular dysfunction [60]. For this reason, patients with confirmed cirrhosis or an increased MELD score of >14 are not eligible for an LVAD (COR II, LOE B).
- ►
- Gastrointestinal imaging: Gastrointestinal bleeding is present in 15–30% of LVAD patients; the rate is higher in patients with previous gastrointestinal bleeding or colon polyposis [61]. Therefore, in patients with precedent gastrointestinal bleeding or with suspected unexplained iron deficiency anemia [62], screening with upper and lower endoscopy should be considered (COR IIa, LOE C).
4.4. Invasive Hemodynamics
An invasive hemodynamics assessment by right heart catheterization should be performed in all patients before LVAD implantation (COR I, LOE C) to measure their intracardiac filling pressures and assess their RV function [63]. The main hemodynamic parameters that predict RVF after LVAD implantation are shown in Table 4. In patients in whom these parameters indicate a high risk of RVF, optimization with diuretics, vasopressors/inotropes, dialysis, or temporary percutaneous mechanical circulatory support should be performed (COR I, LOE C). After optimization, if the RV function remains suboptimal, biventricular assist device support should be considered [64].

Table 4.
Hemodynamics-based parameters associated with right ventricular dysfunction after LVAD implantation. CVP: central venous pressure; PCWP: pulmonary capillary wedge pressure; MAP: mean arterial pressure; PAPi: pulmonary artery pulsatility index; RVSWI: right ventricular stroke work index; SVI: stroke volume index; PACI: pulmonary artery compliance index.
4.5. Psychosocial Evaluation
All patients who are candidates for LVADs should undergo a comprehensive psychosocial assessment (COR I, LOE C) to identify the following:
- ►
- Nonadherence to drug therapy;
- ►
- Unsafe home environment;
- ►
- Absence of social support;
- ►
- Presence of mental illness;
- ►
- Presence of drug or alcohol dependence.
All the above factors are associated with worse outcomes after LVAD implantation, so the candidacy for an LVAD of a patient with one or more of these factors should be carefully considered by a multidisciplinary team (cardiologist, cardiac surgeon, anesthesiologist, psychologist/psychiatrist, internist) [65,66].
5. Conclusions
LVAD implantation represents a valuable therapeutic option in carefully selected patients with advHFrEF. Technological innovations and improved implantation techniques ensure an immediate improvement in symptoms, quality of life, and short- and medium-term survival. However, the high frequency of postimplant adverse events and the high economic and resource costs are still barriers to adopting this treatment for a broader population of patients with advHFrEF. Therefore, improving the patient selection process is crucial to making LVAD implantation an acceptable and cost-effective alternative to orthotopic heart transplantation.
Author Contributions
Conceptualization, D.M.; writing—original draft preparation, D.M., L.F., M.L.M., D.C., F.V., R.G., C.C. and A.P.; writing—review and editing, B.H., R.J.T., M.D.F. and G.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No data were generated in this study.
Conflicts of Interest
The authors report no conflict of interest in this work.
References
- Truby, L.K.; Rogers, J.G. Advanced Heart Failure: Epidemiology, Diagnosis, and Therapeutic Approaches. JACC Heart Fail. 2020, 8, 523–536. [Google Scholar] [PubMed]
- Masarone, D.; Kittleson, M.; Petraio, A.; Pacileo, G. Advanced heart failure: State of the art and future directions. Rev. Cardiovasc. Med. 2022, 23, 48–62. [Google Scholar] [PubMed]
- Bhagra, S.K.; Pettit, S.; Parameshwar, J. Cardiac transplantation: Indications, eligibility and current outcomes. Heart 2019, 105, 252–260. [Google Scholar]
- Frigerio, M. Left Ventricular Assist Device: Indication, Timing, and Management. Heart Fail. Clin. 2021, 17, 619–634. [Google Scholar]
- Masarone, D.; Melillo, E.; Gravino, R.; Errigo, V.; Martucci, M.L.; Caiazzo, A.; Petraio, A.; Pölzl, G.; Pacileo, G. Inotropes in Patients with Advanced Heart Failure: Not Only Palliative Care. Heart Fail. Clin. 2021, 17, 587–598. [Google Scholar]
- Mehra, M.R.; Goldstein, D.J.; Cleveland, J.C.; Cowger, J.A.; Hall, S.; Salerno, C.T.; Naka, Y.; Horstmanshof, D.; Chuang, J.; Wang, A.; et al. Five-Year Outcomes in Patients with Fully Magnetically Levitated vs. Axial-Flow Left Ventricular Assist Devices in the MOMENTUM 3 Randomized Trial. JAMA 2022, 328, 1233–1242. [Google Scholar]
- Plecash, A.R.; Byrne, D.; Flexman, A.; Toma, M.; Field, T.S. Stroke in Patients with Left Ventricular Assist Devices. Cerebrovasc. Dis. 2022, 51, 3–13. [Google Scholar] [CrossRef]
- Inglis, S.S.; Suh, G.A.; Razonable, R.R.; Schettle, S.D.; Spencer, P.J.; Villavicencio, M.A.; Rosenbaum, A.N. Infections in Patients with Left Ventricular Assist Devices: Current State and Future Perspectives. ASAIO J. 2023, 69, 633–641. [Google Scholar]
- Han, J.J.; Acker, M.A.; Atluri, P. Left Ventricular Assist Devices. Circulation 2018, 138, 2841–2851. [Google Scholar]
- Bravo, C.A.; Navarro, A.G.; Dhaliwal, K.K.; Khorsandi, M.; Keenan, J.E.; Mudigonda, P.; O’Brien, K.D.; Mahr, C. Right heart failure after left ventricular assist device: From mechanisms to treatments. Front. Cardiovasc. Med. 2022, 9, 1023549. [Google Scholar]
- Stevenson, L.W.; Pagani, F.D.; Young, J.B.; Jessup, M.; Miller, L.; Kormos, R.L.; Naftel, D.C.; Ulisney, K.; Desvigne-Nickens, P.; Kirklin, J.K. INTERMACS profiles of advanced heart failure: The current picture. J. Heart Lung Transplant. 2009, 28, 535–541. [Google Scholar]
- Kormos, R.L.; Cowger, J.; Pagani, F.D.; Teuteberg, J.J.; Goldstein, D.J.; Jacobs, J.P.; Higgins, R.S.; Stevenson, L.W.; Stehlik, J.; Atluri, P.; et al. The Society of Thoracic Surgeons Intermacs Database Annual Report: Evolving Indications, Outcomes, and Scientific Partnerships. Ann. Thorac. Surg. 2019, 107, 341–353. [Google Scholar] [PubMed]
- Melendo-Viu, M.; Dobarro, D.; Raposeiras Roubin, S.; Llamas Pernas, C.; Moliz Cordón, C.; Vazquez Lamas, M.; Piñón Esteban, M.; Varela Martínez, M.Á.; Abu Assi, E.; Pita Romero, R.; et al. Left Ventricular Assist Device as a Destination Therapy: Current Situation and the Importance of Patient Selection. Life 2023, 13, 1065–1079. [Google Scholar] [PubMed]
- Bansal, N.; Hailpern, S.M.; Katz, R.; Hall, Y.N.; Tamura, M.K.; Kreuter, W.; O’Hare, A.M. Outcomes associated with left ventricular assist devices among recipients with and without end-stage renal disease. JAMA Intern. Med. 2018, 178, 204–209. [Google Scholar]
- Saeed, D.; Feldman, D.; Banayosy, A.E.; Birks, E.; Blume, E.; Cowger, J.; Hayward, C.; Jorde, U.; Kremer, J.; MacGowan, G.; et al. The 2023 International Society for Heart and Lung Transplantation Guidelines for Mechanical Circulatory Support: A 10-Year Update. J. Heart Lung Transplant. 2023, 42, e1–e222. [Google Scholar] [PubMed]
- Crespo-Leiro, M.G.; Metra, M.; Lund, L.H.; Milicic, D.; Costanzo, M.R.; Filippatos, G.; Gustafsson, F.; Tsui, S.; Barge-Caballero, E.; De Jonge, N.; et al. Advanced heart failure: A position statement of the Heart Failure Association of the European Society of Cardiology. Eur. J. Heart Fail. 2018, 20, 1505–1535. [Google Scholar]
- Morris, A.A.; Khazanie, P.; Drazner, M.H.; Albert, N.M.; Breathett, K.; Cooper, L.B.; Eisen, H.J.; O’Gara, P.; Russell, S.D.; American Heart Association Heart Failure and Transplantation Committee of the Council on Clinical Cardiology; et al. Guidance for Timely and Appropriate Referral of Patients With Advanced Heart Failure: A Scientific Statement From the American Heart Association. Circulation 2021, 144, e238–e250. [Google Scholar]
- Metra, M.; Tomasoni, D.; Adamo, M.; Bayes-Genis, A.; Filippatos, G.; Abdelhamid, M.; Adamopoulos, S.; Anker, S.D.; Antohi, L.; Böhm, M.; et al. Worsening of chronic heart failure: Definition, epidemiology, management and prevention. A clinical consensus statement by the Heart Failure Association of the European Society of Cardiology. Eur. J. Heart Fail. 2023, 25, 776–779. [Google Scholar]
- Setoguchi, S.; Stevenson, L.W.; Schneeweiss, S. Repeated hospitalizations predict mortality in the community population with heart failure. Am. Heart J. 2007, 154, 260–266. [Google Scholar]
- Chimed, S.; Stassen, J.; Galloo, X.; Meucci, M.C.; van der Bijl, P.; Knuuti, J.; Delgado, V.; Marsan, N.A.; Bax, J.J. Impact of Worsening Heart Failure on Long-Term Prognosis in Patients with Heart Failure with Reduced Ejection Fraction. Am. J. Cardiol. 2022, 184, 63–71. [Google Scholar]
- Tran, R.H.; Aldemerdash, A.; Chang, P.; Sueta, C.A.; Kaufman, B.; Asafu-Adjei, J.; Vardeny, O.; Daubert, E.; Alburikan, K.A.; Kucharska-Newton, A.M.; et al. Guideline-Directed Medical Therapy and Survival Following Hospitalization in Patients with Heart Failure. Pharmacotherapy 2018, 38, 406–416. [Google Scholar] [PubMed]
- Bhagat, A.A.; Greene, S.J.; Vaduganathan, M.; Fonarow, G.C.; Butler, J. Initiation, Continuation, Switching, and Withdrawal of Heart Failure Medical Therapies During Hospitalization. JACC Heart Fail. 2019, 7, 1–12. [Google Scholar] [PubMed]
- Kittleson, M.; Hurwitz, S.; Shah, M.R.; Nohria, A.; Lewis, E.; Givertz, M.; Fang, J.; Jarcho, J.; Mudge, G.; Stevenson, L.W. Development of circulatory-renal limitations to angiotensin-converting enzyme inhibitors identifies patients with severe heart failure and early mortality. J. Am. Coll. Cardiol. 2003, 41, 2029–2035. [Google Scholar]
- Stevenson, L.W. Clinical use of inotropic therapy for heart failure: Looking backward or forward? Part II: Chronic inotropic therapy. Circulation 2003, 108, 492–497. [Google Scholar]
- Eshaghian, S.; Horwich, T.B.; Fonarow, G.C. Relation of loop diuretic dose to mortality in advanced heart failure. Am. J. Cardiol. 2006, 97, 1759–1764. [Google Scholar] [CrossRef] [PubMed]
- Poole, J.E.; Johnson, G.W.; Hellkamp, A.S.; Anderson, J.; Callans, D.J.; Raitt, M.H.; Reddy, R.K.; Marchlinski, F.E.; Yee, R.; Guarnieri, T.; et al. Prognostic importance of defibrillator shocks in patients with heart failure. N. Engl. J. Med. 2008, 359, 1009–1017. [Google Scholar] [CrossRef]
- Tung, R.; Vaseghi, M.; Frankel, D.S.; Vergara, P.; Di Biase, L.; Nagashima, K.; Yu, R.; Vangala, S.; Tseng, C.H.; Choi, E.K.; et al. Freedom from recurrent ventricular tachycardia after catheter ablation is associated with improved survival in patients with structural heart disease: An International VT Ablation Center Collaborative Group study. Heart Rhythm. 2015, 12, 1997–2007. [Google Scholar]
- Gheorghiade, M.; Abraham, W.T.; Albert, N.M.; Gattis Stough, W.; Greenberg, B.H.; O’Connor, C.M.; She, L.; Yancy, C.W.; Young, J.; Fonarow, G.C.; et al. Relationship between admission serum sodium concentration and clinical outcomes in patients hospitalized for heart failure: An analysis from the OPTIMIZE-HF registry. Eur. Heart J. 2007, 28, 980–988. [Google Scholar]
- O’Connor, C.M.; Abraham, W.T.; Albert, N.M.; Clare, R.; Gattis Stough, W.; Gheorghiade, M.; Greenberg, B.H.; Yancy, C.W.; Young, J.B.; Fonarow, G.C. Predictors of mortality after discharge in patients hospitalized with heart failure: An analysis from the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE-HF). Am. Heart J. 2008, 156, 662–673. [Google Scholar]
- Binanay, C.; Califf, R.M.; Hasselblad, V.; O’Connor, C.M.; Shah, M.R.; Sopko, G.; Stevenson, L.W.; Francis, G.S.; Leier, C.V.; Miller, L.W.; et al. Evaluation study of congestive heart failure and pulmonary artery catheterization effectiveness: The ESCAPE trial. JAMA 2005, 294, 1625–1633. [Google Scholar]
- Rothenburger, M.; Wichter, T.; Schmid, C.; Stypmann, J.; Tjan, T.D.; Berendes, E.; Etz, C.; Pioux, A.; Löher, A.; Wenzelburger, F.; et al. Aminoterminal pro type B natriuretic peptide as a predictive and prognostic marker in patients with chronic heart failure. J. Heart Lung Transplant. 2004, 23, 1189–1197. [Google Scholar]
- Klimczak-Tomaniak, D.; de Bakker, M.; Bouwens, E.; Akkerhuis, K.M.; Baart, S.; Rizopoulos, D.; Mouthaan, H.; van Ramshorst, J.; Germans, T.; Constantinescu, A.; et al. Dynamic personalized risk prediction in chronic heart failure patients: A longitudinal, clinical investigation of 92 biomarkers (Bio-SHiFT study). Sci. Rep. 2022, 12, 2795. [Google Scholar]
- Melillo, E.; Masarone, D.; Oh, J.K.; Verrengia, M.; Valente, F.; Vastarella, R.; Ammendola, E.; Pacileo, R.; Pacileo, G. Echocardiography in Advanced Heart Failure for Diagnosis, Management, and Prognosis. Heart Fail. Clin. 2021, 17, 547–560. [Google Scholar]
- Omar, A.M.; Bansal, M.; Sengupta, P.P. Advances in echocardiographic imaging in heart failure with reduced and preserved ejection fraction. Circ. Res. 2016, 119, 357–374. [Google Scholar]
- Kittleson, M.M.; Prestinenzi, P.; Potena, L. Right Heart Catheterization in Patients with Advanced Heart Failure: When to Perform? How to Interpret? Heart Fail. Clin. 2021, 17, 647–660. [Google Scholar]
- Allen, L.A.; Rogers, J.G.; Warnica, J.W.; Disalvo, T.G.; Tasissa, G.; Binanay, C.; O’Connor, C.M.; Califf, R.M.; Leier, C.V.; Shah, M.R.; et al. High mortality without ESCAPE: The registry of heart failure patients receiving pulmonary artery catheters without randomization. J. Card. Fail. 2008, 14, 661–669. [Google Scholar]
- Bhagra, S.K.; Pettit, S.; Parameshwar, J. Implantable left ventricular assist device: Indications, eligibility and current outcomes. Heart 2022, 108, 233–241. [Google Scholar]
- Sandoval, E.; Carillo, J.A.; Singh, S.K. Prometheus’ predicament: How to address contemporary left ventricular assist devices in patients with liver dysfunction. J. Thorac. Cardiovasc. Surg. 2016, 151, 236–237. [Google Scholar] [CrossRef][Green Version]
- Asleh, R.; Briasoulis, A.; Schettle, S.D.; Tchantchaleishvili, V.; Pereira, N.L.; Edwards, B.S.; Clavell, A.L.; Maltais, S.; Joyce, D.L.; Joyce, L.D.; et al. Impact of Diabetes Mellitus on Outcomes in Patients Supported With Left Ventricular Assist Devices: A Single Institutional 9-Year Experience. Circ. Heart Fail. 2017, 10, e004213. [Google Scholar]
- Kushnir, V.M.; Sharma, S.; Ewald, G.A.; Seccombe, J.; Novak, E.; Wang, I.W.; Joseph, S.M.; Gyawali, C.P. Evaluation of GI bleeding after implantation of left ventricular assist device. Gastrointest. Endosc. 2012, 75, 973–979. [Google Scholar]
- Nozdrzykowski, M.; Bauer, J.M.; Schulz, U.; Jawad, K.; Bireta, C.; Eifert, S.; Sandri, M.; Jozwiak-Nozdrzykowska, J.; Borger, M.A.; Saeed, D. Stroke and pump thrombosis following left ventricular assist device implantation: The impact of the implantation technique. Front. Cardiovasc. Med. 2023, 10, 974527. [Google Scholar]
- Rodenas-Alesina, E.; Brahmbhatt, D.H.; Rao, V.; Salvatori, M.; Billia, F. Prediction, prevention, and management of right ventricular failure after left ventricular assist device implantation: A comprehensive review. Front. Cardiovasc. Med. 2022, 9, 1040251. [Google Scholar]
- Raymer, D.S.; Moreno, J.D.; Sintek, M.A.; Nassif, M.E.; Sparrow, C.T.; Adamo, L.; Novak, E.L.; LaRue, S.J.; Vader, J.M. The combination of Tricuspid Annular Plane Systolic Excursion and HeartMate Risk Score Predicts Right Ventricular Failure after Left Ventricular Assist Device Implantation. ASAIO J. 2019, 6, 247–251. [Google Scholar]
- Chriqui, L.E.; Monney, P.; Kirsch, M.; Tozzi, P. Prediction of right ventricular failure after left ventricular assist device implantation in patients with heart failure: A meta-analysis comparing echocardiographic parameters. Interact. Cardiovasc. Thorac. Surg. 2021, 33, 784–792. [Google Scholar]
- Kato, T.S.; Jiang, J.; Schulze, P.C.; Jorde, U.; Uriel, N.; Kitada, S.; Takayama, H.; Naka, Y.; Mancini, D.; Gillam, L.; et al. Serial echocardiography using tissue Doppler and speckle tracking imaging to monitor right ventricular failure before and after left ventricular assist device surgery. JACC Heart Fail. 2013, 1, 216–222. [Google Scholar]
- Raina, A.; Seetha Rammohan, H.R.; Gertz, Z.M.; Rame, J.E.; Woo, Y.J.; Kirkpatrick, J.N. Postoperative right ventricular failure after left ventricular assist device placement is predicted by preoperative echocardiographic structural, hemodynamic, and functional parameters. J. Card. Fail. 2013, 19, 16–24. [Google Scholar]
- He, Q.; Lin, Y.; Zhu, Y.; Gao, L.; Ji, M.; Zhang, L.; Xie, M.; Li, Y. Clinical Usefulness of Right Ventricle-Pulmonary Artery Coupling in Cardiovascular Disease. J. Clin. Med. 2023, 12, 2526–2534. [Google Scholar]
- Dandel, M.; Potapov, E.; Krabatsch, T.; Stepanenko, A.; Löw, A.; Vierecke, J.; Knosalla, C.; Hetzer, R. Load dependency of right ventricular performance is a major factor to be considered in decision making before ventricular assist device implantation. Circulation 2013, 128, S14–S23. [Google Scholar]
- Dandel, M.; Javier, M.F.D.M.; Javier Delmo, E.M.D.; Hetzer, R. Accurate assessment of right heart function before and after long-term left ventricular assist device implantation. Expert Rev. Cardiovasc. Ther. 2020, 18, 289–308. [Google Scholar]
- Bouabdallaoui, N.; El-Hamamsy, I.; Pham, M.; Giraldeau, G.; Parent, M.C.; Carrier, M.; Rouleau, J.L.; Ducharme, A. Aortic regurgitation in patients with a left ventricular assist device: A contemporary review. J. Heart Lung Transplant. 2018, 37, 1289–1297. [Google Scholar]
- Acharya, D.; Kazui, T.; Al Rameni, D.; Acharya, T.; Betterton, E.; Juneman, E.; Loyaga-Rendon, R.; Lotun, K.; Shetty, R.; Chatterjee, A. Aortic valve disorders and left ventricular assist devices. Front. Cardiovasc. Med. 2023, 10, 1098348. [Google Scholar]
- Wang, T.S.; Hernandez, A.F.; Felker, G.M.; Milano, C.A.; Rogers, J.G.; Patel, C.B. Valvular heart disease in patients supported with left ventricular assist devices. Circ. Heart Fail. 2014, 7, 215–222. [Google Scholar]
- Noly, P.E.; Duggal, N.; Jiang, M.; Nordsletten, D.; Bonini, M.; Lei, I.; Ela, A.A.E.; Haft, J.W.; Pagani, F.D.; Cascino, T.M.; et al. Role of the mitral valve in left ventricular assist device pathophysiology. Front. Cardiovasc. Med. 2022, 9, 1018295. [Google Scholar]
- Kassis, H.; Cherukuri, K.; Agarwal, R.; Kanwar, M.; Elapavaluru, S.; Sokos, G.G.; Moraca, R.J.; Bailey, S.H.; Murali, S.; Benza, R.L.; et al. Significance of Residual Mitral Regurgitation After Continuous Flow Left Ventricular Assist Device Implantation. JACC Heart Fail. 2017, 5, 81–88. [Google Scholar]
- Westaby, S. Tricuspid regurgitation in left ventricular assist device patients. Eur. J. Cardiothorac. Surg. 2012, 41, 217–218. [Google Scholar] [CrossRef]
- Fujino, T.; Imamura, T.; Nitta, D.; Kim, G.; Smith, B.; Kalantari, S.; Nguyen, A.; Chung, B.; Narang, N.; Holzhauser, L.; et al. Effect of Concomitant Tricuspid Valve Surgery with Left Ventricular Assist Device Implantation. Ann. Thorac. Surg. 2020, 110, 918–924. [Google Scholar]
- John, R.; Naka, Y.; Park, S.J.; Sai-Sudhakar, C.; Salerno, C.; Sundareswaran, K.S.; Farrar, D.J.; Milano, C.A. Impact of concurrent surgical valve procedures in patients receiving continuous-flow devices. J. Thorac. Cardiovasc. Surg. 2014, 147, 581–589. [Google Scholar]
- Loforte, A.; Gliozzi, G.; Mariani, C.; Cavalli, G.G.; Martin-Suarez, S.; Pacini, D. Ventricular assist devices implantation: Surgical assessment and technical strategies. Cardiovasc. Diagn. Ther. 2021, 11, 277–291. [Google Scholar]
- Belzile, D.; Turgeon, P.Y.; Leblanc, E.; Massot, M.; Bourgault, C.; Morin, J.; Dupuis, C.; Bilodeau, M.; Laflamme, M.; Charbonneau, E.; et al. Non-invasive diagnostic imaging tests largely underdiagnose cardiac cirrhosis in patients undergoing advanced therapy evaluation: How can we identify the high-risk patient? Clin. Transplant. 2021, 35, e14277. [Google Scholar]
- Critsinelis, A.; Kurihara, C.; Volkovicher, N.; Kawabori, M.; Sugiura, T.; Manon, M., 2nd; Wang, S.; Civitello, A.B.; Morgan, J.A. Model of End-Stage Liver Disease-eXcluding International Normalized Ratio (MELD-XI) Scoring System to Predict Outcomes in Patients Who Undergo Left Ventricular Assist Device Implantation. Ann. Thorac. Surg. 2018, 106, 513–519. [Google Scholar]
- Cushing, K.; Kushnir, V. Gastrointestinal Bleeding Following LVAD Placement from Top to Bottom. Dig. Dis. Sci. 2016, 61, 1440–1447. [Google Scholar]
- Kapuria, D.; Khumri, T.; Shamim, S.; Surana, P.; Khan, S.; Al-Khalisi, N.; Aggarwal, S.; Koh, C.; Chhabra, R. Characterization and timing of gastrointestinal bleeding in continuous flow left ventricular assist device recipients. Heliyon 2020, 6, e04695. [Google Scholar]
- Cochran, J.M.; Alam, A.; Guerrero-Miranda, C.Y. Importance of right heart catheterization in advanced heart failure management. Rev. Cardiovasc. Med. 2022, 23, 12. [Google Scholar] [CrossRef]
- Shehab, S.; Newton, P.J.; Allida, S.M.; Jansz, P.C.; Hayward, C.S. Biventricular mechanical support devices—Clinical perspectives. Expert Rev. Med. Devices 2016, 13, 353–365. [Google Scholar] [CrossRef]
- DeFilippis, E.M.; Breathett, K.; Donald, E.M.; Nakagawa, S.; Takeda, K.; Takayama, H.; Truby, L.K.; Sayer, G.; Colombo, P.C.; Yuzefpolskaya, M.; et al. Psychosocial Risk and Its Association with Outcomes in Continuous-Flow Left Ventricular Assist Device Patients. Circ. Heart Fail. 2020, 13, e006910. [Google Scholar]
- Maukel, L.M.; Weidner, G.; Jan, B.; Spaderna, H. Adverse events after left ventricular assist device implantation linked to psychosocial risk in women and men. J. Heart Lung Transplant. 2023, in press. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).